The Role of Inflammation in Tinnitus: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
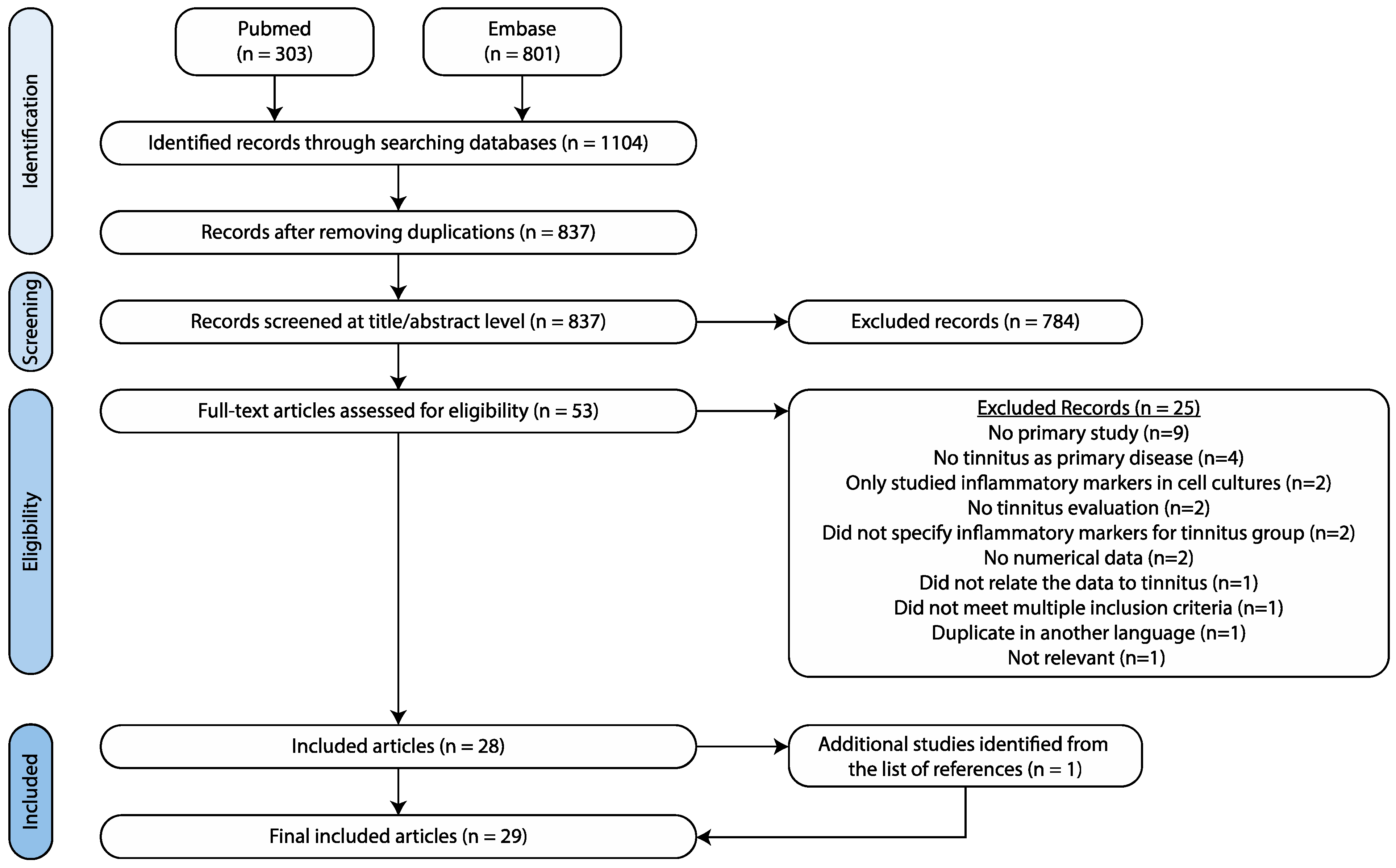
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection and Data Extraction
2.3. Meta-Analysis
3. Results
3.1. Animal Studies
3.1.1. Study Characteristics
3.1.2. Salicylate-Induced Tinnitus
Study Characteristics
Cytokine Levels
Neuroglial Markers
| Study | Animals | n | Tinnitus Induction | Measurement Timing | Tinnitus Evaluation | Tinnitus Present | Controls | Region of Interest | Method | Results | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Significant | Not Significant | |||||||||||
| Salicylate induced tinnitus | Chen and Zheng, 2017 [18] | R | 6 | S 1d | +2 h | GPIAS | No | NM (n = 10) | AC | PCR | TNF-α, IL-6, IFN-γ | |
| Western blot | TNF-α, IL-6, IFN-γ | |||||||||||
| 10 | S 7d | +2 h | Yes | PCR | TNF-α ↑, IFN-γ ↓ | IL-6 | ||||||
| Western blot | TNF-α ↑, IFN-γ ↓ | IL-6 | ||||||||||
| 10 | S 7d | +14 d | No | PCR | TNF-α, IL-6, IFN-γ | |||||||
| Western blot | TNF-α, IL-6, IFN-γ | |||||||||||
| Fang et al., 2016 [22] | R | 10 | S 4d | +4 h | GPIAS | Yes | Saline treated (n = 10) | Ventral CN | Western blot | Iba-1 ↓, GFAP ↑ | ||
| IHC | Iba-1 ↓, GFAP ↑ | |||||||||||
| 10 | S 8d | +4 h | Yes | Western blot | Iba-1 ↓, GFAP ↑ | |||||||
| IHC | Iba-1 ↓, GFAP ↑ | |||||||||||
| 12 | S 8d | +7 d | No | Western blot | Iba-1, GFAP | |||||||
| IHC | Iba-1, GFAP | |||||||||||
| Hu et al., 2014 [17] | R | 6 | S 1d | +1 d | GPIAS | No | No treatment (n = 10) | CN | PCR | TNF-α, IL-6 | ||
| Western blot | TNF-α, IL-6 | |||||||||||
| 10 | S 7d | +1 d | Yes | PCR | TNF-α ↑ | IL-6 | ||||||
| Western blot | TNF-α ↑ | IL-6 | ||||||||||
| 10 | S 14d | +1 d | Yes | PCR | TNF-α ↑ | IL-6 | ||||||
| Western blot | TNF-α ↑ | IL-6 | ||||||||||
| IHC | TNF-α ↑ | IL-6 | ||||||||||
| 6 | S 14d | +15 d | No | PCR | TNF-α, IL-6 | |||||||
| Western blot | TNF-α, IL-6 | |||||||||||
| 6 | S 14d | +29 d | No | PCR | TNF-α, IL-6 | |||||||
| Western blot | TNF-α, IL-6 | |||||||||||
| Hwang et al., 2011a [20] | M | 24 | S 4d | +4 d | Behavioral conditioning | Yes | Saline treated (n = 24) | Cochlea | PCR | TNF-α ↑, IL-1β ↑ Tinnitus scores ↔ TNF-α Tinnitus scores ↔ IL-1β | ||
| IC | PCR | TNF-α ↑, IL-1β ↑ Tinnitus scores ↔ TNF-α Tinnitus scores ↔ IL-1β | ||||||||||
| Salicylate induced tinnitus | Hwang et al., 2011b [23] | M | 24 | S 4d | +4 d | Behavioral conditioning | Yes | Saline treated (n = 24) | Cochlea | PCR | COX-2 | |
| IC | PCR | COX-2 | ||||||||||
| Hwang et al., 2013 [19] | M | 24 | S 4d | +2 h | Behavioral conditioning | Yes | Saline treated (n = 24) | Cochlea | PCR | TNF-α ↑, IL-1β ↑ | COX-2 | |
| IC | PCR | TNF-α ↑, IL-1β ↑ | COX-2 | |||||||||
| Western blot | TNF-α, IL-1β, COX-2 | |||||||||||
| Hwang et al., 2017 [24] | R | 15 | S 3d | +0 d | Behavioral conditioning | Yes | Cochlea | PCR | TNF receptor-1 1.03, TNF receptor-2 66.86 | |||
| Xia et al., 2020 [21] | R | 4 | S 1d | +2 h | GPIAS + PPI | Yes | Saline treated (n = 4) | AC1 | PCR | IL-1β ↑ | ||
| Western blot | GFAP, Iba-1 | |||||||||||
| IHC | astrocyte EP/C ↑, astrocyte LE/C ↑ | GFAP, Iba-1, microglia EP/C, microglia LE/C, IL-1β | ||||||||||
| MGB | PCR | IL-1β | ||||||||||
| Western blot | GFAP, Iba-1 | |||||||||||
| IHC | microglia EP/C ↑, microglia LE/C ↑ | GFAP, Iba-1, astrocyte EP/C, astrocyte LE/C, IL-1β | ||||||||||
| 4 | S 1d | +4 h | Yes | AC1 | PCR | IL-1β ↑ | ||||||
| Western blot | GFAP ↑, Iba-1 ↑ | |||||||||||
| IHC | GFAP ↑, Iba-1 ↑, astrocyte EP/C ↑, astrocyte LE/C ↑, IL-1β ↑ | microglia EP/C, microglia LE/C | ||||||||||
| MGB | PCR | IL-1β | ||||||||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | GFAP ↑, Iba-1 ↑, microglia EP/C ↑, IL-1β ↑ | astrocyte EP/C, astrocyte LE/C, microglia LE/C | ||||||||||
| Salicylate induced tinnitus | Xia et al., 2020 [21] | 4 | S 1d | +8 h | Yes | AC1 | PCR | IL-1β | ||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | Iba-1 ↑, IL-1β ↑, astrocyte EP/C ↑ | GFAP, astrocyte LE/C, microglia EP/C, microglia LE/C | ||||||||||
| MGB | PCR | IL-1β ↑ | ||||||||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | Iba-1 ↑ | GFAP, IL-1β astrocyte EP/C, astrocyte LE/C, microglia EP/C, microglia LE/C | ||||||||||
| 4 | S 1d | +24 h | No | AC1 | PCR | IL-1β | ||||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | Iba-1 ↑, IL-1β ↑ | GFAP, astrocyte EP/C, astrocyte LE/C, microglia EP/C, microglia LE/C | ||||||||||
| MGB | PCR | IL-1β ↑ | ||||||||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | Iba-1 ↑ | GFAP, IL-1β astrocyte EP/C, astrocyte LE/C, microglia EP/C, microglia LE/C | ||||||||||
| 4 | S 7d | +0 d | Yes | AC1 | PCR | IL-1β | ||||||
| Western blot | GFAP ↑, Iba-1 ↑ | |||||||||||
| IHC | GFAP ↑, Iba-1 ↑, astrocyte EP/C ↑, IL-1β ↑ | astrocyte LE/C, microglia EP/C, microglia LE/C | ||||||||||
| MGB | PCR | IL-1β | ||||||||||
| Western blot | Iba-1 ↑ | GFAP | ||||||||||
| IHC | Iba-1 ↑, microglia EP/C ↑ | GFAP, IL-1β astrocyte EP/C, astrocyte LE/C, microglia LE/C | ||||||||||
| Salicylate induced tinnitus | Xia et al., 2020 [21] | 4 | S 7d | +7 d | No | AC1 | PCR | IL-1β | ||||
| Western blot | GFAP, Iba-1 | |||||||||||
| IHC | Iba-1, GFAP, IL-1β astrocyte EP/C, astrocyte LE/C, microglia EP/C, microglia LE/C | |||||||||||
| MGB | PCR | IL-1β | ||||||||||
| Western blot | GFAP, Iba-1 | |||||||||||
| IHC | Iba-1, GFAP, IL-1β astrocyte EP/C, astrocyte LE/C, microglia EP/C, microglia LE/C | |||||||||||
| Noise-induced tinnitus | Wang et al., 2019 [16] | M | 4 | 2-h 8 kHz 112–114 dB unilateral sound (AS) | +12 h | GPIAS + PPI | ? | NM (n = 4) | Bilateral AC1 | PCR | TNF-α ↑ (ipsi > contra), NLRP3 ↑ (bilat.), | IL-1β, IL-18, TNF-α protein |
| 4 | +1 d | ? | PCR | TNF-α ↑ (bilat.), TNF-α protein ↑ (contra > ipsi) | IL-1β, IL-18, NLPR3 | |||||||
| IHC | Soma-to-whole cell size ratio | |||||||||||
| 4 | +3 d | ? | PCR | TNF-α protein ↑ | TNF-α, IL-1β, IL-18, NLPR3 | |||||||
| IHC | Soma-to-whole cell size ratio | |||||||||||
| 4 | +5 d | ? | IHC | Soma-to-whole cell size ratio ↑ (contra > ipsi) | ||||||||
| 4 | +10 d | Yes | PCR | TNF-α ↑ (bilat.), IL-1β ↑ (bilat.), IL-18 ↑ (bilat.), NLRP3 ↑ (bilat.), TNF-α protein ↑ (contra > ipsi) | ||||||||
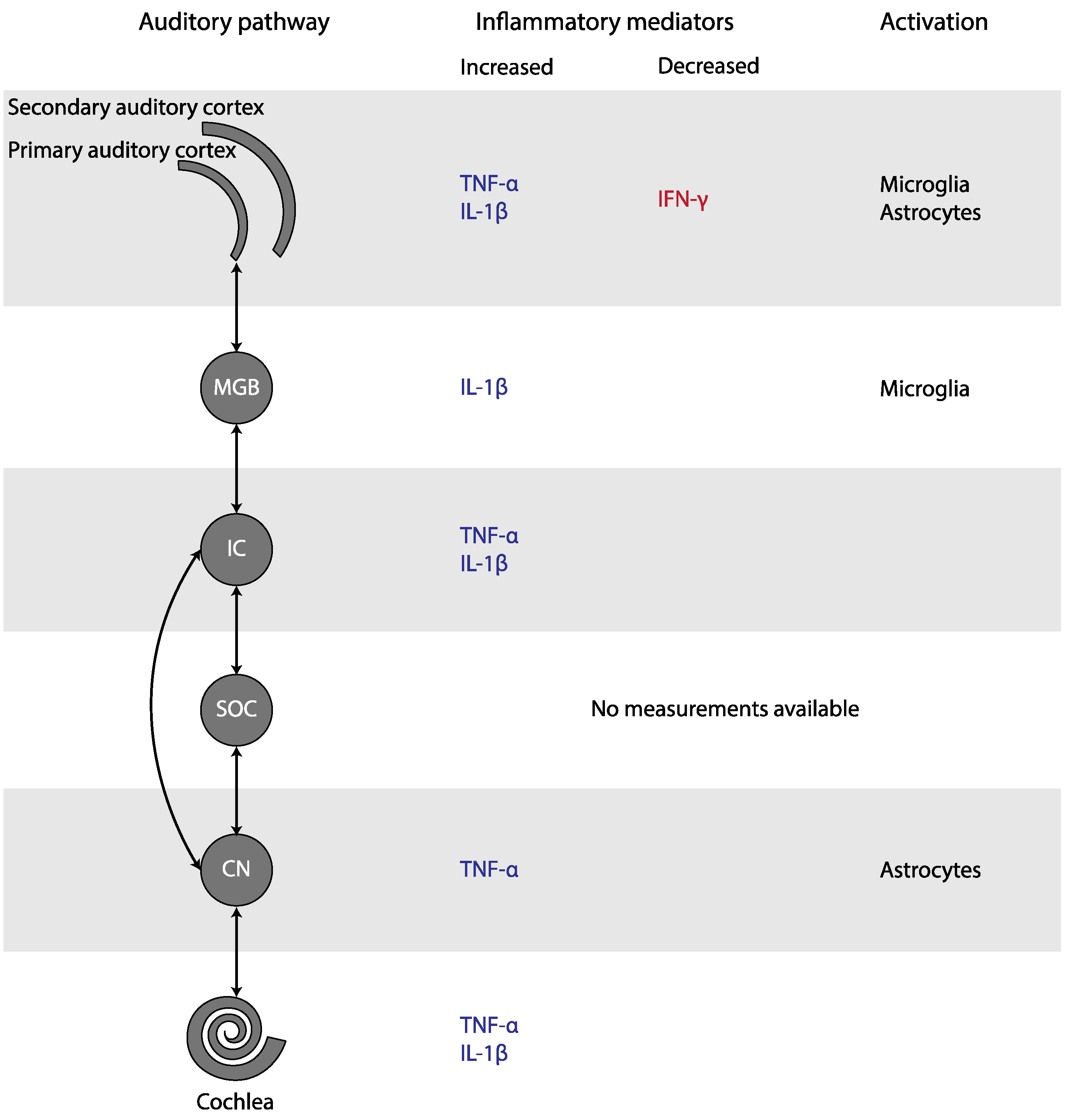
Other Markers of Inflammation
3.1.3. Noise-Induced Tinnitus
3.2. Human Studies
3.2.1. Study Characteristics
3.2.2. Cytokine Levels
| Study | Participants | Controls | Tissue of Interest | Method | Results | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n (M/F) | Age * | Tinnitus Location (uni/bi) | Tinnitus Duration | Tinnitus Score | n (m/f) | Age * | Significant | Not Significant | |||
| Avci, 2020 [33] | 91 (34/57) | 48.03 ± 15.12 y | 67/24 | 7.17 ± 10.52 m | THI: 37.29 ± 15.22 | 65 (24/41) | 47.55 ± 17.49 y | Blood | CBC | MPV ↑ | neutrophils, lymphocytes, NLR, PDW, platelet count, PLR, RCDW, WBC |
| Bayraktar and Taşolar, 2017 [25] | 40 (15/25) | 41.00 ± 9.02 y | 31/9 | ≥3 m | THI: slight (n = 10), mild (n = 11), moderate (n = 7), severe (n = 7), catastrophic (n = 5) | 40 (18/22) | 37.90 ± 8.97 y | Blood | CBC | NLR ↔ tinnitus severity | NLR |
| Bayram et al., 2015 [34] | 51 (26/25) | 43.47 ± 13.74 y | 34/17 | 27.02 ± 34.99 m | TRQ 28.22 ± 19.28. THQ 845.18 ± 565.99 | 42 (24/18) | 45.19 ± 9.71 | Blood | CBC | lymphocytes, MPV, neutrophils, NLR, platelet count, PLR, | |
| Çeçen et al., 2021 [35] | 74 (46/28) | 52.4 ± 13.4 | ? | ≥0.5 m | THI: 51.5 ± 21.1 | 65 (33/30) | 50.6 ± 14.8 | Blood | CBC | MPV ↑, MCV ↑ | NLR, PLR, RCDW |
| Chrbolka et al., 2020 [36] | 40 | Mean 50.86 (incl. controls) | ? | NR | ? | 40 | Mean 50.86 (incl. patients) | Blood | NM | CRP | |
| Demir, 2021 [37] | HFHL-TN 55 (37/18) | 42.20 ± 11.62 y | ? | 0–3 m | ? | 57 (34/23) | 37.42 ± 7.49 y | Blood | CBC | NLR ↑, PLR ↑, MPV ↑ | |
| AFHL-TN 53 (28/25) | 43.30 ± 12.67 y | ? | 0–3 m | ? | NLR ↑, PLR ↑, MPV ↑ | ||||||
| NH-TN 51 (25/26) | 41.23 ± 13.09 y | ? | 0–3 m | ? | MPV, NLR, PLR | ||||||
| Düzenli et al., 2018 [38] | 58 (29/29) | 38.8 ± 9.41 y | 0/58 | ≥6 m | ? | 58 (29/29) | 38.8 ± 9.41 y | Blood | CBC | NLR ↓, PDW ↑ | MPV, Platelet count, RCDW. |
| Gunes et al., 2019 [39] | 52 (25/27) | 41.62 ± 4.76 y | ? | ≥12 m | ? | Blood | CBC | MPV (no hearing loss right ear) 7.52 ± 1.93, MPV (hearing loss right ear) 7.78 ± 1.29, MPV (no hearing loss left ear) 7.52 ± 1.73, MPV (hearing loss left ear) 7.84 ± 1.02, NLR (no hearing loss right ear) 1.99 ± 0.61, NLR (hearing loss right ear) 1.64 ± 0.71, NLR (no hearing loss left ear) 1.89 ± 0.86, NLR (hearing loss left ear) 1.53 ± 0.47 | |||
| Haider et al., 2020 [31] | 92 (46/46) | Median 63 y, p25 59.8 y, p75 68.3 y (incl. controls) | 40/44, 8 ? | 93.6 ± 103.2 m | THI: slight (n = 17), mild (n = 38), moderate (n = 22), severe (n = 14), catastrophic (n = 1) | 22 (8/14) | Median 63 y, p25 59.8 y, p75 68.3 y (incl. tinnitus patients) | Serum | Bead based multiplex assay | IL-10 ↓, IL-10 ↔ tinnitus duration | IFN-γ, IL-1α, IL-1β, IL-2, IL-6, TGF- β, TNF- α |
| Kemal et al., 2016 [28] | 86 (46/40) | 33.58 ± 11.75 y | ? | ? | ? | 84 (36/48) | 32.24 ± 10.18 y | Blood | CBC | MPV ↑ | RCDW, WBC |
| Li et al., 2019 [40] | 100 (59/41) | 41.62 ± 4.76 y | 55/45 | 2.52 ± 0.72 y, range 0.3–10 y | THI: 58.78 ± 11.36 (control n = 50) and 57.26 ± 10.48 (intervention n = 50) | Serum | ELISA | Before treatment: IL-2 (control) 12.4 ± 2.65, IL-2 (intervention): 13.01 ± 3.04 | |||
| Ozbay et al., 2015 [41] | 107 (35/72) | 38.7 ± 12.7 y | ? | ≥0.5 m | THI ≥ grade 3 | 107 (24/83) | 35.8 ± 13.9 y | Blood | CBC | NLR ↑ | Lymphocytes, MPV, neutrophils, platelet count, WBC |
| Sarıkaya et al., 2016 [27] | 101 (47/54) | 40.87 ± 14.13 y | ? | Mean 33.86 m, range 3–240 m | ? | 54 (18/36) | 42.35 ± 8.94 y | Blood | CBC | MPV ↑ | Platelet count |
| Savastano et al., 2006 [30] | 36 (19/17) | 41.0y, range 20–65 y | ? | ? | ? | 20 (11/9) | Mean 43.9 y, range 20–65 y | Blood | ? | Circulating immune complexes ↑ | CRP, Erythrocyte sedimentation rate |
| Savastano et al., 2007 [29] | 85 (46/39) | 48.36 ± 12.57 y | ? | ≥6m | ? | Blood | ? | CD3 70.70 ± 7.27 (nv 68–82), CD4 44.17 ± 7.96 (nv 36–52), CD4/CD3 0.85 ± 0.34 (nv 0.62–1.42), CD3 + CD8 23.27 ± 8.80 (nv 20–34), CD19 12.6 ± 4.20 (nv 5–16), CD16 + CD56NK 15.88 ± 7.25 (nv 1.5–15), CD3 + CD16 + CD56 4.23 ± 4.12 (nv 1.5–2.1). | |||
| Szczepek et al., 2014 [26] | 30 (16/14) | 47y (range 18–67 y) | 6/24 | Mean 60 m, range 9–336 m | TQ: 35.4 ± 17.1 | Blood | ELISA | IL-1β 4.00 ± 0.43, IL-6 0.38 ± 0.006, TNF-α 1.58 ± 1.12 | |||
| Ulusoy et al. (2018) [42] | 64 (33/31) | Median 45, range 18–65 y | ? | ? | ? | 64 (38/26) | Median 41 | Blood | CBC | MPV ↑, PDW ↑ | NLR, platelet count, PLR, WBC count |
| Weber et al., 2002 [32] | 26 (16/10) | 32.2 ± 9.7 y | ? | ? | TQ: 30.50 ± 14.38 | 13 (8/5) | 32.0 ± 6.7 y | Serum | ELISA | IL-6 ↑ | IL-10, TNF- α |
| Yildiz et al., 2020 [43] | 287 (119/168) | 44.89 ± 10.96 | ? | ? | ? | 257 (130/127) | 38.37 ± 10.65 | Blood | CBC | NLR ↑, % of high MPV within the group ↑ | |
| Yüksel and Karataş, 2016 [44] | 100 (57/43) | 50.95 ± 14.6 y | (66/34) | 35.90 ± 44.49 m | THI 41.62 ± 14.65. Slight (n = 2), mild (n = 37), moderate (n = 43), severe (n = 18), catastrophic (n = 0) | 100 (39/61) | 44.39 ± 8.9 y | Blood | CBC | MPV ↓, PDW ↑, Platelet count ↑, CRP ↑, sedimentation ↑ | |
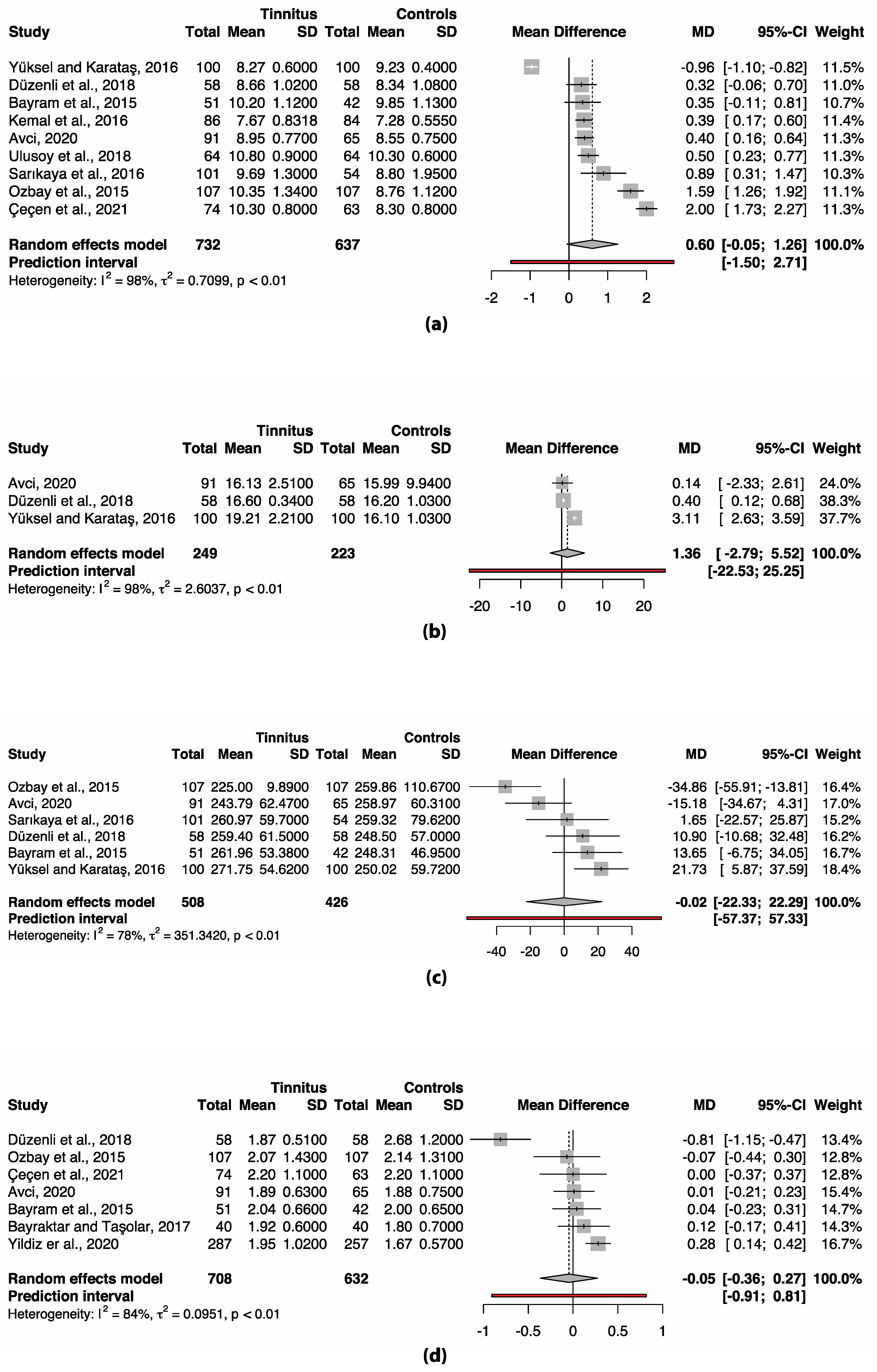
3.3. Meta-Analysis Complete Blood Count (CBC) Markers
| Analysis | MD | 95% CI | p | 95% PI | I2 | 95% CI | Removed Study |
|---|---|---|---|---|---|---|---|
| MPV | |||||||
| Main analysis | 0.602 | −0.052–1.258 | 0.067 | −1.450–2.705 | 98.4% | 97.9–98.8% | |
| Basic outlier removal | 0.426 | 0.297–0.556 | <0.001 * | 0.286–0.567 | 0% | 0–74.6% | Çeçen et al. (2021) [35] Ozbay et al. (2015) [41] Yüksel and Karataş (2016) [44] |
| Influence analysis | 0.807 | 0.261–1.353 | 0.010 * | −0.846–2.459 | 94.9% | 92.1–96.8% | Yüksel and Karataş (2016) [44] |
| PDW | |||||||
| Main analysis | 1.360 | −2.795–5.520 | 0.294 | −22.534–25.255 | 97.8% | 95.9–98.9% | |
| Basic outlier removal | 1.360 | −2.795–5.520 | 0.294 | −22.534–25.255 | 97.8% | 95.9–98.9% | No outliers detected |
| Influence analysis | 0.397 | 0.028–0.765 | 0.047 * | - | 0% | - | Yüksel and Karataş (2016) [44] |
| Platelet count | |||||||
| Main analysis | −0.019 | −22.329–22.291 | 0.998 | −57.369–57.331 | 77.9% | 51.1–90.0% | |
| Basic outlier removal | −0.019 | −22.329–22.291 | 0.998 | −57.369–57.331 | 77.9% | 51.1–90.0% | No outliers detected |
| Influence analysis | 7.155 | −10.966–25.275 | 0.335 | −33.967–48.276 | 55.3 | 0.0–83.5% | Ozbay et al. (2015) [41] |
| NLR | |||||||
| Main analysis | −0.046 | −0.365–0.273 | 0.736 | −0.907–0.815 | 83.6% | 67.7–91.7% | |
| Basic outlier removal | 0.103 | −0.042–0.248 | 0.127 | −0.219–0.425 | 34.6% | 0.0–73.8% | Düzenli et al. (2018) [38] |
| Influence analysis | 0.103 | −0.042–0.248 | 0.127 | −0.219–0.425 | 34.6% | 0.0–73.8% | Düzenli et al. (2018) [38] |
4. Discussion
4.1. Cytokine Involvement in Tinnitus
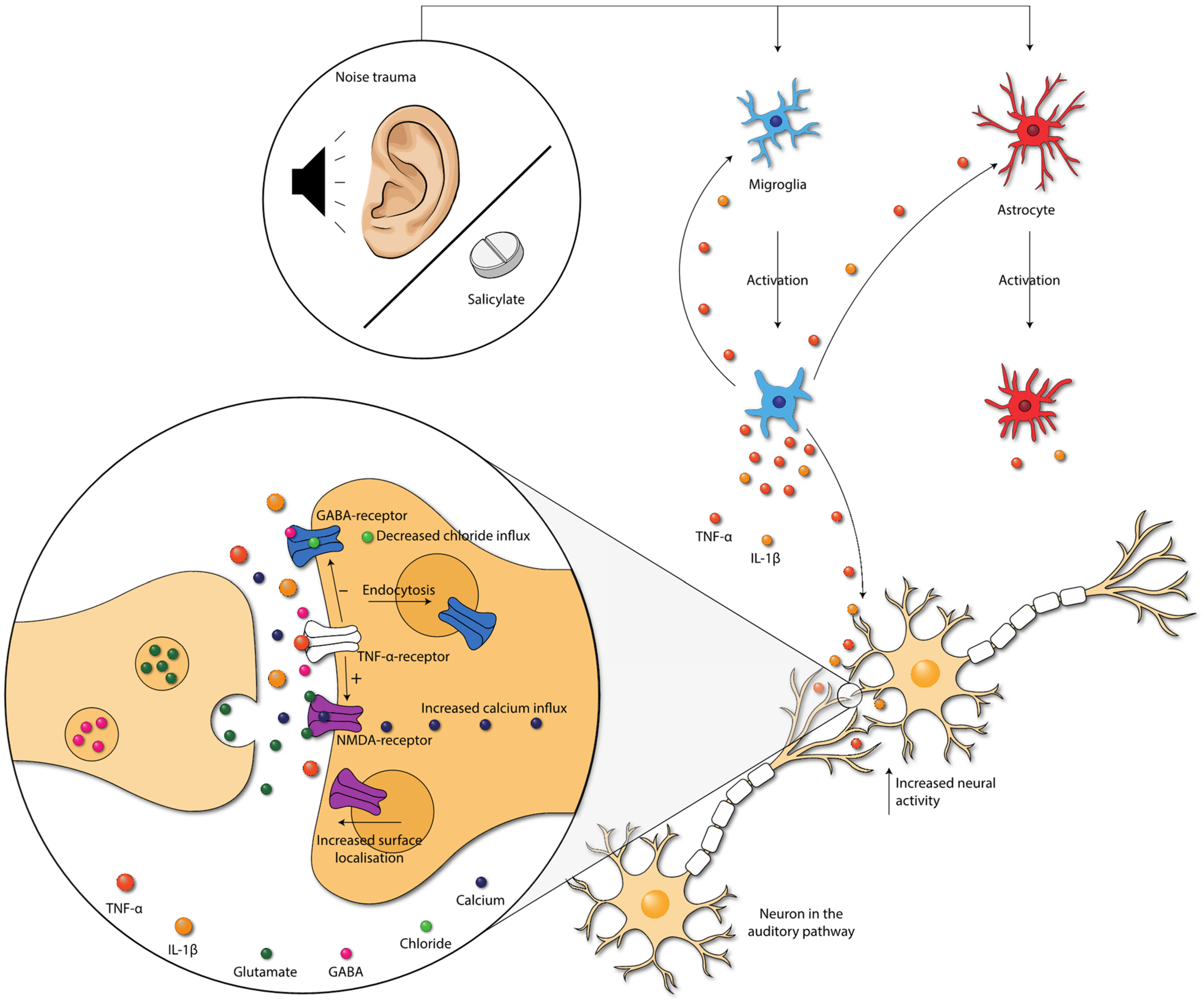
4.1.1. TNF-α in Tinnitus
4.1.2. IL-1β in Tinnitus
4.2. Neuroglial Involvement in Tinnitus
4.3. Platelet Involvement in Tinnitus
4.4. Inflammation in the Pathophysiology of Tinnitus
4.5. Challenges in the Interpretation of Results on Inflammation in Tinnitus
4.5.1. Translation from Animal to Human
4.5.2. Contribution of Hearing Loss
4.6. Potential Treatment Options
4.6.1. Treatments Targeting Cytokines
4.6.2. Treatments Targeting Microglia
4.7. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McCormack, A.; Edmondson-Jones, M.; Somerset, S.; Hall, D. A Systematic Review of the Reporting of Tinnitus Prevalence and Severity. Hear. Res. 2016, 337, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, J.J.; Roberts, L.E. The Neuroscience of Tinnitus. Trends Neurosci. 2004, 27, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Baguley, D.; McFerran, D.; Hall, D. Tinnitus. Lancet 2013, 382, 1600–1607. [Google Scholar] [CrossRef] [Green Version]
- Knipper, M.; Van Dijk, P.; Nunes, I.; Rüttiger, L.; Zimmermann, U. Advances in the Neurobiology of Hearing Disorders: Recent Developments Regarding the Basis of Tinnitus and Hyperacusis. Prog. Neurobiol. 2013, 111, 17–33. [Google Scholar] [CrossRef]
- Fuentes-Santamaría, V.; Alvarado, J.C.; Melgar-Rojas, P.; Gabaldón-Ull, M.C.; Miller, J.M.; Juiz, J.M. The Role of Glia in the Peripheral and Central Auditory System Following Noise Overexposure: Contribution of TNF-α and IL-1β to the Pathogenesis of Hearing Loss. Front. Neuroanat. 2017, 11, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frye, M.D.; Ryan, A.F.; Kurabi, A. Inflammation Associated with Noise-Induced Hearing Loss. J. Acoust. Soc. Am. 2019, 146, 4020–4032. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (Updated February 2021). Available online: www.training.cochrane.org/handbook (accessed on 17 August 2021).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef]
- Viechtbauer, W. Bias and Efficiency of Meta-Analytic Variance Estimators in the Random-Effects Model. J. Educ. Behav. Stat. 2005, 30, 261–293. [Google Scholar] [CrossRef]
- Knapp, G.; Hartung, J. Improved Tests for a Random Effects Meta-Regression with a Single Covariate. Stat. Med. 2003, 22, 2693–2710. [Google Scholar] [CrossRef]
- Steegen, S.; Tuerlinckx, F.; Gelman, A.; Vanpaemel, W. Increasing Transparency through a Multiverse Analysis. Perspect. Psychol. Sci. 2016, 11, 702–712. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to Perform a Meta-Analysis with R: A Practical Tutorial. Evid.-Based Ment. Health 2019, 22, 153–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viechtbauer, W. Conducting Meta-Analyses in R with the Metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef] [Green Version]
- Harrer, M.; Cuijpers, P.; Furukawa, T.; Ebert, D.D. Dmetar: Companion R Package for the Guide “Doing Meta-Analysis in R”. 2019. Available online: http://dmetar.protectlab.org/ (accessed on 22 November 2021).
- van Aert, R.C.M.; Wicherts, J.M.; van Assen, M.A.L.M. Conducting Meta-Analyses Based on p Values: Reservations and Recommendations for Applying p-Uniform and p-Curve. Perspect. Psychol. Sci. 2016, 11, 713–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Zhang, L.S.; Zinsmaier, A.K.; Patterson, G.; Leptich, E.J.; Shoemaker, S.L.; Yatskievych, T.A.; Gibboni, R.; Pace, E.; Luo, H.; et al. Neuroinflammation Mediates Noise-Induced Synaptic Imbalance and Tinnitus in Rodent Models. PLoS Biol. 2019, 17, e3000307. [Google Scholar] [CrossRef]
- Hu, S.S.; Mei, L.; Chen, J.Y.; Huang, Z.W.; Wu, H. Effects of Salicylate on the Inflammatory Genes Expression and Synaptic Ultrastructure in the Cochlear Nucleus of Rats. Inflammation 2014, 37, 365–373. [Google Scholar] [CrossRef]
- Chen, X.H.; Zheng, L.L. Expression of Pro-Inflammatory Cytokines in the Auditory Cortex of Rats with Salicylate-Induced Tinnitus. Mol. Med. Rep. 2017, 16, 5643–5648. [Google Scholar] [CrossRef] [Green Version]
- Hwang, J.-H.; Chen, J.-C.; Chan, Y.-C. Effects of C-Phycocyanin and Spirulina on Salicylate-Induced Tinnitus, Expression of NMDA Receptor and Inflammatory Genes. PLoS ONE 2013, 8, e58215. [Google Scholar] [CrossRef]
- Hwang, J.-H.; Chen, J.-C.; Yang, S.-Y.; Wang, M.-F.; Chan, Y.-C. Expression of Tumor Necrosis Factor-(Alpha) and Interleukin-1(Beta) Genes in the Cochlea and Inferior Colliculus in Salicylate-Induced Tinnitus. J. Neuroinflamm. 2011, 8, 2–7. [Google Scholar] [CrossRef] [Green Version]
- Xia, C.; Yin, M.; Wu, C.; Ji, Y.; Zhou, Y. Neuroglial Activation in the Auditory Cortex and Medial Geniculate Body of Salicylate-Induced Tinnitus Rats. Am. J. Transl. Res. 2020, 12, 6043–6059. [Google Scholar]
- Fang, L.; Fu, Y.; Zhang, T.-Y. Salicylate-Induced Hearing Loss Trigger Structural Synaptic Modifications in the Ventral Cochlear Nucleus of Rats via Medial Olivocochlear (MOC) Feedback Circuit. Neurochem. Res. 2016, 41, 1343–1353. [Google Scholar] [CrossRef]
- Hwang, J.-H.; Chen, J.-C.; Yang, S.-Y.; Wang, M.-F.; Liu, T.-C.; Chan, Y.-C. Expression of COX-2 and NMDA Receptor Genes at the Cochlea and Midbrain in Salicylate-Induced Tinnitus. Laryngoscope 2011, 121, 361–364. [Google Scholar] [CrossRef]
- Hwang, J.-H.; Huang, D.C.-W.; Lu, Y.-C.; Yang, W.-S.; Liu, T.-C. Effects of Tumor Necrosis Factor Blocker on Salicylate-Induced Tinnitus in Mice. Int. Tinnitus J. 2017, 21, 24–29. [Google Scholar] [CrossRef]
- Bayraktar, C.; Taşolar, S. Relationship between Increased Carotid Artery Stiffness and Idiopathic Subjective Tinnitus. Eur. Arch. Oto-Rhino-Laryngol. 2017, 274, 2125–2130. [Google Scholar] [CrossRef]
- Szczepek, A.J.; Haupt, H.; Klapp, B.F.; Olze, H.; Mazurek, B. Biological Correlates of Tinnitus-Related Distress: An Exploratory Study. Hear. Res. 2014, 318, 23–30. [Google Scholar] [CrossRef]
- Sarıkaya, Y.; Bayraktar, C.; Karataş, M.; Doğan, S.; Olt, S.; Kaskalan, E.; Türkbeyler, İ.H. Increased Mean Platelet Volume in Patients with Idiopathic Subjective Tinnitus. Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 3533–3536. [Google Scholar] [CrossRef]
- Kemal, O.; Müderris, T.; Başar, F.; Kutlar, G.; Gül, F. Prognostic Value of Mean Platelet Volume on Tinnitus. J. Laryngol. Otol. 2016, 130, 162–165. [Google Scholar] [CrossRef]
- Savastano, M.; Aita, M.; Barlani, F. Psychological, Neural, Endocrine, and Immune Study of Stress in Tinnitus Patients: Any Correlation between Psychometric and Biochemical Measures? Ann. Otol. Rhinol. Laryngol. 2007, 116, 100–106. [Google Scholar] [CrossRef]
- Savastano, M.; Celadin, M.; Pittoni, M.; Plebani, M.; Marioni, G. Western Blot Immunoassay for HSP-70 Antibodies in Idiopathic Tinnitus: A Preliminary Report. Ann. Otol. Rhinol. Laryngol. 2006, 115, 243–246. [Google Scholar] [CrossRef]
- Haider, H.F.; Ribeiro, S.F.; Martins, C.; Ribeiro, D.; Trigueiros, N.; Szczepek, A.J.; Caria, H.; Hoare, D.J.; Paço, J.; Borrego, L.M. Tinnitus, Hearing Loss and Inflammatory Processes in an Older Portuguese Population. Int. J. Audiol. 2020, 59, 323–332. [Google Scholar] [CrossRef]
- Weber, C.; Arck, P.; Mazurek, B.; Klapp, B.F. Impact of a Relaxation Training on Psychometric and Immunologic Parameters in Tinnitus Sufferers. J. Psychosom. Res. 2002, 52, 29–33. [Google Scholar] [CrossRef]
- Avci, D. Can Mean Platelet Volume Be Used as a Thrombosis Marker in Subjective Tinnitus? Erciyes Med. J. 2020, 42, 157–162. [Google Scholar] [CrossRef]
- Bayram, A.; Yaşar, M.A.; Doğan, M.; Güneri, E.; Özcan, İ. Assessment of Neutrophil-to-Lymphocyte Ratio, Platelet-to-Lymphocyte Ratio and Mean Platelet Volume in Patients with Tinnitus. ENT Updates 2015, 5, 103–106. [Google Scholar] [CrossRef]
- Çeçen, A.; Kemal, Ö.; Yildirim, U.; Kavaz, E.; Terzi, Ö. The Clinical and Prognostic Value of the Neutrophil Lymphocyte Ratio, the Platelet Lymphocyte Ratio and Mean Platelet Volume in Tinnitus Patients. J. Exp. Clin. Med. 2021, 38, 251–254. [Google Scholar] [CrossRef]
- Chrbolka, P.; Alušík, Š.; Kalátová, D.; Paluch, Z. Increased Platelet Activity in Tinnitus Patients. Neuroendocrinol. Lett. 2020, 41, 102–106. [Google Scholar]
- Demir, M. Does Inflammation Play a Role in the Pathophysiology of Tinnitus? Niger. J. Clin. Pract. 2021, 24, 199–204. [Google Scholar] [CrossRef]
- Düzenli, U.; Bozan, N.; Aslan, M.; Özkan, H.; Turan, M.; Kıroğlu, A.F. A Retrospective Analysis of Haemotologic Parameters in Patients with Bilateral Tinnitus. East. J. Med. 2018, 23, 264–268. [Google Scholar] [CrossRef]
- Gunes, A.; Karali, E.; Ural, A.; Ruzgar, F.; Bayatkara, T. The Relationship of High-Frequency Distortion Product Otoacoustic Emission (DPOAE) Values with Hematological Parameters in Tinnitus Patients. Eur. Arch. Oto-Rhino-Laryngol. 2019, 276, 3013–3019. [Google Scholar] [CrossRef]
- Li, J.; Jin, J.; Xi, S.; Zhu, Q.; Chen, Y.; Huang, M.; He, C. Clinical Efficacy of Cognitive Behavioral Therapy for Chronic Subjective Tinnitus. Am. J. Otolaryngol. 2019, 40, 253–256. [Google Scholar] [CrossRef]
- Ozbay, I.; Kahraman, C.; Balikci, H.H.; Kucur, C.; Kahraman, N.K.; Ozkaya, D.P.; Oghan, F. Neutrophil-to-Lymphocyte Ratio in Patients with Severe Tinnitus: Prospective, Controlled Clinical Study. J. Laryngol. Otol. 2015, 129, 544–547. [Google Scholar] [CrossRef]
- Ulusoy, B.; Bozdemir, K.; Akyol, M.; Mişe, H.I.; Kutluhan, A.; Korkmaz, M.H. Investigation of Neutrophil-to-Lymphocyte Ratio, Platelet-to-Lymphocyte Ratio and Mean Platelet Volume in Patients with Tinnitus. J. Laryngol. Otol. 2018, 132, 129–132. [Google Scholar] [CrossRef]
- Yildiz, S.; Karaca, H.; Toros, S.Z. Mean Platelet Volume and Neutrophil to Lymphocyte Ratio in Patients with Tinnitus: A Case-Control Study. Braz. J. Otorhinolaryngol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yüksel, F.; Karataş, D. Can Platelet Indices Be New Biomarkers for Subjective Tinnitus? J. Craniofacial Surg. 2016, 27, e420–e424. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Zadina, J.E.; Harlan, R.E.; Weber, J.T.; Banks, W.A.; Kastin, A.J. Tumor Necrosis Factor-α: A Neuromodulator in the CNS. Neurosci. Biobehav. Rev. 1997, 21, 603–613. [Google Scholar] [CrossRef]
- Jang, C.H.; Lee, S.; Park, I.Y.; Song, A.; Moon, C.; Cho, G.-W. Memantine Attenuates Salicylate-Induced Tinnitus Possibly by Reducing NR2B Expression in Auditory Cortex of Rat. Exp. Neurobiol. 2019, 28, 495–503. [Google Scholar] [CrossRef]
- Nguyen, M.D.; Julien, J.P.; Rivest, S. Innate Immunity: The Missing Link in Neuroprotection and Neurodegeneration? Nat. Rev. Neurosci. 2002, 3, 216–227. [Google Scholar] [CrossRef]
- Shaftel, S.S.; Griffin, W.S.T.; Kerry, K.M. The Role of Interleukin-1 in Neuroinflammation and Alzheimer Disease: An Evolving Perspective. J. Neuroinflamm. 2008, 5, 7. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, C.; Van Dam, A.M.; Lucassen, P.J.; Bol, J.G.J.M.; Winblad, B.; Schultzberg, M. Immunohistochemical Localization of Interleukin-1β, Interleukin-1 Receptor Antagonist and Interleukin-1β Converting Enzyme/Caspase-1 in the Rat Brain after Peripheral Administration of Kainic Acid. Neuroscience 1999, 93, 915–930. [Google Scholar] [CrossRef]
- Dinarello, C.A.; Thompson, R.C. Blocking IL-1: Interleukin 1 Receptor Antagonist in Vivo and in Vitro. Immunol. Today 1991, 12, 404–410. [Google Scholar] [CrossRef]
- Pekny, M.; Wilhelmsson, U.; Tatlisumak, T.; Pekna, M. Astrocyte Activation and Reactive Gliosis—A New Target in Stroke? Neurosci. Lett. 2019, 689, 45–55. [Google Scholar] [CrossRef]
- Zeng, Z.; Roussakis, A.A.; Lao-Kaim, N.P.; Piccini, P. Astrocytes in Parkinson’s Disease: From Preclinical Assays to in Vivo Imaging and Therapeutic Probes. Neurobiol. Aging 2020, 95, 264–270. [Google Scholar] [CrossRef]
- Kane, C.J.M.; Drew, P.D. Neuroinflammatory Contribution of Microglia and Astrocytes in Fetal Alcohol Spectrum Disorders. J. Neurosci. Res. 2020, 99, 1973–1985. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.; Gross, J.; Morest, D.K. Fibroblast Growth Factors (FGFs) in the Cochlear Nucleus of the Adult Mouse Following Acoustic Overstimulation. Hear. Res. 2002, 169, 1–12. [Google Scholar] [CrossRef]
- Parpura, V.; Basarsky, T.A.; Liu, F.; Jeftinija, K.; Jeftinija, S.; Haydon, P.G. Glutamate-Mediated Astrocyte-Neuron Signalling. Nature 1994, 369, 744–747. [Google Scholar] [CrossRef] [PubMed]
- Fellin, T.; Pascual, O.; Gobbo, S.; Pozzan, T.; Haydon, P.G.; Carmignoto, G. Neuronal Synchrony Mediated by Astrocytic Glutamate through Activation of Extrasynaptic NMDA Receptors. Neuron 2004, 43, 729–743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, K.M.; Bowers, W.J. Tumor Necrosis Factor-Alpha Mediated Signaling in Neuronal Homeostasis and Dysfunction. Cell Signal. 2010, 22, 977–983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Zhong, H.; Zhao, Y.; Luo, X.; Gao, W. Role of Platelet Biomarkers in Inflammatory Response. Biomark. Res. 2020, 8, 2–8. [Google Scholar] [CrossRef]
- Morrell, C.N.; Aggrey, A.A.; Chapman, L.M.; Modjeski, K.L. Emerging Roles for Platelets as Immune and Inflammatory Cells. Blood 2014, 123, 2759–2767. [Google Scholar] [CrossRef] [Green Version]
- Aggrey, A.A.; Srivastava, K.; Ture, S.; Field, D.J.; Morrell, C.N. Platelet Induction of the Acute-Phase Response Is Protective in Murine Experimental Cerebral Malaria. J. Immunol. 2013, 190, 4685–4691. [Google Scholar] [CrossRef] [Green Version]
- Behari, M.; Shrivastava, M. Role of Platelets in Neurodegenerative Diseases: A Universal Pathophysiology. Int. J. Neurosci. 2013, 123, 287–299. [Google Scholar] [CrossRef]
- Eggermont, J.J. Tinnitus: Neurobiological Substrates. Drug Discov. Today 2005, 10, 1283–1290. [Google Scholar] [CrossRef]
- Guitton, M.J.; Caston, J.; Ruel, J.; Johnson, R.M.; Pujol, R.; Puel, J.L. Salicylate Induces Tinnitus through Activation of Cochlear NMDA Receptors. J. Neurosci. 2003, 23, 3944–3952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furukawa, K.; Mattson, M.P. The Transcription Factor NF-ΚB Mediates Increases in Calcium Currents and Decreases in NMDA- and AMPA/Kainate-Induced Currents Induced by Tumor Necrosis Factor-α in Hippocampal Neurons. J. Neurochem. 1998, 70, 1876–1886. [Google Scholar] [CrossRef] [PubMed]
- Jara, J.H.; Singh, B.B.; Floden, A.M.; Combs, C.K. Tumor Necrosis Factor Alpha Stimulates NMDA Receptor Activity in Mouse Cortical Neurons Resulting in ERK-Dependent Death. J. Neurochem. 2007, 100, 1407–1420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawasaki, Y.; Zhang, L.; Cheng, J.K.; Ji, R.R. Cytokine Mechanisms of Central Sensitization: Distinct and Overlapping Role of Interleukin-1β, Interleukin-6, and Tumor Necrosis Factor-α in Regulating Synaptic and Neuronal Activity in the Superficial Spinal Cord. J. Neurosci. 2008, 28, 5189–5194. [Google Scholar] [CrossRef] [Green Version]
- Wheeler, D.; Knapp, E.; Bandaru, V.V.R.; Wang, Y.; Knorr, D.; Poirier, C.; Mattson, M.P.; Geiger, J.D.; Haughey, N.J. Tumor Necrosis Factor-α-Induced Neutral Sphingomyelinase-2 Modulates Synaptic Plasticity by Controlling the Membrane Insertion of NMDA Receptors. J. Neurochem. 2009, 109, 1237–1249. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.; Jiang, C.Y.; Fujita, T.; Luo, S.W.; Kumamoto, E. Enhancement by Interleukin-1β of AMPA and NMDA Receptor-Mediated Currents in Adult Rat Spinal Superficial Dorsal Horn Neurons. Mol. Pain 2013, 9, 1. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.T.; Luo, B.; Zhou, K.Q.; Xu, T.L.; Chen, L. Sodium Salicylate Reduces Inhibitory Postsynaptic Currents in Neurons of Rat Auditory Cortex. Hear. Res. 2006, 215, 77–83. [Google Scholar] [CrossRef]
- Jin, Y.; Luo, B.; Su, Y.Y.; Wang, X.X.; Chen, L.; Wang, M.; Wang, W.W.; Chen, L. Sodium Salicylate Suppresses GABAergic Inhibitory Activity in Neurons of Rodent Dorsal Raphe Nucleus. PLoS ONE 2015, 10, e0126956. [Google Scholar] [CrossRef]
- Brozoski, T.J.; Spires, T.J.D.; Bauer, C.A. Vigabatrin, a GABA Transaminase Inhibitor, Reversibly Eliminates Tinnitus in an Animal Model. JARO J. Assoc. Res. Otolaryngol. 2007, 8, 105–118. [Google Scholar] [CrossRef] [Green Version]
- Stellwagen, D.; Beattie, E.C.; Seo, J.Y.; Malenka, R.C. Differential Regulation of AMPA Receptor and GABA Receptor Trafficking by Tumor Necrosis Factor-α. J. Neurosci. 2005, 25, 3219–3228. [Google Scholar] [CrossRef] [Green Version]
- Gerrow, K.; Triller, A. Synaptic Stability and Plasticity in a Floating World. Curr. Opin. Neurobiol. 2010, 20, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Shore, S.E.; Roberts, L.E.; Langguth, B. Maladaptive Plasticity in Tinnitus-Triggers, Mechanisms and Treatment. Nat. Rev. Neurol. 2016, 12, 150–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, F.R.; Musella, A.; De Vito, F.; Fresegna, D.; Bullitta, S.; Vanni, V.; Guadalupi, L.; Stampanoni Bassi, M.; Buttari, F.; Mandolesi, G.; et al. Tumor Necrosis Factor and Interleukin-1β Modulate Synaptic Plasticity during Neuroinflammation. Neural Plast. 2018, 2018, 8430123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakamoto, K.; Karelina, K.; Obrietan, K. CREB: A Multifaceted Regulator of Neuronal Plasticity and Protection. J. Neurochem. 2011, 116, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vezzani, A.; Viviani, B. Neuromodulatory Properties of Inflammatory Cytokines and Their Impact on Neuronal Excitability. Neuropharmacology 2015, 96, 70–82. [Google Scholar] [CrossRef]
- Roberts, L.E. Neural Plasticity and Its Initiating Conditions in Tinnitus. HNO 2018, 66, 172–178. [Google Scholar] [CrossRef]
- Wu, C.; Stefanescu, R.A.; Martel, D.T.; Shore, S.E. Tinnitus: Maladaptive Auditory-Somatosensory Plasticity. Hear. Res. 2016, 334, 20–29. [Google Scholar] [CrossRef] [Green Version]
- Elarbed, A.; Fackrell, K.; Baguley, D.M.; Hoare, D.J. Tinnitus and Stress in Adults: A Scoping Review. Int. J. Audiol. 2021, 60, 171–182. [Google Scholar] [CrossRef]
- Baigi, A.; Oden, A.; Almlid-Larsen, V.; Barrenäs, M.L.; Holgers, K.M. Tinnitus in the General Population with a Focus on Noise and Stress: A Public Health Study. Ear Hear. 2011, 32, 787–789. [Google Scholar] [CrossRef]
- Rohleder, N. Stress and Inflammation—The Need to Address the Gap in the Transition between Acute and Chronic Stress Effects. Psychoneuroendocrinology 2019, 105, 164–171. [Google Scholar] [CrossRef]
- Stolzberg, D.; Salvi, R.J.; Allman, B.L. Salicylate Toxicity Model of Tinnitus. Front. Syst. Neurosci. 2012, 6, 28. [Google Scholar] [CrossRef] [Green Version]
- Koops, E.A.; Eggermont, J.J. The Thalamus and Tinnitus: Bridging the Gap between Animal Data and Findings in Humans. Hear. Res. 2021, 407, 108280. [Google Scholar] [CrossRef]
- Manohar, S.; Dahar, K.; Adler, H.J.; Dalian, D.; Salvi, R. Noise-Induced Hearing Loss: Neuropathic Pain via Ntrk1 Signaling. Mol. Cell. Neurosci. 2016, 75, 101–112. [Google Scholar] [CrossRef] [Green Version]
- Smith, J.A.; Das, A.; Ray, S.K.; Banik, N.L. Role of Pro-Inflammatory Cytokines Released from Microglia in Neurodegenerative Diseases. Brain Res. Bull. 2012, 87, 10–20. [Google Scholar] [CrossRef]
- Ledeboer, A.; Sloane, E.M.; Milligan, E.D.; Frank, M.G.; Mahony, J.H.; Maier, S.F.; Watkins, L.R. Minocycline Attenuates Mechanical Allodynia and Proinflammatory Cytokine Expression in Rat Models of Pain Facilitation. Pain 2005, 115, 71–83. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mennink, L.M.; Aalbers, M.W.; van Dijk, P.; van Dijk, J.M.C. The Role of Inflammation in Tinnitus: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 1000. https://doi.org/10.3390/jcm11041000
Mennink LM, Aalbers MW, van Dijk P, van Dijk JMC. The Role of Inflammation in Tinnitus: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2022; 11(4):1000. https://doi.org/10.3390/jcm11041000
Chicago/Turabian StyleMennink, Lilian M., Marlien W. Aalbers, Pim van Dijk, and J. Marc C. van Dijk. 2022. "The Role of Inflammation in Tinnitus: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 11, no. 4: 1000. https://doi.org/10.3390/jcm11041000
APA StyleMennink, L. M., Aalbers, M. W., van Dijk, P., & van Dijk, J. M. C. (2022). The Role of Inflammation in Tinnitus: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 11(4), 1000. https://doi.org/10.3390/jcm11041000







