The Role of Thyroid Disorders, Obesity, Diabetes Mellitus and Estrogen Exposure as Potential Modifiers for Pulmonary Hypertension
Abstract
1. Introduction
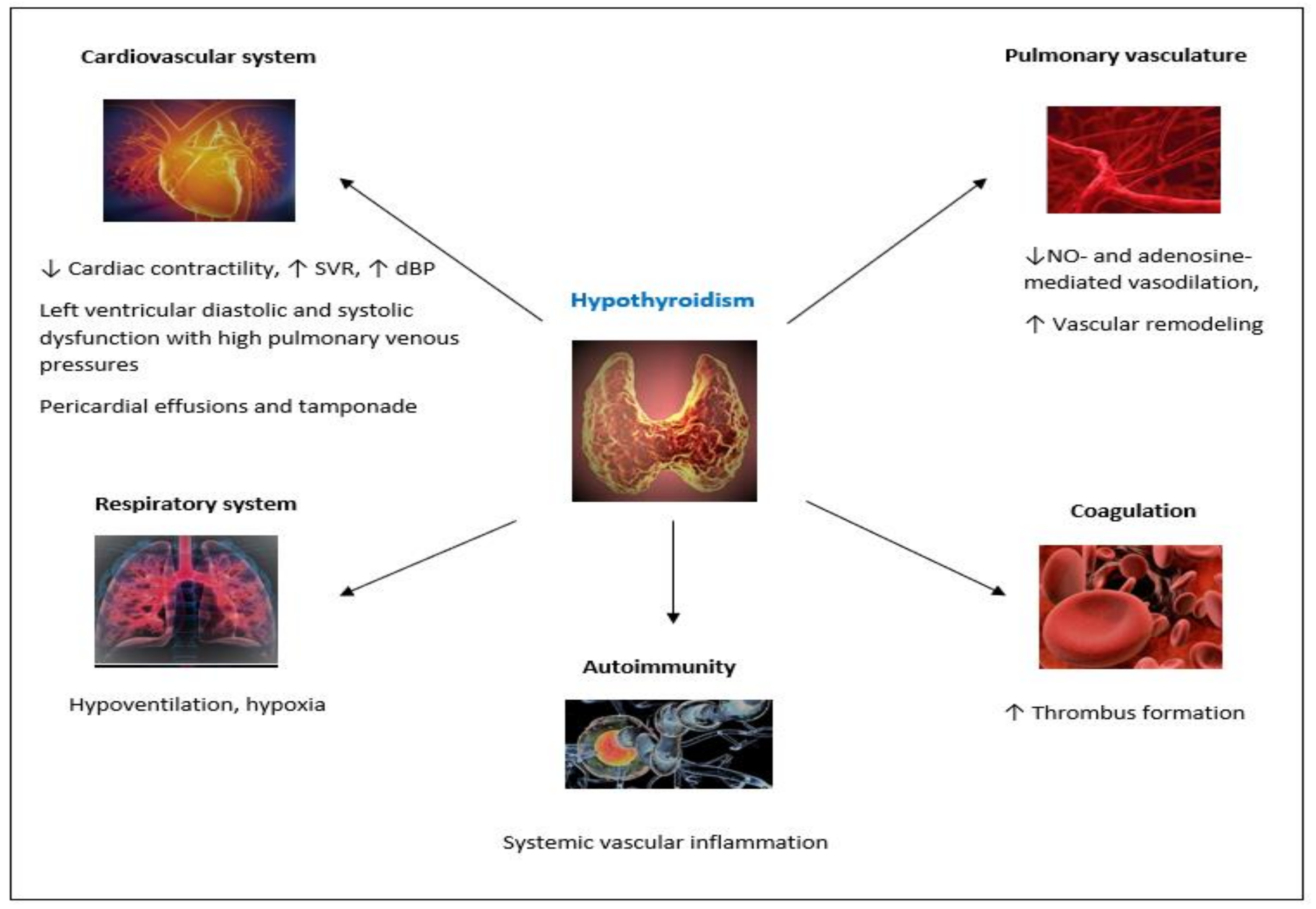
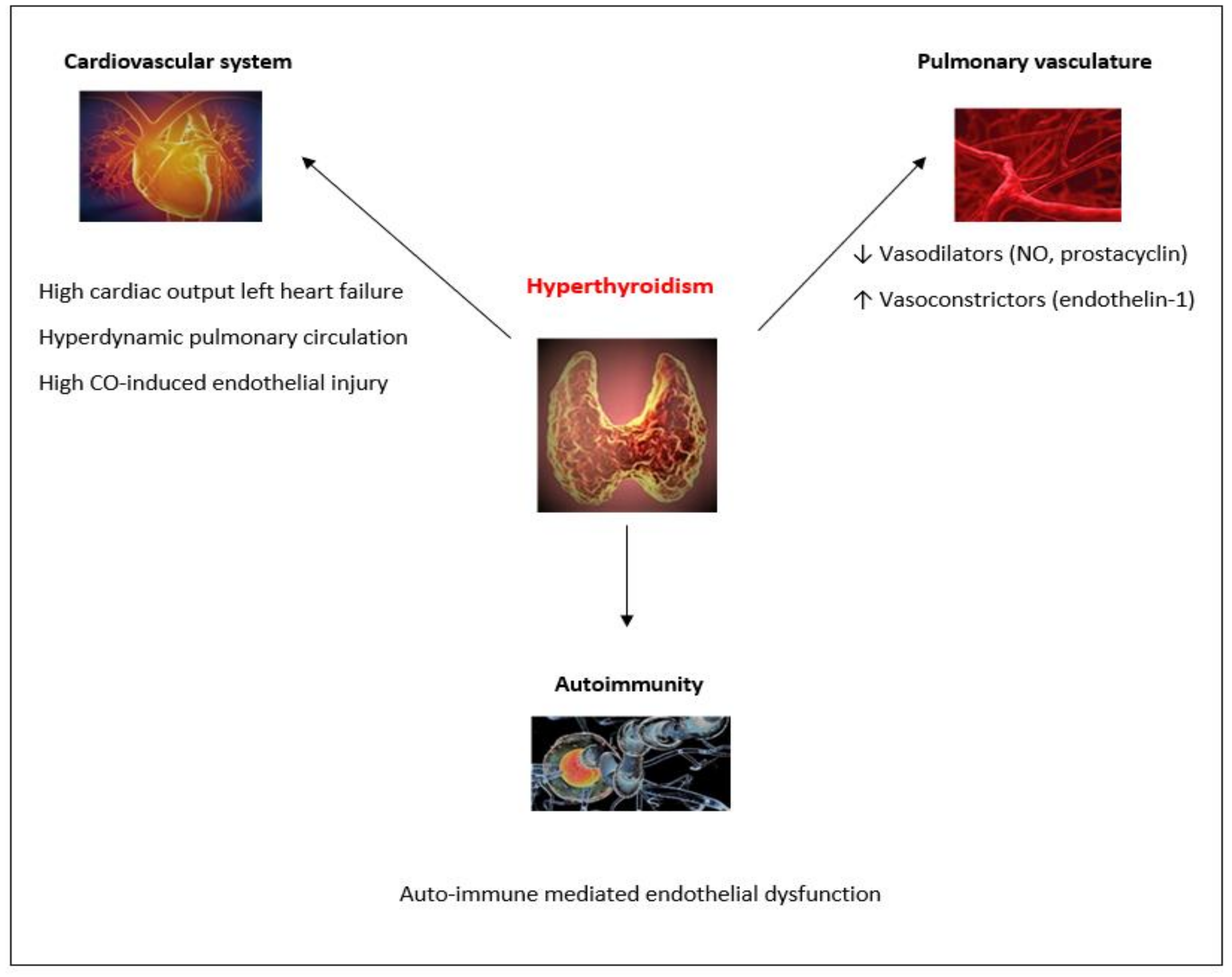
2. Thyroid Dysfunction and PH
2.1. Thyroid Hormones
2.2. Thyroid Diseases in PH Patients
2.3. Biologically Plausible Pathogenetic Mechanisms Linking Thyroid Disorders with PH
3. Obesity
4. Diabetes Mellitus
5. Estrogen Exposure
6. Other Endocrine Disorders and PH
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Galiè, N.; Humbert, M.; Vachiery, J.L.; Gibbs, S.; Lang, I.; Torbicki, A.; Simonneau, G.; Peacock, A.; Vonk Noordegraaf, A.; Beghetti, M.; et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). Eur. Heart J. 2016, 37, 67–119. [Google Scholar] [CrossRef] [PubMed]
- Simonneau, G.; Montani, D.; Celermajer, D.S.; Denton, C.P.; Gatzoulis, M.A.; Krowka, M.; Williams, P.G.; Souza, R. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur. Respir. J. 2019, 53, 1801913. [Google Scholar] [CrossRef] [PubMed]
- Huber, L.C.; Bye, H.; Brock, M. Swiss Society of Pulmonary Hypertension. The pathogenesis of pulmonary hypertension—An update. Swiss Med. Wkly. 2015, 145, w14202. [Google Scholar] [PubMed]
- Lan, N.S.H.; Massam, B.D.; Kulkarni, S.S.; Lang, C.C. Pulmonary Arterial Hypertension: Pathophysiology and Treatment. Diseases 2018, 6, 38. [Google Scholar] [CrossRef]
- Simonneau, G.; Galiè, N.; Rubin, L.J.; Langleben, D.; Seeger, W.; Domenighetti, G.; Gibbs, S.; Lebrec, D.; Speich, R.; Beghetti, M. Clinical classification of pulmonary hypertension. J. Am. Coll. Cardiol. 2004, 43, S5–S12. [Google Scholar] [CrossRef]
- Ferris, A.; Jacobs, T.; Widlitz, A.; Barst, R.J.; Morse, J.H. Pulmonary arterial hypertension and thyroid disease. Chest 2001, 119, 1980–1981. [Google Scholar] [CrossRef]
- Badesch, D.B.; Raskob, G.E.; Elliott, C.G.; Krichman, A.M.; Farber, H.W.; Frost, A.E.; Barst, R.J.; Benza, R.L.; Liou, T.G.; Turner, M.; et al. Pulmonary arterial hypertension: Baseline characteristics from the REVEAL Registry. Chest 2010, 137, 376–387. [Google Scholar] [CrossRef]
- Bogaard, H.J.; Al Husseini, A.; Farkas, L.; Farkas, D.; Gomez-Arroyo, J.; Abbate, A.; Voelkel, N.F. Severe pulmonary hypertension: The role of metabolic and endocrine disorders. Pulm. Circ. 2012, 2, 148–154. [Google Scholar] [CrossRef]
- Chin, R.G.; Fisher, J. Hypothyroidism and pulmonary hypertension: Observation and speculation. Cardiovasc. Rev. Rep. 1986, 9, 789–796. [Google Scholar]
- Lang, I.M.; Palazzini, M. The burden of comorbidities in pulmonary arterial hypertension. Eur. Heart J. Suppl. 2019, 21, K21–K28. [Google Scholar] [CrossRef]
- Kahaly, G.J.; Dillmann, W.H. Thyroid hormone action in the heart. Endocr. Rev. 2005, 26, 704–728. [Google Scholar] [CrossRef]
- Jabbar, A.; Pingitore, A.; Pearce, S.H.S.; Zaman, A.; Iervasi, G.; Razvi, S. Thyroid hormones and cardiovascular disease. Nat. Rev. Cardiol. 2017, 14, 39–55. [Google Scholar] [CrossRef]
- Senese, R.; Lasala, P.; Leanza, C.; de Lange, P. New avenues for regulation of lipid metabolism by thyroid hormones and analogs. Front. Physiol. 2014, 5, 475. [Google Scholar] [CrossRef]
- Li, J.H.; Safford, R.E.; Aduen, J.F.; Heckman, M.G.; Crook, J.E.; Burger, C.D. Pulmonary hypertension and thyroid disease. Chest 2007, 132, 793–797. [Google Scholar] [CrossRef]
- Curnock, A.L.; Dweik, R.A.; Higgins, B.H.; Saadi, H.F.; Arroliga, A.C. High prevalence of hypothyroidism in patients with primary pulmonary hypertension. Am. J. Med. Sci. 1999, 318, 289–292. [Google Scholar] [CrossRef]
- Silva, D.R.; Gazzana, M.B.; John, A.B.; Siqueira, D.R.; Maia, A.L.; Barreto, S.S. Pulmonary arterial hypertension and thyroid disease. J. Bras. Pneumol. Publicacao Soc. Bras. Pneumol. Tisilogia 2009, 35, 179–185. [Google Scholar]
- Krieg, V.J.; Hobohm, L.; Liebetrau, C.; Guth, S.; Kölmel, S.; Troidl, C.; Mayer, E.; Konstantinides, S.V.; Wiedenroth, C.B.; Lankeit, M. Risk factors for chronic thromboembolic pulmonary hypertension-Importance of thyroid disease and function. Thromb. Res. 2020, 185, 20–26. [Google Scholar] [CrossRef]
- Bonderman, D.; Wilkens, H.; Wakounig, S.; Schäfers, H.J.; Jansa, P.; Lindner, J.; Simkova, I.; Martischnig, A.M.; Dudczak, J.; Sadushi, R.; et al. Risk factors for chronic thromboembolic pulmonary hypertension. Eur. Respir. J. 2009, 33, 325–331. [Google Scholar] [CrossRef]
- Delcroix, M.; Kerr, K.; Fedullo, P. Chronic Thromboembolic Pulmonary Hypertension. Epidemiology and Risk Factors. Ann. Am. Thorac. Soc. 2016, 13, S201–S206. [Google Scholar] [CrossRef]
- Robledo, G.C.; Lucas, S.A.G.; Pola, G.B. Evaluation of thyroid function in pulmonary arterial hypertension. Eur. Respir. J. 2017, 50, PA2420. [Google Scholar]
- Vakilian, F.; Attaran, D.; Shegofte, M.; Lari, S.; Ghare, S. Assessment of Thyroid Function in Idiopathic Pulmonary Hypertension. Res. Cardiovasc. Med. 2016, 5, e29361. [Google Scholar] [CrossRef]
- Richter, M.J.; Sommer, N.; Schermuly, R.; Grimminger, B.; Seeger, W.; Tello, K.; Ghofrani, H.A.; Gall, H. The prognostic impact of thyroid function in pulmonary hypertension. J. Heart Lung Transplant. 2016, 35, 1427–1434. [Google Scholar] [CrossRef]
- Chu, J.W.; Kao, P.N.; Faul, J.L.; Doyle, R.L. High prevalence of autoimmune thyroid disease in pulmonary arterial hypertension. Chest 2002, 122, 1668–1673. [Google Scholar] [CrossRef]
- Satoh, M.; Aso, K.; Nakayama, T.; Naoi, K.; Ikehara, S.; Uchino, Y.; Shimada, H.; Takatsuki, S.; Matsuura, H.; Saji, T. Autoimmune thyroid disease in children and adolescents with idiopathic pulmonary arterial hypertension. Circ. J. 2010, 74, 371–374. [Google Scholar] [CrossRef]
- Marvisi, M.; Brianti, M.; Marani, G.; Del Borello, R.; Bortesi, M.L.; Guariglia, A. Hyperthyroidism and pulmonary hypertension. Respir. Med. 2002, 96, 215–220. [Google Scholar] [CrossRef][Green Version]
- Vallabhajosula, S.; Radhi, S.; Cevik, C.; Alalawi, R.; Raj, R.; Nugent, K. Hyperthyroidism and pulmonary hypertension: An important association. Am. J. Med. Sci. 2011, 342, 507–512. [Google Scholar] [CrossRef]
- Siu, C.W.; Zhang, X.H.; Yung, C.; Kung, A.W.; Lau, C.P.; Tse, H.F. Hemodynamic changes in hyperthyroidism-related pulmonary hypertension: A prospective echocardiographic study. J. Clin. Endocrinol. Metab. 2007, 92, 1736–1742. [Google Scholar] [CrossRef]
- Armigliato, M.; Paolini, R.; Aggio, S.; Zamboni, S.; Galasso, M.P.; Zonzin, P.; Cella, G. Hyperthyroidism as a cause of pulmonary arterial hypertension: A prospective study. Angiology 2006, 57, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Trapp, C.M.; Elder, R.W.; Gerken, A.T.; Sopher, A.B.; Lerner, S.; Aranoff, G.S.; Rosenzweig, E.B. Pediatric pulmonary arterial hypertension and hyperthyroidism: A potentially fatal combination. J. Clin. Endocrinol. Metab. 2012, 97, 2217–2222. [Google Scholar] [CrossRef]
- Merce, J.; Ferras, S.; Oltra, C.; Sanz, E.; Vendrell, J.; Simon, I.; Camprubı, M.; Bardajı, A.; Ridao, C. Cardiovascular abnormalities in hyperthyroidism: A prospective Doppler echocardiographic study. Am. J. Med. 2005, 118, 126–131. [Google Scholar] [CrossRef]
- Marvisi, M.; Zambrelli, P.; Brianti, M.; Civardi, G.; Lampugnani, R.; Delsignore, R. Pulmonary hypertension is frequent in hyperthyroidism and normalizes after therapy. Eur. J. Intern. Med. 2006, 17, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Chadha, C.; Pritzker, M.; Mariash, C.N. Effect of epoprostenol on the thyroid gland: Enlargement and secretion of thyroid hormone. Endocr. Pract. 2009, 15, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Ferris, A.M.; Morse, J.H.; Jacobs, T.; Rosenzweig, E.B.; Aranoff, G.S.; Barst, R.J. Thyroid disease and pulmonary arterial hypertension (PAH) in patients treated with prostacyclin. Circulation 2000, 102, 426. [Google Scholar]
- Marvisi, M.; Balzarini, L.; Mancini, C.; Mouzakiti, P. Thyroid gland and pulmonary hypertension. What’s the link? Panminerva Med. 2013, 55, 93–97. [Google Scholar]
- Klein, I.; Danzi, S. Thyroid disease and the heart. Circulation 2007, 116, 1725–1735. [Google Scholar] [CrossRef]
- Vargas, F.; Moreno, J.M.; Rodriguez-Gomez, I.; Wangensteen, R.; Osuna, A.; Alvarez-Guerra, M.; Garcia-Estan, J. Vascular and renal function in experimental thyroid disorders. Eur. J. Endocrinol. 2006, 154, 197–212. [Google Scholar] [CrossRef]
- Osmak-Tizon, L.; Poussier, M.; Cottin, Y.; Rochette, L. Non-genomic actions of thyroid hormones: Molecular aspects. Arch. Cardiovasc. Dis. 2014, 107, 207–211. [Google Scholar] [CrossRef]
- Tamajusuku, A.S.; Carrillo-Sepulveda, M.A.; Braganhol, E.; Wink, M.R.; Sarkis, J.J.; Barreto-Chaves, M.L.; Battastini, A.M. Activity and expression of ecto-50-nucleotidase/CD73 are increased by thyroid hormones in vascular smooth muscle cells. Mol. Cell. Biochem. 2006, 289, 65–72. [Google Scholar] [CrossRef]
- Fukuyama, K.; Ichiki, T.; Imayama, I.; Ohtsubo, H.; Ono, H.; Hashiguchi, Y.; Takeshita, A.; Sunagawa, K. Thyroid hormone inhibits vascular remodeling through suppression of cAMP response element binding protein activity. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2049–2055. [Google Scholar] [CrossRef]
- Badesch, D.B.; Wynne, K.M.; Bonvallet, S.; Voelkel, N.F.; Ridgway, C.; Groves, B.M. Hypothyroidism and primary pulmonary hypertension: An autoimmune pathogenetic link? Ann. Intern. Med. 1993, 119, 44–46. [Google Scholar] [CrossRef]
- Hostiuc, M.; Curca, G.C.; Dermengiu, D.; Sinescu, C.; Hostiuc, S. Can subclinical hypothyroidism explain some sudden deaths due to pulmonary embolism without evident risk factors? Med. Hypotheses 2011, 76, 855–857. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.E.; Barst, R.J.; McElroy, J.J.; Widlitz, A.; Chada, K.; Knowles, J.A.; Morse, J.H. Bone morphogenetic protein receptor 2 mutations in adults and children with idiopathic pulmonary arterial hypertension: Association with thyroid disease. Chest 2005, 128, S618. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, T.; Yamanaka, S.; Takeuchi, H.; Morimoto, N.; Kamioka, M.; Matsumura, Y. Autoimmunity and pulmonary hypertension in patients with Graves’ disease. Heart Vessel. 2015, 30, 642–646. [Google Scholar] [CrossRef] [PubMed]
- Osuna, P.M.; Udovcic, M.; Sharma, M.D. Hyperthyroidism and the Heart. Methodist Debakey Cardiovasc. J. 2017, 13, 60–63. [Google Scholar] [CrossRef]
- Nicolls, M.R.; Taraseviciene-Stewart, L.; Rai, P.R.; Badesch, D.B.; Voelkel, N.F. Autoimmunity and pulmonary hypertension: A perspective. Eur. Respir. J. 2005, 26, 1110–1118. [Google Scholar] [CrossRef]
- D’Andrea, A.; Naeije, R.; Grunig, E.; Caso, P.; D’Alto, M.; Di Palma, E.; Nunziata, L.; Riegler, L.; Scarafile, R.; Cocchia, R.; et al. Echocardiography of the pulmonary circulation and right ventricular function: Exploring the physiologic spectrum in 1,480 normal subjects. Chest 2014, 145, 1071–1078. [Google Scholar] [CrossRef]
- McQuillan, B.M.; Picard, M.H.; Leavitt, M.; Weyman, A.E. Clinical correlates and reference intervals for pulmonary artery systolic pressure among echocardiographically normal subjects. Circulation 2001, 104, 2797–2802. [Google Scholar] [CrossRef]
- Frank, R.C.; Min, J.; Abdelghany, M.; Paniagua, S.; Bhattacharya, R.; Bhambhani, V.; Pomerantsev, E.; Ho, J.E. Obesity Is Associated with Pulmonary Hypertension and Modifies Outcomes. J. Am. Heart Assoc. 2020, 9, e014195. [Google Scholar] [CrossRef]
- Weatherald, J.; Huertas, A.; Boucly, A.; Guignabert, C.; Taniguchi, Y.; Adir, Y.; Jevnikar, M.; Savale, L.; Jaïs, X.; Peng, M.; et al. Association Between BMI and Obesity With Survival in Pulmonary Arterial Hypertension. Chest 2018, 154, 872–881. [Google Scholar] [CrossRef]
- Friedman, S.E.; Andrus, B.W. Obesity and pulmonary hypertension: A review of pathophysiologic mechanisms. J. Obes. 2012, 2012, 505274. [Google Scholar] [CrossRef]
- Sieminska, L.; Wojciechowska, C.; Walczak, K.; Borowski, A.; Marek, B.; Nowak, M.; Kajdaniuk, D.; Foltyn, W.; Kos-Kudła, B. Associations between metabolic syndrome, serum thyrotropin, and thyroid antibodies status in postmenopausal women, and the role of interleukin-6. Endokrynol. Pol. 2015, 66, 394–403. [Google Scholar]
- Sweeney, L.; Voelkel, N.F. Estrogen exposure, obesity and thyroid disease in women with severe pulmonary hypertension. Eur. J. Med. Res. 2009, 14, 433–442. [Google Scholar] [CrossRef]
- Mair, K.M.; Gaw, R.; MacLean, M.R. Obesity, estrogens and adipose tissue dysfunction-implications for pulmonary arterial hypertension. Pulm. Circ. 2020, 10, 1–21. [Google Scholar] [CrossRef]
- Zafrir, B.; Adir, Y.; Shehadeh, W.; Shteinberg, M.; Salman, N.; Amir, O. The association between obesity, mortality and filling pressures in pulmonary hypertension patients; the “obesity paradox”. Respir. Med. 2013, 7, 139–146. [Google Scholar] [CrossRef]
- McLean, L.L.; Pellino, K.; Brewis, M.; Peacock, A.; Johnson, M.; Church, A.A. The obesity paradox in pulmonary arterial hypertension: The Scottish perspective. ERJ Open Res. 2019, 5, 00241. [Google Scholar] [CrossRef]
- Movahed, M.R.; Hashemzadeh, M.; Jamal, M.M. The prevalence of pulmonary embolism and pulmonary hypertension in patients with type II diabetes mellitus. Chest 2005, 128, 3568–3571. [Google Scholar] [CrossRef]
- Abernethy, A.D.; Stackhouse, K.; Hart, S.; Devendra, G.; Bashore, T.M.; Dweik, R.; Krasuski, R.A. Impact of diabetes in patients with pulmonary hypertension. Pulm. Circ. 2015, 5, 117–123. [Google Scholar] [CrossRef]
- Trammell, A.W.; Hemnes, A.R.; Tseng, V.; Shah, A.J.; Phillips, L.S.; Hart, C.M. Influence of Body Weight and Diabetes Mellitus in Patients with Pulmonary Hypertension. Am. J. Cardiol. 2020, 134, 130–137. [Google Scholar] [CrossRef]
- Whitaker, M.E.; Nair, V.; Sinari, S.; Dherange, P.A.; Natarajan, B.; Trutter, L.; Brittain, E.L.; Hemnes, A.R.; Austin, E.D.; Patel, K.; et al. Diabetes Mellitus Associates with Increased Right Ventricular Afterload and Remodeling in Pulmonary Arterial Hypertension. Am. J. Med. 2018, 131, 702.e7–702.e13. [Google Scholar] [CrossRef]
- Benson, L.; Brittain, E.L.; Pugh, M.E.; Austin, E.D.; Fox, K.; Wheeler, L.; Robbins, I.M.; Hemnes, A.R. Impact of diabetes on survival and right ventricular compensation in pulmonary arterial hypertension. Pulm. Circ. 2014, 4, 311–318. [Google Scholar] [CrossRef]
- Disoteo, O.E.; Zampetti, B.; Garascia, A.; Attanasio, R.; Cozzi, R. Autoimmune Polyendocrine Syndrome Complicated by Pulmonary Hypertension. Endocr. Metab. Immune Disord. Drug Targets 2021, 21, 561–565. [Google Scholar] [CrossRef]
- Iijima, T.; Niitani, T.; Tanaka, S.; Yanagi, K.; Jojima, T.; Suzuki, K.; Usui, I.; Aso, Y. Concurrent variant type 3 autoimmune polyglandular syndrome and pulmonary arterial hypertension in a Japanese woman. Endocr. J. 2018, 65, 493–498. [Google Scholar] [CrossRef]
- García-Hernández, F.J.; Ocaña-Medina, C.; González-León, R.; Garrido-Rasco, R.; Sánchez-Román, J. Autoimmune polyglandular syndrome and pulmonary arterial hypertension. Eur. Respir. J. 2006, 27, 657. [Google Scholar] [CrossRef]
- Zamanian, R.T.; Hansmann, G.; Snook, S. Lilienfeld, D.; Rappaport, K.M.; Reaven, G.M.; Rabinovitch, M.; Doyle, R.L. Insulin resistance in pulmonary arterial hypertension. Eur. Respir. J. 2009, 33, 318–324. [Google Scholar] [CrossRef]
- Xinping, C.; Austin, E.D.; Talati, M.; Fessel, J.P.; Farber-Eger, E.H.; Brittain, E.L.; Hemnes, A.R.; Loyd, J.E.; West, J. Oestrogen inhibition reverses pulmonary arterial hypertension and associated metabolic defects. Eur. Respir. J. 2017, 50, 1602337. [Google Scholar]
- Sun, Y.; Sangam, S.; Guo, Q.; Wang, J.; Tang, H.; Black, S.M.; Desai, A.A. Sex Differences, Estrogen Metabolism and Signaling in the Development of Pulmonary Arterial Hypertension. Front. Cardiovasc. Med. 2021, 8, 719058. [Google Scholar] [CrossRef]
- Liu, A.; Schreier, D.; Tian, L.; Eickhoff, J.C.; Wang, Z.; Hacker, T.A.; Chesler, N.C. Direct and indirect protection of right ventricular function by estrogen in an experimental model of pulmonary arterial hypertension. Am. J. Physiol. Heart Circul. Physiol. 2014, 307, H273–H283. [Google Scholar] [CrossRef]
- Mair, K.M.; Yang, X.D.; Long, L.; White, K.; Wallace, E.; Ewart, M.A.; Docherty, C.K.; Morrell, N.W.; MacLean, M.R. Sex affects bone morphogenetic protein type II receptor signaling in pulmonary artery smooth muscle cells. Am. J. Respir. Crit. Care Med. 2015, 19, 693–703. [Google Scholar] [CrossRef]
- White, K.; Johansen, A.K.; Nilsen, M.; Ciuclan, L.; Wallace, E.; Paton, L.; Campbell, A.; Morecroft, I.; Loughlin, L.; McClure, J.D.; et al. Activity of the estrogen-metabolizing enzyme cytochrome P450 1B1 influences the development of pulmonary arterial hypertension. Circulation 2012, 126, 1087–1098. [Google Scholar] [CrossRef]
- Dempsie, Y.; Nilsen, M.; White, K.; Mair, K.M.; Loughlin, L.; Ambartsumian, N.; Rabinovitch, M.; MacLean, M.R. Development of pulmonary arterial hypertension in mice over-expressing S100A4/Mts1 is specific to females. Respir. Res. 2011, 12, 159. [Google Scholar] [CrossRef]
- Lahm, T.; Albrecht, M.; Fisher, A.J.; Selejm, M.; Patel, N.G.; Brown, J.A.; Justice, M.J.; Brown, M.B.; Van Demark, M.; Trulock, K.M.; et al. 17β-Estradiol attenuates hypoxic pulmonary hypertension via estrogen receptor-mediated effects. Am. J. Respir. Crit. Care Med. 2012, 185, 965–980. [Google Scholar] [CrossRef] [PubMed]
- Zacharia, L.C.; Gogos, J.A.; Karayiorgou, M.; Jackson, E.K.; Gillespie, D.G.; Barchiesi, F.; Dubey, R.K. Methoxyestradiols mediate the antimitogenic effects of 17beta-estradiol: Direct evidence from catechol-O-methyltransferase-knockout mice. Circulation 2003, 108, 2974–2978. [Google Scholar] [CrossRef] [PubMed]
- Lahm, T.; Crisostomo, P.R.; Markel, T.A.; Wang, M.; Wang, Y.; Weil, B.; Meldrum, D.R. Exogenous estrogen rapidly attenuates pulmonary artery vasoreactivity and acute hypoxic pulmonary vasoconstriction. Shock 2008, 30, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Lahm, T.; Patel, K.M.; Crisostomo, P.R.; Markel, T.A.; Wang, M.; Herring, C.; Meldrum, D.R. Endogenous estrogen attenuates pulmonary artery vasoreactivity and acute hypoxic pulmonary vasoconstriction: The effects of sex and menstrual cycle. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E865–E871. [Google Scholar] [CrossRef]
- Speyer, C.L.; Rancilio, N.J.; McClintock, S.D.; Crawford, J.D.; Gao, H.; Sarma, J.; Ward, P.A. Regulatory effects of estrogen on acute lung inflammation in mice. Am. J. Physiol. Cell Physiol. 2005, 288, C881–C890. [Google Scholar] [CrossRef]
- Tofovic, S.P.; Jackson, E.K. Estradiol Metabolism: Crossroads in Pulmonary Arterial Hypertension. Int. J. Mol. Sci. 2019, 21, 116. [Google Scholar] [CrossRef]
- Kawut, S.M.; Archer-Chicko, C.L.; DeMichele, A.; Fritz, J.S.; Klinger, J.R.; Ky, B.; Palevsky, H.I.; Palmisciano, A.J.; Patel, M.; Pinder, D.; et al. Anastrozole in Pulmonary Arterial Hypertension. A Randomized, Double-Blind, Placebo-controlled Trial. Am. J. Respir. Crit. Care Med. 2017, 195, 360–368. [Google Scholar] [CrossRef]
- Tofovic, S.P. Estrogens and development of pulmonary hypertension: Interaction of estradiol metabolism and pulmonary vascular disease. J. Cardiovasc. Pharmacol. 2010, 56, 696–708. [Google Scholar] [CrossRef]
- Limoges, M.; Langleben, D.; Fox, B.D.; Shear, R.; Wieczorek, P.; Rudski, L.G.; Hirsch, A.M.; Schlesinger, R.D.; Lesenko, L. Pregnancy as a possible trigger for heritable pulmonary arterial hypertension. Pulm. Circ. 2016, 6, 381–383. [Google Scholar] [CrossRef]
- Genctoy, G.; Arikan, S.; Gedik, O. Secondary hyperparathyroidism is associated with pulmonary hypertension in older patients with chronic kidney disease and proteinuria. Int. Urol. Nephrol. 2014, 47, 353–358. [Google Scholar] [CrossRef]
- Alhamad, E.H.; Al-Ghonaim, M.; Alfaleh, H.F.; Cal, J.P.; Said, N. Pulmonary hypertension in end-stage renal disease and post renal transplantation patients. J. Thorac. Dis. 2014, 6, 606–616. [Google Scholar]
- Saliba, W.R.; Rock, W.; Elias, M. Pulmonary hypertension in a patient with Schmidt syndrome. Am. J. Emerg. Med. 2009, 27, 1025. [Google Scholar] [CrossRef]
- Hassoun, P.M. Pulmonary Arterial Hypertension. N. Engl. J. Med. 2021, 385, 2361–2376. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrigkou, E.; Vassilatou, E.; Dima, E.; Langleben, D.; Kotanidou, A.; Tzanela, M. The Role of Thyroid Disorders, Obesity, Diabetes Mellitus and Estrogen Exposure as Potential Modifiers for Pulmonary Hypertension. J. Clin. Med. 2022, 11, 921. https://doi.org/10.3390/jcm11040921
Vrigkou E, Vassilatou E, Dima E, Langleben D, Kotanidou A, Tzanela M. The Role of Thyroid Disorders, Obesity, Diabetes Mellitus and Estrogen Exposure as Potential Modifiers for Pulmonary Hypertension. Journal of Clinical Medicine. 2022; 11(4):921. https://doi.org/10.3390/jcm11040921
Chicago/Turabian StyleVrigkou, Eleni, Evangeline Vassilatou, Effrosyni Dima, David Langleben, Anastasia Kotanidou, and Marinella Tzanela. 2022. "The Role of Thyroid Disorders, Obesity, Diabetes Mellitus and Estrogen Exposure as Potential Modifiers for Pulmonary Hypertension" Journal of Clinical Medicine 11, no. 4: 921. https://doi.org/10.3390/jcm11040921
APA StyleVrigkou, E., Vassilatou, E., Dima, E., Langleben, D., Kotanidou, A., & Tzanela, M. (2022). The Role of Thyroid Disorders, Obesity, Diabetes Mellitus and Estrogen Exposure as Potential Modifiers for Pulmonary Hypertension. Journal of Clinical Medicine, 11(4), 921. https://doi.org/10.3390/jcm11040921






