Elevated Serum CCL23 Levels at Admission Predict Delayed Cerebral Ischemia and Functional Outcome after Aneurysmal Subarachnoid Hemorrhage
Abstract
1. Introduction
2. Patients and Methods
2.1. Participants and Study Design
2.2. Data Collection
2.3. Blood Sample Collection and Serum CCL23 Assay
2.4. Statistical Analysis
3. Results
3.1. Study Population
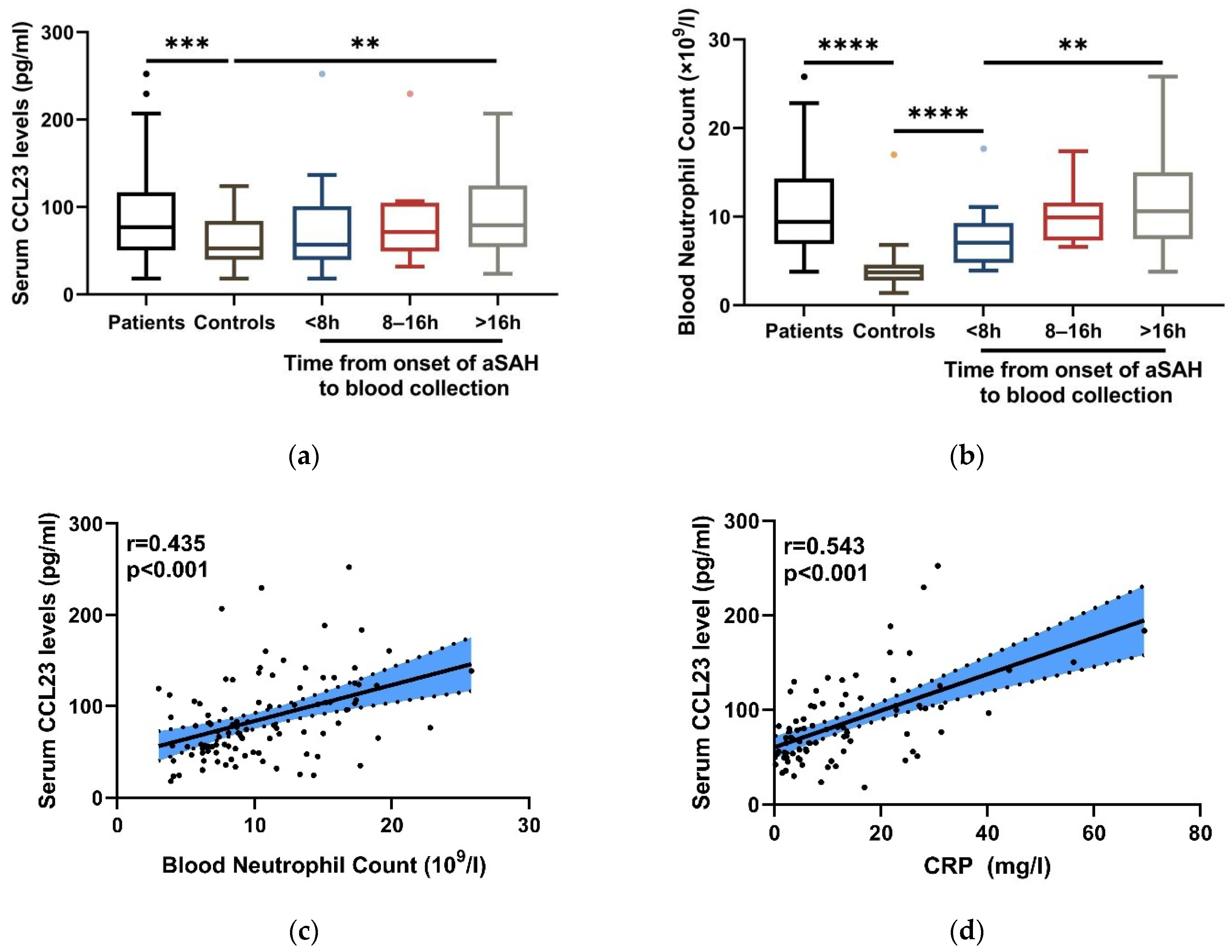
3.2. Changes in Serum CCL23 Levels after aSAH
3.3. Association of Serum CCL23 Levels with Blood Neutrophils in Inflammation after aSAH
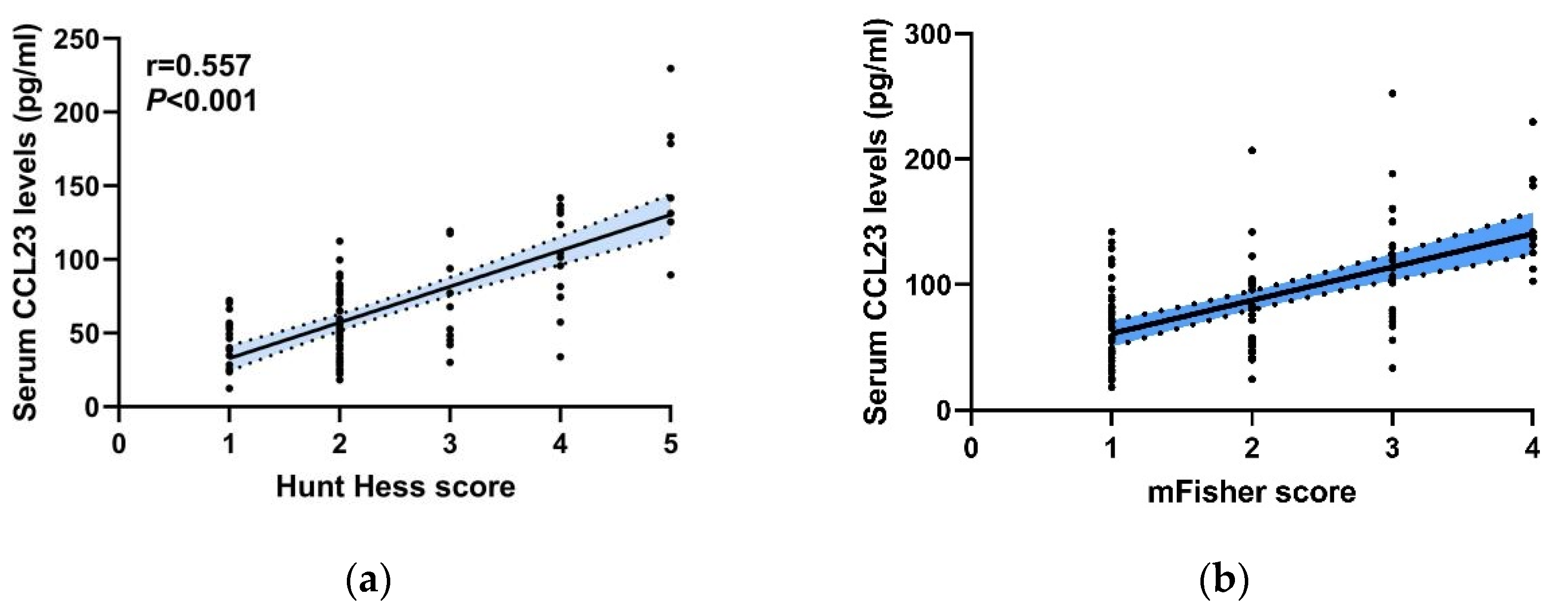
3.4. Correlation between Serum CCL23 Levels with the Initial Clinical Severity after aSAH
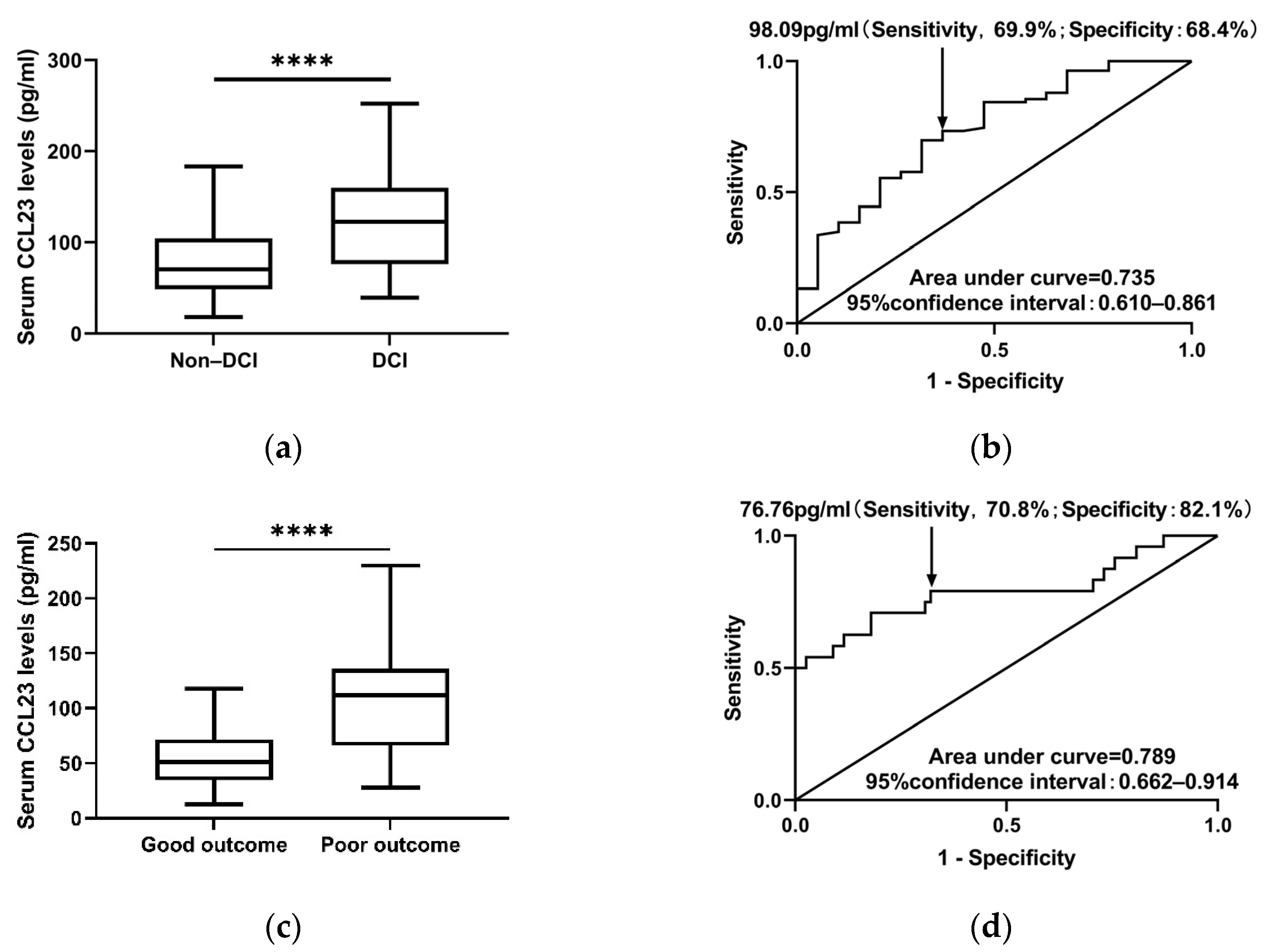
3.5. Serum CCL23 Levels Predicts DCI after aSAH
3.6. Serum CCL23 Is an Independent Predictor of Outcome in aSAH
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neifert, S.N.; Chapman, E.K.; Martini, M.L.; Shuman, W.H.; Schupper, A.J.; Oermann, E.K.; Mocco, J.; Macdonald, R.L. Aneurysmal Subarachnoid Hemorrhage: The Last Decade. Transl. Stroke Res. 2021, 12, 428–446. [Google Scholar] [CrossRef]
- Mayer, S.A.; Kreiter, K.T.; Copeland, D.; Bernardini, G.L.; Bates, J.E.; Peery, S.; Claassen, J.; Du, Y.E.; Connolly, E.S. Global and domain-specific cognitive impairment and outcome after subarachnoid hemorrhage. Neurology 2002, 59, 1750–1758. [Google Scholar] [CrossRef]
- Rouanet, C.; Silva, G.S. Aneurysmal subarachnoid hemorrhage: Current concepts and updates. Arq. Neuro-Psiquiatria 2019, 77, 806–814. [Google Scholar] [CrossRef]
- Ingall, T.; Asplund, K.; Mähönen, M.; Bonita, R. A Multinational Comparison of Subarachnoid Hemorrhage Epidemiology in the WHO MONICA Stroke Study. Stroke 2000, 31, 1054–1061. [Google Scholar] [CrossRef]
- Song, J.-P.; Ni, W.; Gu, Y.-X.; Zhu, W.; Chen, L.; Xu, B.; Leng, B.; Tian, Y.-L.; Mao, Y. Epidemiological Features of Nontraumatic Spontaneous Subarachnoid Hemorrhage in China: A Nationwide Hospital-based Multicenter Study. Chin. Med. J. 2017, 130, 776–781. [Google Scholar] [CrossRef]
- Lucke-Wold, B.P.; Logsdon, A.F.; Manoranjan, B.; Turner, R.C.; McConnell, E.; Vates, G.E.; Huber, J.D.; Rosen, C.L.; Simard, J.M. Aneurysmal Subarachnoid Hemorrhage and Neuroinflammation: A Comprehensive Review. Int. J. Mol. Sci. 2016, 17, 497. [Google Scholar] [CrossRef]
- Mocco, J.; Ransom, E.R.; Komotar, R.J.; Schmidt, J.M.; Sciacca, R.R.; Mayer, S.A.; Connolly, E.S. Preoperative Prediction of Long-term Outcome in Poor-grade Aneurysmal Subarachnoid Hemorrhage. Neurosurgery 2006, 59, 529–538. [Google Scholar] [CrossRef]
- Vergouwen, M.D.; Vermeulen, M.; van Gijn, J.; Rinkel, G.J.; Wijdicks, E.F.; Muizelaar, J.P.; Mendelow, A.D.; Juvela, S.; Yonas, H.; Terbrugge, K.G.; et al. Definition of Delayed Cerebral Ischemia After Aneurysmal Subarachnoid Hemorrhage as an Outcome Event in Clinical Trials and Observational Studies: Proposal of a multidisciplinary research group. Stroke 2010, 41, 2391–2395. [Google Scholar] [CrossRef] [PubMed]
- De Rooij, N.K.; Rinkel, G.J.; Dankbaar, J.W.; Frijns, C.J. Delayed Cerebral Ischemia After Subarachnoid Hemorrhage: A systematic review of clinical, laboratory, and radiological predictors. Stroke 2013, 44, 43–54. [Google Scholar] [CrossRef]
- Osgood, M.L. Aneurysmal Subarachnoid Hemorrhage: Review of the Pathophysiology and Management Strategies. Curr. Neurol. Neurosci. Rep. 2021, 21, 50. [Google Scholar] [CrossRef]
- Ahn, S.-H.; Savarraj, J.P.J.; Parsha, K.; Hergenroeder, G.W.; Chang, T.R.; Kim, D.H.; Kitagawa, R.S.; Blackburn, S.L.; Choi, H.A. Inflammation in delayed ischemia and functional outcomes after subarachnoid hemorrhage. J. Neuroinflamm. 2019, 16, 213. [Google Scholar] [CrossRef]
- Fujii, M.; Yan, J.; Rolland, W.B.; Soejima, Y.; Caner, B.; Zhang, J.H. Early Brain Injury, an Evolving Frontier in Subarachnoid Hemorrhage Research. Transl. Stroke Res. 2013, 4, 432–446. [Google Scholar] [CrossRef]
- Zhang, Z.; Fang, Y.; Lenahan, C.; Chen, S. The role of immune inflammation in aneurysmal subarachnoid hemorrhage. Exp. Neurol. 2021, 336, 113535. [Google Scholar] [CrossRef]
- Wu, F.; Liu, Z.; Li, G.; Zhou, L.; Huang, K.; Wu, Z.; Zhan, R.; Shen, J. Inflammation and Oxidative Stress: Potential Targets for Improving Prognosis After Subarachnoid Hemorrhage. Front. Cell. Neurosci. 2021, 15, 739506. [Google Scholar] [CrossRef]
- Schall, T.J.; Bacon, K.B. Chemokines, leukocyte trafficking, and inflammation. Curr. Opin. Immunol. 1994, 6, 865–873. [Google Scholar] [CrossRef]
- Yao, Y.; Tsirka, S.E. Chemokines and Their Receptors in Intracerebral Hemorrhage. Transl. Stroke Res. 2012, 3, 70–79. [Google Scholar] [CrossRef]
- Arruda-Silva, F.; Bianchetto-Aguilera, F.; Gasperini, S.; Polletti, S.; Cosentino, E.; Tamassia, N.; Cassatella, M.A. Human Neutrophils Produce CCL23 in Response to Various TLR-Agonists and TNFα. Front. Cell. Infect. Microbiol. 2017, 7, 176. [Google Scholar] [CrossRef]
- Poposki, J.A.; Keswani, A.; Kim, J.K.; Klingler, A.I.; Suh, L.A.; Norton, J.; Carter, R.G.; Peters, A.T.; Hulse, K.E.; Grammer, L.C.; et al. Tissue proteases convert CCL23 into potent monocyte chemoattractants in patients with chronic rhinosinusitis. J. Allergy Clin. Immunol. 2016, 137, 1274–1277. [Google Scholar] [CrossRef]
- Poposki, J.A.; Uzzaman, A.; Nagarkar, D.R.; Chustz, R.T.; Peters, A.T.; Suh, L.A.; Carter, R.; Norton, J.; Harris, K.E.; Grammer, L.C.; et al. Increased expression of the chemokine CCL23 in eosinophilic chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2011, 128, 73–81. [Google Scholar] [CrossRef]
- Van Damme, J.; Proost, P.; Lenaerts, J.P.; Opdenakker, G. Structural and functional identification of two human, tumor-derived monocyte chemotactic proteins (MCP-2 and MCP-3) belonging to the chemokine family. J. Exp. Med. 1992, 176, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Kamat, K.; Krishnan, V.; Dorigo, O. Macrophage-derived CCL23 upregulates expression of T-cell exhaustion markers in ovarian cancer. Br. J. Cancer 2022, 127, 1026–1033. [Google Scholar] [CrossRef]
- Zweemer, A.; Toraskar, J.; Heitman, L.; Ijzerman, A.P. Bias in chemokine receptor signalling. Trends Immunol. 2014, 35, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Berahovich, R.D.; Miao, Z.; Wang, Y.; Premack, B.; Howard, M.C.; Schall, T.J. Proteolytic Activation of Alternative CCR1 Ligands in Inflammation. J. Immunol. 2005, 174, 7341–7351. [Google Scholar] [CrossRef] [PubMed]
- Yanaba, K.; Yoshizaki, A.; Muroi, E.; Ogawa, F.; Asano, Y.; Kadono, T.; Sato, S. Serum CCL23 levels are increased in patients with systemic sclerosis. Arch. Dermatol. Res. 2011, 303, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, K.; Myśliwiec, M.; Pawlak, D. Effect of diabetes and oxidative stress on plasma CCL23 levels in patients with severe chronic kidney disease. Pol. Arch. Intern. Med. 2014, 124, 459–466. [Google Scholar] [CrossRef]
- Kim, C.-S.; Kang, J.-H.; Cho, H.-R.; Blankenship, T.N.; Erickson, K.L.; Kawada, T.; Yu, R. Potential involvement of CCL23 in atherosclerotic lesion formation/progression by the enhancement of chemotaxis, adhesion molecule expression, and MMP-2 release from monocytes. Agents Actions 2011, 60, 889–895. [Google Scholar] [CrossRef]
- Maglinger, B.; Frank, J.A.; McLouth, C.J.; Trout, A.L.; Roberts, J.M.; Grupke, S.; Turchan-Cholewo, J.; Stowe, A.M.; Fraser, J.F.; Pennypacker, K.R. Proteomic changes in intracranial blood during human ischemic stroke. J. NeuroInterv. Surg. 2021, 13, 395–399. [Google Scholar] [CrossRef]
- Faura, J.; Bustamante, A.; Penalba, A.; Giralt, D.; Simats, A.; Martínez-Sáez, E.; Alcolea, D.; Fortea, J.; Lleó, A.; Teunissen, C.E.; et al. CCL23: A Chemokine Associated with Progression from Mild Cognitive Impairment to Alzheimer’s Disease. J. Alzheimer’s Dis. 2020, 73, 1585–1595. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Y.; Zhao, Z.; Li, P.; Ma, C.; Zhu, B. Diagnostic Value of Serum MIF and CCL23 in the Patients with Acute Cerebral Infarction. Clin. Lab. 2020, 66. [Google Scholar] [CrossRef]
- Francoeur, C.L.; Mayer, S.A. Management of delayed cerebral ischemia after subarachnoid hemorrhage. Crit. Care 2016, 20, 277. [Google Scholar] [CrossRef]
- Badjatia, N.; Monahan, A.; Carpenter, A.; Zimmerman, J.; Schmidt, J.M.; Claassen, J.; Connolly, E.S.; Mayer, S.A.; Karmally, W.; Seres, D. Inflammation, negative nitrogen balance, and outcome after aneurysmal subarachnoid hemorrhage. Neurology 2015, 84, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, Y.S.; Ko, J. CKβ8/CCL23 induces cell migration via the Gi/Go protein/PLC/PKCδ/NF-κB and is involved in inflammatory responses. Life Sci. 2010, 86, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Li, F.; Zhang, C.; Na Li, N.; Huang, H.; Shao, Z.; Zhang, M.; Zhan, X.; He, Y.; Ju, Z.; et al. Eosinophil-derived chemokine (hCCL15/23, mCCL6) interacts with CCR1 to promote eosinophilic airway inflammation. Signal Transduct. Target. Ther. 2021, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Simats, A.; García-Berrocoso, T.; Penalba, A.; Giralt, D.; Llovera, G.; Jiang, Y.; Ramiro, L.; Bustamante, A.; Martinez-Saez, E.; Canals, F.; et al. CCL23: A newCCchemokine involved in human brain damage. J. Intern. Med. 2018, 283, 461–475. [Google Scholar] [CrossRef]
- Zhan, Y.; Krafft, P.R.; Lekic, T.; Ma, Q.; Souvenir, R.; Zhang, J.H.; Tang, J. Imatinib preserves blood-brain barrier integrity following experimental subarachnoid hemorrhage in rats. J. Neurosci. Res. 2015, 93, 94–103. [Google Scholar] [CrossRef]
- Sokol, C.L.; Luster, A.D. The Chemokine System in Innate Immunity. Cold Spring Harb. Perspect. Biol. 2015, 7, a016303. [Google Scholar] [CrossRef]
- van Gijn, J.; Kerr, R.S.; Rinkel, G.J. Subarachnoid haemorrhage. Lancet 2007, 369, 306–318. [Google Scholar] [CrossRef]
- Macdonald, R.L.; Schweizer, T.A. Spontaneous subarachnoid haemorrhage. Lancet 2017, 389, 655–666. [Google Scholar] [CrossRef]
- Hwang, J.; Son, K.-N.; Kim, C.W.; Ko, J.; Na, D.S.; Kwon, B.S.; Gho, Y.S.; Kim, J. Human CC chemokine CCL23, a ligand for CCR1, induces endothelial cell migration and promotes angiogenesis. Cytokine 2005, 30, 254–263. [Google Scholar] [CrossRef]
- Patel, V.P.; Kreider, B.L.; Li, Y.; Li, H.; Leung, K.; Salcedo, T.; Nardelli, B.; Pippalla, V.; Gentz, S.; Thotakura, R.; et al. Molecular and Functional Characterization of Two Novel Human C-C Chemokines as Inhibitors of Two Distinct Classes of Myeloid Progenitors. J. Exp. Med. 1997, 185, 1163–1172. [Google Scholar] [CrossRef]
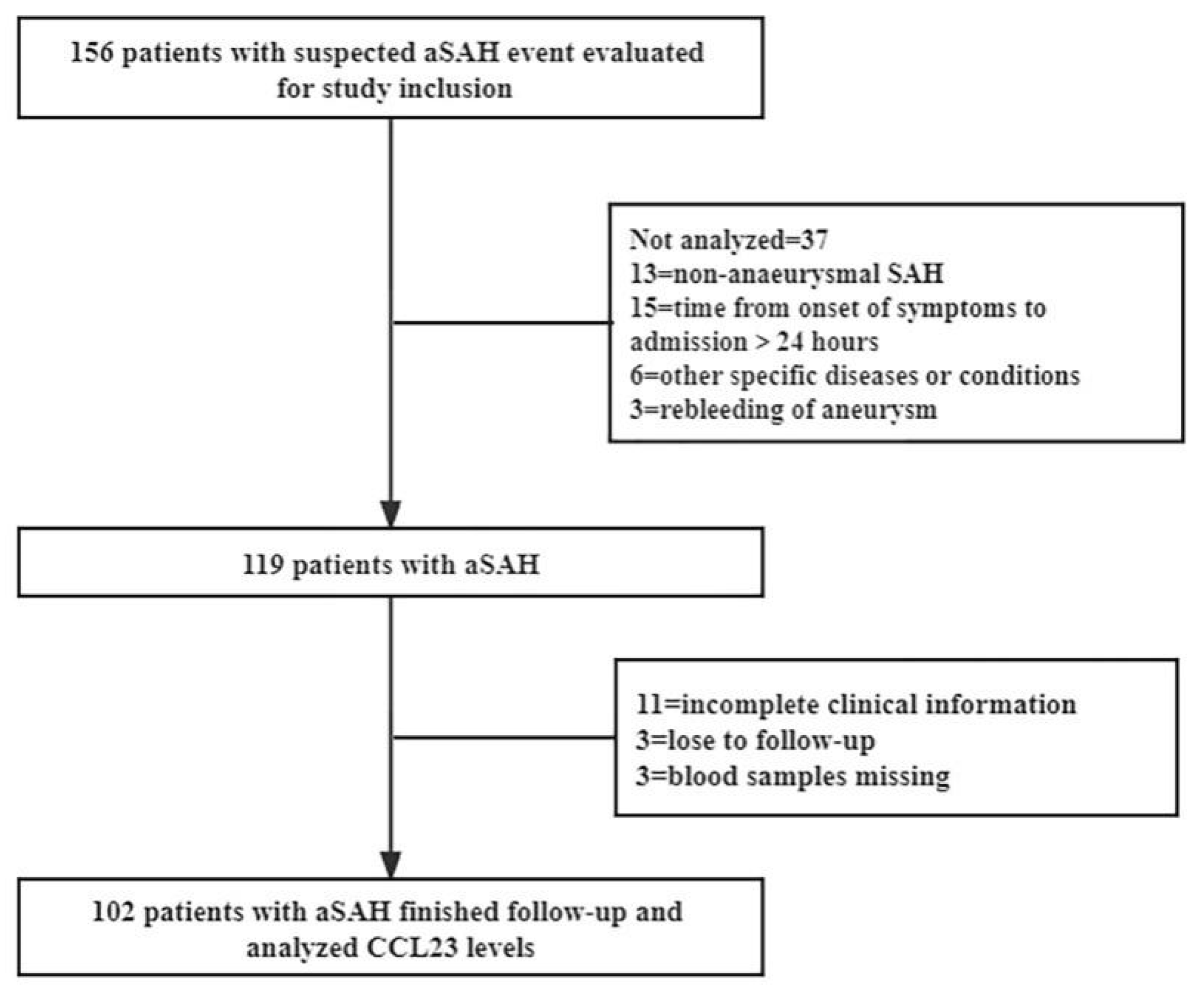
| Characteristics | Patients with aSAH (n = 102) | Controls (n = 61) | p Value |
|---|---|---|---|
| Age, years | 55 (46.5–65) | 59 (51–66) | 0.135 |
| Gender, female | 56 (54.9%) | 30 (49.2%) | 0.616 |
| BMI, kg/m2 | 23.4 (21.6–26.1) | 22.3 (20.9–24.7) | 0.244 |
| Prior medical history | |||
| Smoker | 28 (27.5%) | 10 (9.8%) | 0.107 |
| Alcohol consumption | 21 (20.6%) | 11 (18.0%) | 0.693 |
| Hypertension | 47 (46.1%) | 16 (16.4%) | 0.012 |
| Diabetes mellitus | 9 (8.8%) | 4 (6.6%) | 0.608 |
| Cardiovascular comorbidities | 7 (6.9%) | 2 (3.3%) | 0.335 |
| Admission status | |||
| Hunt–Hess grade | - | - | |
| 1 | 22 (21.6%) | ||
| 2 | 31 (30.4%) | ||
| 3 | 25 (24.5%) | ||
| 4 | 14 (13.7%) | ||
| 5 | 10 (9.8%) | ||
| Modified Fisher grade | - | - | |
| 1 | 43 (42.2%) | ||
| 2 | 27 (26.5%) | ||
| 3 | 22 (21.6%) | ||
| 4 | 10 (9.8%) | ||
| In-hospital data | |||
| Aneurysmal position, anterior circulation | 89 (87.3%) | - | - |
| Aneurysmal shape, cystic | 87 (85.3%) | - | - |
| Aneurysm size, mm | 5.0 (3.7–8.0) | - | - |
| Aneurysm size > 10 mm | 18 (17.6%) | - | - |
| Surgery, endovascular coiling | 50 (49.0%) | - | - |
| Acute hydrocephalus | 26 (25.5%) | - | - |
| DCI | 19 (18.6%) | - | - |
| External ventricular drain | 21 (20.6%) | - | - |
| Laboratory findings at admission | |||
| CRP, mg/L | 8.6 (2.3–21.9) | 4.9 (4.3–5.7) | <0.001 |
| Neutrophil count, ×109/L | 8.1 (5.6–10.5) | 9.4 (6.9–14.3) | <0.001 |
| CCL23, pg/mL | 76.8 (50.4–117.0) | 52.5 (39.7–84.1) | <0.001 |
| Characteristics | Patients without DCI (n = 83) | Patients with DCI (n = 19) | OR | p-Value |
|---|---|---|---|---|
| Age, years | 55 (49–64) | 55 (43–71) | 1.001 (0.960–1.042) | 0.981 |
| Gender (female) | 38 (45.8%) | 8 (42.1%) | 0.861 (0.341–2.360) | 0.771 |
| BMI, kg/m2 | 23.5 (21.7–26.1) | 23.3 (19.9–25.6) | 1.097 (0.921–1.307) | 0.299 |
| Current smokers | 23 (27.7%) | 5 (26.3%) | 0.872 (0.673–1.130) | 0.300 |
| Alcohol consumption | 18 (21.7%) | 3 (15.8%) | 0.677 (0.177–2.583) | 0.568 |
| Hypertension | 39 (47.0%) | 8 (42.1%) | 0.821 (0.300–2.247) | 0.700 |
| Diabetes mellitus | 8 (9.6%) | 1 (5.3%) | 0.521 (0.061–4.434) | 0.551 |
| Cardiovascular comorbidities | 4 (4.8%) | 3 (15.8%) | 3.703 (0.755–18.168) | 0.107 |
| Hunt–Hess grade | 2 (2–3) | 4 (2–4) | 1.694 (1.120–2.562) | 0.012 |
| Modified Fisher grade | 2 (1–3) | 2 (2–3) | 1.452 (0.900–2.342) | 0.126 |
| Aneurysmal position (anterior circulation) | 74 (89.2%) | 15 (78.9%) | 2.193 (0.596–8.061) | 0.237 |
| Aneurysmal shape (cystic) | 74 (89.2%) | 13 (68.4%) | 0.264 (0.080–0.866) | 0.028 |
| Aneurysm size, mm | 5.0 (3.6–8.0) | 6.5 (4.5–9.0) | 1.034 (0.927–1.152) | 0.551 |
| Aneurysm size > 10 mm | 15 (18.1%) | 3 (15.8%) | 0.850 (0.219–3.292) | 0.814 |
| Endovascular coiling | 40 (48.2%) | 10 (52.6%) | 1.194 (0.440–3.241) | 0.727 |
| Acute hydrocephalus | 23 (27.7%) | 8 (42.1%) | 1.897 (0.677–5.313) | 0.223 |
| External ventricular drain | 6 (7.2%) | 4 (21.1%) | 3.422 (0.860–13.614) | 0.081 |
| CRP, mg/L | 7.8 (3.2–14.1) | 22.6 (7.3–28.7) | 1.048 (1.006–1.092) | 0.025 |
| Neutrophil count, ×109/L | 8.8 (6.6–13.7) | 11.1 (8.4–16.9) | 1.106 (0.996–1.228) | 0.061 |
| CCL23, pg/mL | 70.6 (48.5–104.4) | 122.5 (76.4–160.2) | 1.020 (1.009–1.032) | 0.001 |
| Characteristics | Good Outcome (n = 60) | Poor Outcome (n = 42) | OR | p-Value |
|---|---|---|---|---|
| Age, years | 54 (46–62) | 63 (49.1–69) | 1.027 (0.993–1.063) | 0.117 |
| Gender (female) | 33 (55.0%) | 23 (54.8%) | 1.010 (0.457–2.230) | 0.981 |
| BMI, kg/m2 | 22.8 (21.1–26.0) | 23.8 (21.7–26.6) | 1.097 (0.921–1.307) | 0.299 |
| Current smokers | 15 (25.0%) | 13 (31.0%) | 1.345 (0.559–3.233) | 0.508 |
| Alcohol consumption | 12 (20.0%) | 9 (21.4%) | 1.091 (0.413–2.881) | 0.861 |
| Hypertension | 22 (36.7%) | 25 (59.5%) | 2.540 (1.131–5.707) | 0.024 |
| Diabetes mellitus | 4 (6.7%) | 5 (11.9%) | 1.892 (0.477–7.511) | 0.365 |
| Cardiovascular comorbidities | 2 (3.3%) | 5 (11.9%) | 3.919 (0.722–21.257) | 0.113 |
| Hunt–Hess grade | 2 (1–3) | 3 (2–4) | 2.663 (1.734–4.089) | <0.001 |
| Modified Fisher grade | 1 (1–2) | 3 (2–3) | 2.521 (1.596–3.983) | <0.001 |
| Aneurysmal position (anterior circulation) | 54 (90.0%) | 35 (83.3%) | 1.800 (0.558–5.802) | 0.325 |
| Aneurysmal shape (cystic) | 52 (86.7%) | 35 (83.3%) | 1.300 (0.432–3.910) | 0.641 |
| Aneurysm size, mm | 5 (3.5–7.0) | 6.2 (3.9–10.7) | 1.176 (1.044–1.325) | 0.008 |
| Aneurysm size > 10 mm | 7 (11.7%) | 11 (26.2%) | 2.687 (0.944–7.648) | 0.064 |
| Endovascular coiling | 34 (56.7%) | 16 (38.1%) | 0.471 (0.210–1.053) | 0.067 |
| Acute hydrocephalus | 12 (20.0%) | 19 (45.2%) | 3.304 (1.375–7.944) | 0.008 |
| DCI | 6 (10.0%) | 13 (31.0%) | 1.027 (0.993–1.063) | 0.036 |
| External ventricular drain | 2 (3.3%) | 8 (19.0%) | 6.824 (1.369–34.010) | 0.019 |
| CRP, mg/L | 5.6 (2.6–13.0) | 14.8 (6.7–27.4) | 1.084 (1.032–1.140) | 0.001 |
| Neutrophil count, ×109/L | 8.7 (6.5–13.4) | 10.6 (7.5–15.4) | 1.084 (0.990–1.186) | 0.080 |
| CCL23, pg/mL | 61.5 (45.1–86.6) | 103.7 (78.0–139.5) | 1.024 (1.012–1.036) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, H.; Shen, J.; Zhu, Y.; Zhou, L.; Wu, F.; Liu, Z.; Zhang, S.; Zhan, R. Elevated Serum CCL23 Levels at Admission Predict Delayed Cerebral Ischemia and Functional Outcome after Aneurysmal Subarachnoid Hemorrhage. J. Clin. Med. 2022, 11, 6879. https://doi.org/10.3390/jcm11236879
Lin H, Shen J, Zhu Y, Zhou L, Wu F, Liu Z, Zhang S, Zhan R. Elevated Serum CCL23 Levels at Admission Predict Delayed Cerebral Ischemia and Functional Outcome after Aneurysmal Subarachnoid Hemorrhage. Journal of Clinical Medicine. 2022; 11(23):6879. https://doi.org/10.3390/jcm11236879
Chicago/Turabian StyleLin, Hongwei, Jie Shen, Yu Zhu, Lihui Zhou, Fan Wu, Zongchi Liu, Shengxiang Zhang, and Renya Zhan. 2022. "Elevated Serum CCL23 Levels at Admission Predict Delayed Cerebral Ischemia and Functional Outcome after Aneurysmal Subarachnoid Hemorrhage" Journal of Clinical Medicine 11, no. 23: 6879. https://doi.org/10.3390/jcm11236879
APA StyleLin, H., Shen, J., Zhu, Y., Zhou, L., Wu, F., Liu, Z., Zhang, S., & Zhan, R. (2022). Elevated Serum CCL23 Levels at Admission Predict Delayed Cerebral Ischemia and Functional Outcome after Aneurysmal Subarachnoid Hemorrhage. Journal of Clinical Medicine, 11(23), 6879. https://doi.org/10.3390/jcm11236879






