Relationship between Cardiac Acoustic Biomarkers and Pulmonary Artery Pressure in Patients with Heart Failure
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
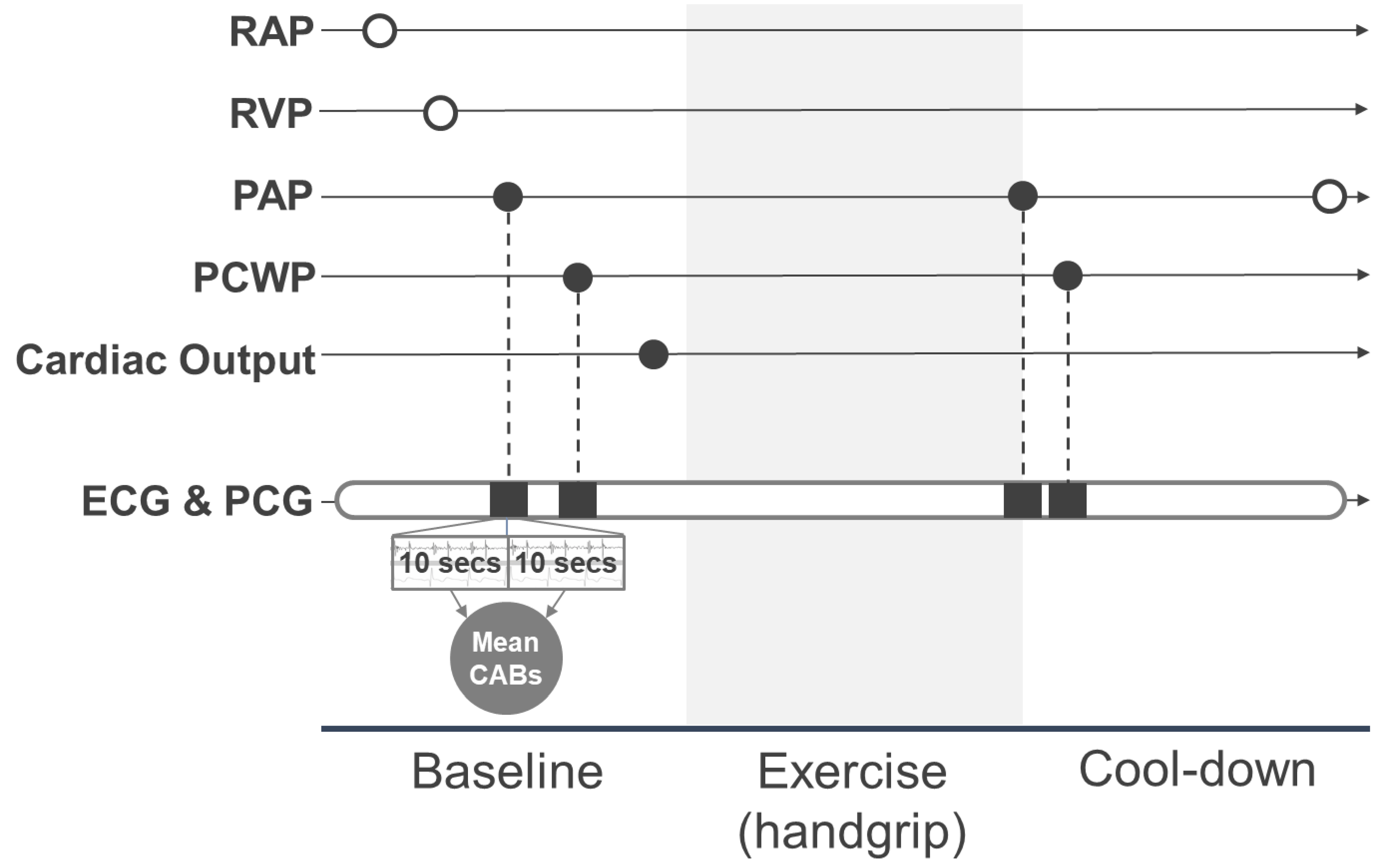
2.2. Data Collection
2.2.1. Right Heart Catheterization
2.2.2. CABs
2.3. Statistical Analysis
3. Results
3.1. Patient Demographics
3.2. Correlation between Hemodynamic Parameters and CABs
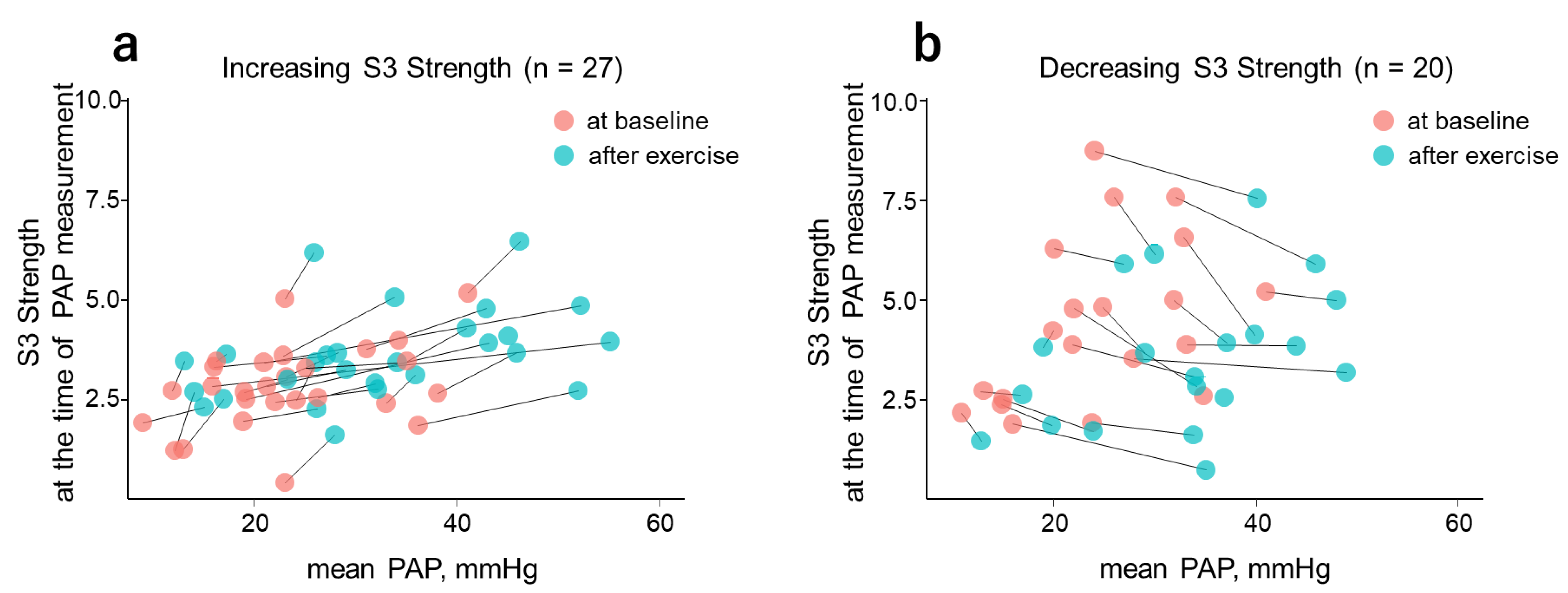
3.3. Differences in Background Factors between Exercise-Induced Increases and Decreases in CABs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure Developed by the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC) With the Special Contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Zile, M.R.; Bennett, T.D.; St. John Sutton, M.; Cho, Y.K.; Adamson, P.B.; Aaron, M.F.; Aranda, J.M.; Abraham, W.T.; Smart, F.W.; Stevenson, L.W.; et al. Transition from Chronic Compensated to Acute Compensated Heart Failure: Pathophysiological Insights Obtained from Continuous Monitoring of Intracardiac Pressures. Circulation 2008, 118, 1433–1441. [Google Scholar] [CrossRef]
- Desai, A.S.; Bhimaraj, A.; Bharmi, R.; Jermyn, R.; Bhatt, K.; Shavelle, D.; Redfield, M.M.; Hull, R.; Pelzel, J.; Davis, K.; et al. Ambulatory Hemodynamic Monitoring Reduces Heart Failure Hospitalizations in “Real-World” Clinical Practice. J. Am. Coll Cardiol. 2017, 69, 2357–2365. [Google Scholar] [CrossRef] [PubMed]
- Boehmer, J.P.; Hariharan, R.; Devecchi, F.G.; Smith, A.L.; Molon, G.; Capucci, A.; An, Q.; Averina, V.; Stolen, C.M.; Thakur, P.H.; et al. A Multisensor Algorithm Predicts Heart Failure Events in Patients With Implanted Devices: Results From the MultiSENSE Study. JACC Heart Fail. 2017, 5, 216–225. [Google Scholar] [CrossRef]
- Erath, J.W.; Wanczura, P.; Wranicz, J.; Linke, A.; Rohrer, U.; Scherr, D. Influence of Decompensated Heart Failure on Cardiac Acoustic Biomarkers: Impact on Early Readmissions. ESC Heart Fail 2020, 7, 4198–4205. [Google Scholar] [CrossRef] [PubMed]
- Burkhoff, D.; Bailey, G.; Gimbel, J.R. Characterization of Cardiac Acoustic Biomarkers in Patients with Heart Failure. Ann. Noninvasive Electrocardiol. 2020, 25, e12717. [Google Scholar] [CrossRef] [PubMed]
- Dillier, R.; Zuber, M.; Arand, P.; Erne, S.; Erne, P. Assessment of Systolic and Diastolic Function in Asymptomatic Subjects Using Ambulatory Monitoring With Acoustic Cardiography. Clin. Cardiol. 2011, 34, 384–388. [Google Scholar] [CrossRef]
- Collins, S.P.; Kontos, M.C.; Michaels, A.D.; Zuber, M.; Kipfer, P.; Jost, C.A.; Roos, M.; Jamshidi, P.; Erne, P.; Lindsell, C.J. Utility of a Bedside Acoustic Cardiographic Model to Predict Elevated Left Ventricular Filling Pressure. Emerg. Med. J. 2010, 27, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Marcus, G.M.; Gerber, I.L.; McKeown, B.H.; Vessey, J.C.; Jordan, M.V.; Huddleston, M.; McCulloch, C.E.; Foster, E.; Chatterjee, K.; Michaels, A.D. Association between Phonocardiographic Third and Fourth Heart Sounds and Objective Measures of Left Ventricular Function. JAMA 2005, 293, 2238–2244. [Google Scholar] [CrossRef]
- Shah, S.J.; Marcus, G.M.; Gerber, I.L.; McKeown, B.H.; Vessey, J.C.; Jordan, M.V.; Huddleston, M.; Foster, E.; Chatterjee, K.; Michaels, A.D. Physiology of The Third Heart Sound: Novel Insights from Tissue Doppler Imaging. J. Am. Soc. Echocardiogr. 2008, 21, 394–400. [Google Scholar] [CrossRef]
- Chan, W.; Woldeyohannes, M.; Colman, R.; Arand, P.; Michaels, A.D.; Parker, J.D.; Granton, J.T.; Mak, S. Haemodynamic and Structural Correlates of the First and Second Heart Sounds in Pulmonary Arterial Hypertension: An Acoustic Cardiography Cohort Study. BMJ Open 2013, 3, e002660. [Google Scholar] [CrossRef] [PubMed]
- Yamakawa, N.; Kotooka, N.; Kato, T.; Kuroda, T.; Node, K. Cardiac Acoustic Biomarkers as Surrogate Markers to Diagnose the Phenotypes of Pulmonary Hypertension: An Exploratory Study. Heart Vessel. 2022, 37, 593–600. [Google Scholar] [CrossRef]
- Manson, A.L.; Nudelman, S.P.; Hagley, M.T.; Hall, A.F.; Kovács, S.J. Relationship of the Third Heart Sound to Transmitral Flow Velocity Deceleration. Circulation 1995, 92, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Zuber, M.; Kipfer, P.; Jost, A.C. Systolic dysfunction: Correlation of acoustic cardiography with Doppler echocardiography. Congest. Heart Fail. 2006, 12 (Suppl. 1), 14–18. [Google Scholar] [CrossRef] [PubMed]
- Roos, M.; Toggweiler, S.; Zuber, M.; Jamshidi, P.; Erne, P. Acoustic cardiographic parameters and their relationship to invasive hemodynamic measurements in patients with left ventricular systolic dysfunction. Congest. Heart Fail. 2006, 12 (Suppl. 1), 19–24. [Google Scholar] [CrossRef] [PubMed]
- Moyers, B.; Shapiro, M.; Marcus, G.M.; Gerber, I.L.; McKeown, B.H.; Vessey, J.C.; Jordan, M.V.; Huddleston, M.; Foster, E.; Chatterjee, K.; et al. Performance of Phonoelectrocardiographic Left Ventricular Systolic Time Intervals and B-Type Natriuretic Peptide Levels in the Diagnosis of Left Ventricular Dysfunction. Ann. Noninvasive Electrocardiol. 2007, 12, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Colman, R.; Whittingham, H.; Tomlinson, G.; Granton, J. Utility of the Physical Examination in Detecting Pulmonary Hypertension. A Mixed Methods Study. PLoS ONE 2014, 9, e108499. [Google Scholar] [CrossRef] [PubMed]
- Shitara, J.; Kasai, T.; Murata, N.; Yamakawa, N.; Yatsu, S.; Murata, A.; Matsumoto, H.; Kato, T.; Suda, S.; Matsue, Y.; et al. Temporal Changes of Cardiac Acoustic Biomarkers and Cardiac Function in Acute Decompensated Heart Failure. ESC Heart Fail. 2021, 8, 4037–4047. [Google Scholar] [CrossRef]
- Gensini, G.F.; Alderighi, C.; Rasoini, R.; Mazzanti, M.; Casolo, G. Value of Telemonitoring and Telemedicine in Heart Failure Management. Card Fail. Rev. 2017, 3, 116. [Google Scholar] [CrossRef]
- Veenis, J.F.; Radhoe, S.P.; Hooijmans, P.; Brugts, J.J. Remote Monitoring in Chronic Heart Failure Patients: Is Non-Invasive Remote Monitoring the Way to Go? Sensors 2021, 21, 887. [Google Scholar] [CrossRef]
- Koehler, F.; Koehler, K.; Deckwart, O.; Prescher, S.; Wegscheider, K.; Kirwan, B.A.; Winkler, S.; Vettorazzi, E.; Bruch, L.; Oeff, M.; et al. Efficacy of Telemedical Interventional Management in Patients with Heart Failure (TIM-HF2): A Randomised, Controlled, Parallel-Group, Unmasked Trial. Lancet 2018, 392, 1047–1057. [Google Scholar] [CrossRef]
- Stehlik, J.; Schmalfuss, C.; Bozkurt, B.; Nativi-Nicolau, J.; Wohlfahrt, P.; Wegerich, S.; Rose, K.; Ray, R.; Schofield, R.; Deswal, A.; et al. Continuous Wearable Monitoring Analytics Predict Heart Failure Hospitalization. Circ. Heart Fail. 2020, 13, e006513. [Google Scholar] [CrossRef]
- Tanaka, A.; Node, K. Call for Fine Remote Monitoring of Prognostic Signs and Symptoms in Heart Failure. JACC Heart Fail. 2021, 9, 530. [Google Scholar] [CrossRef]

| Variable | Overall (n = 49) |
|---|---|
| Age, year, mean ± SD | 68.1 ± 11.2 |
| Male, number (%) | 37 (75.5) |
| BMI, kg/m2, mean ± SD | 23.8 ± 4.3 |
| eGFR, mL/min/1.73 m2, mean ± SD | 52.6 ± 20.1 |
| NT-proBNP, median [inter quartile range] | 1353.0 [429.1, 2044.0] |
| LVEF, %, mean ± SD | 36.3 ± 16.5 |
| <40%, number (%) | 32 (65.3) |
| 40–49%, number (%) | 7 (14.3) |
| ≥50%, number (%) | 10 (20.4) |
| E/e’, mean ± SD | 13.8 ± 4.8 |
| NYHA Functional classification, number (%) | |
| 1 | 4 (8.2) |
| 2 | 10 (20.4) |
| 3 | 31 (63.3) |
| 4 | 4 (8.2) |
| Etiology, number (%) | |
| Dilated cardiomyopathy | 8 (16.3) |
| Ischemia | 7 (14.3) |
| Arrhythmia | 6 (12.2) |
| Valvular | 5 (10.2) |
| Hypertensive | 2 (4.1) |
| Amyloidosis | 2 (4.1) |
| Sarcoidosis | 1 (2.0) |
| Others/unknown | 18 (36.7) |
| Implantable device, number (%) | 5 (10.2) |
| Comorbidity, number (%) | |
| Coronary artery disease | 5 (10.2) |
| Atrial fibrillation | 20 (38.8) |
| Stroke | 4 (8.2) |
| Diabetes mellitus | 17 (34.7) |
| Hypertension | 28 (57.1) |
| Medication, number (%) | |
| RAS inhibitor | 41 (83.7) |
| Diuretic | 39 (79.6) |
| β-blocker | 34 (69.4) |
| Calcium channel blocker | 8 (16.3) |
| At Baseline | After Exercise | p Value | |
|---|---|---|---|
| Right Heart Catheterization | |||
| Mean PAP, mmHg | 23.52 ± 8.41 | 32.45 ± 11.28 | <0.001 |
| Mean PCWP, mmHg | 15.39 ± 8.02 | 16.96 ± 7.96 | 0.335 |
| CABs at PAP measurement | |||
| HR, bpm | 72.92 ± 13.05 | 75.93 ± 11.47 | 0.469 |
| S1 Intensity, mV | 7.68 ± 4.71 | 8.02 ± 4.13 | 0.704 |
| S1 Width, ms | 175.72 ± 33.92 | 168.36 ± 32.71 | 0.500 |
| S1 Complexity | 2.92 ± 0.94 | 2.98 ± 0.71 | 0.511 |
| S2 Intensity, mV | 6.17 ± 3.21 | 6.49 ± 2.97 | 0.829 |
| S2 Width, ms | 114.95 ± 21.14 | 111.77 ± 18.94 | 0.656 |
| S2 Complexity | 1.93 ± 1.04 | 2.05 ± 1.13 | 0.430 |
| S3 Strength | 3.51 ± 1.72 | 3.60 ± 1.42 | 0.849 |
| S3 Intensity, mV | 1.30 ± 0.46 | 1.39 ± 0.39 | 0.464 |
| S4 Strength | 3.93 ± 1.61 | 4.67 ± 1.93 | 0.221 |
| S4 Intensity, mV | 1.09 ± 0.46 | 1.54 ± 0.92 | 0.044 |
| S2/S1 Intensity | 0.93 ± 0.53 | 0.93 ± 0.49 | 0.916 |
| S2/S1 Complexity | 0.69 ± 0.36 | 0.73 ± 0.45 | 0.408 |
| CABs at PCWP measurement | |||
| HR, bpm | 71.91 ± 11.44 | 74.06 ± 10.96 | 0.347 |
| S1 Intensity, mV | 7.56 ± 4.16 | 8.63 ± 4.87 | 0.248 |
| S1 Width, ms | 175.89 ± 33.33 | 171.94 ± 31.17 | 0.548 |
| S1 Complexity | 3.03 ± 0.91 | 2.75 ± 0.93 | 0.143 |
| S2 Intensity, mV | 6.25 ± 3.26 | 7.10 ± 4.10 | 0.262 |
| S2 Width, ms | 116.21 ± 23.74 | 110.85 ± 18.71 | 0.221 |
| S2 Complexity | 2.07 ± 1.22 | 1.66 ± 1.01 | 0.080 |
| S3 Strength | 3.75 ± 1.72 | 3.63 ± 1.58 | 0.720 |
| S3 Intensity, mV | 1.28 ± 0.39 | 1.82 ± 2.65 | 0.169 |
| S4 Strength | 4.20 ± 1.80 | 4.13 ± 1.98 | 0.892 |
| S4 Intensity, mV | 1.21 ± 0.82 | 1.42 ± 0.73 | 0.287 |
| S2/S1 Intensity | 0.96 ± 0.57 | 0.96 ± 0.56 | 0.959 |
| S2/S1 Complexity | 0.81 ± 0.84 | 0.62 ± 0.34 | 0.159 |
| Mean PAP | ||||
|---|---|---|---|---|
| At Baseline | After Exercise | |||
| n | r (95% Confidence Interval) | n | r (95% Confidence Interval) | |
| HR, bpm | 48 | 0.212 (−0.077, 0.468) | 49 | 0.159 (−0.127, 0.422) |
| S1 Intensity, mV | 48 | −0.153 (−0.419, 0.137) | 49 | −0.176 (−0.435, 0.111) |
| S1 Width, ms | 48 | −0.27 (−0.514, 0.016) | 49 | −0.189 (−0.446, 0.098) |
| S1 Complexity | 48 | −0.207 (−0.464, 0.082) | 49 | −0.251 (−0.497, 0.032) |
| S2 Intensity, mV | 48 | 0.247 (−0.040, 0.496) | 49 | 0.185 (−0.102, 0.443) |
| S2 Width, ms | 48 | 0.354 (0.078, 0.580) * | 49 | 0.363 (0.091, 0.584) * |
| S2 Complexity | 48 | 0.249 (−0.038, 0.498) | 49 | 0.312 (0.034, 0.545) * |
| S3 Strength | 48 | 0.375 (0.102, 0.596) * | 48 | 0.386 (0.114, 0.604) * |
| S3 Intensity, mV | 47 | 0.335 (0.053, 0.568) * | 46 | 0.270 (−0.022, 0.520) |
| S4 Strength | 34 | −0.075 (−0.403, 0.270) | 29 | −0.166 (−0.502, 0.214) |
| S4 Intensity, mV | 34 | 0.109 (−0.238, 0.431) | 27 | −0.079 (−0.446, 0.31) |
| S2/S1 Intensity | 48 | 0.296 (0.013, 0.535) * | 49 | 0.259 (−0.024, 0.504) |
| S2/S1 Complexity | 48 | 0.267 (−0.019, 0.512) | 49 | 0.360 (0.087, 0.582) * |
| Mean PAP | ||
|---|---|---|
| n | r (95% Confidence Interval) | |
| HR, bpm | 48 | 0.205 (−0.084, 0.462) |
| S1 Intensity, mV | 48 | 0.009 (−0.276, 0.293) |
| S1 Width, ms | 48 | 0.029 (−0.258, 0.310) |
| S1 Complexity | 48 | 0.051 (−0.237, 0.330) |
| S2 Intensity, mV | 48 | −0.027 (−0.309, 0.259) |
| S2 Width, ms | 48 | −0.058 (−0.337, 0.230) |
| S2 Complexity | 48 | 0.173 (−0.117, 0.436) |
| S3 Strength | 47 | 0.089 (−0.204, 0.367) |
| S3 Intensity, mV | 45 | −0.005 (−0.299, 0.289) |
| S4 Strength | 28 | −0.057 (−0.421, 0.323) |
| S4 Intensity, mV | 27 | −0.033 (−0.408, 0.351) |
| S2/S1 Intensity | 48 | −0.011 (−0.294, 0.274) |
| S2/S1 Complexity | 48 | 0.145 (−0.146, 0.412) |
| S3 Strength Response | |||||
|---|---|---|---|---|---|
| Overall | Decreasing (n = 20) | Increasing (n = 27) | p Value | ||
| Age, number (%) | |||||
| (median) | <69 years | 24 (51.1) | 12 (60.0) | 12 (44.4) | 0.380 |
| ≥69 years | 23 (48.9) | 8 (40.0) | 15 (55.6) | ||
| BMI, number (%) | |||||
| (median) | <24.0 kg/m2 | 24 (51.1) | 12 (60.0) | 12 (44.4) | 0.380 |
| ≥24.0 kg/m2 | 23 (48.9) | 8 (40.0) | 15 (55.6) | ||
| eGFR, number (%) | |||||
| <60.0 mL/min/1.73 m2 | 32 (68.1) | 12 (60.0) | 20 (74.1) | 0.355 | |
| ≥60.0 mL/min/1.73 m2 | 15 (31.9) | 8 (40.0) | 7 (25.9) | ||
| NT-proBNP, number (%) | |||||
| (median) | <1353 pg/mL | 15 (51.7) | 6 (50.0) | 9 (52.9) | 1.000 |
| ≥1353 pg/mL | 14 (48.3) | 6 (50.0) | 8 (47.1) | ||
| Atrial fibrillation, number (%) | |||||
| Yes | 18 (38.3) | 5 (25.0) | 13 (48.1) | 0.137 | |
| No | 29 (61.7) | 15 (75.0) | 14 (51.9) | ||
| Diabetes mellitus, number (%) | |||||
| Yes | 16 (34.0) | 6 (30.0) | 10 (37.0) | 0.758 | |
| No | 31 (66.0) | 14 (70.0) | 17 (63.0) | ||
| Hypertension, number (%) | |||||
| Yes | 26 (55.3) | 9 (45.0) | 17 (63.0) | 0.250 | |
| No | 21 (44.7) | 11 (55.0) | 10 (37.0) | ||
| β-blocker use, number (%) | |||||
| Yes | 34 (72.3) | 6 (30.0) | 7 (25.9) | 1.000 | |
| No | 13 (27.7) | 14 (70.0) | 20 (74.1) | ||
| LVEF, number (%) | |||||
| <40% | 30 (63.8) | 13 (65.0) | 17 (63.0) | 0.573 | |
| 40–49% | 7 (14.9) | 4 (20.0) | 3 (11.1) | ||
| ≥50% | 10 (21.3) | 3 (15.0) | 7 (25.9) | ||
| E/e’, number (%) | |||||
| <14 | 29 (61.7) | 11 (55) | 18 (66.7) | 0.546 | |
| ≥14 | 18 (38.3) | 9 (45) | 9 (33.3) | ||
| Cardiac index, number (%) | |||||
| <2.2 mL/m2 | 24 (51.1) | 14 (70.0) | 10 (37.0) | 0.039 | |
| ≥2.2 mL/m2 | 23 (48.9) | 6 (30.0) | 17 (63.0) | ||
| Mean PCWP, number (%) | |||||
| <15 mmHg | 23 (48.9) | 8 (40.0) | 15 (55.6) | 0.380 | |
| ≥15 mmHg | 24 (51.1) | 12 (60.0) | 12 (44.4) | ||
| Mean PAP, number (%) | |||||
| ≤20 mmHg | 17 (36.2) | 7 (35) | 10 (37) | 1.000 | |
| >20 mmHg | 30 (63.8) | 13 (65) | 17 (63) | ||
| PH, number (%) | |||||
| Yes (Ipc-PH or Cpc-PH) | 23 (51.1) | 11 (57.9) | 12 (46.2) | 0.550 | |
| No | 22 (48.9) | 8 (42.1) | 14 (53.8) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaneko, T.; Tanaka, A.; Jojima, K.; Yoshida, H.; Yajima, A.; Asaka, M.; Yamakawa, N.; Kato, T.; Kotooka, N.; Node, K. Relationship between Cardiac Acoustic Biomarkers and Pulmonary Artery Pressure in Patients with Heart Failure. J. Clin. Med. 2022, 11, 6373. https://doi.org/10.3390/jcm11216373
Kaneko T, Tanaka A, Jojima K, Yoshida H, Yajima A, Asaka M, Yamakawa N, Kato T, Kotooka N, Node K. Relationship between Cardiac Acoustic Biomarkers and Pulmonary Artery Pressure in Patients with Heart Failure. Journal of Clinical Medicine. 2022; 11(21):6373. https://doi.org/10.3390/jcm11216373
Chicago/Turabian StyleKaneko, Tetsuya, Atsushi Tanaka, Kota Jojima, Hisako Yoshida, Ayumu Yajima, Machiko Asaka, Nobuhide Yamakawa, Tomoyuki Kato, Norihiko Kotooka, and Koichi Node. 2022. "Relationship between Cardiac Acoustic Biomarkers and Pulmonary Artery Pressure in Patients with Heart Failure" Journal of Clinical Medicine 11, no. 21: 6373. https://doi.org/10.3390/jcm11216373
APA StyleKaneko, T., Tanaka, A., Jojima, K., Yoshida, H., Yajima, A., Asaka, M., Yamakawa, N., Kato, T., Kotooka, N., & Node, K. (2022). Relationship between Cardiac Acoustic Biomarkers and Pulmonary Artery Pressure in Patients with Heart Failure. Journal of Clinical Medicine, 11(21), 6373. https://doi.org/10.3390/jcm11216373






