Clinical Outcomes and Prevalence of Sarcopenia in Patients with Moderate to Severe COVID-19
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Characteristics of Patients
2.3. Rehabilitation Program
2.4. Assessment of Physical Functions and Activities of Daily Living (ADLs)
2.4.1. Physical Functions
2.4.2. ADLs
2.5. Statistical Analysis
3. Results
3.1. Characteristics of Patients
3.2. Safety of Physiotherapy in Patients with Severe COVID-19: Complications of Physiotherapy in This Series
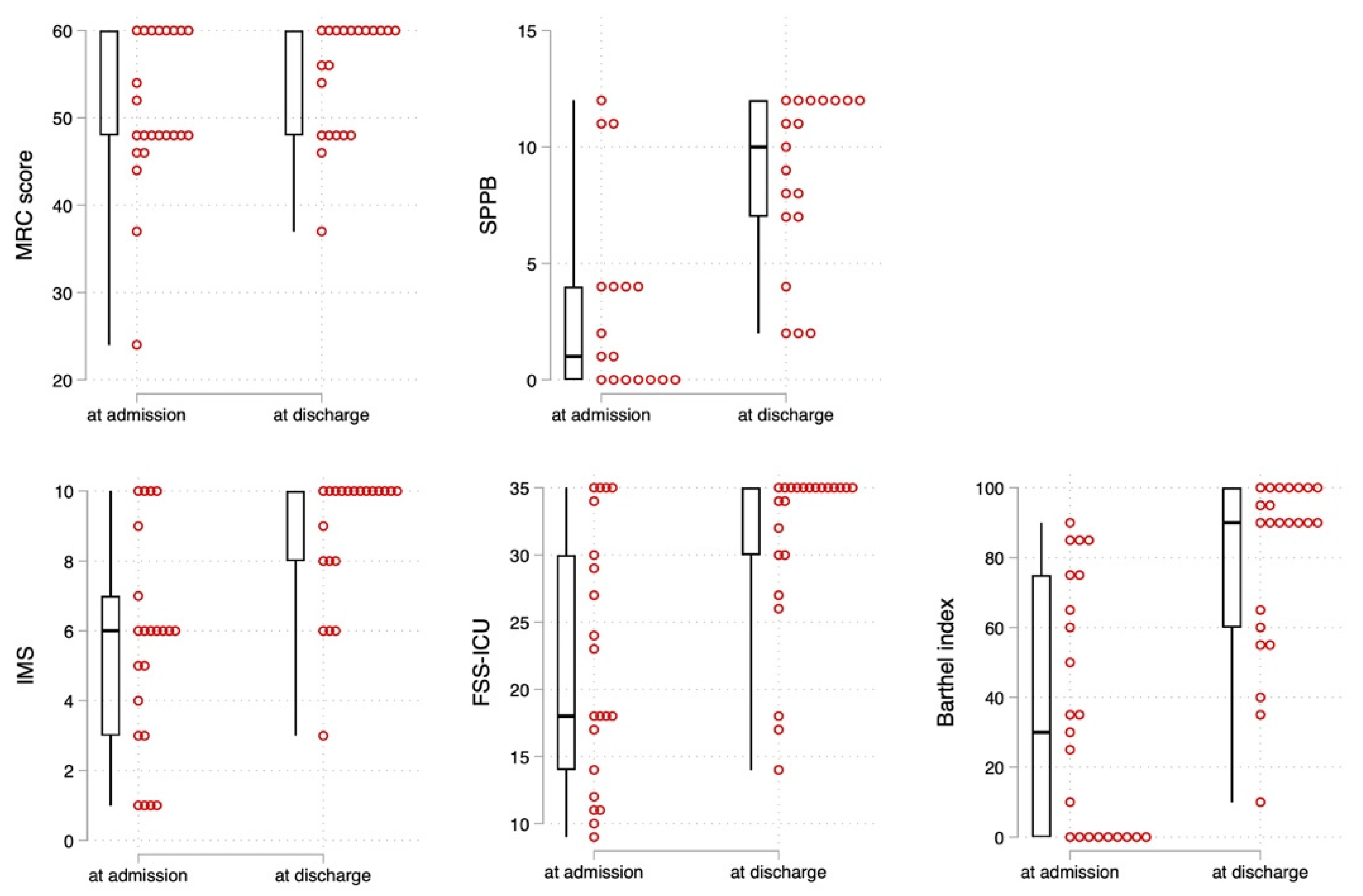
3.3. Effects of Rehabilitation on Physical Functions for Hospitalized Patients with COVID-19
| Admission (n = 23) | Discharge (n = 23) | p Value | Effect Size (95% CI) | |
|---|---|---|---|---|
| Laboratory data | ||||
| White blood cell (×103/μL) | 8.21 [4.60, 10.84] | 7.94 [4.72, 9.90] | 0.345 | −0.11 (−0.69 to 0.47) |
| C-reactive protein (mg/dL) | 8.15 [4.04, 10.35] | 0.31 [0.01, 0.79] | <0.001 | −1.86 (−2.54 to −1.15) |
| Albumin (g/dL) | 3.0 [2.8, 3.3] | 3.0 [2.8, 3.2] | 0.963 | 0.01 (−0.57 to 0.59) |
| AST (U/L) | 35.0 [29.0, 54.0] | 23.0 [21.0, 31.0] | 0.012 | −0.56 (−1.14 to 0.03) |
| ALT (U/L) | 39.0 [22.0, 68.0] | 52.0 [30.0, 76.0] | 0.247 | 0.31 (−0.27 to 0.89) |
| D-dimer (μg/mL) | 1.4 [1.1, 2.5] | 1.3 [0.9, 2.0] | 0.169 | −0.52 (−1.12 to 0.08) |
| Creatinine (mg/dL) | 0.76 [0.62, 0.98] | 0.70 [0.59, 0.98] | 0.513 | −0.14 (−0.72 to 0.44) |
| BUN (mg/dL) | 19.6 [14.2, 28.7] | 21.5 [9.8, 23.4] | 0.361 | −0.32 (−0.90 to 0.27) |
| eGFR (mL/min/1.73 m2) | 78.0 [61.0, 95.0] | 80.0 [66.0, 96.0] | 0.951 | −0.02 (−0.59 to 0.56) |
| Physical functions | ||||
| MRC score (points) | 48.0 [48.0, 60.0] | 60.0 [48.0, 60.0] | 0.014 | 0.50 (−0.11 to 1.10) |
| SPPB (points) | 1.0 [0.0, 4.0] | 10.0 [7.0, 12.0] | <0.001 | 1.39 (0.65 to 2.11) |
| ADL | ||||
| IMS (points) | 6.0 [3.0, 7.0] | 10.0 [8.0, 10.0] | <0.001 | 1.25 (0.59 to 1.90) |
| FSS-ICU (points) | 18.0 [14.0, 30.0] | 35.0 [30.0, 35.0] | <0.001 | 1.14 (0.49 to 1.77) |
| Barthel index (points) | 30.0 [0.0, 75.0] | 90.0 [60.0, 100.0] | <0.001 | 1.46 (0.80 to 2.11) |

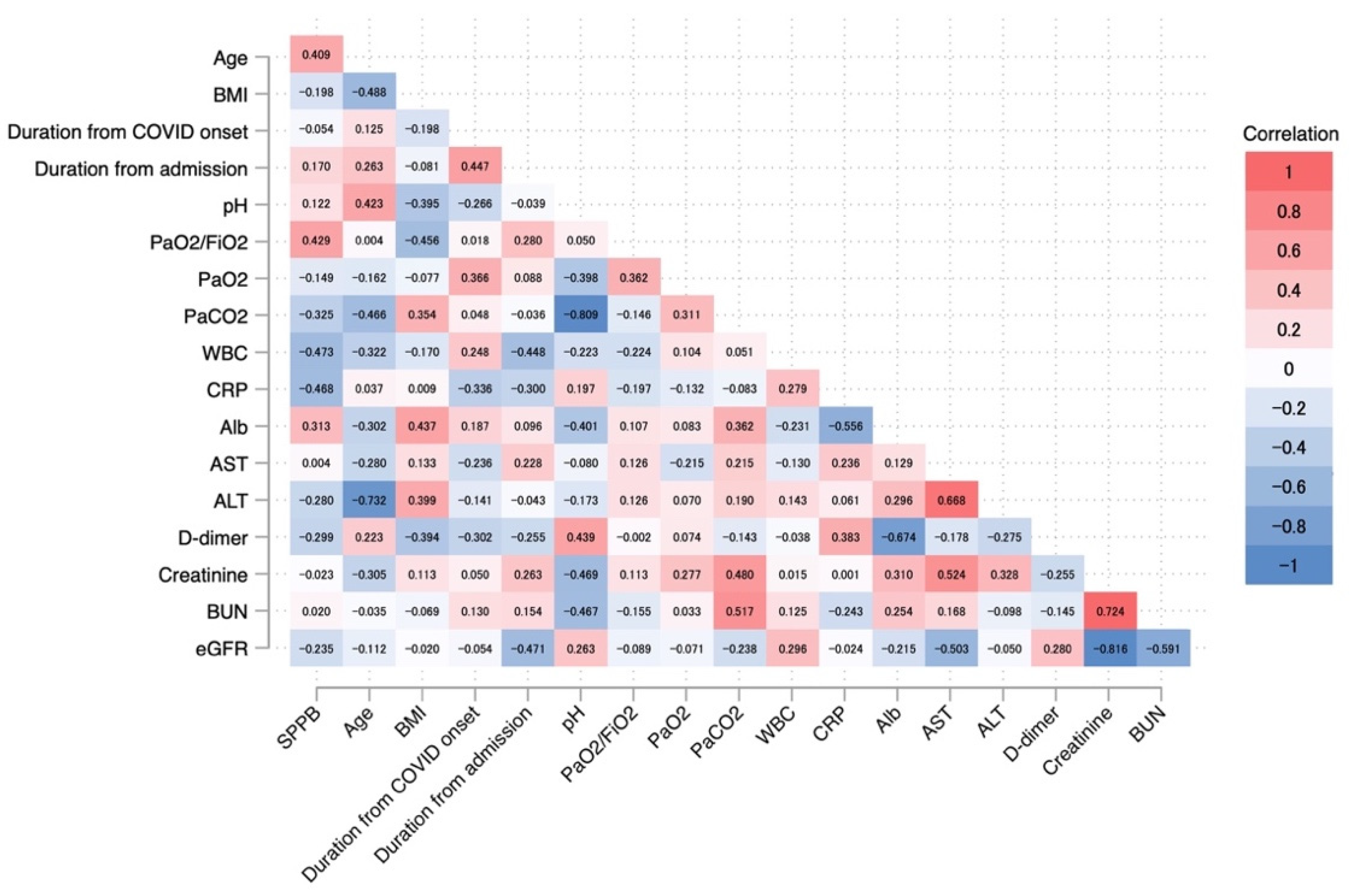
3.4. Association of SPPB at Discharge with Background Factors and Laboratory Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Heesakkers, H.; van der Hoeven, J.G.; Corsten, S.; Janssen, I.; Ewalds, E.; Simons, K.S.; Westerhof, B.; Rettig, T.C.; Jacobs, C.; van Santen, S.; et al. Clinical Outcomes Among Patients With 1-Year Survival Following Intensive Care Unit Treatment for COVID-19. JAMA 2022, 327, 559–565. [Google Scholar] [CrossRef]
- Maley, J.H.; Sandsmark, D.K.; Trainor, A.; Bass, G.D.; Dabrowski, C.L.; Magdamo, B.A.; Durkin, B.; Hayes, M.M.; Schwartzstein, R.M.; Stevens, J.P.; et al. Six-Month Impairment in Cognition, Mental Health, and Physical Function Following COVID-19-Associated Respiratory Failure. Crit. Care Explor. 2022, 4, e0673. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Morris, P.E.; Goad, A.; Thompson, C.; Taylor, K.; Harry, B.; Passmore, L.; Ross, A.; Anderson, L.; Baker, S.; Sanchez, M.; et al. Early intensive care unit mobility therapy in the treatment of acute respiratory failure. Crit. Care Med. 2008, 36, 2238–2243. [Google Scholar] [CrossRef]
- Schweickert, W.D.; Pohlman, M.C.; Pohlman, A.S.; Nigos, C.; Pawlik, A.J.; Esbrook, C.L.; Spears, L.; Miller, M.; Franczyk, M.; Deprizio, D.; et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: A randomised controlled trial. Lancet 2009, 373, 1874–1882. [Google Scholar] [CrossRef]
- Spruit, M.A.; Holland, A.E.; Singh, S.J.; Tonia, T.; Wilson, K.C.; Troosters, T. COVID-19: Interim Guidance on Rehabilitation in the Hospital and Post-Hospital Phase from a European Respiratory Society and American Thoracic Society-coordinated International Task Force. Eur. Respir. J. 2020, 56, 2002197. [Google Scholar] [CrossRef]
- Thomas, P.; Baldwin, C.; Bissett, B.; Boden, I.; Gosselink, R.; Granger, C.L.; Hodgson, C.; Jones, A.Y.; Kho, M.E.; Moses, R.; et al. Physiotherapy management for COVID-19 in the acute hospital setting: Clinical practice recommendations. J. Physiother. 2020, 66, 73–82. [Google Scholar] [CrossRef]
- Ambrose, A.F.; Kurra, A.; Tsirakidis, L.; Hunt, K.C.; Ayers, E.; Gitkind, A.; Yerra, S.; Lo, Y.; Ortiz, N.; Jamal, F.; et al. Rehabilitation and In-Hospital Mortality in COVID-19 patients. J. Gerontol. A Biol. Sci. Med. Sci. 2021, 77, e148–e154. [Google Scholar] [CrossRef]
- Tsukakoshi, D.; Yamamoto, S.; Wada, Y.; Terashima, S.; Osawa, R.; Matsumori, K.; Ito, S.; Nakamura, Y.; Nagamine, K.; Ikegami, S.; et al. Rehabilitation of a Patient with Severe COVID-19 Using Extracorporeal Membrane Oxygenation: A Case Report. Jpn. J. Rehabil. Med. 2022, 59, 92–98. [Google Scholar] [CrossRef]
- Hermans, G.; Clerckx, B.; Vanhullebusch, T.; Segers, J.; Vanpee, G.; Robbeets, C.; Casaer, M.P.; Wouters, P.; Gosselink, R.; Van Den Berghe, G. Interobserver agreement of Medical Research Council sum-score and handgrip strength in the intensive care unit. Muscle Nerve 2012, 45, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, C.; Needham, D.; Haines, K.; Bailey, M.; Ward, A.; Harrold, M.; Young, P.; Zanni, J.; Buhr, H.; Higgins, A.; et al. Feasibility and inter-rater reliability of the ICU Mobility Scale. Heart Lung 2014, 43, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Zanni, J.M.; Korupolu, R.; Fan, E.; Pradhan, P.; Janjua, K.; Palmer, J.B.; Brower, R.G.; Needham, D.M. Rehabilitation therapy and outcomes in acute respiratory failure: An observational pilot project. J. Crit. Care 2010, 25, 254–262. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Chen, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Chou, M.Y.; Iijima, K.; Jang, H.C.; Kang, L.; Kim, M.; Kim, S.; et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307.e2. [Google Scholar] [CrossRef]
- Grasselli, G.; Greco, M.; Zanella, A.; Albano, G.; Antonelli, M.; Bellani, G.; Bonanomi, E.; Cabrini, L.; Carlesso, E.; Castelli, G.; et al. Risk Factors Associated with Mortality Among Patients with COVID-19 in Intensive Care Units in Lombardy, Italy. JAMA Intern. Med. 2020, 180, 1345–1355. [Google Scholar] [CrossRef]
- Zampogna, E.; Paneroni, M.; Belli, S.; Aliani, M.; Gandolfo, A.; Visca, D.; Bellanti, M.T.; Ambrosino, N.; Vitacca, M. Pulmonary Rehabilitation in Patients Recovering from COVID-19. Respiration 2021, 100, 416–422. [Google Scholar] [CrossRef]
- Xu, Y.; Xu, J.W.; You, P.; Wang, B.L.; Liu, C.; Chien, C.W.; Tung, T.H. Prevalence of Sarcopenia in Patients with COVID-19: A Systematic Review and Meta-Analysis. Front. Nutr. 2022, 9, 925606. [Google Scholar] [CrossRef]
- Musheyev, B.; Borg, L.; Janowicz, R.; Matarlo, M.; Boyle, H.; Singh, G.; Ende, V.; Babatsikos, I.; Hou, W.; Duong, T.Q. Functional status of mechanically ventilated COVID-19 survivors at ICU and hospital discharge. J. Intensive Care 2021, 9, 31. [Google Scholar] [CrossRef]
- Paliogiannis, P.; Mangoni, A.A.; Dettori, P.; Nasrallah, G.K.; Pintus, G.; Zinellu, A. D-Dimer Concentrations and COVID-19 Severity: A Systematic Review and Meta-Analysis. Front. Public Health 2020, 8, 432. [Google Scholar] [CrossRef] [PubMed]
- Debi, H.; Itu, Z.T.; Amin, M.T.; Hussain, F.; Hossain, M.S. Association of serum C-reactive protein (CRP) and D-dimer concentration on the severity of COVID-19 cases with or without diabetes: A systematic review and meta-analysis. Expert Rev. Endocrinol. Metab. 2022, 17, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.N.; Eggelbusch, M.; Naddaf, E.; Gerrits, K.H.; van der Schaaf, M.; van den Borst, B.; Wiersinga, W.J.; van Vugt, M.; Weijs, P.J.; Murray, A.J.; et al. Skeletal muscle alterations in patients with acute COVID-19 and post-acute sequelae of COVID-19. J. Cachexia Sarcopenia Muscle 2022, 13, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, V.A.; Allan, L.; Bethel, A.; Cowley, A.; Cross, J.L.; Day, J.; Drummond, A.; Hall, A.J.; Howard, M.; Morley, N.; et al. Rehabilitation to enable recovery from COVID-19: A rapid systematic review. Physiotherapy 2021, 111, 4–22. [Google Scholar] [CrossRef]

| Overall (n = 23) | |
|---|---|
| Age (years) | 59 [47, 67] |
| Male gender (n, %) | 17 (73.9) |
| BMI (kg/m2) | 24.4 [22.4, 28.6] |
| Comorbidities (n, %) | |
| Hypertension | 7 (30.4) |
| Dyslipidemia | 5 (21.7) |
| Diabetes | 8 (34.8) |
| Atrial fibrillation | 1 (4.3) |
| Coronary artery disease | 4 (17.4) |
| COPD | 1 (4.3) |
| Interstitial pneumonia | 1 (6.2) |
| Stroke | 2 (8.7) |
| Cancer | 2 (8.7) |
| Mechanical ventilation (n, %) | 10 (43.5) |
| Duration of mechanical ventilation, days | 4 [1, 7] |
| pH | 7.42 [7.36, 7.47] |
| PaO2/FiO2 | 172.0 [123.0, 209.0] |
| PaO2 (mmHg) | 90.5 [75.3, 107.0] |
| PaCO2 (mmHg) | 35.4 [31.2, 39.2] |
| Duration from COVID-19 onset to start of rehabilitation (days) | 8.0 [4.0, 11.0] |
| Duration from admission to start of rehabilitation (days) | 3.0 [2.0, 4.0] |
| Duration of rehabilitation (days) | 12 [7.0, 20.0] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamamoto, S.; Sakai, Y.; Matsumori, K.; Osawa, R.; Ito, S.; Tsukakoshi, D.; Ohno, T.; Ohta, H.; Ichiyama, T.; Komatsu, M.; et al. Clinical Outcomes and Prevalence of Sarcopenia in Patients with Moderate to Severe COVID-19. J. Clin. Med. 2022, 11, 6578. https://doi.org/10.3390/jcm11216578
Yamamoto S, Sakai Y, Matsumori K, Osawa R, Ito S, Tsukakoshi D, Ohno T, Ohta H, Ichiyama T, Komatsu M, et al. Clinical Outcomes and Prevalence of Sarcopenia in Patients with Moderate to Severe COVID-19. Journal of Clinical Medicine. 2022; 11(21):6578. https://doi.org/10.3390/jcm11216578
Chicago/Turabian StyleYamamoto, Shuhei, Yasunari Sakai, Keiji Matsumori, Ryuji Osawa, Shun Ito, Daichi Tsukakoshi, Tomoki Ohno, Hiroaki Ohta, Takashi Ichiyama, Masamichi Komatsu, and et al. 2022. "Clinical Outcomes and Prevalence of Sarcopenia in Patients with Moderate to Severe COVID-19" Journal of Clinical Medicine 11, no. 21: 6578. https://doi.org/10.3390/jcm11216578
APA StyleYamamoto, S., Sakai, Y., Matsumori, K., Osawa, R., Ito, S., Tsukakoshi, D., Ohno, T., Ohta, H., Ichiyama, T., Komatsu, M., Wada, Y., Hanaoka, M., Ikegami, S., & Horiuchi, H. (2022). Clinical Outcomes and Prevalence of Sarcopenia in Patients with Moderate to Severe COVID-19. Journal of Clinical Medicine, 11(21), 6578. https://doi.org/10.3390/jcm11216578








