Medication Adherence in Medicare-Enrolled Older Adults with Chronic Obstructive Pulmonary Disease before and during the COVID-19 Pandemic
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Sources
2.3. Statistical Analysis
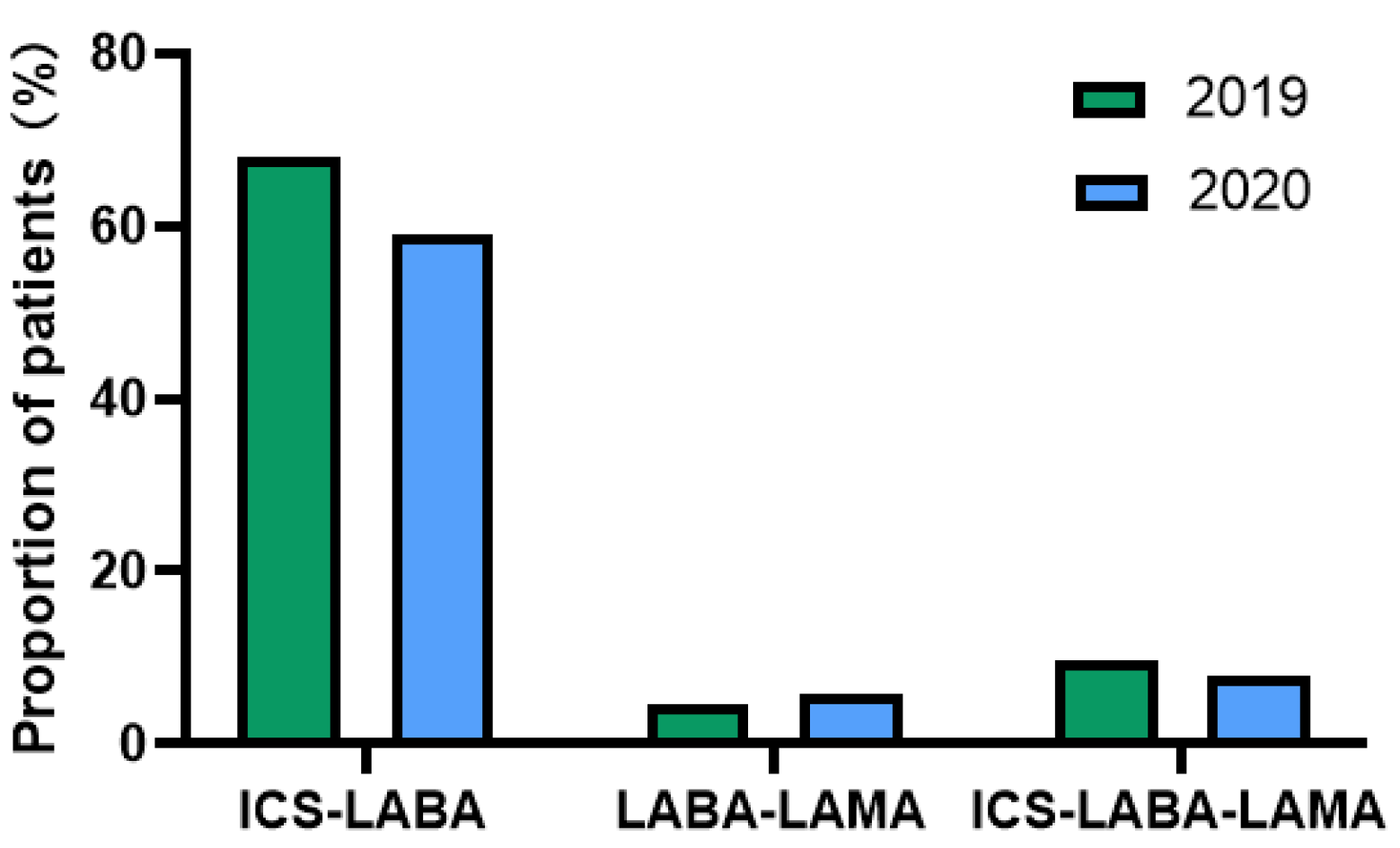
3. Results
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murphy, S.L.; Kochanek, K.D.; Xu, J.; Arias, E. Mortality in the United States, 2020. NCHS Data Brief. 2021, 427, 1–8. [Google Scholar]
- Iheanacho, I.; Zhang, S.; King, D.; Rizzo, M.; Ismaila, A.S. Economic Burden of Chronic Obstructive Pulmonary Disease (COPD): A Systematic Literature Review. Int. J. Chronic Obstr. Pulm. Dis. 2020, ume 15, 439–460. [Google Scholar] [CrossRef]
- Chen, W.; Thomas, J.; Sadatsafavi, M.; FitzGerald, J.M. Risk of cardiovascular comorbidity in patients with chronic obstructive pulmonary disease: A systematic review and meta-analysis. Lancet Respir. Med. 2015, 3, 631–639. [Google Scholar] [CrossRef]
- Fry, J.S.; Hamling, J.S.; Lee, P.N. Systematic review with meta-analysis of the epidemiological evidence relating FEV1decline to lung cancer risk. BMC Cancer 2012, 12, 498. [Google Scholar] [CrossRef] [PubMed]
- Higham, A.; Mathioudakis, A.; Vestbo, J.; Singh, D. COVID-19 and COPD: A narrative review of the basic science and clinical outcomes. Eur. Respir. Rev. 2020, 29, 200199. [Google Scholar] [CrossRef] [PubMed]
- Kühl, K.; Schürmann, W.; Rief, W. Mental disorders and quality of life in COPD patients and their spouses. Int. J. Chronic Obstr. Pulm. Dis. 2008, 3, 727–736. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis m, and Prevention of Chronic Obstructive Pulmonary Disease. 2022. Available online: https://goldcopd.org/2022-gold-reports-2/ (accessed on 20 July 2022).
- Humenberger, M.; Horner, A.; Labek, A.; Kaiser, B.; Frechinger, R.; Brock, C.; Lichtenberger, P.; Lamprecht, B. Adherence to inhaled therapy and its impact on chronic obstructive pulmonary disease (COPD). BMC Pulm. Med. 2018, 18, 163. [Google Scholar] [CrossRef]
- Mannino, D.M.; Yu, T.-C.; Zhou, H.; Higuchi, K. Effects of GOLD-Adherent Prescribing on COPD Symptom Burden, Exacerbations, and Health Care Utilization in a Real-World Setting. Chronic Obstr. Pulm. Dis. J. COPD Found. 2015, 2, 223–235. [Google Scholar] [CrossRef]
- Albrecht, J.S.; Park, Y.; Hur, P.; Huang, T.-Y.; Harris, I.; Netzer, G.; Lehmann, S.W.; Langenberg, P.; Khokhar, B.; Wei, Y.-J.; et al. Adherence to Maintenance Medications among Older Adults with Chronic Obstructive Pulmonary Disease. The Role of Depression. Ann. Am. Thorac. Soc. 2016, 13, 1497–1504. [Google Scholar] [CrossRef]
- Soones, T.N.; Lin, J.L.; Wolf, M.S.; O’Conor, R.; Martynenko, M.; Wisnivesky, J.P.; Federman, A.D. Pathways linking health literacy, health beliefs, and cognition to medication adherence in older adults with asthma. J. Allergy Clin. Immunol. 2016, 139, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.-C.; Chen, J.-H.; Kuo, H.-K.; We, C.-J.; Lu, I.-S.; Chiu, L.-S.; Wu, S.-C. Drug-related problems (DRPs) identified from geriatric medication safety review clinics. Arch. Gerontol. Geriatr. 2012, 54, 168–174. [Google Scholar] [CrossRef]
- Elbeddini, A.; Tayefehchamani, Y. Amid COVID-19 pandemic: Challenges with access to care for COPD patients. Res. Soc. Adm. Pharm. 2020, 17, 1934–1937. [Google Scholar] [CrossRef] [PubMed]
- Kahnert, K.; Lutter, J.I.; Welte, T.; Alter, P.; Behr, J.; Herth, F.; Kauczor, H.-U.; Söhler, S.; Pfeifer, M.; Watz, H.; et al. Impact of the COVID-19 pandemic on the behaviour and health status of patients with COPD: Results from the German COPD cohort COSYCONET. ERJ Open Res. 2021, 7, 00242–2021. [Google Scholar] [CrossRef]
- Halpin, D.M.G.; Criner, G.J.; Papi, A.; Singh, D.; Anzueto, A.; Martinez, F.J.; Agusti, A.A.; Vogelmeier, C.F. Global Initiative for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease. The 2020 GOLD Science Committee Report on COVID-19 and Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2021, 203, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Kaye, L.; Theye, B.; Smeenk, I.; Gondalia, R.; Barrett, M.A.; Stempel, D.A. Changes in medication adherence among patients with asthma and COPD during the COVID-19 pandemic. J. Allergy Clin. Immunol. Pr. 2020, 8, 2384–2385. [Google Scholar] [CrossRef] [PubMed]
- Yıldız, M.; Aksu, F.; Yıldız, N.; Aksu, K. Clinician’s perspective regarding medication adherence in patients with obstructive lung diseases and the impact of COVID-19. Rev. Assoc. Med. Bras. 2021, 67, 97–101. [Google Scholar] [CrossRef]
- Barrett, R.; Barrett, R. Asthma and COPD medicines prescription-claims: A time-series analysis of England’s national prescriptions during the COVID-19 pandemic (Jan 2019 to Oct 2020). Expert Rev. Respir. Med. 2021, 15, 1605–1612. [Google Scholar] [CrossRef]
- Zhang, H.-Q.; Lin, J.-Y.; Guo, Y.; Pang, S.; Jiang, R.; Cheng, Q.-J. Medication adherence among patients with chronic obstructive pulmonary disease treated in a primary general hospital during the COVID-19 pandemic. Ann. Transl. Med. 2020, 8, 1179. [Google Scholar] [CrossRef]
- Nishi, S.P.E.; Maslonka, M.; Zhang, W.; Kuo, Y.-F.; Sharma, G. Pattern and Adherence to Maintenance Medication Use in Medicare Beneficiaries with Chronic Obstructive Pulmonary Disease: 2008–2013. Chronic Obstr. Pulm. Dis. J. COPD Found. 2018, 5, 16–26. [Google Scholar] [CrossRef]
- Le, T.T.; Bjarnadóttir, M.; Qato, D.M.; Magder, L.; Zafari, Z.; Simoni-Wastila, L. Prediction of treatment nonadherence among older adults with chronic obstructive pulmonary disease using Medicare real-world data. J. Manag. Care Spec. Pharm. 2022, 28, 631–644. [Google Scholar]
- Bell, J.S.; Reynolds, L.; Freeman, C.; Jackson, J.K. Strategies to promote access to medications during the COVID-19 pandemic. Aust. J. Gen. Pr. 2020, 49, 530–532. [Google Scholar] [CrossRef]
- Ismail, H.; Marshall, V.D.; Patel, M.; Tariq, M.; Mohammad, R.A. The impact of the COVID-19 pandemic on medical conditions and medication adherence in people with chronic diseases. J. Am. Pharm. Assoc. 2022, 62, 834–839.e1. [Google Scholar] [CrossRef] [PubMed]
- Vordenberg, S.E.; Zikmund-Fisher, B.J. Older adults’ strategies for obtaining medication refills in hypothetical scenarios in the face of COVID-19 risk. J. Am. Pharm. Assoc. 2020, 60, 915–922.e4. [Google Scholar] [CrossRef] [PubMed]
- Khera, A.; Baum, S.; Gluckman, T.J.; Gulati, M.; Martin, S.S.; Michos, E.D.; Navar, A.M.; Taub, P.R.; Toth, P.P.; Virani, S.S.; et al. Continuity of care and outpatient management for patients with and at high risk for cardiovascular disease during the COVID-19 pandemic: A scientific statement from the American Society for Preventive Cardiology. Am. J. Prev. Cardiol. 2020, 1, 100009. [Google Scholar] [CrossRef] [PubMed]
- Rymer, J.A.; Fonseca, E.; Bhandary, D.D.; Kumar, D.; Khan, N.D.; Wang, T.Y. Difference in Medication Adherence Between Patients Prescribed a 30-Day Versus 90-Day Supply After Acute Myocardial Infarction. J. Am. Hear. Assoc. 2021, 10, e016215. [Google Scholar] [CrossRef]
- Ameli, N.; Jones, W.N. Evaluating Medication Day-Supply for Improving Adherence and Clinical Biomarkers of Hemoglobin A1c, Blood Pressure, and Low-Density Lipoprotein. J. Pharm. Pract. 2022. [Google Scholar] [CrossRef]
- Centers for Medicare & Medicaid Services. Information related to coronavirus disease 2019-COVID-19. 22 May 2020. Available online: https://www.cms.gov/files/document/covid-19-updated-guidance-ma-and-part-d-plan-sponsors-52220.pdf (accessed on 6 October 2022).
- López-Pintor, E.; Grau, J.; González, I.; Bernal-Soriano, M.J.; Quesada, J.; Lumbreras, B. Impact of patients’ perception of COPD and treatment on adherence and health-related quality of life in real-world: Study in 53 community pharmacies. Respir. Med. 2020, 176, 106280. [Google Scholar] [CrossRef]
- Kumamaru, H.; Lee, M.P.; Choudhry, N.K.; Dong, Y.-H.; Krumme, A.A.; Khan, N.; Brill, G.; Kohsaka, S.; Miyata, H.; Schneeweiss, S.; et al. Using Previous Medication Adherence to Predict Future Adherence. J. Manag. Care Spec. Pharm. 2018, 24, 1146–1155. [Google Scholar] [CrossRef]
- Fan, V.S.; Gylys-Colwell, I.; Locke, E.; Sumino, K.; Nguyen, H.Q.; Thomas, R.M.; Magzamen, S. Overuse of short-acting beta-agonist bronchodilators in COPD during periods of clinical stability. Respir. Med. 2016, 116, 100–106. [Google Scholar] [CrossRef]
- Gondalia, R.; Bender, B.G.; Theye, B.; Stempel, D.A. Higher short-acting beta-agonist use is associated with greater COPD burden. Respir. Med. 2019, 158, 110–113. [Google Scholar] [CrossRef]
- Haney, S.; Hancox, R.J. Recovery From Bronchoconstriction and Bronchodilator Tolerance. Clin. Rev. Allergy Immunol. 2006, 31, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Sarc, I.; Dolinar, A.L.; Morgan, T.; Sambt, J.; Ziherl, K.; Gavric, D.; Selb, J.; Rozman, A.; Bonca, P.D. Mortality, seasonal variation, and susceptibility to acute exacerbation of COPD in the pandemic year: A nationwide population study. Ther. Adv. Respir. Dis. 2022, 16. [Google Scholar] [CrossRef] [PubMed]
- Faria, N.; Costa, M.I.; Gomes, J.; Sucena, M. Reduction of Severe Exacerbations of COPD during COVID-19 Pandemic in Portugal: A Protective Role of Face Masks? COPD: J. Chronic Obstr. Pulm. Dis. 2021, 18, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Plácido, A.I.; Herdeiro, M.T.; Morgado, M.; Figueiras, A.; Roque, F. Drug-related Problems in Home-dwelling Older Adults: A Systematic Review. Clin. Ther. 2020, 42, 559–572.e14. [Google Scholar] [CrossRef]
- Somers, A.; Petrovic, M. Major drug related problems leading to hospital admission in the elderly. J. Pharm. Belg. 2014, 2, 34–38. [Google Scholar]
- Liu, M.; Liu, J.; Geng, Z.; Bai, S. Evaluation of outcomes of medication therapy management (MTM) services for patients with chronic obstructive pulmonary disease (COPD). Pak. J. Med Sci. 2021, 37, 1832–1836. [Google Scholar] [CrossRef]


| Characteristic | Overall Cohort | With High Adherence | With Nonadherence | p Value | B Value | Adjusted Odds Ratio |
|---|---|---|---|---|---|---|
| N = 1533 | N = 1141 | N = 392 | (95% confidence interval) | |||
| N (%) | N (%) | N (%) | ||||
| Age, year | 0.36 | |||||
| 65–74 (reference) | 644 (42.0) | 484 (42.4) | 160 (40.8) | |||
| 75–84 | 656 (42.8) | 496 (43.5) | 160 (40.8) | 0.81 | 0.03 | 1.04 (0.78–1.37) |
| ≥85 | 233 (15.2) | 161 (14.1) | 72 (18.4) | 0.23 | −0.23 | 0.80 (0.55–1.15) |
| Sex | ||||||
| Male (reference) | 629 (41.0) | 466 (40.8) | 163 (41.6) | |||
| Female | 904 (59.0) | 675 (59.2) | 229 (58.4) | 0.51 | −0.09 | 0.509 (0.71–1.19) |
| Number of medications | 0.31 | |||||
| 8–10 (reference) | 456 (29.7) | 315 (27.6) | 141 (36.0) | |||
| 11–13 | 400 (26.1) | 293 (25.7) | 107 (27.3) | 0.82 | 0.04 | 1.04 (0.75–1.44) |
| 14–16 | 299 (19.5) | 228 (20.0) | 71 (18.1) | 0.51 | 0.13 | 1.14 (0.78–1.65) |
| 17–19 | 200 (13.0) | 159 (13.9) | 41 (10.5) | 0.08 | 0.39 | 1.48 (0.95–2.31) |
| ≥20 | 178 (11.6) | 146 (12.8) | 32 (8.2) | 0.11 | 0.42 | 1.52 (0.91–2.53) |
| Number of medication-related problems | 0.007 | |||||
| 0 | 179 (11.7) | 144 (12.6) | 35 (8.9) | 0.01 | −0.64 | 1.90 (1.16–3.10) |
| 1 | 288 (18.8) | 232 (20.3) | 56 (14.3) | <0.001 | −0.52 | 2.10 (1.38–3.18) |
| 2 | 294 (19.2) | 216 (18.9) | 78 (19.9) | 0.11 | −0.49 | 1.37 (0.93–2.03) |
| 3 | 220 (14.4) | 161 (14.1) | 59 (15.1) | 0.47 | −0.32 | 1.17 (0.77–1.77) |
| 4 | 176 (11.5) | 122 (10.7) | 54 (13.8) | 0.57 | 0.10 | 1.13 (0.74–1.75) |
| ≥5 (reference) | 376 (24.5) | 266 (23.3) | 110 (28.1) | |||
| Number of prescribers | 0.66 | |||||
| 1–5 (reference) | 743 (48.5) | 536 (47.0) | 207 (52.8) | |||
| 6–10 | 635 (41.4) | 479 (42.0) | 156 (39.8) | 0.55 | 0.09 | 1.089 (0.82–1.44) |
| 11–15 | 131 (8.5) | 106 (9.3) | 25 (6.4) | 0.29 | 0.29 | 1.329 (0.78–2.26) |
| ≥16 | 24 (1.6) | 20 (1.8) | 4 (1.0) | 0.44 | 0.47 | 1.592 (0.49–5.21) |
| Number of pharmacies | 0.92 | |||||
| 1 (reference) | 450 (29.4) | 336 (29.4) | 114 (29.1) | |||
| 2 | 449 (29.3) | 336 (29.4) | 113 (28.8) | 0.95 | −0.01 | 0.99 (0.71–1.37) |
| 3 | 322 (21.0) | 244 (21.4) | 78 (19.9) | 0.63 | 0.09 | 1.10 (0.76–1.58) |
| ≥4 | 312 (20.4) | 225 (19.7) | 87 (22.2) | 0.82 | −0.04 | 0.96 (0.66–1.39) |
| Depression | ||||||
| Yes | 259 (16.9) | 197 (17.3) | 62 (15.8) | 0.41 | 0.15 | |
| No (reference) | 1274 (83.1) | 944 (82.7) | 330 (84.2) | |||
| Number of controlled medication classes | 0.001 | |||||
| 1 (reference) | 339 (22.1) | 228 (20.0) | 111 (28.3) | |||
| 2 | 598 (39.0) | 380 (33.3) | 218 (55.6) | 0.15 | −0.22 | 0.81 (0.60–1.09) |
| 3 | 480 (31.3) | 423 (37.1) | 57 (14.5) | <0.001 | 1.06 | 2.89 (1.99–4.21) |
| ≥4 | 116 (7.6) | 110 (9.6) | 6 (1.5) | <0.001 | 1.86 | 6.44 (2.68–15.46) |
| Number of oral corticosteroid fills | 0.53 | |||||
| 0 (reference) | 1040 (67.8) | 767 (67.2) | 273 (69.6) | |||
| 1 | 264 (17.2) | 194 (17.0) | 71 (18.1) | 0.35 | −0.16 | 0.85 (0.61–1.20) |
| 2 | 98 (6.4) | 72 (6.3) | 26 (6.6) | 0.35 | −0.25 | 0.78 (0.46–1.31) |
| ≥3 | 131 (8.5) | 108 (9.5) | 22 (5.6) | 0.54 | 0.17 | 1.18 (0.69–2.03) |
| Number of albuterol inhalers | ||||||
| ≤2 (reference) | 647 (42.2) | 414 (36.3) | 233 (59.4) | |||
| ≥3 | 886 (57.8) | 727 (63.7) | 159 (40.6) | <0.001 | 0.81 | 2.25 (1.74–2.91) |
| 90-day supply of inhalers | ||||||
| No (reference) | 1088 (71.0) | 759 (66.5) | 329 (83.9) | |||
| Yes | 445 (29.0) | 382 (33.5) | 63 (16.1) | <0.001 | 0.95 | 2.57 (1.88–3.53) |
| Characteristic | Overall Cohort | With High Adherence | With Non-Adherence | p Value | B Value | Adjusted Odds Ratio |
|---|---|---|---|---|---|---|
| N = 1533 | N = 891 (58.1) | N = 642 (42.9) | (95% confidence interval) | |||
| N (%) | N (%) | N (%) | ||||
| Age, year | 0.88 | |||||
| 65–74 (reference) | 644 (42.0) | 371 (41.6) | 273 (42.5) | |||
| 75–84 | 656 (42.8) | 401 (45.0) | 255 (39.7) | 0.77 | 0.05 | 1.05 (0.76–1.45) |
| ≥85 | 233 (15.2) | 119 (13.4) | 114 (17.8) | 0.79 | −0.1 | 0.94 (0.61–1.46) |
| Sex | ||||||
| Male (reference) | 629 (41.0) | 367 (41.2) | 262 (40.8) | |||
| Female | 904 (59.0) | 524 (58.8) | 380 (59.2) | 0.25 | 0.18 | 1.19 (0.88–1.62) |
| Number of medications | 0.47 | |||||
| 8–10 (reference) | 456 (29.7) | 250 (28.1) | 206 (32.1) | |||
| 11–13 | 400 (26.1) | 235 (26.4) | 165 (25.7) | 0.56 | 0.12 | 1.12 (0.76–1.66) |
| 14–16 | 299 (19.5) | 181 (20.3) | 118 (18.4) | 0.83 | −0.05 | 0.95 (0.62–1.47) |
| 17–19 | 200 (13.0) | 113 (12.7) | 87 (13.6) | 0.20 | −0.32 | 0.72 (0.44–1.18) |
| ≥20 | 178 (11.6) | 112 (12.6) | 66 (10.3) | 0.62 | 0.14 | 1.15 (0.66–2.01) |
| Number of medication-related problems | 0.03 | |||||
| 0 | 179 (11.7) | 117 (13.1) | 62 (9.7) | 0.50 | −0.20 | 1.22 (0.68–2.21) |
| 1 | 288 (18.8) | 181 (20.3) | 107 (16.7) | 0.35 | 0.21 | 0.80 (0.50–1.28) |
| 2 | 294 (19.2) | 154 (17.3) | 140 (21.8) | 0.08 | −0.52 | 0.66 (0.41–1.05) |
| 3 | 220 (14.4) | 123 (13.8) | 97 (15.1) | 0.19 | −0.62 | 0.73 (0.45–1.18) |
| 4 | 176 (11.5) | 104 (11.7) | 72 (11.2) | 0.14 | −0.42 | 1.51 (0.87–2.63) |
| ≥5 (reference) | 376 (24.5) | 212 (23.8) | 164 (25.5) | |||
| Number of prescribers | 0.13 | |||||
| 1–5 (reference) | 874 (57.0) | 475 (53.3) | 399 (62.1) | |||
| 6–10 | 544 (35.5) | 335 (37.6) | 209 (32.6) | 0.80 | −0.04 | 0.96 (0.70–1.32) |
| 11–15 | 98 (6.4) | 72 (8.1) | 26 (4.0) | 0.61 | 0.17 | 1.18 (0.63–2.22) |
| ≥16 | 17 (1.1) | 9 (1.0) | 8 (1.2) | 0.02 | −1.35 | 0.26 (0.08–0.83) |
| Number of pharmacies | 0.08 | |||||
| 1 (reference) | 450 (29.4) | 254 (28.5) | 196 (30.5) | |||
| 2 | 449 (29.3) | 271 (30.4) | 178 (27.7) | 0.05 | 0.38 | 1.46 (1.00–2.14) |
| 3 | 322 (21.0) | 180 (20.2) | 142 (22.1) | 0.87 | 0.03 | 1.03 (0.69–1.55) |
| ≥4 | 312 (20.4) | 186 (20.9) | 126 (19.6) | 0.05 | 0.44 | 1.56 (1.01–2.41) |
| Depression | ||||||
| Yes | 259 (16.9) | 152 (17.1) | 107 (16.7) | 0.38 | 0.19 | 1.21 (0.79–1.85) |
| No (reference) | 1274 (83.1) | 739 (82.9) | 535 (83.3) | |||
| Number of controlled medication classes | <0.001 | |||||
| 0–1 (reference) | 595 (38.8) | 175 (19.6) | 420 (65.4) | |||
| 2 | 432 (28.2) | 272 (30.5) | 160 (24.9) | 0.18 | −0.25 | 0.78 (0.54 -1.12) |
| 3 | 397 (25.9) | 340 (38.2) | 57 (8.9) | <0.001 | 0.82 | 2.28 (1.50–3.47) |
| ≥4 | 109 (7.1) | 104 (11.7) | 5 (0.8) | <0.001 | 1.88 | 6.55 (2.47–17.35) |
| Number of oral corticosteroids fills | 0.57 | |||||
| 0 (reference) | 1139 (74.3) | 641 (71.9) | 498 (77.6) | |||
| 1 | 187 (12.2) | 115 (12.9) | 72 (11.2) | 0.21 | −0.29 | 0.75 (0.48–1.18) |
| 2 | 85 (5.5) | 55 (6.2) | 30 (4.7) | 0.60 | 0.20 | 1.22 (0.59–2.51) |
| ≥3 | 122 (8.0) | 80 (9.0) | 42 (6.5) | 0.91 | −0.03 | 0.97 (0.56–1.68) |
| Number of albuterol inhalers | ||||||
| <3 (reference) | 729 (47.6) | 324 (36.4) | 405 (63.1) | |||
| ≥3 | 804 (52.4) | 567 (63.6) | 237 (36.9) | 0.01 | 0.39 | 1.48 (1.09–1.99) |
| 90-day supply of inhalers | ||||||
| No (reference) | 1025 (66.9) | 509 (57.1) | 516 (80.4) | |||
| Yes | 508 (33.1) | 382 (42.9) | 126 (19.6) | <0.001 | 0.89 | 2.44 (1.73–3.46) |
| High adherence to any inhalers in 2019 | ||||||
| No (reference) | 392 (25.6) | 155 (17.4) | 237 (36.9) | |||
| Yes | 1141 (74.4) | 736 (82.6) | 405 (63.1) | <0.001 | 0.81 | 2.24 (1.63–3.08) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Almodóvar, A.S.; Nahata, M.C. Medication Adherence in Medicare-Enrolled Older Adults with Chronic Obstructive Pulmonary Disease before and during the COVID-19 Pandemic. J. Clin. Med. 2022, 11, 6985. https://doi.org/10.3390/jcm11236985
Liu L, Almodóvar AS, Nahata MC. Medication Adherence in Medicare-Enrolled Older Adults with Chronic Obstructive Pulmonary Disease before and during the COVID-19 Pandemic. Journal of Clinical Medicine. 2022; 11(23):6985. https://doi.org/10.3390/jcm11236985
Chicago/Turabian StyleLiu, Ligang, Armando Silva Almodóvar, and Milap C. Nahata. 2022. "Medication Adherence in Medicare-Enrolled Older Adults with Chronic Obstructive Pulmonary Disease before and during the COVID-19 Pandemic" Journal of Clinical Medicine 11, no. 23: 6985. https://doi.org/10.3390/jcm11236985
APA StyleLiu, L., Almodóvar, A. S., & Nahata, M. C. (2022). Medication Adherence in Medicare-Enrolled Older Adults with Chronic Obstructive Pulmonary Disease before and during the COVID-19 Pandemic. Journal of Clinical Medicine, 11(23), 6985. https://doi.org/10.3390/jcm11236985








