Influence of Renal Dysfunction on the Differential Behaviour of Procalcitonin for the Diagnosis of Postoperative Infection in Cardiac Surgery
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Definition of Impairment in Renal Function
2.3. Procalcitonin and C-Reactive Protein Quantification
2.4. Statistical Analysis
3. Results
3.1. Clinical Characteristics
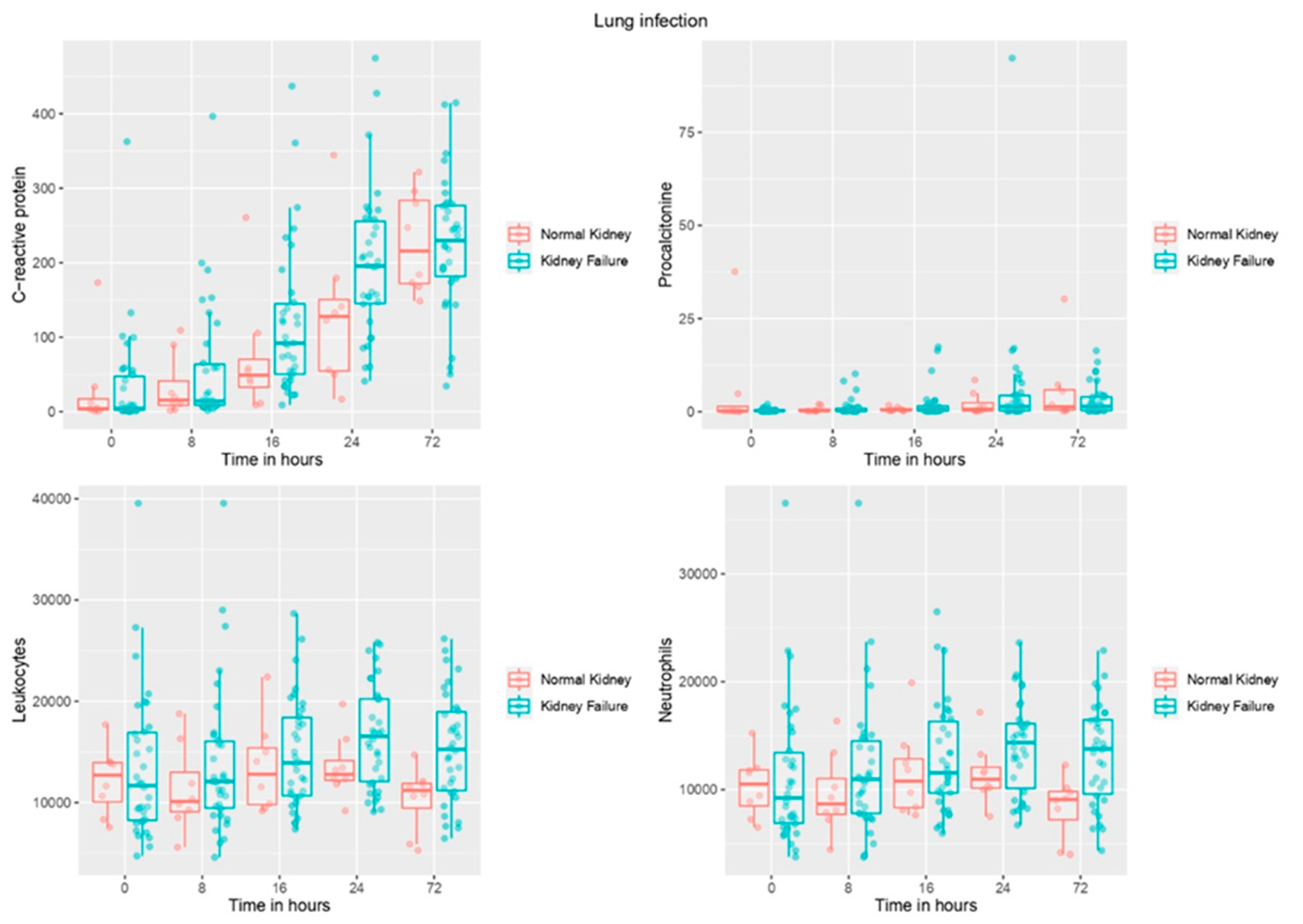
3.2. Biomarker Levels Based on Presence/Absence of Renal Failure and over Time in Patients with Infection
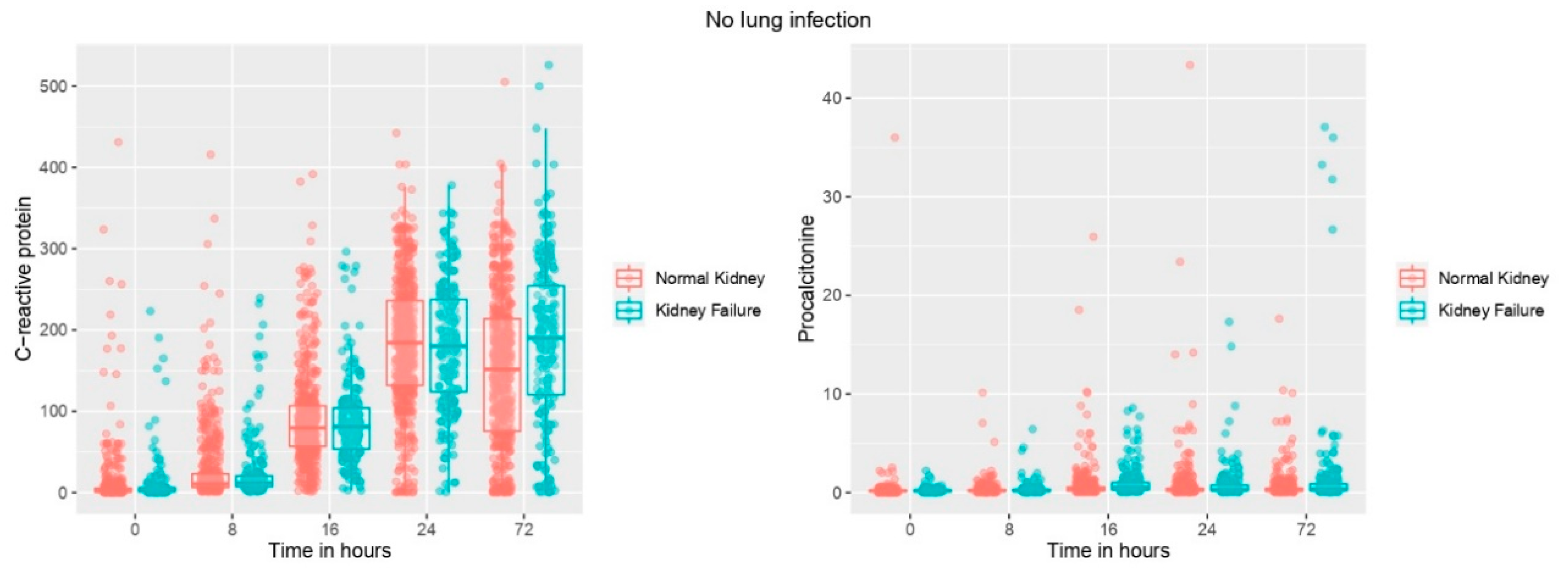
3.3. Biomarker Levels Based on the Presence/Absence of Renal Failure and over Time in Patients without Infection
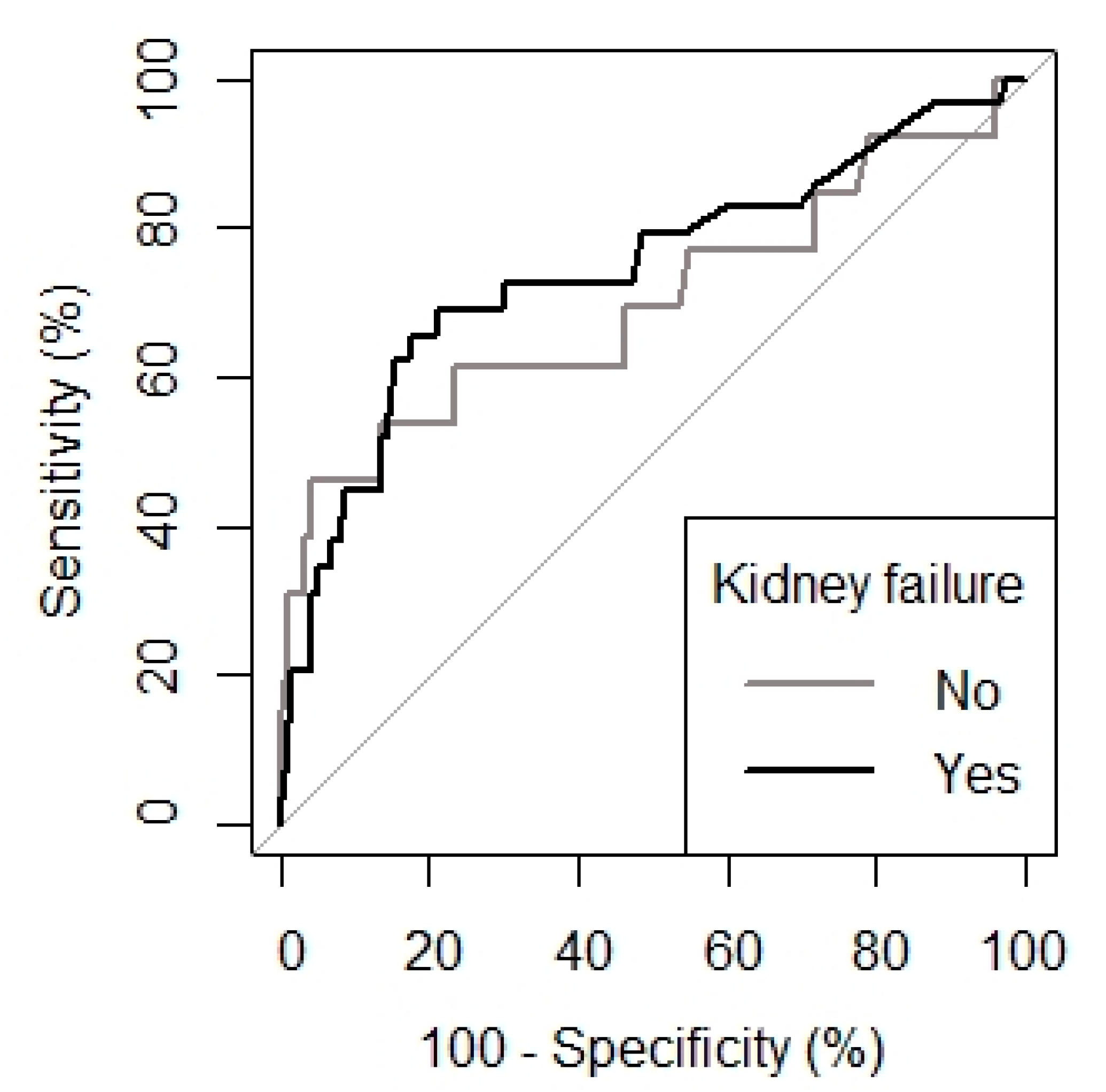
3.4. Biomarkers Association with Presence of Renal Failure over Time in Patients with Infection
3.5. Biomarker Association with Presence of Renal Failure over Time in Patients without Infection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Perrotti, A.; Chenevier-Gobeaux, C.; Ecarnot, F.; Barrucand, B.; Lassalle, P.; Dorigo, E.; Chocron, S. Relevance of Endothelial Cell-Specific Molecule 1 (Endocan) Plasma Levels for Predicting Pulmonary Infection after Cardiac Surgery in Chronic Kidney Disease Patients: The Endolung Pilot Study. Cardiorenal Med. 2017, 8, 1–8. [Google Scholar]
- Tamayo, E.; Álvarez, F.J.; Martínez-Rafael, B.; Bustamante, J.; Bermejo-Martin, J.F.; Fierro, I.; Eiros, J.M.; Castrodeza, J.; Heredia, M.; Gómez-Herreras, J.I. Ventilator-associated pneumonia is an important risk factor for mortality after major cardiac surgery. J. Crit. Care 2012, 27, 18–25. [Google Scholar] [CrossRef]
- Cunnion, K.M.; Weber, D.J.; Broadhead, W.E.; Hanson, L.C.; Pieper, C.F.; Rutala, W.A. Risk factors for nosocomial pneumonia: Comparing adult critical-care populations. Am. J. Respir. Crit. Care Med. 1996, 153, 158–162. [Google Scholar]
- Melsen, W.G.; Rovers, M.M.; Groenwold, R.H.H.; Bergmans, D.C.J.J.; Camus, C.; Bauer, T.T.; Hanisch, E.; Klarin, B.; Koeman, M.; A Krueger, W.; et al. Attributable mortality of ventilator-associated pneumonia: A meta-analysis of individual patient data from randomised prevention studies. Lancet Infect. Dis. 2013, 13, 665–671. [Google Scholar]
- Nguile-Makao, M.; Zahar, J.-R.; Français, A.; Tabah, A.; Garrouste-Orgeas, M.; Allaouchiche, B.; Goldgran-Toledano, D.; Azoulay, E.; Adrie, C.; Jamali, S.; et al. Attributable mortality of ventilator-associated pneumonia: Respective impact of main characteristics at ICU admission and VAP onset using conditional logistic regression and multi-state models. Intensive Care Med. 2010, 36, 781–789. [Google Scholar]
- Amour, J.; Birenbaum, A.; Langeron, O.; Le Manach, Y.; Bertrand, M.; Coriat, P.; Riou, B.; Bernard, M.; Hausfater, P. Influence of renal dysfunction on the accuracy of procalcitonin for the diagnosis of postoperative infection after vascular surgery. Crit. Care Med. 2008, 36, 1147–1154. [Google Scholar]
- Yang, Y.; Zhang, Y.; Li, S.; Zheng, X.; Wong, M.-H.; Sak Leung, K.; Cheng, L. A robust and generalizable inmune-related signature for sepsis diagnostics. IEEE/ACM Trans. Comput. Biol. Bioinform. 2021. early access. [Google Scholar] [CrossRef]
- Zheng, X.; Leung, K.-S.; Wong, M.-H.; Cheng, L. Long non-coding RNA pairs to assist in diagnosing sepsis. BMC Genom. 2021, 22, 275. [Google Scholar]
- Jebali, M.A.; Hausfater, P.; Abbes, Z.; Aouni, Z.; Riou, B.; Ferjani, M. Assessment of the accuracy of procalcitonin to diagnose postoperative infection after cardiac surgery. Anesthesiology 2007, 107, 232–238. [Google Scholar]
- Rothenburger, M.; Markewitz, A.; Lenz, T.; Kaulbach, H.G.; Marohl, K.; Kuhlmann, W.D.; Weinhold, C. Detection of acute phase response and infection. The role of procalcitonin and C-reactive protein. Clin. Chem. Lab Med. 1999, 37, 275–279. [Google Scholar]
- Uchino, S.; Kellum, J.A.; Bellomo, R.; Doig, G.S.; Morimatsu, H.; Morgera, S.; Schetz, M.; Tan, I.; Bouman, C.; Macedo, E.; et al. Acute renal failure in critically ill patients: A multinational, multicenter study. JAMA 2005, 294, 813–818. [Google Scholar]
- Cubero-Gallego, H.; Heredia-Rodriguez, M.; Tamayo, E. Influence of impairment in renal function on the accuracy of high-sensitivity cardiac troponin T for the diagnosis of perioperative myocardial infarction after heart valve surgery. Interact. Cardiovasc. Thorac. Surg. 2018, 27, 234–237. [Google Scholar]
- Freda, B.J.; Tang, W.H.W.; Van Lente, F.; Peacock, W.F.; Francis, G.S. Cardiac troponins in renal insufficiency: Review and clinical implications. J. Am. Coll. Cardiol. 2002, 40, 2065–2071. [Google Scholar]
- Kellum, J.A.; Sileanu, F.E.; Murugan, R.; Lucko, N.; Shaw, A.D.; Clermont, G. Classifying AKI by Urine Output versus Serum Creatinine Level. J. Am. Soc. Nephrol. 2015, 26, 2231–2238. [Google Scholar]
- Murashima, M.; Nishimoto, M.; Kokubu, M.; Hamano, T.; Matsui, M.; Eriguchi, M.; Samejima, K.-I.; Akai, Y.; Tsuruya, K. Inflammation as a predictor of acute kidney injury and mediator of higher mortality after acute kidney injury in non-cardiac surgery. Sci. Rep. 2019, 9, 20260. [Google Scholar]
- Han, S.S.; Kim, D.K.; Kim, S.; Chin, H.J.; Chae, D.-W.; Na, K.Y. C-Reactive Protein Predicts Acute Kidney Injury and Death after Coronary Artery Bypass Grafting. Ann. Thorac. Surg. 2017, 104, 804–810. [Google Scholar]
- Seoane, L.A.; Burgos, L.; Espinoza, J.C.; Furmento, J.F.; Benzadón, M.N.; Vrancic, J.M.; Piccinini, F.; Navia, D. Prognostic Value of the Leuko-Glycaemic Index in the Postoperative Period of Coronary Artery Bypass Grafting. Braz. J. Cardiovasc. Surg. 2021, 36, 484–491. [Google Scholar]
- Usta, S.; Abanoz, M. Can Peroperative Neutrophil To Lymphocyte Ratio Change (Deltanlr) Be Used as a Parameter in Predicting Acute Renal Failure Following Coronary Bypass Operations With Cardiopulmonary Bypass? Heart Surg. Forum. 2021, 24, E194–E200. [Google Scholar]
- Chang, Y.-C.; Tsai, P.-H.; Chou, Y.-C.; Lu, K.-C.; Chang, F.-Y.; Wu, C.-C. Biomarkers Linked with Dynamic Changes of Renal Function in Asymptomatic and Mildly Symptomatic COVID-19 Patients. J. Pers. Med. 2021, 11, 432. [Google Scholar]
- Sakaki, K.; Kitamura, T.; Kohira, S.; Torii, S.; Mishima, T.; Hanayama, N.; Kobayashi, K.; Ohkubo, H.; Miyaji, K. Regional thigh tissue oxygen saturation during cardiopulmonary bypass predicts acute kidney injury after cardiac surgery. J. Artif. Organs. 2020, 23, 315–320. [Google Scholar]
- Bi, J.-B.; Zhang, J.; Ren, Y.-F.; Du, Z.-Q.; Wu, Z.; Lv, Y.; Wu, R.-Q. Neutrophil-to-lymphocyte ratio predicts acute kidney injury occurrence after gastrointestinal and hepatobiliary surgery. World J. Gastrointest. Surg. 2020, 12, 326–335. [Google Scholar]
- Huang, W.; Liu, X.; Liu, X.; Lu, Y.; Li, Y.; Zhang, Y.; Kuang, Z.-M.; Cao, D.; Chen, A.F.; Yuan, H. Value of Neutrophil Counts in Predicting Surgery-Related Acute Kidney Injury and the Interaction of These Counts With Diabetes in Chronic Kidney Disease Patients With Hypertension. Medicine 2015, 94, e1780. [Google Scholar]
- Cao, C.; Yao, Y.; Zeng, R. Lymphocytes: Versatile Participants in Acute Kidney Injury and Progression to Chronic Kidney Disease. Front. Physiol. 2021, 12, 729084. [Google Scholar]
- Cubero-Gallego, H.; Lorenzo, M.; Heredia, M.; Gómez, I.; Tamayo, E. Diagnosis of perioperative myocardial infarction after heart valve surgery with new cut-off point of high-sensitivity troponin T and new electrocardiogram or echocardiogram changes. J. Thorac. Cardiovasc. Surg. 2017, 154, 895–903. [Google Scholar]
- Garner, J.S.; Jarvis, W.R.; Emori, T.G.; Horan, T.C.; Hughes, J.M. CDC definitions for nosocomial infections. Am. J. Infect. Control 1988, 16, 128–140. [Google Scholar]
- Aouifi, A.; Piriou, V.; Blanc, P.; Barrier, H.; Bastien, O.; Chiari, P.; Rousson, R.; Evans, R.; Lehot, J.J. Effect of cardiopulmonary by pass serum procalcitonin and C-reactive protein concentrations. Br. J. Anaesth. 1999, 83, 602–607. [Google Scholar]
- Samama, C.M.; Thiry, D.; Elalamy, I.; Diaby, M.; Guillosson, J.-J.; Kieffer, E.; Coriat, P. Perioperative activation of hemostasis in vascular surgery patients. Anesthesiology 2001, 94, 74–78. [Google Scholar]
- Aouifi, A.; Piriou, V.; Bastien, O.; Blanc, P.; Bouvier, H.; Evans, R.; Célard, M.; Vandenesch, F.; Rousson, R.; Lehot, J.J. Usefulness of procalcitonin for diagnosis of infection in cardiac surgical patients. Crit. Care Med. 2000, 28, 3171–3176. [Google Scholar]
- Al-Nawas, B.; Krammer, I.; Shah, P.M. Procalcitonin in diagnosis of severe infections. Eur. J. Med. Res. 1996, 1, 331–333. [Google Scholar]
- Lemiale, V.; Renaud, B.; Moutereau, S. N’Gako, A.; Salloum, M.; Calmettes, M.J.; Hervé, J.; Boraud, C.; Santin, A.; Grégo, J.-C.; et al. A single procalcitonin level does not predict adverse outcomes of women with pyelonephritis. Eur. Urol. 2007, 51, 1394–1401. [Google Scholar]

| No Infection (1) (n = 761) | Infection (2) (n = 42) | p Value (1 vs. 2) | |
|---|---|---|---|
| Characteristics | |||
| Age [years, median (IQR)] | 14 (70) | 11 (72.50) | 0.25 |
| Male [n, (%)] | 364 (47.8) | 23 (54.8) | 0.38 |
| Functional classification (NYHA) | |||
| I | 1 (2) | 1 (2) | 0.07 |
| II | 70.4 (536) | 59.5 (25) | 0.13 |
| III-IV | 29.6 (225) | 40.5 (17) | 0.13 |
| Comorbidities, (n, (%)) | |||
| Peripheral arterial disease | 20 (2.6) | 2 (4.8) | 0.41 |
| Chronic respiratory disease | 61 (8) | 3 (7.1) | 0.84 |
| High Blood Pressure | 669 (87.9) | 39 (92.9) | 0.33 |
| Chronic renal failure | 42 (5.5) | 4 (9.5) | 0.28 |
| Diabetes mellitus | 148 (19.4) | 14 (33.3) | 0.029 |
| Dyslipidaemia | 557 (73.2) | 35 (83.3) | 0.15 |
| Stroke | 32 (4.2) | 6 (14.3) | 0.003 |
| Chronic ischemic cardiac disease | 54 (7.1) | 4 (9.5) | 0.55 |
| Previous valve intervention | 88 (11.6) | 8 (19) | 0.15 |
| Atrial fibrillation | 266 (35) | 27 (64.3) | <0.001 |
| EuroSCORE II, % | 0.8 (1.65) | 1.1 (2.06) | 0.001 |
| Intraoperative characteristics | |||
| Time of CPB, min | 95 (38) | 122.50 (67) | <0.001 |
| Time of aortic cross-clamp, min | 69 (30) | 91 (47) | <0.001 |
| Defibrillation | 165 (21.7) | 10 (23.8) | 0.75 |
| Laboratory assessments, (median (IQR)) | |||
| Weight (kg) | 74 (15) | 70 (16) | 0.42 |
| Height (cm) | 161 (15) | 156 (19) | 0.020 |
| Troponin T hs (pg/mL) | 520.90 (449.3) | 1129.50 (2640.1) | <0.001 |
| Total bilirubin (mg/dL) | 0.8 (0.7) | 1.20 (1.7) | <0.001 |
| GOT (U/L) | 51.55 (34.4) | 92.95 (139.6) | 0.002 |
| Glucose (mg/dL) | 167.2 (44) | 188.35 (79.2) | 0.002 |
| Creatinine (mg/dL) | 0.95 (0.45) | 1.34 (0.9) | <0.001 |
| Na (mmol/L) | 138.91 (3.5) | 138.48 (6.9) | 0.52 |
| K (mmol/L) | 4 (0.62) | 4.43 (0.7) | <0.001 |
| Ca (mg/dL) | 8.01 (0.5) | 8 (0.7) | 0.83 |
| Mg (mg/dL) | 1.66 (0.3) | 1.62 (0.3) | 0.43 |
| Cl (mmol/L) | 101 (4) | 102 (7.6) | 0.40 |
| Urea (mg/dL) | 37.9 (19.7) | 56.9 (20.6) | <0.001 |
| CPK (U/mL) | 544 (412) | 632.5 (904) | 0.12 |
| CK-MB (U/L) | 22.24 (26.7) | 39.25 (62.1) | <0.001 |
| LDH (U/L) | 355.5 (141) | 490 (390) | <0.001 |
| Lactate (mmol/L) | 0.91 (1.5) | 0.44 (1.83) | 0.09 |
| Procalcitonin (ng/mL) | 0.34 (0.4) | 0.51 (1.2) | 0.014 |
| C-Reactive Protein (mg/L) | 79.79 (49.7) | 82.49 (98.6) | 0.72 |
| Erythrocytes (cells/mm3) | 3710 (620) | 3675 (490) | 0.52 |
| Haematocrit (%) | 32.7 (5.3) | 32 (4.9) | 0.37 |
| Haemoglobin (g/dL) | 10.9 (2) | 10.85 (1.7) | 0.24 |
| Platelet count (cells/mm3) | 136,000 (57000) | 135,000 (59000) | 0.30 |
| White Blood cells (cells/mm3) | 11,565 (4857.5) | 13,925 (7717.5) | 0.003 |
| Neutrophils (cells/mm3) | 9822.19 (4404.1) | 11,535 (7124.1) | 0.002 |
| SOFA score | 3 (3) | 10 (3.50) | <0.001 |
| Outcome | |||
| Length of hospital stay, [days, median (IQR)] | 13 (8) | 20.5 (21) | <0.001 |
| Length of ICU stay, [days, median (IQR)] | 3 (3) | 11 (13) | <0.001 |
| Length of mechanical ventilation, [hours, median (IQR)] | 6 (3) | 120 (171) | <0.001 |
| Mortality, [n (%)] | 24 (3.2) | 61.9 (26) | <0.001 |
| No Renal Function Impairment (n = 13) | Renal Function Impairment (n = 29) | p Value | |
|---|---|---|---|
| Procalcitonin 0 h (ng/mL) | 0.15 (0.13) | 0.30 (0.25) | 0.021 |
| C-Reactive Protein 0 h (mg/L) | 2.98 (21.80) | 4.95 (53.29) | 0.06 |
| White Blood cells 0 h (cells/mm3) | 11640 (8150) | 11,860 (8745) | 0.47 |
| Neutrophils 0 h (cells/mm3) | 9463.32 (7378.21) | 10,223.32 (5991.08) | 0.52 |
| Procalcitonin 8 h (ng/mL) | 0.20 (0.85) | 0.30 (0.86) | 0.40 |
| C-Reactive Protein 8 h (mg/L) | 10.62 (39.47) | 15.19 (95.39) | 0.19 |
| White Blood cells 8 h (cells/mm3) | 11,990 (6365) | 11,280 (7530) | 0.42 |
| Neutrophils 8 h (cells/mm3) | 10,611.15 (5698.08) | 9956.25 (7014.15) | 0.36 |
| Procalcitonin 16 h (ng/mL) | 0.40 (0.96) | 0.90 (1.65) | 0.63 |
| C-Reactive Protein 16 h (mg/L) | 89.55 (80.04) | 75.43 (136.98) | 0.63 |
| White Blood cells 16 h (cells/mm3) | 14,060 (8625) | 13,820 (7565) | 0.82 |
| Neutrophils 16 h (cells/mm3) | 11,838.52 (8051.52) | 11,233.25 (6893) | 0.73 |
| Procalcitonin 24 h (ng/mL) | 0.25 (2.98) | 1.50 (4.40) | 0.09 |
| C-Reactive Protein 24 h (mg/L) | 179.33 (148.41) | 160.78 (132.95) | 0.73 |
| White Blood cells 24 h (cells/mm3) | 13,970 (9360) | 15850 (8295) | 0.84 |
| Neutrophils 24 h (cells/mm3) | 12,838.43 (8834.61) | 13,730.84 (6143.56) | 0.99 |
| Procalcitonin 48 h (ng/mL) | 0.81 (2.55) | 0.84 (2.42) | 0.88 |
| C-Reactive Protein 48 h (mg/L) | 265.09 (127.48) | 221.95 (134.19) | 0.78 |
| White Blood cells 48 h (cells/mm3) | 14,120 (11855) | 16,140 (7055) | 0.48 |
| Neutrophils 48 h (cells/mm3) | 12,623.28 (10,918.98) | 13,769.67 (5675.79) | 0.67 |
| Procalcitonin 72 h (ng/mL) | 0.79 (6.17) | 1.90 (3.82) | 0.34 |
| C-Reactive Protein 72 h (mg/L) | 195.10 (89.37) | 243.78 (104.55) | 0.35 |
| White Blood cells 72 h (cells/mm3) | 11820 (3710) | 15410 (9465) | 0.12 |
| Neutrophils 72 h (cells/mm3) | 10,160.29 (4708.34) | 13,851 (8306.30) | 0.08 |
| No Renal Function Impairment (n = 562) | Renal Function Impairment (n = 199) | p Value | |
|---|---|---|---|
| Procalcitonin 0 h (ng/mL) | 0.20 (0.13) | 0.19 (0.13) | 0.847 |
| C-Reactive Protein 0 h (mg/L) | 1.84 (4.39) | 2.37 (6.16) | 0.091 |
| White Blood cells 0 h (cells/mm3) | 10,810 (4945) | 10,360 (5550) | 0.782 |
| Neutrophils 0 h (cells/mm3) | 8969.94 (4343.85) | 8675.04 () | 0.893 |
| Procalcitonin 8 h (ng/mL) | 0.20 (0.15) | 0.20 (0.20) | 0.143 |
| C-Reactive Protein 8 h (mg/L) | 11.21 (14.55) | 12.22 (18.71) | 0.383 |
| White Blood cells 8 h (cells/mm3) | 11,380 (4755) | 11,210 (5090) | 0.824 |
| Neutrophils 8 h (cells/mm3) | 9624.36 (4430.27) | 9459.84 (4881.14) | 0.809 |
| Procalcitonin 16 h (ng/mL) | 0.30 (0.36) | 0.59 (0.89) | <0.001 |
| C-Reactive Protein 16 h (mg/L) | 79.64 (48.31) | 80.82 (57.13) | 0.885 |
| White Blood cells 16 h (cells/mm3) | 11,430 (4645) | 12,210 (5490) | 0.036 |
| Neutrophils 16 h (cells/mm3) | 9713.34 (4140.28) | 10,350.64 (5139.92) | 0.031 |
| Procalcitonin 24 h (ng/mL) | 0.24 (0.24) | 0.33 (0.62) | <0.001 |
| C-Reactive Protein 24 h (mg/L) | 182.65 (104.10) | 186.61 (110.81) | 0.578 |
| White Blood cells 24 h (cells/mm3) | 11,960 (5080) | 12,860 (5800) | 0.003 |
| Neutrophils 24 h (cells/mm3) | 9877 (4785.94) | 11,025 (5498.64) | 0.001 |
| Procalcitonin 48 h (ng/mL) | 0.28 (0.38) | 0.51 (0.96) | <0.001 |
| C-Reactive Protein 48 h (mg/L) | 173.11 (134.90) | 219.90 (133.24) | <0.001 |
| White Blood cells 48 h (cells/mm3) | 11,180 (5265) | 11,570 (5622.50) | 0.199 |
| Neutrophils 48 h (cells/mm3) | 8964.78 (4680.39) | 9487.68 (4892.40) | 0.118 |
| Procalcitonin 72 h (ng/mL) | 0.25 (0.25) | 0.47 (0.82) | <0.001 |
| C-Reactive Protein 72 h (mg/L) | 153.80 (140.62) | 188.79 (126.82) | <0.001 |
| White Blood cells 72 h (cells/mm3) | 10,270 (5155) | 10,230 (5562.50) | 0.517 |
| Neutrophils 72 h (cells/mm3) | 8113.74 (4697.66) | 8364.55 (5509.24) | 0.311 |
| Estimate | CI 95% min | CI 95% max | p-Value | |
|---|---|---|---|---|
| Age | −0.9334000000 | −1.59 | −0.28 | 0.009050 |
| Gender | 0.8350000000 | −9.82 | 11.49 | 0.88058 |
| Time | −0.0162300000 | −0.05 | 0.02 | 0.412307 |
| CRP | −0.0378300000 | −0.05 | −0.03 | 2.31 × 10−10 |
| PCT | −0.3307000000 | −0.43 | −0.23 | 1.08 × 10−10 |
| Leukocytes | −0.0015920000 | 0.00 | 0.00 | 0.000438 |
| Neutrophils | 0.0008293000 | 0.00 | 0.00 | 0.087382 |
| Estimate | CI 95% min | CI 95% max | p-Value | |
|---|---|---|---|---|
| Age | 125.5000000000 | 78.30 | 172.98 | 7.81 × 10−6 |
| Gender | −0.9157000000 | −1.57 | −0.27 | 0.01010 |
| Time | 0.9311000000 | −9.58 | 11.43 | 0.865820 |
| CRP | −0.0082450000 | −0.10 | 0.09 | 0.863720 |
| PCT | 0.0005547000 | 0.00 | 0.00 | 0.611500 |
| Leukocytes | −0.0011590000 | 0.00 | 0.00 | 0.249550 |
| Neutrophils | −0.0419700000 | −0.07 | −0.02 | 0.00299 |
| Renal Function Impairment (n = 228) | No Renal Function Impairment (n = 574) | |
|---|---|---|
| Threshold of procalcitonin (ng/mL) | 1.00 | 0.78 |
| AUROC | 0.74 (0.63–0.85) | 0.70 (0.51–0.88) |
| p-value vs. no discrimination curve | <0.001 | 0.01 |
| Variable | Procalcitonin 0.78 ng/mL | Adapted Procalcitonin 1.00 ng/mL |
|---|---|---|
| Sensitivity | 0.65 (0.47–0.80) | 0.62 (0.44–0.77) |
| Specificity | 0.67 (0.60–0.73) | 0.74 (0.67–0.79) |
| Positive predictive value | 0.23 (0.15–0.33) | 0.26 (0.17–0.37) |
| Negative predictive value | 0.93 (0.88–0.96) | 0.92 (0.88–0.96) |
| Accuracy | 0.67 | 0.72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Varga-Martínez, O.; Martín-Fernández, M.; Heredia-Rodríguez, M.; Ceballos, F.; Cubero-Gallego, H.; Priede-Vimbela, J.M.; Bardají-Carrillo, M.; Sánchez-de Prada, L.; López-Herrero, R.; Jorge-Monjas, P.; et al. Influence of Renal Dysfunction on the Differential Behaviour of Procalcitonin for the Diagnosis of Postoperative Infection in Cardiac Surgery. J. Clin. Med. 2022, 11, 7274. https://doi.org/10.3390/jcm11247274
de la Varga-Martínez O, Martín-Fernández M, Heredia-Rodríguez M, Ceballos F, Cubero-Gallego H, Priede-Vimbela JM, Bardají-Carrillo M, Sánchez-de Prada L, López-Herrero R, Jorge-Monjas P, et al. Influence of Renal Dysfunction on the Differential Behaviour of Procalcitonin for the Diagnosis of Postoperative Infection in Cardiac Surgery. Journal of Clinical Medicine. 2022; 11(24):7274. https://doi.org/10.3390/jcm11247274
Chicago/Turabian Stylede la Varga-Martínez, Olga, Marta Martín-Fernández, María Heredia-Rodríguez, Francisco Ceballos, Hector Cubero-Gallego, Juan Manuel Priede-Vimbela, Miguel Bardají-Carrillo, Laura Sánchez-de Prada, Rocío López-Herrero, Pablo Jorge-Monjas, and et al. 2022. "Influence of Renal Dysfunction on the Differential Behaviour of Procalcitonin for the Diagnosis of Postoperative Infection in Cardiac Surgery" Journal of Clinical Medicine 11, no. 24: 7274. https://doi.org/10.3390/jcm11247274
APA Stylede la Varga-Martínez, O., Martín-Fernández, M., Heredia-Rodríguez, M., Ceballos, F., Cubero-Gallego, H., Priede-Vimbela, J. M., Bardají-Carrillo, M., Sánchez-de Prada, L., López-Herrero, R., Jorge-Monjas, P., Tamayo, E., & Gómez-Sánchez, E. (2022). Influence of Renal Dysfunction on the Differential Behaviour of Procalcitonin for the Diagnosis of Postoperative Infection in Cardiac Surgery. Journal of Clinical Medicine, 11(24), 7274. https://doi.org/10.3390/jcm11247274





