Quo Vadis Anesthesiologist? The Value Proposition of Future Anesthesiologists Lies in Preserving or Restoring Presurgical Health after Surgical Insult
1. Introduction
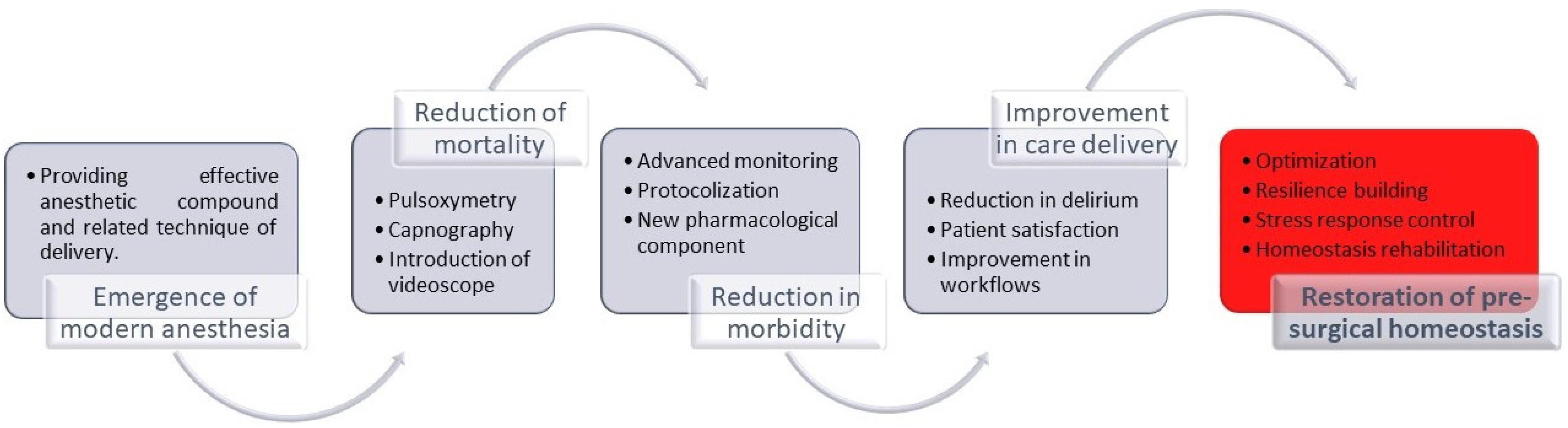
2. New Paradigm of Anesthesia Care
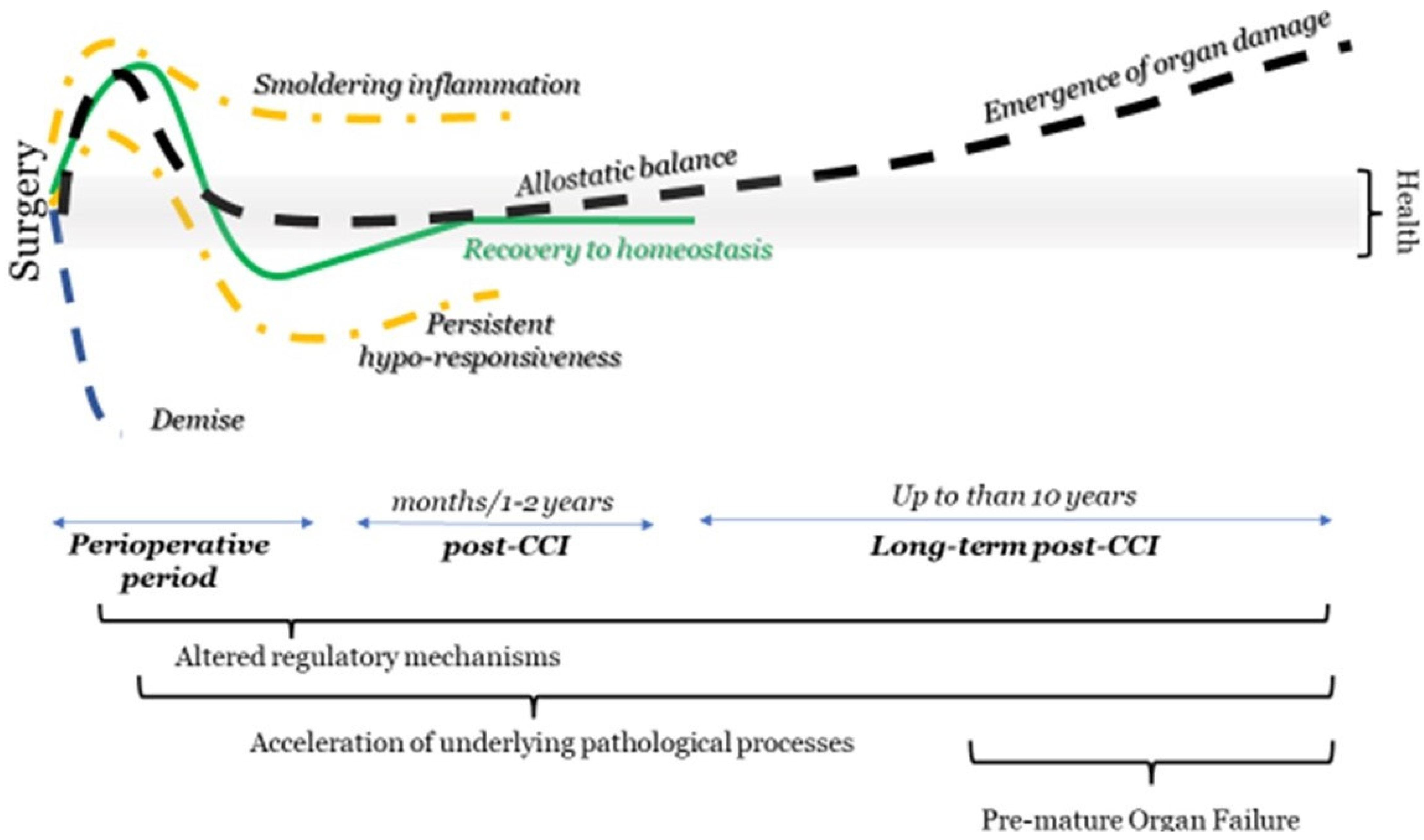
3. The Effect of Anesthesia on Long-Term Postoperative Outcomes
4. Long-Term Effects Intervention
4.1. Preoperative Period
4.2. Operative Period
4.3. Postoperative Period
5. Innovation in Service
6. The Market Value of the New Paradigm of an Anesthesiologist
Funding
Acknowledgments
Conflicts of Interest
References
- Robinson, D.H.; Toledo, A.H. Historical Development of Modern Anesthesia. J. Investig. Surg. 2012, 25, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Saunders, R.; Struys, M.M.R.F.; Pollock, R.F.; Mestek, M.; Lightdale, J.R. Patient safety during procedural sedation using capnography monitoring: A systematic review and meta-analysis. BMJ Open 2017, 7, e013402. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, T.; Moller, A.M.; Pedersen, B.D. Pulse oximetry for perioperative monitoring: Systematic review of randomized, controlled trials. Anesth. Analg. 2003, 96, 426–431. [Google Scholar] [PubMed]
- Li, J.; Krishna, R.; Zhang, Y.; Lam, D.; Vadivelu, N. Ultrasound-Guided Neuraxial Anesthesia. Curr. Pain Headache Rep. 2020, 24, 59. [Google Scholar] [CrossRef]
- Bignami, E.; Maffezzoni, M.; Bellini, V. Lung Ultrasound in Thoracic Anesthesia: Which Uses? J. Cardiothorac. Vasc. Anesth. 2021, 35, 374–375. [Google Scholar] [CrossRef]
- Carron, M.; Linassi, F.; De Cassai, A. Role of sugammadex in accelerating postoperative discharge: An updated meta-analysis. J. Clin. Anesth. 2020, 65, 109895. [Google Scholar] [CrossRef]
- van Veen-Berkx, E.; van Dijk, M.V.; Cornelisse, D.C.; Kazemier, G.; Mokken, F.C. Scheduling Anesthesia Time Reduces Case Cancellations and Improves Operating Room Workflow in a University Hospital Setting. J. Am. Coll. Surg. 2016, 223, 343–351. [Google Scholar] [CrossRef]
- Behrens, V.; Dudaryk, R.; Nedeff, N.; Tobin, J.M.; Varon, A.J. The Ryder Cognitive Aid Checklist for Trauma Anesthesia. Anesth. Analg. 2016, 122, 1484–1487. [Google Scholar] [CrossRef]
- Jelacic, S.; Bowdle, A.; Nair, B.G.; Togashi, K.; Boorman, D.J.; Cain, K.C.; Lang, J.D.; Dellinger, E.P. Aviation-Style Computerized Surgical Safety Checklist Displayed on a Large Screen and Operated by the Anesthesia Provider Improves Checklist Performance. Anesth. Analg. 2020, 130, 382–390. [Google Scholar] [CrossRef]
- Arbous, M.S.; Grobbee, D.E.; van Kleef, J.W.; de Lange, J.J.; Spoormans, H.H.A.J.M.; Touw, P.; Werner, F.M.; Meursing, A.E.E. Mortality associated with anaesthesia: A qualitative analysis to identify risk factors. Anaesthesia 2001, 56, 1141–1153. [Google Scholar] [CrossRef]
- Arbous, M.S.; Grobbee, D.E.; van Kleef, J.W.; Meursing, A.E. Dutch case-control study of anaesthesia-related morbidity and mortality. Rationale and methods. Anaesthesia 1998, 53, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Arbous, M.S.; Meursing, A.E.E.; Van Kleef, J.W.; De Lange, J.J.; Spoormans, H.H.A.J.M.; Touw, P.; Werner, F.M.; Grobbee, D.E. Impact of Anesthesia Management Characteristics on Severe Morbidity and Mortality. Anesthesiology 2005, 102, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.Y.; Warner, D.O.; Schroeder, D.R.; Offord, K.P.; Warner, M.A.; Maxson, P.M.; Whisnant, J.P. Risk of Surgery and Anesthesia for Ischemic Stroke. Anesthesiology 2000, 92, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.-B. Perioperative stroke: Pathophysiology and management. Korean J. Anesthesiol. 2018, 71, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Hendén, P.L.; Rentzos, A.; Karlsson, J.-E.; Rosengren, L.; Leiram, B.; Sundeman, H.; Dunker, D.; Schnabel, K.; Wikholm, G.; Hellström, M.; et al. General Anesthesia Versus Conscious Sedation for Endovascular Treatment of Acute Ischemic Stroke. Stroke 2017, 48, 1601–1607. [Google Scholar] [CrossRef]
- Tarhan, S.; Moffitt, E.A.; Taylor, W.F.; Giuliani, E.R. Myocardial Infarction after General Anesthesia. Anesth. Analg. 1977, 56, 455–461. [Google Scholar] [CrossRef][Green Version]
- Harwin, B.; Formanek, B.; Spoonamore, M.; Robertson, D.; Buser, Z.; Wang, J.C. The incidence of myocardial infarction after lumbar spine surgery. Eur. Spine J. 2019, 28, 2070–2076. [Google Scholar] [CrossRef]
- Cannon, W. Homeostasis. 1 September 2014. Available online: http://www.panarchy.org/cannon/homeostasis.1932.html (accessed on 15 January 2022).
- Eisenstein, T.K. The Role of Opioid Receptors in Immune System Function. Front. Immunol. 2019, 10, 2904. [Google Scholar] [CrossRef]
- Osuka, A.; Ogura, H.; Ueyama, M.; Shimazu, T.; Lederer, J.A. Immune response to traumatic injury: Harmony and discordance of immune system homeostasis. Acute Med. Surg. 2014, 1, 63–69. [Google Scholar] [CrossRef]
- Laudanski, K.; Wyczechowska, D. Monocyte-related immunopathologies in trauma patients. Arch. Immunol. Ther. Exp. 2005, 53, 321–328. [Google Scholar]
- Bilotta, F.; Evered, L.A.; Gruenbaum, S.E. Neurotoxicity of anesthetic drugs: An update. Curr. Opin. Anaesthesiol. 2017, 30, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, P.; Faure, E.; Kipnis, E. Inflammasomes in Tissue Damages and Immune Disorders after Trauma. Front. Immunol. 2018, 9, 1900. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.L.; Latz, E.; Ontiveros, F.; Kono, H. The Sterile Inflammatory Response. Annu. Rev. Immunol. 2010, 28, 321–342. [Google Scholar] [CrossRef] [PubMed]
- Zindel, J.; Kubes, P. DAMPs, PAMPs, and LAMPs in Immunity and Sterile Inflammation. Annu. Rev. Pathol. Mech. Dis. 2020, 15, 493–518. [Google Scholar] [CrossRef]
- Feldman, N.; Rotter-Maskowitz, A.; Okun, E. DAMPs as mediators of sterile inflammation in aging-related pathologies. Ageing Res. Rev. 2015, 24, 29–39. [Google Scholar] [CrossRef]
- Frye, C.C.; Bery, A.I.; Kreisel, D.; Kulkarni, H.S. Sterile inflammation in thoracic transplantation. Cell Mol. Life Sci. 2021, 78, 581–601. [Google Scholar] [CrossRef]
- Li, J.-Y.; Yao, Y.-M.; Tian, Y.-P. Ferroptosis: A Trigger of Proinflammatory State Progression to Immunogenicity in Necroinflammatory Disease. Front. Immunol. 2021, 12, 701163. [Google Scholar] [CrossRef]
- Aron-Wisnewsky, J.; Doré, J.; Clement, K. The importance of the gut microbiota after bariatric surgery. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 590–598. [Google Scholar] [CrossRef]
- Simsek, T.; Simsek, H.U.; Cantürk, N.Z. Response to trauma and metabolic changes: Posttraumatic metabolism. Turk. J. Surg. 2014, 30, 153–159. [Google Scholar] [CrossRef]
- Relja, B.; Mörs, K.; Marzi, I. Danger signals in trauma. Eur. J. Trauma Emerg. Surg. 2018, 44, 301–316. [Google Scholar] [CrossRef]
- Hauser, C.; Otterbein, L.E. Danger signals from mitochondrial DAMPS in trauma and post-injury sepsis. Eur. J. Trauma Emerg. Surg. 2018, 44, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Yamanashi, T.; Nagao, T.; Wahba, N.E.; Marra, P.S.; Crutchley, K.J.; Meyer, A.A.; Andreasen, A.J.; Hellman, M.M.; Jellison, S.S.; Hughes, C.G.; et al. DNA methylation in the inflammatory genes after neurosurgery and diagnostic ability of post-operative delirium. Transl. Psychiatry 2021, 11, 627. [Google Scholar] [CrossRef] [PubMed]
- Relja, B.; Land, W.G. Damage-associated molecular patterns in trauma. Eur. J. Trauma Emerg. Surg. 2019, 46, 751–775. [Google Scholar] [CrossRef]
- Wilmore, D.W. Metabolic Response to Severe Surgical Illness: Overview. World J. Surg. 2000, 24, 705–711. [Google Scholar] [CrossRef]
- McBride, M.A.; Owen, A.M.; Stothers, C.L.; Hernandez, A.; Luan, L.; Burelbach, K.R.; Patil, T.K.; Bohannon, J.K.; Sherwood, E.R.; Patil, N.K. The Metabolic Basis of Immune Dysfunction Following Sepsis and Trauma. Front. Immunol. 2020, 11, 1043. [Google Scholar] [CrossRef] [PubMed]
- Logan, J.G.; Barksdale, D.J. Allostasis and allostatic load: Expanding the discourse on stress and cardiovascular disease. J. Clin. Nurs. 2008, 17, 201–208. [Google Scholar] [CrossRef]
- Ramsay, D.S.; Woods, S.C. Clarifying the roles of homeostasis and allostasis in physiological regulation. Psychol. Rev. 2014, 121, 225–247. [Google Scholar] [CrossRef]
- Laudanski, K. Persistence of alterations in lipoproteins and cholesterol during and after septic episode—Review of current evidence of long-term post septic lipid profile aberrations and their implication for allostasis. Int. J. Mol. Sci. 2021, 22, 10517. [Google Scholar] [CrossRef]
- Yoshimura, A.; Ito, M. Resolution of inflammation and repair after ischemic brain injury. Neuroimmunol. Neuroinflamm. 2020, 7, 264–276. [Google Scholar] [CrossRef]
- Zhang, T.; Yan, L.L.; Chen, H.-S.; Jin, H.-Y.; Wu, C. Association between allostatic load and mortality among Chinese older adults: The Chinese Longitudinal Health and Longevity Study. BMJ Open 2021, 11, e045369. [Google Scholar] [CrossRef]
- Placek, K.; Schultze, J.L.; Aschenbrenner, A.C. Epigenetic reprogramming of immune cells in injury, repair, and resolution. J. Clin. Investig. 2019, 129, 2994–3005. [Google Scholar] [CrossRef] [PubMed]
- Roquilly, A.; Jacqueline, C.; Davieau, M.; Mollé, A.; Sadek, A.; Fourgeux, C.; Rooze, P.; Broquet, A.; Misme-Aucouturier, B.; Chaumette, T.; et al. Alveolar macrophages are epigenetically altered after inflammation, leading to long-term lung immunoparalysis. Nat. Immunol. 2020, 21, 636–648. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P. Post-transcriptional regulons coordinate the initiation and resolution of inflammation. Nat. Rev. Immunol. 2010, 10, 24–35. [Google Scholar] [CrossRef]
- Ji, R.-R.; Xu, Z.-Z.; Strichartz, G.; Serhan, C.N. Emerging roles of resolvins in the resolution of inflammation and pain. Trends Neurosci. 2011, 34, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Sen, C.K. miRNA in Wound Inflammation and Angiogenesis. Microcirculation 2012, 19, 224–232. [Google Scholar] [CrossRef]
- Cox, A.; Coles, A.; Nortje, J.; Bradley, P.; Chatfield, D.; Thompson, S.; Menon, D. An investigation of auto-reactivity after head injury. J. Neuroimmunol. 2006, 174, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Rahtes, A.; Li, L. Polarization of Low-Grade Inflammatory Monocytes through TRAM-Mediated Up-Regulation of Keap1 by Super-Low Dose Endotoxin. Front. Immunol. 2020, 11, 1478. [Google Scholar] [CrossRef]
- Pradhan, K.; Yi, Z.; Geng, S.; Li, L. Development of Exhausted Memory Monocytes and Underlying Mechanisms. Front. Immunol. 2021, 12, 778830. [Google Scholar] [CrossRef]
- Bandyopadhyay, G.; De, A.; Laudanski, K.; Li, F.; Lentz, C.; Bankey, P.; Miller-Graziano, C. Negative signaling contributes to T-cell anergy in trauma patients. Crit. Care Med. 2007, 35, 794–801. [Google Scholar] [CrossRef]
- Beckie, T.M. A Systematic Review of Allostatic Load, Health, and Health Disparities. Biol. Res. Nurs. 2012, 14, 311–346. [Google Scholar] [CrossRef]
- Sessler, D.I. Long-term Consequences of Anesthetic Management. Anesthesiology 2009, 111, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Mestas, J.; Hughes, C.C.W. Of mice and not men: Differences between mouse and human immunology. J. Immunol. 2004, 172, 2731–2738. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.B.; Bartal, I.; Goldfarb, Y.; Levi, B.; Avraham, R.; Raz, A.; Ben-Eliyahu, S. Perioperative Use of β-blockers and COX-2 Inhibitors May Improve Immune Competence and Reduce the Risk of Tumor Metastasis. Ann. Surg. Oncol. 2008, 15, 2042–2052. [Google Scholar] [CrossRef]
- Bar-Yosef, S.; Melamed, R.; Page, G.G.; Shakhar, G.; Shakhar, K.; Ben-Eliyahu, S. Attenuation of the Tumor-promoting Effect of Surgery by Spinal Blockade in Rats. Anesthesiology 2001, 94, 1066–1073. [Google Scholar] [CrossRef]
- Afsharimani, B.; Cabot, P.J.; Parat, M.-O. Morphine Use in Cancer Surgery. Front. Pharmacol. 2011, 2, 46. [Google Scholar] [CrossRef]
- Demarco, G.J.; Nunamaker, E.A. A Review of the Effects of Pain and Analgesia on Immune System Function and Inflammation: Relevance for Preclinical Studies. Comp. Med. 2019, 69, 520–534. [Google Scholar] [CrossRef]
- Kabon, B.; Fleischmann, E.; Treschan, T.; Taguchi, A.; Kapral, S.; Kurz, A. Thoracic Epidural Anesthesia Increases Tissue Oxygenation during Major Abdominal Surgery. Anesth. Analg. 2003, 97, 1812–1817. [Google Scholar] [CrossRef]
- Hopkins, P.M. Does regional anaesthesia improve outcome? Br. J. Anaesth. 2015, 115, ii26–ii33. [Google Scholar] [CrossRef]
- Muncey, A.R.; Patel, S.Y.; Whelan, C.J.; Ackerman, R.S.; Gatenby, R.A. The Intersection of Regional Anesthesia and Cancer Progression: A Theoretical Framework. Cancer Control 2020, 27. [Google Scholar] [CrossRef]
- Sessler, D.I.; Riedel, B. Anesthesia and Cancer Recurrence: Context for Divergent Study Outcomes. Anesthesiology 2019, 130, 3–5. [Google Scholar] [CrossRef]
- Cakmakkaya, O.S.; Kolodzie, K.; Apfel, C.C.; Pace, N.L. Anaesthetic techniques for risk of malignant tumour recurrence. Cochrane Database Syst. Rev. 2014, 11, CD008877. [Google Scholar] [CrossRef] [PubMed]
- Jakobsson, J.G.; Johnson, M.Z. Perioperative regional anaesthesia and postoperative longer-term outcomes. F1000Research 2016, 5, 2501. [Google Scholar] [CrossRef] [PubMed]
- Sessler, D.I.; Pei, L.; Huang, Y.; Fleischmann, E.; Marhofer, P.; Kurz, A.; Mayers, D.B.; Meyer-Treschan, T.A.; Grady, M.; Tan, E.Y.; et al. Recurrence of breast cancer after regional or general anaesthesia: A randomised controlled trial. Lancet 2019, 394, 1807–1815. [Google Scholar] [CrossRef]
- Sieber, F.E.; Zakriya, K.J.; Gottschalk, A.; Blute, M.-R.; Lee, H.B.; Rosenberg, P.B.; Mears, S.C. Sedation Depth During Spinal Anesthesia and the Development of Postoperative Delirium in Elderly Patients Undergoing Hip Fracture Repair. Mayo Clin. Proc. 2010, 85, 18–26. [Google Scholar] [CrossRef]
- Sieber, F.E.; Neufeld, K.; Gottschalk, A.; Bigelow, G.E.; Oh, E.S.; Rosenberg, P.B.; Mears, S.C.; Stewart, K.J.; Ouanes, J.-P.P.; Jaberi, M.; et al. Effect of Depth of Sedation in Older Patients Undergoing Hip Fracture Repair on Postoperative Delirium: The STRIDE Randomized Clinical Trial. JAMA Surg. 2018, 153, 987–995. [Google Scholar] [CrossRef]
- Fritz, B.A.; King, C.R.; Mickle, A.M.; Wildes, T.S.; Budelier, T.P.; Oberhaus, J.; Park, D.; Maybrier, H.R.; Ben Abdallah, A.; Kronzer, A.; et al. Effect of electroencephalogram-guided anaesthesia administration on 1-yr mortality: Follow-up of a randomised clinical trial. Br. J. Anaesth. 2021, 127, 386–395. [Google Scholar] [CrossRef]
- Wallace, A.W.; Galindez, D.; Salahieh, A.; Layug, E.L.; Lazo, E.A.; Haratonik, K.A.; Boisvert, D.M.; Kardatzke, D. Effect of Clonidine on Cardiovascular Morbidity and Mortality after Noncardiac Surgery. Anesthesiology 2004, 101, 284–293. [Google Scholar] [CrossRef]
- Wallace, A.W. Clonidine and modification of perioperative outcome. Curr. Opin. Anaesthesiol. 2006, 19, 411–417. [Google Scholar] [CrossRef]
- Duncan, D.; Sankar, A.; Beattie, W.S.; Wijeysundera, D.N. Alpha-2 adrenergic agonists for the prevention of cardiac complications among adults undergoing surgery. Cochrane Database Syst. Rev. 2018, 2018, CD004126. [Google Scholar] [CrossRef]
- Li, H.; Liu, J.; Shi, H. Effect of Dexmedetomidine on Perioperative Hemodynamics and Myocardial Protection in Thoracoscopic-Assisted Thoracic Surgery. Med Sci. Monit. 2021, 27, e929949. [Google Scholar] [CrossRef]
- Elgebaly, A.S.; Fathy, S.M.; Sallam, A.A.; Elbarbary, Y. Cardioprotective effects of propofol-dexmedetomidine in open-heart surgery: A prospective double-blind study. Ann. Card. Anaesth. 2020, 23, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Narita, M. Postoperative outcome among elderly patients after general anesthesia. Acta Anaesthesiol. Scand. 2005, 50, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.E.; Torp-Pedersen, C.; Gislason, G.H.; Jensen, P.F.; Berger, S.M.; Christiansen, C.B.; Overgaard, C.; Schmiegelow, M.D.; Andersson, C. Time Elapsed After Ischemic Stroke and Risk of Adverse Cardiovascular Events and Mortality Following Elective Noncardiac Surgery. JAMA 2014, 312, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, P.; Rossi, M.G.; Anghelescu, D.L.; Liu, W.; Breazeale, A.M.; Reddick, W.E.; Glass, J.O.; Phillips, N.S.; Jacola, L.M.; Sabin, N.D.; et al. Association Between Anesthesia Exposure and Neurocognitive and Neuroimaging Outcomes in Long-term Survivors of Childhood Acute Lymphoblastic Leukemia. JAMA Oncol. 2019, 5, 1456–1463. [Google Scholar] [CrossRef]
- Williams, R.K.; Black, I.H.; Howard, D.B.; Adams, D.C.; Mathews, D.M.; Friend, A.F.; Meyers, H.W.B. Cognitive Outcome After Spinal Anesthesia and Surgery During Infancy. Anesth. Analg. 2014, 119, 651–660. [Google Scholar] [CrossRef]
- Sanders, R.D.; Jørgensen, M.E.; Mashour, G.A. Perioperative stroke: A question of timing? Br. J. Anaesth. 2015, 115, 11–13. [Google Scholar] [CrossRef]
- Matthews, C.R.; Hartman, T.; Madison, M.; Villelli, N.W.; Namburi, N.; Colgate, C.L.; Faiza, Z.; Lee, L.S. Preoperative stroke before cardiac surgery does not increase risk of postoperative stroke. Sci. Rep. 2021, 11, 9025. [Google Scholar] [CrossRef]
- Ancelin, M.-L.; de Roquefeuil, G.; Scali, J.; Bonnel, F.; Adam, J.-F.; Cheminal, J.-C.; Cristol, J.-P.; Dupuy, A.-M.; Carrière, I.; Ritchie, K. Long-Term Post-Operative Cognitive Decline in the Elderly: The Effects of Anesthesia Type, Apolipoprotein E Genotype, and Clinical Antecedents. J. Alzheimer’s Dis. 2010, 22, S105–S113. [Google Scholar] [CrossRef]
- Humeidan, M.L.; Reyes, J.-P.C.; Mavarez-Martinez, A.; Roeth, C.; Nguyen, C.M.; Sheridan, E.; Zuleta-Alarcon, A.; Otey, A.; Abdel-Rasoul, M.; Bergese, S.D. Effect of Cognitive Prehabilitation on the Incidence of Postoperative Delirium Among Older Adults Undergoing Major Noncardiac Surgery: The Neurobics Randomized Clinical Trial. JAMA Surg. 2021, 156, 148–156. [Google Scholar] [CrossRef]
- Mbagwu, C.; Sloan, M.; Neuwirth, A.L.; Charette, R.S.; Baldwin, K.D.; Kamath, A.F.; Mason, B.S.; Nelson, C.L. Preoperative Albumin, Transferrin, and Total Lymphocyte Count as Risk Markers for Postoperative Complications After Total Joint Arthroplasty: A Systematic Review. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2020, 4, e19.00057. [Google Scholar] [CrossRef]
- Vlisides, P.E.; Das, A.R.; Thompson, A.M.; Kunkler, B.; Zierau, M.; Cantley, M.J.; McKinney, A.M.; Giordani, B. Home-based Cognitive Prehabilitation in Older Surgical Patients: A Feasibility Study. J. Neurosurg. Anesthesiol. 2019, 31, 212–217. [Google Scholar] [CrossRef] [PubMed]
- O’Gara, B.P.; Mueller, A.; Gasangwa, D.V.I.; Patxot, M.; Shaefi, S.; Khabbaz, K.; Banner-Goodspeed, V.; Pascal-Leone, A.; Marcantonio, E.R.; Subramaniam, B. Prevention of Early Postoperative Decline: A Randomized, Controlled Feasibility Trial of Perioperative Cognitive Training. Anesth. Analg. 2020, 130, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Ishizawa, Y. Does Preoperative Cognitive Optimization Improve Postoperative Outcomes in the Elderly? J. Clin. Med. 2022, 11, 445. [Google Scholar] [CrossRef] [PubMed]
- Vaage, J.; Valen, G. Preconditioning and cardiac surgery. Ann. Thorac. Surg. 2003, 75, S709–S714. [Google Scholar] [CrossRef]
- Späth, M.R.; Koehler, F.C.; Hoyer-Allo, K.J.R.; Grundmann, F.; Burst, V.; Müller, R.-U. Preconditioning strategies to prevent acute kidney injury. F1000Research 2020, 9, 237. [Google Scholar] [CrossRef]
- Petrowsky, H.; McCormack, L.; Trujillo, M.; Selzner, M.; Jochum, W.; Clavien, P.-A. A Prospective, Randomized, Controlled Trial Comparing Intermittent Portal Triad Clamping Versus Ischemic Preconditioning With Continuous Clamping for Major Liver Resection. Ann. Surg. 2006, 244, 921–930. [Google Scholar] [CrossRef]
- Andoh, T.; Chock, P.B.; Chiueh, C.C. Preconditioning-Mediated Neuroprotection: Role of nitric oxide, cGMP, and new protein expression. Ann. N. Y. Acad. Sci. 2002, 962, 1–7. [Google Scholar] [CrossRef]
- Casanova, J.; Garutti, I.; Simon, C.; Giraldez, A.; Martin, B.; Gonzalez, G.; Azcarate, L.; Garcia, C.; Vara, E. The Effects of Anesthetic Preconditioning with Sevoflurane in an Experimental Lung Autotransplant Model in Pigs. Anesth. Analg. 2011, 113, 742–748. [Google Scholar] [CrossRef]
- Beck-Schimmer, B.; Breitenstein, S.; Urech, S.; De Conno, E.; Wittlinger, M.; Puhan, M.; Jochum, W.; Spahn, D.R.; Graf, R.; Clavien, P.-A. A Randomized Controlled Trial on Pharmacological Preconditioning in Liver Surgery Using a Volatile Anesthetic. Ann. Surg. 2008, 248, 909–918. [Google Scholar] [CrossRef]
- Liang, R.; Tang, Q.; Song, W.; Zhang, M.; Teng, L.; Kang, Y.; Zhu, L. Electroacupuncture Preconditioning Reduces Oxidative Stress in the Acute Phase of Cerebral Ischemia-Reperfusion in Rats by Regulating Iron Metabolism Pathways. Evid.-Based Complement. Altern. Med. 2021, 2021, 3056963. [Google Scholar] [CrossRef]
- Fei, L.; Jingyuan, X.; Fangte, L.; Huijun, D.; Liu, Y.; Ren, J.; Jinyuan, L.; Linghui, P. Preconditioning with rHMGB1 ameliorates lung ischemia–reperfusion injury by inhibiting alveolar macrophage pyroptosis via the Keap1/Nrf2/HO-1 signaling pathway. J. Transl. Med. 2020, 18, 301. [Google Scholar] [CrossRef] [PubMed]
- Fensterheim, B.A.; Young, J.D.; Luan, L.; Kleinbard, R.R.; Stothers, C.L.; Patil, N.K.; McAtee-Pereira, A.G.; Guo, Y.; Trenary, I.; Hernandez, A.; et al. The TLR4 Agonist Monophosphoryl Lipid A Drives Broad Resistance to Infection via Dynamic Reprogramming of Macrophage Metabolism. J. Immunol. 2018, 200, 3777–3789. [Google Scholar] [CrossRef] [PubMed]
- Watts, B.A.; George, T.; Sherwood, E.R.; Good, D.W. Monophosphoryl lipid A induces protection against LPS in medullary thick ascending limb through a TLR4-TRIF-PI3K signaling pathway. Am. J. Physiol. 2017, 313, F103–F115. [Google Scholar] [CrossRef] [PubMed]
- Weighardt, H.; Feterowski, C.; Veit, M.; Rump, M.; Wagner, H.; Holzmann, B. Increased Resistance Against Acute Polymicrobial Sepsis in Mice Challenged with Immunostimulatory CpG Oligodeoxynucleotides Is Related to an Enhanced Innate Effector Cell Response. J. Immunol. 2000, 165, 4537–4543. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Van Dyke, T.E. Resolving inflammation: Dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef]
- Marcheselli, V.L.; Mukherjee, P.K.; Arita, M.; Hong, S.; Antony, R.; Sheets, K.; Winkler, J.W.; Petasis, N.A.; Serhan, C.N.; Bazan, N.G. Neuroprotectin D1/protectin D1 stereoselective and specific binding with human retinal pigment epithelial cells and neutrophils. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 27–34. [Google Scholar] [CrossRef]
- Wu, Z.; Zhang, L.; Zhao, X.; Li, Z.; Lu, H.; Bu, C.; Wang, R.; Wang, X.; Cai, T.; Wu, D. Protectin D1 protects against lipopolysaccharide-induced acute lung injury through inhibition of neutrophil infiltration and the formation of neutrophil extracellular traps in lung tissue. Exp. Ther. Med. 2021, 22, 1074. [Google Scholar] [CrossRef]
- Perez-Hernandez, J.; Chiurchiù, V.; Perruche, S.; You, S. Regulation of T-Cell Immune Responses by Pro-Resolving Lipid Mediators. Front. Immunol. 2021, 12, 768133. [Google Scholar] [CrossRef]
- Marcon, R.; Bento, A.F.; Dutra, R.; Bicca, M.A.; Leite, D.F.P.; Calixto, J.B. Maresin 1, a Proresolving Lipid Mediator Derived from Omega-3 Polyunsaturated Fatty Acids, Exerts Protective Actions in Murine Models of Colitis. J. Immunol. 2013, 191, 4288–4298. [Google Scholar] [CrossRef]
- Mandwie, M.; Karunia, J.; Niaz, A.; Keay, K.A.; Musumeci, G.; Rennie, C.; McGrath, K.; Al-Badri, G.; Castorina, A. Metformin Treatment Attenuates Brain Inflammation and Rescues PACAP/VIP Neuropeptide Alterations in Mice Fed a High-Fat Diet. Int. J. Mol. Sci. 2021, 22, 13660. [Google Scholar] [CrossRef]
- Qin, Z.; Zhou, C.; Xiao, X.; Guo, C. Metformin attenuates sepsis-induced neuronal injury and cognitive impairment. BMC Neurosci. 2021, 22, 78. [Google Scholar] [CrossRef]
- Zhou, C.; Peng, B.; Qin, Z.; Zhu, W.; Guo, C. Metformin attenuates LPS-induced neuronal injury and cognitive impairments by blocking NF-κB pathway. BMC Neurosci. 2021, 22, 73. [Google Scholar] [CrossRef]
- Bijker, J.B.; van Klei, W.A.; Kappen, T.H.; van Wolfswinkel, L.; Moons, K.G.; Kalkman, C.J. Incidence of intraoperative hypotension as a function of the chosen definition: Literature definitions applied to a retrospective cohort using automated data collection. Anesthesiology 2007, 107, 213–220. [Google Scholar] [CrossRef]
- Bijker, J.B.; van Klei, W.A.; Vergouwe, Y.; Eleveld, D.J.; van Wolfswinkel, L.; Moons, K.G.M.; Kalkman, C. Intraoperative Hypotension and 1-Year Mortality after Noncardiac Surgery. Anesthesiology 2009, 111, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; MacKenzie, C.R.; Gold, J.P.; Ales, K.L.; Topkins, M.; Shires, G.T. Preoperative characteristics predicting intraoperative hypotension and hypertension among hypertensives and diabetics undergoing noncardiac surgery. Ann. Surg. 1990, 212, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, A.; McCormick, P.; Sessler, D.; Reich, D.; You, J.; Mascha, E.; Castillo, J.; Levin, M.; Duncan, A. Prolonged concurrent hypotension and low bispectral index (‘double low’) are associated with mortality, serious complications, and prolonged hospitalization after cardiac surgery. Br. J. Anaesth. 2017, 119, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Casanova, J.; Simon, C.; Vara, E.; Sanchez, G.; Rancan, L.; Abubakra, S.; Calvo, A.; Gonzalez, F.J.; Garutti, I. Sevoflurane anesthetic preconditioning protects the lung endothelial glycocalyx from ischemia reperfusion injury in an experimental lung autotransplant model. J. Anesth. 2016, 30, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Rancan, L.; Huerta, L.; Cusati, G.; Erquicia, I.; Isea, J.; Paredes, S.D.; García, C.; Garutti, I.; Simón, C.; Vara, E. Sevoflurane Prevents Liver Inflammatory Response Induced by Lung Ischemia-Reperfusion. Transplantation 2014, 98, 1151–1157. [Google Scholar] [CrossRef]
- Resnick, B.; Beaupre, L.; McGilton, K.S.; Galik, E.; Liu, W.; Neuman, M.D.; Gruber-Baldini, A.L.; Orwig, D.; Magaziner, J. Rehabilitation Interventions for Older Individuals with Cognitive Impairment Post-Hip Fracture: A Systematic Review. J. Am. Med. Dir. Assoc. 2015, 17, 200–205. [Google Scholar] [CrossRef]
- Laudanski, K.; Miller-Graziano, C.; Xiao, W.; Mindrinos, M.N.; Richards, D.R.; De, A.; Moldawer, L.L.; Maier, R.V.; Bankey, P.; Baker, H.V.; et al. Cell-specific expression and pathway analyses reveal alterations in trauma-related human T cell and monocyte pathways. Proc. Natl. Acad. Sci. USA 2006, 103, 15564–15569. [Google Scholar] [CrossRef]
- Chiang, N.; Serhan, C.N.; Dahlén, S.-E.; Drazen, J.M.; Hay, D.W.P.; Rovati, G.; Shimizu, T.; Yokomizo, T.; Brink, C. The Lipoxin Receptor ALX: Potent Ligand-Specific and Stereoselective Actions in Vivo. Pharmacol. Rev. 2006, 58, 463–487. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.A.; Vago, J.P.; Teixeira, M.M.; Sousa, L.P. Annexin A1 and the Resolution of Inflammation: Modulation of Neutrophil Recruitment, Apoptosis, and Clearance. J. Immunol. Res. 2016, 2016, 8239258. [Google Scholar] [CrossRef] [PubMed]
- Headland, S.E.; Norling, L.V. The resolution of inflammation: Principles and challenges. Semin. Immunol. 2015, 27, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Lopez, N.; Krzyzaniak, M.; Costantini, T.; De Maio, A.; Baird, A.; Eliceiri, B.; Coimbra, R. Vagal Nerve Stimulation Blocks Peritoneal Macrophage Inflammatory Responsiveness After Severe Burn Injury. Shock 2012, 38, 294–300. [Google Scholar] [CrossRef]
- Andersson, U.; Tracey, K.J. Neural reflexes in inflammation and immunity. J. Exp. Med. 2012, 209, 1057–1068. [Google Scholar] [CrossRef]
- Laudanski, K.; Zawadka, M.; Polosak, J.; Modi, J.; DiMeglio, M.; Gutsche, J.; Szeto, W.Y.; Puzianowska-Kuznicka, M. Acquired immunological imbalance after surgery with cardiopulmonary bypass due to epigenetic over-activation of PU.1/M-CSF. J. Transl. Med. 2018, 16, 143. [Google Scholar] [CrossRef]
- DiMeglio, M.; Furey, W.; Hajj, J.; Lindekens, J.; Patel, S.; Acker, M.; Bavaria, J.; Szeto, W.Y.; Atluri, P.; Haber, M.; et al. Observational study of long-term persistent elevation of neurodegeneration markers after cardiac surgery. Sci. Rep. 2019, 9, 7177. [Google Scholar] [CrossRef] [PubMed]
- Kox, M.; Pompe, J.C.; Pickkers, P.; Hoedemaekers, C.W.; Van Vugt, A.B.; Van Der Hoeven, J.G. Increased vagal tone accounts for the observed immune paralysis in patients with traumatic brain injury. Neurology 2008, 70, 480–485. [Google Scholar] [CrossRef]
- Heinonen, K.; Strandvik, T. Reframing service innovation: COVID-19as a catalyst for imposed service innovation. J. Serv. Manag. 2020, 32, 101–112. [Google Scholar] [CrossRef]
- COVID Innovations. 2021. Available online: https://www.covidinnovations.com/ (accessed on 18 December 2021).
- Gutierrez, G. Artificial Intelligence in the Intensive Care Unit. Crit. Care 2020, 24, 2–9. [Google Scholar] [CrossRef]
- Matheny, M.E.; Whicher, D.; Israni, S.T. Artificial Intelligence in Health Care: A Report from the National Academy of Medicine. JAMA 2020, 323, 509. [Google Scholar] [CrossRef] [PubMed]
- Loftus, T.J.; Vlaar, A.P.; Hung, A.J.; Bihorac, A.; Dennis, B.M.; Juillard, C.; Hashimoto, D.A.; Kaafarani, H.M.; Tighe, P.J.; Kuo, P.C.; et al. Executive summary of the artificial intelligence in surgery series. Surgery, 2021, in press. [CrossRef] [PubMed]
- Holzinger, A.; Mohammadzadeh, N.; Park, S.J.; Lee, E.J.; Kim, S.I.; Kong, S.-H.; Jeong, C.W.; Kim, H.S. Clinical Desire for an Artificial Intelligence-Based Surgical Assistant System: Electronic Survey-Based Study. JMIR Med. Inform. 2020, 8, e17647. [Google Scholar] [CrossRef]
- Suri, J.S.; Puvvula, A.; Biswas, M.; Majhail, M.; Saba, L.; Faa, G.; Singh, I.M.; Oberleitner, R.; Turk, M.; Chadha, P.S.; et al. COVID-19 pathways for brain and heart injury in comorbidity patients: A role of medical imaging and artificial intelligence-based COVID severity classification: A review. Comput. Biol. Med. 2020, 124, 103960. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, Y.; Byrne, R.; Schneider, G.; Yang, S. Concepts of Artificial Intelligence for Computer-Assisted Drug Discovery. Chem. Rev. 2019, 119, 10520–10594. [Google Scholar] [CrossRef]
- Yuan, K.-C.; Tsai, L.-W.; Lee, K.-H.; Cheng, Y.-W.; Hsu, S.-C.; Lo, Y.-S.; Chen, R.-J. The development an artificial intelligence algorithm for early sepsis diagnosis in the intensive care unit. Int. J. Med. Inform. 2020, 141, 104176. [Google Scholar] [CrossRef]
- Jaynes, T.L. “Compoundless Anaesthesia”, Controlled Administration, and Post-Operative Recovery Acceleration: Musings on Theoretical Nanomedicine Applications. J. Clin. Med. 2022, 11, 256. [Google Scholar] [CrossRef]
- Anjum, S.; Ishaque, S.; Fatima, H.; Farooq, W.; Hano, C.; Abbasi, B.; Anjum, I. Emerging Applications of Nanotechnology in Healthcare Systems: Grand Challenges and Perspectives. Pharmaceuticals 2021, 14, 707. [Google Scholar] [CrossRef]
- Hampson, G.; Towse, A.; Pearson, S.D.; Dreitlein, W.B.; Henshall, C. Gene therapy: Evidence, value and affordability in the US health care system. J. Comp. Eff. Res. 2018, 7, 15–28. [Google Scholar] [CrossRef]
- Shah, S.A.; Garrett, R.A. CRISPR/Cas and Cmr modules, mobility and evolution of adaptive immune systems. Res. Microbiol. 2011, 162, 27–38. [Google Scholar] [CrossRef]
- Zhang, J.; Qi, X.; Yi, F.; Cao, R.; Gao, G.; Zhang, C. Comparison of Clinical Efficacy and Safety Between da Vinci Robotic and Laparoscopic Intersphincteric Resection for Low Rectal Cancer: A Meta-Analysis. Front. Surg. 2021, 8, 615. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Liu, H.-D.; Yang, K.; Chen, Z.-H.; Zhang, Y.-X.; Hu, J.-K. Effectiveness and safety of robotic gastrectomy versus laparoscopic gastrectomy for gastric cancer: A meta-analysis of 12,401 gastric cancer patients. Updat. Surg. 2021, 74, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Garas, G.; Cingolani, I.; Panzarasa, P.; Darzi, A.; Athanasiou, T. Network analysis of surgical innovation: Measuring value and the virality of diffusion in robotic surgery. PLoS ONE 2017, 12, e0183332. [Google Scholar] [CrossRef] [PubMed]
- Koh, M.H.; Yen, S.-C.; Leung, L.Y.; Gans, S.; Sullivan, K.; Adibnia, Y.; Pavel, M.; Hasson, C.J. Exploiting telerobotics for sensorimotor rehabilitation: A locomotor embodiment. J. Neuroeng. Rehabil. 2021, 18, 66. [Google Scholar] [CrossRef]
- Sadeghi, A.H.; el Mathari, S.; Abjigitova, D.; Maat, A.P.M.; Taverne, Y.J.J.; Bogers, A.J.C.; Mahtab, E.A. Current and Future Applications of Virtual, Augmented, and Mixed Reality in Cardiothoracic Surgery. Ann. Thorac. Surg. 2020, 113, 681–691. [Google Scholar] [CrossRef]
- Goudra, B.; Singh, P.M. Failure of Sedasys: Destiny or Poor Design? Anesth. Analg. 2017, 124, 686–688. [Google Scholar] [CrossRef]
- Xiao, X.; Poon, H.; Lim, C.M.; Meng, M.Q.-H.; Ren, H. Pilot Study of Trans-oral Robotic-Assisted Needle Direct Tracheostomy Puncture in Patients Requiring Prolonged Mechanical Ventilation. Front. Robot. AI 2020, 7, 575445. [Google Scholar] [CrossRef]
- Lederman, D. Endotracheal Intubation Confirmation Based on Video Image Classification Using a Parallel GMMs Framework: A Preliminary Evaluation. Ann. Biomed. Eng. 2010, 39, 508–516. [Google Scholar] [CrossRef]
- Hemmerling, T.M.; Wehbe, M.; Zaouter, C.; Taddei, R.; Morse, J. The Kepler Intubation System. Anesth. Analg. 2012, 114, 590–594. [Google Scholar] [CrossRef]
- Kissin, I. Depth of Anesthesia and Bispectral Index Monitoring. Anesth. Analg. 2000, 90, 1114–1117. [Google Scholar] [CrossRef]
- Li, C.; Wei, J.; Huang, X.; Duan, Q.; Zhang, T. Effects of a Brain-Computer Interface-Operated Lower Limb Rehabilitation Robot on Motor Function Recovery in Patients with Stroke. J. Health Eng. 2021, 2021, 4710044. [Google Scholar] [CrossRef] [PubMed]
- Birbaumer, N. Breaking the silence: Brain-computer interfaces (BCI) for communication and motor control. Psychophysiology 2006, 43, 517–532. [Google Scholar] [CrossRef] [PubMed]
- Parks, N. Brain chips and future of human evolution. In Proceedings of the Harvard Model Congress Boston 2022, Boston, MA, USA, 24–27 February 2022. [Google Scholar]
- Venkatesan, M.; Mohan, H.; Ryan, J.R.; Schürch, C.M.; Nolan, G.P.; Frakes, D.H.; Coskun, A.F. Virtual and augmented reality for biomedical applications. Cell Rep. Med. 2021, 2, 100348. [Google Scholar] [CrossRef] [PubMed]
- Hayes-O’Neil, T.; Dixon, K. Hospital Markets and the Effects of Consolidation. AAF—American Action Forum. 2022. Available online: https://www.americanactionforum.org/research/hospital-markets-and-the-effects-of-consolidation/ (accessed on 5 February 2022).
- Stein, E.J.; Mesrobian, J.R.; Szokol, J.W.; Abouleish, A.E. The 2016 job market for graduating anesthesiology residents. ASA Monit. 2016, 81, 56–62. [Google Scholar]
- Erstad, B.L. Value-Based Medicine: Dollars and Sense. Crit. Care Med. 2016, 44, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Rochaix, L. Information asymmetry and search in the market for physicians’ services. J. Health Econ. 1989, 8, 53–84. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laudanski, K. Quo Vadis Anesthesiologist? The Value Proposition of Future Anesthesiologists Lies in Preserving or Restoring Presurgical Health after Surgical Insult. J. Clin. Med. 2022, 11, 1135. https://doi.org/10.3390/jcm11041135
Laudanski K. Quo Vadis Anesthesiologist? The Value Proposition of Future Anesthesiologists Lies in Preserving or Restoring Presurgical Health after Surgical Insult. Journal of Clinical Medicine. 2022; 11(4):1135. https://doi.org/10.3390/jcm11041135
Chicago/Turabian StyleLaudanski, Krzysztof. 2022. "Quo Vadis Anesthesiologist? The Value Proposition of Future Anesthesiologists Lies in Preserving or Restoring Presurgical Health after Surgical Insult" Journal of Clinical Medicine 11, no. 4: 1135. https://doi.org/10.3390/jcm11041135
APA StyleLaudanski, K. (2022). Quo Vadis Anesthesiologist? The Value Proposition of Future Anesthesiologists Lies in Preserving or Restoring Presurgical Health after Surgical Insult. Journal of Clinical Medicine, 11(4), 1135. https://doi.org/10.3390/jcm11041135




