Human Respiratory Syncytial Virus Infections among Hospitalized Children in Poland during 2010–2020: Study Based on the National Hospital Registry
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Methods
2.2. Statistical Analysis
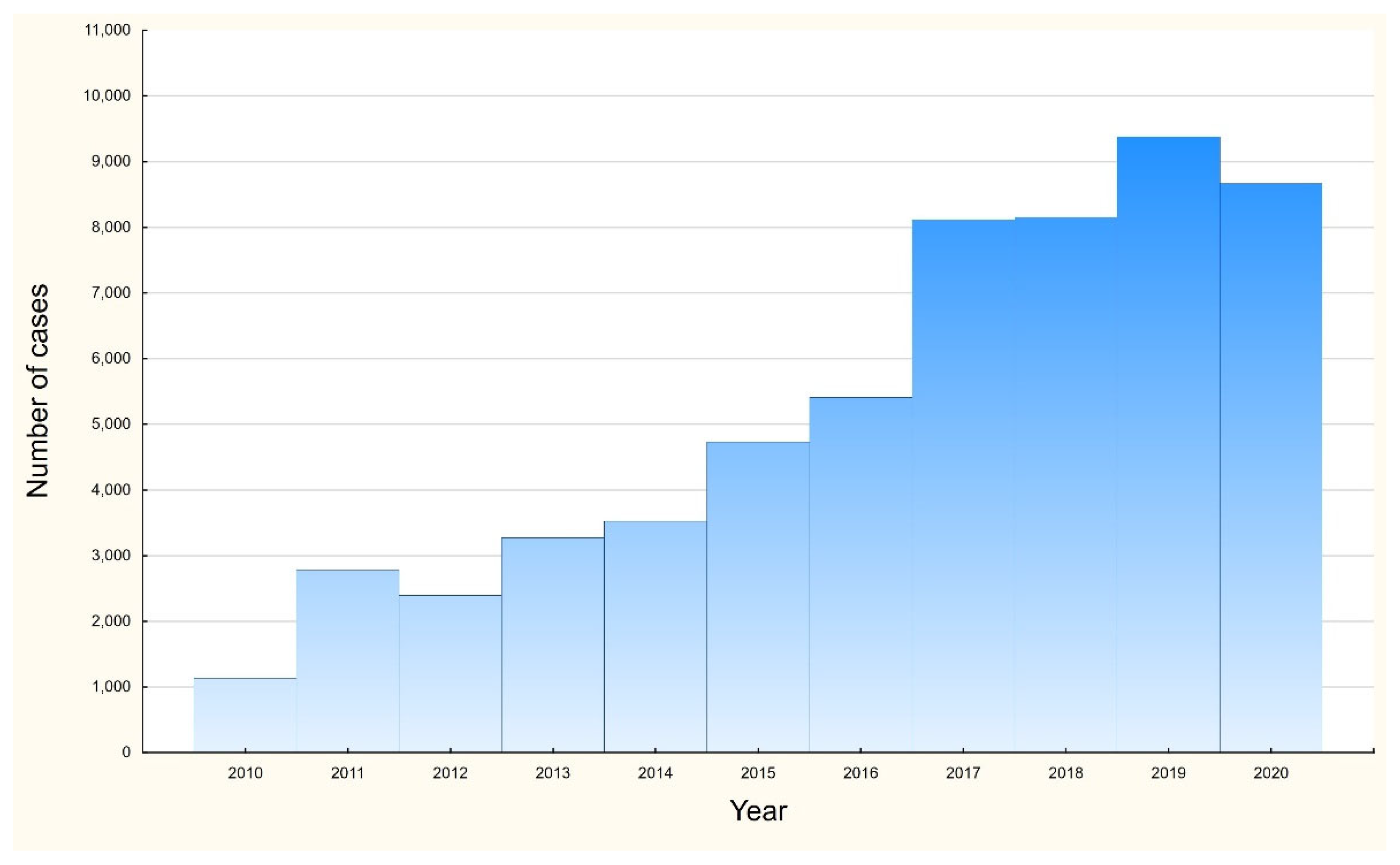
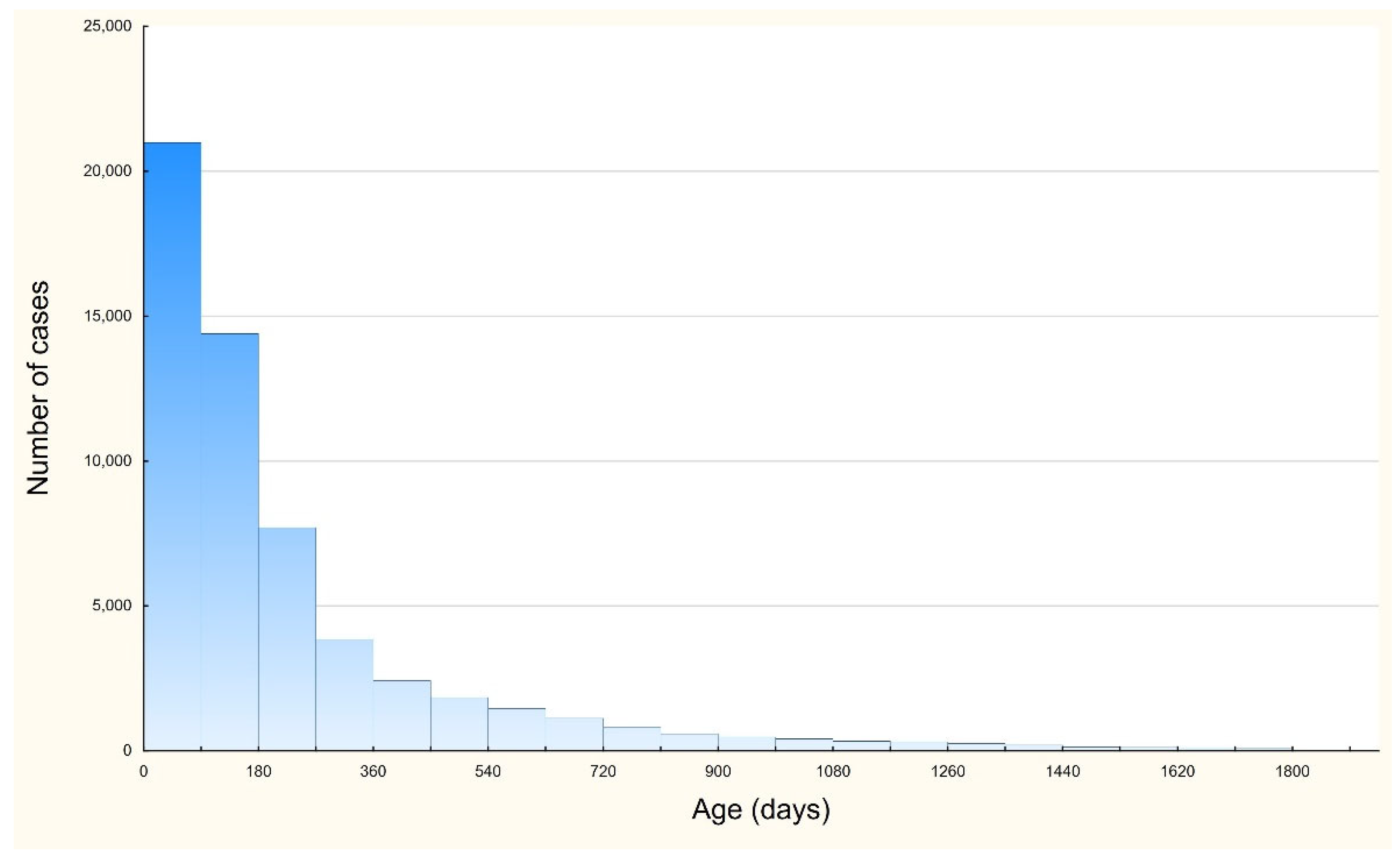
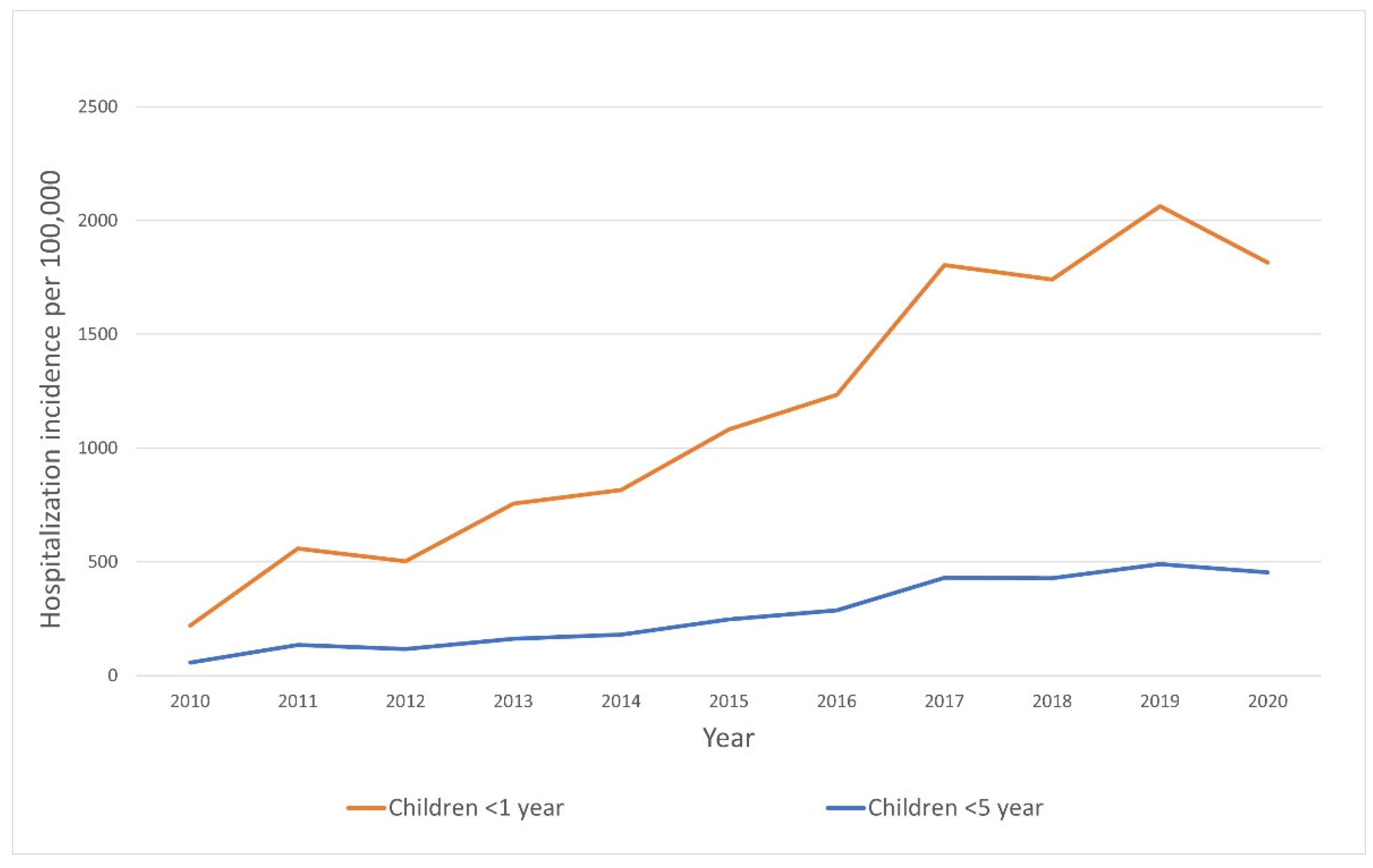
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manti, S.; Piedimonte, G. An overview on the RSV-mediated mechanisms in the onset of non-allergic asthma. Front. Pediatr. 2022, 10, 998296. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.; Drews, S.J.; Marchant, D.J. Respiratory Syncytial Virus: Infection, Detection, and New Options for Prevention and Treatment. Clin. Microbiol. Rev. 2017, 30, 277–319. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Wang, T. Laboratory epidemiology of respiratory viruses in a large children’s hospital: A STROBE-compliant article. Medicine 2018, 97, e11385. [Google Scholar] [CrossRef]
- Borchers, A.T.; Chang, C.; Gershwin, M.E.; Gershwin, L.J. Respiratory syncytial virus—A comprehensive review. Clin. Rev. Allergy Immunol. 2013, 45, 331–379. [Google Scholar] [CrossRef]
- Barr, R.; Green, C.A.; Sande, C.J.; Drysdale, S.B. Respiratory syncytial virus: Diagnosis, prevention and management. Ther. Adv. Infect. Dis. 2019, 6, 2049936119865798. [Google Scholar] [CrossRef]
- Vandini, S.; Biagi, C.; Lanari, M. Respiratory Syncytial Virus: The Influence of Serotype and Genotype Variability on Clinical Course of Infection. Int. J. Mol. Sci. 2017, 18, 1717. [Google Scholar] [CrossRef]
- Nair, H.; Nokes, D.J.; Gessner, B.D.; Dherani, M.; Madhi, S.A.; Singleton, R.J.; O’Brien, K.L.; Roca, A.; Wright, P.F.; Bruce, N.; et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: A systematic review and meta-analysis. Lancet 2010, 375, 1545–1555. [Google Scholar] [CrossRef]
- Wrotek, A.; Kobiałka, M.; Grochowski, B.; Kamińska, I.; Pędziwiatr, K.; Skoczek-Wojciechowska, A.; Godek, M.; Jackowska, T. Respiratory Complications in Children Hospitalized with Respiratory Syncytial Virus Infection. Adv. Exp. Med. Biol. 2020, 1279, 113–120. [Google Scholar] [CrossRef]
- Régnier, S.A.; Huels, J. Association between respiratory syncytial virus hospitalizations in infants and respiratory sequelae: Systematic review and meta-analysis. Pediatr. Infect. Dis. J. 2013, 32, 820–826. [Google Scholar] [CrossRef]
- Shi, T.; McAllister, D.A.; O’Brien, K.L.; Simoes, E.A.; Madhi, S.A.; Gessner, B.D.; Polack, F.P.; Balsells, E.; Acacio, S.; Aguayo, C.; et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: A systematic review and modelling study. Lancet 2017, 390, 946–958. [Google Scholar] [CrossRef]
- Stein, R.T.; Bont, L.J.; Zar, H.; Polack, F.P.; Park, C.; Claxton, A.; Borok, G.; Butylkova, Y.; Wegzyn, C. Respiratory syncytial virus hospitalization and mortality: Systematic review and meta-analysis. Pediatr. Pulmonol. 2017, 52, 556–569. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Stobart, C.C.; Hotard, A.L.; Moore, M.L. An overview of respiratory syncytial virus. PLoS Pathog. 2014, 10, e1004016. [Google Scholar] [CrossRef] [PubMed]
- Góra, D. Hospitalization of children in Poland due to selected respiratory diseases in 2012–2017 in hospitals of different referral levels. Otorhinolaryngol.–Clin. Rev. 2018, 17, 20–28. (In Polish) [Google Scholar]
- Hallmann-Szelińska, E.; Bednarska, K.; Kondratiuk, K.; Rabczenko, D.; Brydak, L.B. Viral Infections in Children in the 2014/2015 Epidemic Season in Poland. Adv. Exp. Med. Biol. 2016, 912, 51–56. [Google Scholar] [CrossRef]
- Teirlinck, A.C.; Broberg, E.K.; Berg, A.S.; Campbell, H.; Reeves, R.M.; Carnahan, A.; Lina, B.; Pakarna, G.; Bøås, H.; Nohynek, H.; et al. Recommendations for respiratory syncytial virus surveillance at the national level. Eur. Respir. J. 2021, 58, 2003766. [Google Scholar] [CrossRef]
- TIBCO Software Inc. Statistica (Data Analysis Software System), Version 13. 2017. Available online: http://statistica.io (accessed on 1 August 2022).
- Abramson, J.H. WINPEPI Updated: Computer Programs for Epidemiologists, and Their Teaching Potential. Epidemiol. Persp. 2011, 8, 1. [Google Scholar] [CrossRef]
- Statistics Poland. Available online: https://stat.gov.pl (accessed on 4 August 2022).
- Lumley, T.; Diehr, P.; Emerson, S.; Chen, L. The importance of the normality assumption in large public health data sets. Annu. Rev. Public Health 2002, 23, 151–169. [Google Scholar] [CrossRef]
- Johannesen, C.K.; van Wijhe, M.; Tong, S.; Fernández, L.V.; Heikkinen, T.; van Boven, M.; Wang, X.; Bøås, H.; Li, Y.; Campbell, H.; et al. Age-Specific Estimates of Respiratory Syncytial Virus-Associated Hospitalizations in 6 European Countries: A Time Series Analysis. J. Infect. Dis. 2022, 226 (Suppl. 1), S29–S37. [Google Scholar] [CrossRef]
- Bruning, A.H.; Leeflang, M.M.; Vos, J.M.; Spijker, R.; de Jong, M.D.; Wolthers, K.C.; Pajkrt, D. Rapid Tests for Influenza, Respiratory Syncytial Virus, and Other Respiratory Viruses: A Systematic Review and Meta-analysis. Clin. Infect. Dis. 2017, 65, 1026–1032. [Google Scholar] [CrossRef]
- Anderson, E.J.; Carbonell-Estrany, X.; Blanken, M.; Lanari, M.; Sheridan-Pereira, M.; Rodgers-Gray, B.; Fullarton, J.; Rouffiac, E.; Vo, P.; Notario, G.; et al. Burden of Severe Respiratory Syncytial Virus Disease Among 33-35 Weeks’ Gestational Age Infants Born During Multiple Respiratory Syncytial Virus Seasons. Pediatr. Infect. Dis. J. 2017, 36, 160–167. [Google Scholar] [CrossRef]
- Yassine, H.M.; Sohail, M.U.; Younes, N.; Nasrallah, G.K. Systematic Review of the Respiratory Syncytial Virus (RSV) Prevalence, Genotype Distribution, and Seasonality in Children from the Middle East and North Africa (MENA) Region. Microorganisms 2020, 8, 713. [Google Scholar] [CrossRef]
- Salimi, V.; Tavakoli-Yaraki, M.; Yavarian, J.; Bont, L.; Mokhtari-Azad, T. Prevalence of human respiratory syncytial virus circulating in Iran. J. Infect. Public Health 2016, 9, 125–135. [Google Scholar] [CrossRef]
- Hasegawa, K.; Goto, T.; Hirayama, A.; Laham, F.R.; Mansbach, J.M.; Piedra, P.A.; Camargo, C.A., Jr. Respiratory Virus Epidemiology Among US Infants With Severe Bronchiolitis: Analysis of 2 Multicenter, Multiyear Cohort Studies. Pediatr. Infect. Dis. J. 2019, 38, e180–e183. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Balsells, E.; Wastnedge, E.; Singleton, R.; Rasmussen, Z.A.; Zar, H.J.; Rath, B.A.; Madhi, S.A.; Campbell, S.; Vaccari, L.C.; et al. Risk factors for respiratory syncytial virus associated with acute lower respiratory infection in children under five years: Systematic review and meta-analysis. J. Glob. Health 2015, 5, 020416. [Google Scholar] [CrossRef] [PubMed]
- Suleiman-Martos, N.; Caballero-Vázquez, A.; Gómez-Urquiza, J.L.; Albendín-García, L.; Romero-Béjar, J.L.; Cañadas-De la Fuente, G.A. Prevalence and Risk Factors of Respiratory Syncytial Virus in Children under 5 Years of Age in the WHO European Region: A Systematic Review and Meta-Analysis. J. Pers. Med. 2021, 11, 416. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.L.; Chaves, S.S.; Demont, C.; Viboud, C. Mortality Associated With Influenza and Respiratory Syncytial Virus in the US, 1999-2018. JAMA Netw. Open 2022, 5, e220527. [Google Scholar] [CrossRef]
- Nitsch-Osuch, A.; Kuchar, E.; Topczewska-Cabanek, A.; Wardyn, K.; Życińska, K.; Brydak, L. Incidence and Clinical Course of Respiratory Viral Coinfections in Children Aged 0-59 Months. Adv. Exp. Med. Biol. 2016, 905, 17–23. [Google Scholar] [CrossRef]
- Zhang, S.; Akmar, L.Z.; Bailey, F.; Rath, B.A.; Alchikh, M.; Schweiger, B.; Lucero, M.G.; Nillos, L.T.; Kyaw, M.H.; Kieffer, A.; et al. Cost of Respiratory Syncytial Virus-Associated Acute Lower Respiratory Infection Management in Young Children at the Regional and Global Level: A Systematic Review and Meta-Analysis. J. Infect. Dis. 2020, 222 (Suppl. 7), S680–S687. [Google Scholar] [CrossRef]
- Reeves, R.M.; Hardelid, P.; Gilbert, R.; Warburton, F.; Ellis, J.; Pebody, R.G. Estimating the burden of respiratory syncytial virus (RSV) on respiratory hospital admissions in children less than five years of age in England, 2007–2012. Influenza Other Respir. Viruses 2017, 11, 122–129. [Google Scholar] [CrossRef]
- Bardsley, M.; Morbey, R.A.; Hughes, H.E.; Beck, C.R.; Watson, C.H.; Zhao, H.; Ellis, J.; Smith, G.E.; Elliot, A.J. Epidemiology of respiratory syncytial virus in children younger than 5 years in England during the COVID-19 pandemic, measured by laboratory, clinical, and syndromic surveillance: A retrospective observational study. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef]
- Bögli, J.; Güsewell, S.; Strässle, R.; Kahlert, C.R.; Albrich, W.C. Pediatric hospital admissions, case severity, and length of hospital stay during the first 18 months of the COVID-19 pandemic in a tertiary children’s hospital in Switzerland. Infection 2022, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Batinović, E.; Milić, P.; Markić, J. The role of birth month in the burden of hospitalisations for acute lower respiratory infections due to respiratory syncytial virus in young children in Croatia. PLoS ONE 2022, 17, e0273962. [Google Scholar] [CrossRef]
- Checchia, P.A.; Nalysnyk, L.; Fernandes, A.W.; Mahadevia, P.J.; Xu, Y.; Fahrbach, K.; Welliver Sr, R.C. Mortality and morbidity among infants at high risk for severe respiratory syncytial virus infection receiving prophylaxis with palivizumab: A systematic literature review and meta-analysis. Pediatr. Crit. Care Med. 2011, 12, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Blanken, M.O.; Frederix, G.W.; Nibbelke, E.E.; Koffijberg, H.; Sanders, E.A.; Rovers, M.M.; Bont, L. Cost-effectiveness of rule-based immunoprophylaxis against respiratory syncytial virus infections in preterm infants. Eur. J. Pediatr. 2018, 177, 133–144. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rząd, M.; Kanecki, K.; Lewtak, K.; Tyszko, P.; Szwejkowska, M.; Goryński, P.; Nitsch-Osuch, A. Human Respiratory Syncytial Virus Infections among Hospitalized Children in Poland during 2010–2020: Study Based on the National Hospital Registry. J. Clin. Med. 2022, 11, 6451. https://doi.org/10.3390/jcm11216451
Rząd M, Kanecki K, Lewtak K, Tyszko P, Szwejkowska M, Goryński P, Nitsch-Osuch A. Human Respiratory Syncytial Virus Infections among Hospitalized Children in Poland during 2010–2020: Study Based on the National Hospital Registry. Journal of Clinical Medicine. 2022; 11(21):6451. https://doi.org/10.3390/jcm11216451
Chicago/Turabian StyleRząd, Michał, Krzysztof Kanecki, Katarzyna Lewtak, Piotr Tyszko, Martyna Szwejkowska, Paweł Goryński, and Aneta Nitsch-Osuch. 2022. "Human Respiratory Syncytial Virus Infections among Hospitalized Children in Poland during 2010–2020: Study Based on the National Hospital Registry" Journal of Clinical Medicine 11, no. 21: 6451. https://doi.org/10.3390/jcm11216451
APA StyleRząd, M., Kanecki, K., Lewtak, K., Tyszko, P., Szwejkowska, M., Goryński, P., & Nitsch-Osuch, A. (2022). Human Respiratory Syncytial Virus Infections among Hospitalized Children in Poland during 2010–2020: Study Based on the National Hospital Registry. Journal of Clinical Medicine, 11(21), 6451. https://doi.org/10.3390/jcm11216451





