Pathological Findings in COVID-19 and Non-COVID-19 Vaccine-Associated Lymphadenopathy: A Systematic Review
Abstract
1. Introduction
2. Material and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Analysis
3. Results
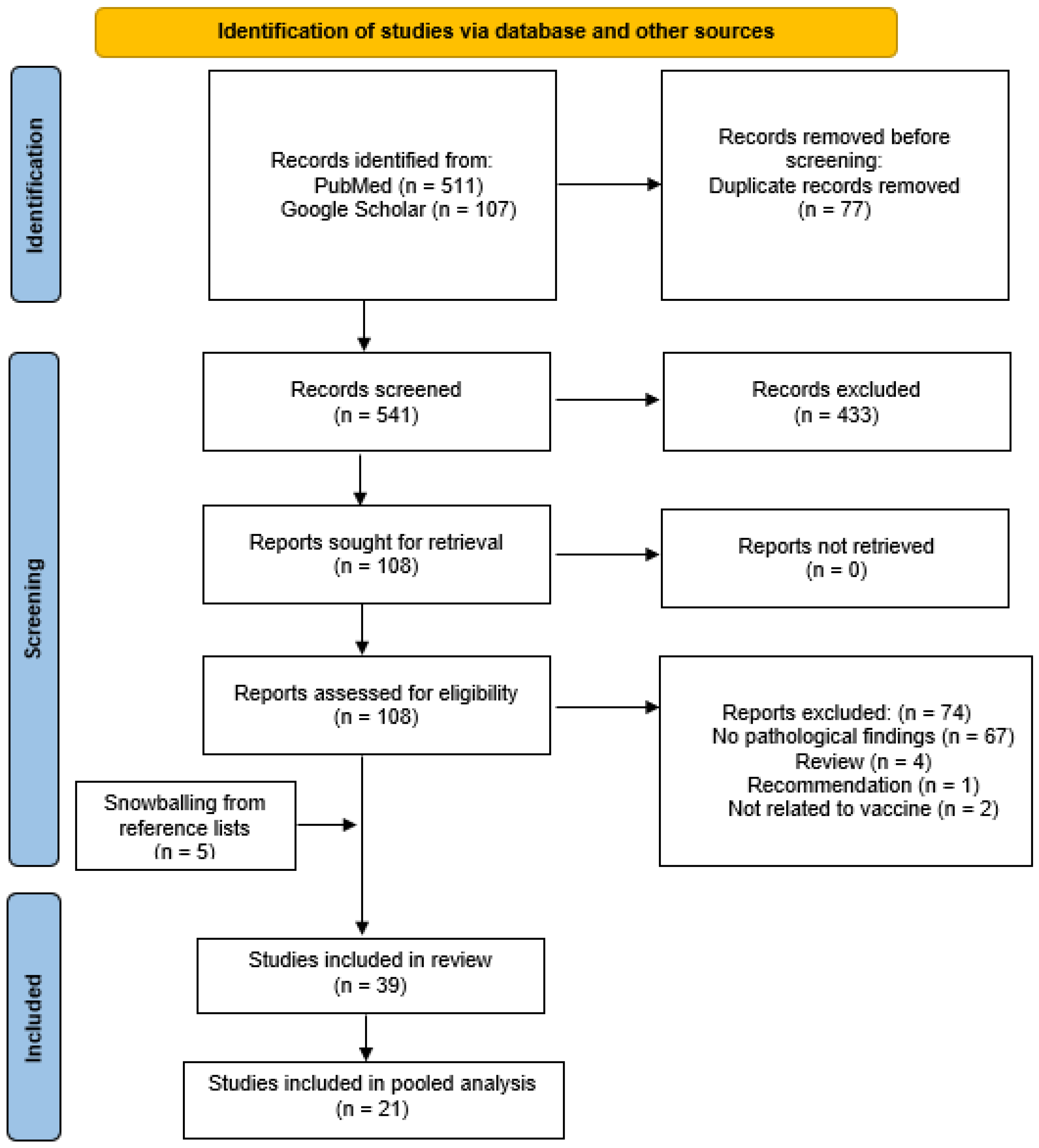
3.1. Literature Search
3.2. COVID-19 Vaccine-Associated Lymphadenopathy Study Characteristics
3.3. Pooled Analysis of COVID-19 Vaccine-Associated Lymphadenopathy
3.4. Clinical Lymphadenopathy (CL)
3.5. Subclinical Lymphadenopathy Detected on Imaging (SLDI)
3.6. Non-COVID-19 Vaccine-Associated Lymphadenopathy
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Johns Hopkins Coronavirus Resource Center. COVID-19 Map. 2021. Available online: https://coronavirus.jhu.edu/map.html (accessed on 4 January 2022).
- Administration FaD. FDA Approves First COVID-19 Vaccine. 2021. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-covid-19-vaccine (accessed on 4 January 2022).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Thomas, S.J.; Moreira, E.D., Jr.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Polack, F.P.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine through 6 Months. N. Engl. J. Med. 2021, 385, 1761–1773. [Google Scholar] [CrossRef]
- Prevention CfDCa. Vaccines and Immunizations: Reactions and Adverse Reactions. 2021. Available online: https://www.cdc.gov/vaccines/covid-19/info-by-product/moderna/reactogenicity.html (accessed on 4 January 2022).
- Hartsock, R.J. Postvaccinial lymphadenitis. Hyperplasia of lymphoid tissue that simulates malignant lymphomas. Cancer 1968, 21, 632–649. [Google Scholar] [CrossRef]
- Aelami, M.H.; Alborzi, A.; Pouladfar, G.; Geramizadeh, B.; Pourabbas, B.; Mardaneh, J. Post-Vaccination Disseminated Bacillus Calmette Guerin Infection Among Children in Southern Iran. Jundishapur J. Microbiol. 2015, 8, e25663. [Google Scholar] [CrossRef]
- Dotlic, S.; Vranic, S.; Jakovljevic, G.; Ilic, I.; Kardum-Paro, M.M.; Dojcinov, S.D. Neonatal hyperimmune T-cell reaction mimicking T-cell non-Hodgkin’s lymphoma following BCG and hepatitis B co-vaccination. Virchows Arch. 2012, 461, 601–605. [Google Scholar] [CrossRef]
- Watanabe, T.; Hashidate, H.; Hirayama, Y.; Iinuma, Y. Kikuchi-Fujimoto disease following vaccination against human papilloma virus infection and Japanese encephalitis. Eur. J. Pediatr. 2012, 171, 1409–1411. [Google Scholar] [CrossRef]
- White, C.K.; Al-Saleem, T.; Skarbnik, A.P.; Smith, M.R. Tetanus toxoid reactive lymphadenopathy masquerading as T-cell lymphoma. Future Oncol. 2012, 8, 631–634. [Google Scholar] [CrossRef]
- Bettini, E.; Locci, M. SARS-CoV-2 mRNA Vaccines: Immunological Mechanism and Beyond. Vaccines 2021, 9, 147. [Google Scholar] [CrossRef]
- Turner, J.S.; O’Halloran, J.A.; Kalaidina, E.; Kim, W.; Schmitz, A.J.; Zhou, J.Q.; Lei, T.; Thapa, M.; Chen, R.E.; Case, J.B.; et al. SARS-CoV-2 mRNA vaccines induce persistent human germinal centre responses. Nature 2021, 596, 109–113. [Google Scholar] [CrossRef]
- Lederer, K.; Castaño, D.; Atria, D.G.; Oguin, T.H., III; Wang, S.; Manzoni, T.B.; Muramatsu, H.; Hogan, M.J.; Amanat, F.; Cherubin, P.; et al. SARS-CoV-2 mRNA Vaccines Foster Potent Antigen-Specific Germinal Center Responses Associated with Neutralizing Antibody Generation. Immunity 2020, 53, 1281–1295.e5. [Google Scholar] [CrossRef]
- Lehman, C.D.; D’Alessandro, H.A.; Mendoza, D.P.; Succi, M.D.; Kambadakone, A.; Lamb, L.R. Unilateral Lymphadenopathy After COVID-19 Vaccination: A Practical Management Plan for Radiologists Across Specialties. J. Am. Coll. Radiol. 2021, 18, 843–852. [Google Scholar] [CrossRef]
- Treglia, G.; Cuzzocrea, M.; Giovanella, L.; Elzi, L.; Muoio, B. Prevalence and Significance of Hypermetabolic Lymph Nodes Detected by 2-[(18)F]FDG PET/CT after COVID-19 Vaccination: A Systematic Review and a Meta-Analysis. Pharmaceuticals 2021, 14, 762. [Google Scholar] [CrossRef]
- Keshavarz, P.; Yazdanpanah, F.; Rafiee, F.; Mizandari, M. Lymphadenopathy Following COVID-19 Vaccination: Imaging Findings Review. Acad. Radiol. 2021, 28, 1058–1071. [Google Scholar] [CrossRef]
- Placke, J.-M.; Reis, H.; Hadaschik, E.; Roesch, A.; Schadendorf, D.; Stoffels, I.; Klode, J. Coronavirus disease 2019 vaccine mimics lymph node metastases in patients undergoing skin cancer follow-up: A monocentre study. Eur. J. Cancer 2021, 154, 167–174. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Moher, D. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J. Clin. Epidemiol. 2021, 134, 103–112. [Google Scholar] [CrossRef]
- Cardoso, F.; Reis, A.; Osório, C.; Scigliano, H.; Nora, M. A Case of Cervical Lymphadenopathy After Vaccination Against COVID-19. Cureus 2021, 13, e15050. [Google Scholar] [CrossRef]
- Hagen, C.; Nowack, M.; Messerli, M.; Saro, F.; Mangold, F.; Bode, P.K. Fine needle aspiration in COVID-19 vaccine-associated lymphadenopathy. Swiss Med. Wkly. 2021, 151, w20557. [Google Scholar] [CrossRef]
- Faermann, R.; Nissan, N.; Halshtok-Neiman, O.; Shalmon, A.; Gotlieb, M.; Yagil, Y.; Samoocha, D.; Friedman, E.; Sklair-Levy, M. COVID-19 Vaccination Induced Lymphadenopathy in a Specialized Breast Imaging Clinic in Israel: Analysis of 163 cases. Acad. Radiol. 2021, 28, 1191–1197. [Google Scholar] [CrossRef]
- Felices-Farias, J.M.; Martínez-Martínez, J.F.; Guzmán-Aroca, F. Unusual lymphadenopathies secondary to the BNT162b2 mRNA COVID-19 vaccine. Med. Clin. 2021, 158, 343. [Google Scholar] [CrossRef]
- Fernández-Prada, M.; Rivero-Calle, I.; Calvache-González, A.; Martinón-Torres, F. Acute onset supraclavicular lymphadenopathy coinciding with intramuscular mRNA vaccination against COVID-19 may be related to vaccine injection technique, Spain, January and February 2021. Eurosurveillance 2021, 26, 2100193. [Google Scholar] [CrossRef] [PubMed]
- Ganga, K.; Solyar, A.Y.; Ganga, R. Massive Cervical Lymphadenopathy Post-COVID-19 Vaccination. Ear Nose Throat J. 2021. [Google Scholar] [CrossRef]
- Kado, S.; Kamiya, K.; Iwabuchi, S.; Kajii, E.; Ohtsuki, M. Unilateral lymphadenopathy associated with COVID-19 vaccination. J. Cutan. Immunol. Allergy 2021, 5, 100–101. [Google Scholar] [CrossRef]
- Kim, B.; Park, Y.; Kim, E.K.; Lee, S.H. Supraclavicular lymphadenopathy after COVID-19 vaccination in Korea: Serial follow-up using ultrasonography. Clin. Imaging 2021, 79, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Larkin, K.; Sharma, A.; Salaru, G.; Drachtman, R. Supraclavicular Lymphadenopathy after COVID-19 vaccination. Authorea Prepr. 2021. Available online: https://www.authorea.com/users/444098/articles/543906-supraclavicular-lymphadenopathy-after-covid-19-vaccination (accessed on 4 January 2022). [CrossRef]
- Özütemiz, C.; Krystosek, L.A.; Church, A.L.; Chauhan, A.; Ellermann, J.M.; Domingo-Musibay, E.; Steinberger, D. Lymphadenopathy in COVID-19 vaccine recipients: Diagnostic dilemma in oncologic Patients. Radiology 2021, 300, E296–E300. [Google Scholar] [CrossRef]
- Park, J.Y.; Yi, S.Y. Rare case of contralateral supraclavicular lymphadenopathy after COVID-19 vaccination: Computed tomography and ultrasonography findings. Radiol. Case Rep. 2021, 16, 3879–3881. [Google Scholar] [CrossRef]
- Al Soub, H.; Ibrahim, W.; Al Maslamani, M.; Ali, G.A.; Ummer, W. Kikuchi-Fujimoto disease following SARS CoV2 vaccination: Case report. IDCases 2021, 25, e01253. [Google Scholar] [CrossRef]
- Tan, N.J.H.; Tay, K.X.J.; Wong, S.B.J.; Nga, M.E. COVID-19 post-vaccination lymphadenopathy: Report of cytological findings from fine needle aspiration biopsy. Diagn. Cytopathol. 2021, 49, E467–E470. [Google Scholar] [CrossRef]
- Tan, H.M.; Hue, S.S.; Wee, A.; See, K.C. Kikuchi-Fujimoto Disease Post COVID-19 Vaccination: Case Report and Review of Literature. Vaccines 2021, 9, 1251. [Google Scholar] [CrossRef]
- Tintle, S.; Chen, M. Lymphadenopathy with florid lymphoid and Langerhans cell hyperplasia and hemophagocytosis mimicking lymphoma after COVID-19 mRNA vaccination. eJHaem 2021, 2, 845–847. [Google Scholar] [CrossRef] [PubMed]
- Aalberg, J.J.; Collins, T.P.; Dobrow, E.M. Axillary lymphadenopathy in a renal cell carcinoma patient after COVID-19 Vaccination. Radiol. Case Rep. 2021, 16, 2164–2167. [Google Scholar] [CrossRef] [PubMed]
- Ashoor, A.; Shephard, J.; Lissidini, G.; Nicosia, L. Axillary Adenopathy in Patients with Recent COVID-19 Vaccination: A New Diagnostic Dilemma. Korean J. Radiol. 2021, 22, 2124–2126. [Google Scholar] [CrossRef]
- Eifer, M.; Tau, N.; Alhoubani, Y.; Kanana, N.; Domachevsky, L.; Shams, J.; Keret, N.; Gorfine, M.; Eshet, Y. COVID-19 mRNA Vaccination: Age and Immune Status and its Association with Axillary Lymph Node PET/CT Uptake. J. Nucl. Med. 2021, 63, 134–139. [Google Scholar] [CrossRef]
- Lane, D.L.; Neelapu, S.S.; Xu, G.; Weaver, O. COVID-19 Vaccine-Related Axillary and Cervical Lymphadenopathy in Patients with Current or Prior Breast Cancer and Other Malignancies: Cross-Sectional Imaging Findings on MRI, CT, and PET-CT. Korean J. Radiol. 2021, 22, 1938–1945. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Lee, S.A.; Khil, E.K.; Byeon, S.J.; Kang, H.J.; Choi, J.A. COVID-19 vaccine-related axillary lymphadenopathy in breast cancer patients: Case series with a review of literature. Semin. Oncol. 2021, 48, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Prieto, P.A.; Mannava, K.; Sahasrabudhe, D.M. COVID-19 mRNA vaccine-related adenopathy mimicking metastatic melanoma. Lancet Oncol. 2021, 22, e281. [Google Scholar] [CrossRef]
- Pudis, M.; Vercher Conejero, J.L.; Martín Marcuartu, J.J.; Cortés Romera, M. 68Ga-DOTATOC-avid lymphadenopathies induced from COVID-19 mRNA vaccination. Jpn. J. Clin. Oncol. 2021, 51, 1765. [Google Scholar] [CrossRef]
- Trikannad, A.; Vellanki, S.; Kunapareddy, G. Mediastinal Lymphadenopathy after COVID-19 Vaccine: Staging Dilemma in Oncology Patients. Chest 2021, 160, A1460. [Google Scholar] [CrossRef]
- Robinson, K.A.; Maimone, S.; Gococo-Benore, D.A.; Li, Z.; Advani, P.P.; Chumsri, S. Incidence of Axillary Adenopathy in Breast Imaging After COVID-19 Vaccination. JAMA Oncol. 2021, 7, 1395–1397. [Google Scholar] [CrossRef]
- Barouni, A.S.; Augusto, C.; Queiroz, M.V.; Lopes, M.T.; Zanini, M.S.; Salas, C.E. BCG lymphadenopathy detected in a BCG-vaccinated infant. Braz. J. Med. Biol. Res. 2004, 37, 697–700. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Biers, S.M.; Di Marco, A.; Mostafid, A.H. Case report: Differential diagnosis of isolated iliac lymphadenopathy following Bacillus Calmette-Guérin treatment for high-risk superficial bladder cancer. Int. Urol. Nephrol. 2007, 39, 1039–1041. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Chakarabarti, S.; Phukan, J.P.; Biswas, S.; Sinha, A.; Sinha, R. Role of needle aspiration in diagnosis and management of suppurative bacille calmette-guerin adenitis: An institutional study of 30 cases. J. Lab. Physicians 2015, 7, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.C.; Wu, H.J.; Yong, S.B. Bacillus Calmette-Guérin vaccination-associated axillary lymphadenopathy in a 2-year-old girl: Case report. J. Formos. Med. Assoc. 2019, 118, 533–534. [Google Scholar] [CrossRef] [PubMed]
- Toy, H.; Karasoy, D.; Keser, M. Lymphadenitis caused by H1N1 vaccination: Case report. Vaccine 2010, 28, 2158–2160. [Google Scholar] [CrossRef]
- Pereira, M.P.; Flores, P.; Neto, A.S. Neck and supraclavicular lymphadenopathy secondary to 9-valent human papillomavirus vaccination. BMJ Case Rep. 2019, 12, e231582. [Google Scholar] [CrossRef]
- Dorfman, R.F.; Herweg, J.C. Live, attenuated measles virus vaccine. Inguinal lymphadenopathy complicating administration. JAMA 1966, 198, 320–321. [Google Scholar] [CrossRef]
- Sumaya, C.V.; Cherry, J.D.; Gohd, R. Exaggerated antibody response following rubella vaccination in a child with sinus histiocytosis with massive lymphadenopathy. J. Pediatr. 1976, 89, 81–83. [Google Scholar] [CrossRef]
- White, C. Tetanus vaccine Supraclavicular lymphadenopathy: Case report. Reactions 2012, 1415, 18. [Google Scholar]
- Gupta, K.; Singh, N.; Bhatia, A.; Arora, V.K.; Singh, U.R.; Singh, B. Cytomorphologic patterns in Calmette Guerin bacillus lymphadenitis. Acta Cytol. 1997, 41, 348–350. [Google Scholar] [CrossRef]
- Bosch, X.; Guilabert, A.; Miquel, R.; Campo, E. Enigmatic Kikuchi-Fujimoto disease: A comprehensive review. Am. J. Clin. Pathol. 2004, 122, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, C.B.; Wang, E. Kikuchi-Fujimoto disease. Arch. Pathol. Lab. Med. 2010, 134, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Bosch, X.; Guilabert, A. Kikuchi-Fujimoto disease. Orphanet J. Rare Dis. 2006, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Garreffa, E.; Hamad, A.; O’Sullivan, C.C.; Hazim, A.Z.; York, J.; Puri, S.; Turnbull, A.; Robertson, J.F.; Goetz, M.P. Regional lymphadenopathy following COVID-19 vaccination: Literature review and considerations for patient management in breast cancer care. Eur. J. Cancer 2021, 159, 38–51. [Google Scholar] [CrossRef]
- Bshesh, K.; Khan, W.; Vattoth, A.L.; Janjua, E.; Nauman, A.; Almasri, M.; Ali, A.M.; Ramadorai, V.; Mushannen, B.; Al Subaie, M.; et al. Lymphadenopathy post-COVID-19 vaccination with increased FDG uptake may be falsely attributed to oncological disorders: A systematic review. J. Med. Virol. 2022, 94, 1833–1845. [Google Scholar] [CrossRef]
- Brown, A.; Shah, S.; Dluzewski, S.; Musaddaq, B.; Wagner, T.; Szyszko, T.; Wan, S.; Groves, A.; Mokbel, K.; Malhotra, A. Unilateral axillary adenopathy following COVID-19 vaccination: A multimodality pictorial illustration and review of current guidelines. Clin. Radiol. 2021, 76, 553–558. [Google Scholar] [CrossRef]
- Schiaffino, S.; Pinker, K.; Magni, V.; Cozzi, A.; Athanasiou, A.; Baltzer, P.A.; Camps Herrero, J.; Clauser, P.; Fallenberg, E.M.; Forrai, G.; et al. Axillary lymphadenopathy at the time of COVID-19 vaccination: Ten recommendations from the European Society of Breast Imaging (EUSOBI). Insights Imaging 2021, 12, 119. [Google Scholar] [CrossRef]
- Samkowski, J.; Sklinda, K.; Walecki, J.M. Lymphadenopathy in the era of COVID-19 vaccination—An oncological dilemma in diagnostic imaging. Pol. J. Radiol. 2022, 87, e304–e310. [Google Scholar] [CrossRef]
- Van Nijnatten, T.J.A.; Jochelson, M.S.; Lobbes, M.B.I. Axillary lymph node characteristics in breast cancer patients versus post-COVID-19 vaccination: Overview of current evidence per imaging modality. Eur. J. Radiol. 2022, 152, 110334. [Google Scholar] [CrossRef]
- McIntosh, L.J.; Bankier, A.A.; Vijayaraghavan, G.R.; Licho, R.; Rosen, M.P. COVID-19 Vaccination-Related Uptake on FDG PET/CT: An Emerging Dilemma and Suggestions for Management. AJR Am. J. Roentgenol. 2021, 217, 975–983. [Google Scholar] [CrossRef]
- Pollard, A.J.; Bijker, E.M. A guide to vaccinology: From basic principles to new developments. Nat. Rev. Immunol. 2021, 21, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Sadarangani, M.; Marchant, A.; Kollmann, T.R. Immunological mechanisms of vaccine-induced protection against COVID-19 in humans. Nat. Rev. Immunol. 2021, 21, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Mascellino, M.T.; Di Timoteo, F.; de Angelis, M.; Oliva, A. Overview of the Main Anti-SARS-CoV-2 Vaccines: Mechanism of Action, Efficacy and Safety. Infect. Drug Resist. 2021, 14, 3459–3476. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A.J.; Netea, M.G. BCG-induced trained immunity: Can it offer protection against COVID-19? Nat. Rev. Immunol. 2020, 20, 335–337. [Google Scholar] [CrossRef] [PubMed]
- Curtis, N.; Sparrow, A.; Ghebreyesus, T.A.; Netea, M.G. Considering BCG vaccination to reduce the impact of COVID-19. Lancet 2020, 395, 1545–1546. [Google Scholar] [CrossRef]
- Sekizawa, A.; Hashimoto, K.; Kobayashi, S.; Kozono, S.; Kobayashi, T.; Kawamura, Y.; Kimata, M.; Fujita, N.; Ono, Y.; Obuchi, Y.; et al. Rapid progression of marginal zone B-cell lymphoma after COVID-19 vaccination (BNT162b2): A case report. Front. Med. 2022, 9, 963393. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.-R.; Hsu, C.-W.; Lee, C.-C.; Huang, W.-L.; Lin, C.-Y.; Hsu, Y.-T.; Chang, C.; Tsai, M.-T.; Hu, Y.-N.; Hsu, C.-H.; et al. A Case Report of Posttransplant Lymphoproliferative Disorder After AstraZeneca Coronavirus Disease 2019 Vaccine in a Heart Transplant Recipient. Transplant. Proc. 2022, 54, 1575–1578. [Google Scholar] [CrossRef]
- Goldman, S.; Bron, D.; Tousseyn, T.; Vierasu, I.; Dewispelaere, L.; Heimann, P.; Cogan, E.; Goldman, M. Rapid Progression of Angioimmunoblastic T Cell Lymphoma Following BNT162b2 mRNA Vaccine Booster Shot: A Case Report. Front. Med. 2021, 8, 798095. [Google Scholar] [CrossRef]
- Rocco, J.M.; Mallarino-Haeger, C.; Randolph, A.H.; Ray, S.M.; Schechter, M.C.; Zerbe, C.S.; Holland, S.M.; Sereti, I. Hyperinflammatory Syndromes After Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Messenger RNA vaccination in Individuals With Underlying Immune Dysregulation. Clin. Infect. Dis. 2022, 75, e912–e915. [Google Scholar] [CrossRef]
- Buscarlet, M.; Provost, S.; Zada, Y.F.; Barhdadi, A.; Bourgoin, V.; Lépine, G.; Mollica, L.; Szuber, N.; Dubé, M.-P.; Busque, L. DNMT3A and TET2 dominate clonal hematopoiesis and demonstrate benign phenotypes and different genetic predispositions. Blood 2017, 130, 753–762. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Update on Omicron. 2021. Available online: https://www.who.int/news/item/28-11-2021-update-on-omicron (accessed on 4 January 2022).
- Collie, S.; Champion, J.; Moultrie, H.; Bekker, L.G.; Gray, G. Effectiveness of BNT162b2 Vaccine against Omicron Variant in South Africa. N. Engl. J. Med. 2021, 386, 494–496. [Google Scholar] [CrossRef] [PubMed]
- Barda, N.; Dagan, N.; Cohen, C.; Hernán, M.A.; Lipsitch, M.; Kohane, I.S.; Reis, B.Y.; Balicer, R.D. Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: An observational study. Lancet 2021, 398, 2093–2100. [Google Scholar] [CrossRef]
| Total | CL | SLDI | ||||
|---|---|---|---|---|---|---|
| Overall | Reactive Changes or Negative for Malignancy | Florid Lymphoid Hyperplasia | Kikuchi–Fujimoto Disease | Overall | ||
| Number of patients | 37 | 18 | 13 | 2 | 3 | 19 |
| Mean age ± SD (years) | 47.8 ± 19.1 | 37.8 ± 15.6 | 44.2 ± 13.1 | 18.0 ± 5.0 * | 23.3 ± 7.5 * | 57.2 ± 17.3 ** |
| Number of females (%) | 23 (62.2) | 9 (50.0) | 7 (53.8) | 1 (50.0) | 1 (33.3) | 14 (73.7) |
| Medical history, n (%) | ||||||
| No history | 9 (24.3) | 9 (50.0) | 7 (53.8) | 1 (50.0) | 1 (33.3) | 0 (0.0) |
| Family history of breast cancer | 2 (5.4) | 2 (11.1) | 2 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Personal history of breast cancer | 7 (18.9) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 7 (36.8) |
| Personal history of lung cancer | 2 (5.4) | 2 (11.1) | 2 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Personal history of cecum/appendix NET | 2 (5.4) | 1 (5.5) | 1 (5.5) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Personal history of melanoma | 8 (21.6) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 8 (42.1) |
| Personal history of Merkel cell carcinoma | 2 (5.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (10.5) |
| Personal history of RCC | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Non-neoplastic/malignant history | 3 (8.1) | 3 (16.7) | 0 (0.0) | 1 (50.0) | 2 (66.6) | 0 (0.0) |
| Not reported | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0 | 0 (0.0) | 0 (0.0) |
| Dose and type of last vaccine, n (%) | ||||||
| 1st dose of Pfizer-Bio-Ntech | 13 (35.1) | 7 (38.9) | 6 (46.2) | 0 (0.0) | 1 (33.3) | 6 (31.6) |
| 2nd dose of Pfizer-Bio-Ntech | 7 (18.9) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 6 (31.6) |
| Unspecified dose of Pfizer-Bio-Ntech | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| 1st dose of Moderna | 3 (8.1) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| 2nd dose of Moderna | 3 (8.1) | 2 (11.1) | 1 (7.7) | 1 (50.0) | 0 (0.0) | 1 (5.3) |
| Unspecified dose of Moderna | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 1st dose of AstraZeneca | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 2nd dose of AstraZeneca | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| 1st dose of unspecified vaccine | 3 (8.1) | 3 (16.7) | 1 (7.7) | 0 (0.0) | 2 (66.6) | 0 (0.0) |
| 1st dose of Vaxzevria | 2 (5.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (10.5) |
| 2nd dose of CureVac | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Not reported | 1 (2.7) | 1 (5.5) | 0 (0.0) | 1 (50.0) | 0 (0.0) | 0 (0.0) |
| Duration from last vaccination to CL or SLDI, mean ± SD (days) | 14.5 ± 11.0 | 12.5 ± 7.9 | 10.9 ± 6.3 | 10.5 ± 3.5 | 20.7 ± 10.5 * | 16.5 ± 12.9 |
| Laterality of lymphadenopathy compared with site of vaccination, n (%) | ||||||
| Ipsilateral | 23 (62.2) | 10 (55.6) | 9 (69.2) | 1 (50.0) | 0 (0.0) | 13 (68.4) |
| Contralateral | 2 (5.4) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Not reported | 12 (32.4) | 6 (33.3) | 2 (15.4) | 1 (50.0) | 3 (100.0) | 6 (31.6) |
| Site of lymphadenopathy, n (%) | out of 32 | out of 13 | out of 8 | |||
| Cervical | 4 (12.5) | 3 (23.1) | 2 (25.0) | 0 (0.0) | 1 (33.3) | 1 (5.3) |
| Axilla | 18 (56.3) | 4 (30.8) | 1 (12.5) | 1 (50.0) | 2 (66.6) | 14 (73.7) ** |
| Supraclavicular | 7 (21.9) | 6 (46.2) | 5 (62.5) | 1 (50.0) | 0 (0.0) | 1 (5.3) ** |
| Others | 2 (6.3) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Not reported | 5 (15.6) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 5 (26.3) |
| Additional clinical symptoms aside from lymphadenopathy, n (%) | ||||||
| No other symptoms | 2 (5.4) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Pain | 4 (10.8) | 4 (30.8) | 3 (23.1) | 1 (50.0) | 0 (0.0) | 0 (0.0) |
| Fever | 6 (16.2) | 6 (46.2) | 2 (15.4) | 1 (50.0) | 3 (100.0) | 0 (0.0) |
| Fatigue/malaise | 2 (5.4) | 2 (11.1) | 1 (7.7) | 1 (50.0) | 0 (0.0) | 0 (0.0) |
| Myalgia | 2 (5.4) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Dysphagia | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Chills | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Others | 2 (5.4) | 2 (11.1) | 1 (7.7) | 1 (50.0) | 0 (0.0) | 0 (0.0) |
| Not reported | 24 (64.9) | 5 (27.8) | 5 (38.5) | 0 (0.0) | 0 (0.0) | 19 (100.0) |
| Largest dimension of lymph node, mean ± SD (mm) | 20.8 ± 13.3 | 21.1 ± 14.7 | 22.1 ± 18.2 | 15.5 ± 5.5 | 22.3 ± 7.1 | 19.7 ± 2.9 |
| Imaging modality with abnormal findings, n (%) | ||||||
| US | 11 (29.7) | 7 (38.9) | 7 (53.8) | 0 (0.0) | 0 (0.0) | 4 (21.1) |
| PET/CT | 8 (21.6) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 6 (31.6) |
| CT/MRI | 11 (29.7) | 8 (44.4) | 4 (30.8) | 1 (50.0) | 3 (100.0) | 3 (15.8) |
| Not reported | 10 (27.0) | 2 (11.1) | 1 (7.7) | 1 (50.0) | 0 (0.0) | 8 (42.1) |
| Indication for aspiration or biopsy, n (%) | ||||||
| Suspicion of malignancy | 15 (40.5) | 8 (44.4) | 7 (53.8) | 1 (50.0) | 0 (0.0) | 7 (36.8) |
| Family history of malignancy | 1 (2.7) | 1 (5.5) | 1 (7.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Palpable mass | 2 (5.4) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Suspicion of lymphadenitis and/or KFD | 2 (5.4) | 2 (11.1) | 2 (15.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Patient’s preference | 2 (5.4) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (10.5) |
| Oncologic management | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Not reported | 15 (40.5) | 6 (46.2) | 2 (15.4) | 1 (50.0) | 3 (100.0) | 9 (47.4) |
| Type of procedure, n (%) | ||||||
| FNA | 9 (24.3) | 7 (38.9) | 7 (53.8) | 0 (0.0) | 0 (0.0) | 2 (10.5) |
| Core needle biopsy | 12 (32.4) | 7 (38.9) | 5 (38.5) | 0 (0.0) | 2 (66.6) | 5 (26.3) |
| Excision biopsy | 12 (32.4) | 5 (27.8) | 2 (15.4) | 2 (100.0) | 1 (33.3) | 7 (36.8) |
| Others | 4 (10.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 4 (21.1) |
| Not reported | 1 (2.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (5.3) |
| Pathological diagnosis, n (%) | ||||||
| Reactive/negative for malignancy | 28 (75.7) | 13 (72.2) | - | - | - | 15 (78.9) |
| Florid lymphoid hyperplasia | 2 (5.4) | 2 (11.1) | - | - | - | 0 (0.0) |
| Kikuchi–Fujimoto Disease | 3 (8.1) | 3 (16.7) | - | - | - | 0 (0.0) |
| Granulomatous inflammation | 2 (5.4) | 0 (0.0) | - | - | - | 2 (10.5) |
| Metastatic malignancy | 2 (5.4) | 0 (0.0) | - | - | - | 2 (10.5) |
| Author (Country) [Type of Publication] | Number of Patients with Pathological Findings | Age (Sex) | Significant History | Type and Dose of Most Recent Vaccine | Site of Vaccine | Lymphadenopathy (Palpable or Painful) | Other Clinical Symptoms | Duration from Last Vaccination to Lymphadenopathy (Days) | Laterality of Lymphadenopathy Compared with Site of Vaccination | Site of Lymphadenopathy | Largest Dimension of Lymph Node (mm) | Ultrasound Finding(s) | PET/CT/MRI Finding(s) | Indication for Aspiration or Biopsy | Type of Pathological Specimen | Pathological Finding(s) | Management | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cardoso et al. (Portugal) [Case report] | 1 | 48 (F) | Family history of breast cancer | 1st dose of Pfizer-BioNTech (2nd dose given after lymphadenopathy) | NR | Palpable | No other systemic symptoms | 14 | NR | Posterior edge of lower third of right sternocleidomastoid muscle | 14 | Increase in echogenicity and sphericity index without a defined hilum | CT: right lateral cervical adenopathies | Family history of malignancy | FNA and excision biopsy | FNA: atypical lymphoid cytology Biopsy: reactive follicular hyperplasia | NR | NR |
| Faermann et al. (Israel) [Retrospective] | 7 | NR (F) | Breast cancer, family history, BRCA carrier | Pfizer-Bio-Ntech | NR | NR | NR | NR | Ipsilateral | Axilla | NR | NR | NR | Clinical suspicion of metastases | US-guided core needle biopsy | Reactive | NR | NR |
| Felices-Farias et al. (Spain) [NR] | 11 | NR (NR) | NR | Pfizer-Bio-Ntech | Arm | Painful | NR | NR | Ipsilateral | Axilla and supraclavicular | NR | NR | NR | Suspicion of malignancy | US-guided core needle biopsy | Reactive paracortical/interfollicular hyperplasia | NR | NR |
| Fernandez-Prada et al. (Spain) [Case series] | 5 | NR (NR) | NR | Pfizer-Bio-Ntech and Moderna | NR | Palpable | Pain, swelling | 1 to 9 | Ipsilateral | Supraclavicular | NR | NR | NR | NR | Aspiration | Reactive; lymphocytic infiltrate and active germinal centers | NR | NR |
| Ganga et al. (USA) [Case report] | 1 | 58 (M) | Nil | Moderna | NR | Palpable | Fever, fatigue, myalgia, dysphagia | 2 | NR | Left neck | 59 | Mass effect upon left internal jugular vein | CT: marked irregular thickening and inflammation of sternocleidomastoid area; | NR | US-guided biopsy | Negative for malignancy | Empiric antibiotics, dexamethasone | Significant reduction in swelling on 2nd day of admission; no residual symptoms 2 weeks later |
| Hagen et al. (Switzerland) [Case series] | 5 | 66 (M) | Lung cancer | 2nd dose of Moderna | Left arm | Palpable | NR | 22 | Ipsilateral | Cervical level IV, supra-, infra-, or retroclavicular, axilla | 10 to 24 | Ovoid to rounded shapes, sharp borders, partially detectable hilum; some with suspicious findings such as spherical shape with loss of hilum | PET/CT: enlarged and very highly FDG-active axillary lymph nodes | Clinical suspicion of metastases | FNA | Reactive; no evidence of metastases | NR | Complete regression at 2-month follow-up |
| 41 (F) | Nil | 1st dose of Moderna | Left arm | Palpable | NR | 3 | Ipsilateral | NR | Palpable mass | FNA | Reactive; no evidence of metastases | NR | ||||||
| 47 (F) | Nil | 1st dose of Pfizer-BioNTech | Left arm | Palpable | NR | 19 | Ipsilateral | NR | Palpable mass | FNA | Reactive; no evidence of metastases | NR | ||||||
| 47 (F) | Appendix NET | 1st dose of Moderna | Left arm | Palpable | NR | 8 | Ipsilateral | NR | Clinical suspicion of metastases | FNA | Reactive; no evidence of metastases | NR | ||||||
| 52 (M) | Lung cancer | 2nd dose of Pfizer-BioNTech | Right arm | Palpable | NR | 12 | Contralateral | PET/CT: moderate and very high FDG activity | Clinical suspicion of metastases | FNA | No evidence of metastases or lymphoma | NR | ||||||
| Kado et al. (Japan) [Case report] | 1 | 31 (F) | NR | 1st dose of Pfizer-Bio-Ntech | Left arm | Palpable | Pain | 8 | Ipsilateral | Left-upper clavicle, left-lower scapula | 15 | Rounded and fatty hilum not observed in some lymph nodes | CT: deep cervical lymphadenopathies on the left side | Suspicion of malignancy | Needle biopsy | Follicular hyperplasia, no evidence of malignancy | NR | Decrease in size and number of unilateral lymphadenopathies after 6 weeks; impalpable subcutaneous nodules after 3 months |
| Kim et al. (South Korea) [Case report] | 1 | 36 (F) | Nil | 1st dose of Pfizer-Bio-Ntech | Left arm | Palpable, pain | Discomfort, swelling, pain | 17 | Ipsilateral | Left supraclavicular, level V | 7 | More than 5 lymph nodes, round, thickened cortex, loss of normal fatty hilum, ill-defined border with perinodal fat hyper echogenicity | NR | Possibility of lymphadenitis and Kikuchi disease | US-guided core needle biopsy | Reactive; predominantly small mature T-lymphocytes with small mature B-lymphocytes; negative EBV-encoded small RNA | Symptom relief; advised for second dose of vaccine in the contralateral arm | Reduction in size and extent of lymphadenopathy; newly developed palpable lesions in the right supraclavicular region after 2nd dose of vaccine in the right arm; lymphadenopathy eventually subsided |
| Larkin et al. (USA) [Case series] | 1 | 16 (M) | Nil | 1st dose | Left arm | Palpable | No other symptoms | 14 | Ipsilateral | Left supraclavicular | 10 | NR | NR | Suspicion of malignancy | Excision biopsy | Reactive follicular hyperplasia and focal increased EBV-positive cells (serum negative EBV PCR, EBER-ISH suggestive of prior infection, EBV IgG positive) | NR | NR |
| Ozutemiz et al. (USA) [Case series] | 1 | 38 (F) | Family history of breast cancer | 1st dose of Pfizer-BioNTech | Left arm | Pain | Axillary pain | 8 | Ipsilateral | Left axilla | NR | Cortical thickness of 6 mm | NR | NR | US-guided core needle biopsy | Reactive follicular hyperplasia; no evidence of malignancy | NR | NR |
| Park et al. (South Korea) [Case report] | 1 | 61 (M) | Nil | 1st dose of AstraZeneca | Left arm | Palpable | Fever, chills, muscle pain | 14 | Contralateral | Right supraclavicular | 40 | NR | CT: enlarged, clusters and conglomerated lymph nodes with perinodal infiltration in the right supraclavicular area; no necrosis | Clinical suspicion of malignancy and Kikuchi disease | US-guided core needle biopsy | Reactive hyperplasia with capsular and trabecular fibrosis; negative for malignancy | NR | Improvement of lymphadenopathy |
| Tan et al. (Singapore) [Case report] | 1 | 34 (M) | Nil | 1st dose of Pfizer-BioNTech | Left arm | Palpable, pain | Soreness over vaccination site | 1 | Ipsilateral | Left supraclavicular | 10 | Minimal internal vascularity and no calcification. Hilum was not clearly visualized; however, no sonographically suspicious features | NR | To exclude an occult metastatic malignancy | FNA | Reactive follicular hyperplasia | NR | Complete resolution of lymphadenopathy |
| Larkin et al. (USA) [Case series] | 1 | 13 (M) | Nil | NR | Left arm | Palpable | Transiently painful | 14 | Ipsilateral | Left supraclavicular | 10 | NR | NR | Suspicion of malignancy | Excision biopsy | Florid, reactive follicular hyperplasia, with foci of follicular lysis, increased immunoblasts and progressive transformation of germinal centers | NR | NR |
| Tintle et al. (USA) [Case report] | 1 | 23 (F) | Asthma, eczema, hypothyroidism | 2nd dose of Moderna | NR | Palpable | Fever, malaise, vomiting, acute kidney injury | 7 | NR | Left axilla and abdomen | 21 | NR | CT: left axillary lymphadenopathy and multiple enlarged lymph nodes in the abdomen | NR | Excision biopsy | Florid follicular and interfollicular lymphoid and Langerhans cell hyperplasia | Dexamethasone and anakinra | Recovered within 2 weeks |
| Soub et al. (Qatar) [Case report] | 1 | 18 (M) | Steroid dependent minimal change renal disease | 1st dose of Pfizer-BioNTech | NR | Palpable | Fever | 10 | NR | Left neck | 15 | NR | CT: multiple left cervical and axillary lymph nodes, with the largest one in left supraclavicular region measuring 11 × 10 mm | NR | Excision biopsy | Kikuchi–Fujimoto disease | NR | Discharged in good condition |
| Tan et al. (Singapore) [Case series] | 2 | 18 (F) | Nil | 1st dose | NR | NR | Fever | 35 | NR | Left axilla | 20 | NR | CT: enlarged left supraclavicular, subpectoral and axillary lymph nodes | NR | US-guided core biopsy | Kikuchi–Fujimoto disease | Symptom relief | Resolution of symptoms by day 58 |
| 34 (M) | Diabetes mellitus, hypertension | 1st dose | NR | NR | Fever | 17 | NR | Left axilla | 32 | NR | CT: enlarged left axillary lymph nodes | NR | US-guided core biopsy | Kikuchi–Fujimoto disease | Symptom relief | Resolution of symptoms by day 38 |
| Author (Country) [Type of Publication] | Number of Patients with Pathological Findings | Age (Sex) | Significant History | Type and Dose of Most Recent Vaccine | Site of Vaccine | Duration from Last Vaccination to Development of Lymphadenopathy (Days) | Laterality of Lymphadenopathy Compared with Site of Vaccination | Site of Lymphadenopathy | Largest Dimension of Lymph Node (mm) | Ultrasound Finding(s) | PET/CT/MRI Finding(s) | Indication for Aspiration or Biopsy | Site of Aspiration or Biopsy | Type of Pathological Specimen | Pathological Finding(s) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aalberg et al. (USA) [Case report] | 1 | 74 (M) | Stage IV clear cell RCC with bone and lung metasatses | 2nd dose of Moderna | Left deltoid | 2 | Ipsilateral | Left axilla | 23 × 12 mm | NR | Standardized uptake value of 9.7, additional sub-centimeter left axillary lymph nodes with maximum SUV of 4.1 | Known history of metastatic malignancy and variable PET findings to known metastatic lesions in lung | Left axilla | FNA | Polymorphous lymphoid population compatible with reactive lymph node; negative for metastatic carcinoma |
| Ashoor et al. (UK) [Case series] | 1 | 61 (F) | High-grade DCIS | 2nd dose of AstraZeneca | Left arm | 1 | Ipsilateral | Left axilla | NR | Thickened cortex > 3 mm, intact fatty hilum; eccentrically thickened cortex that measured 4.9 mm in one lymph node | NR | Indeterminate US finding and extensive malignant-appearing calcification on screening mammography | Left axilla | Biopsy | Benign reactive changes |
| Eifer et al. (Israel) [Retrospective] | 1 | 41 (F) | Breast cancer | 2nd dose of Pfizer-BioNTech | Both arms | 1 | Ipsilateral | Left axilla | NR | NR | PET/CT: Increased FDG uptake | Clinical suspicion of metastases | Left axilla | US-guided core needle biopsy | Reactive; no evidence of metastases |
| Lane et al. (USA) [Case series] | 1 | 44 (F) | Left-breast high-grade DCIS | 1st dose of Pfizer-Bio-Ntech | Left arm | 4 | Ipsilateral | Left axilla | NR | NR | MRI: asymmetrical left axillary nodes compared with the right | Oncologic management | Left axilla | US-guided biopsy | Benign; no metastases |
| Lim et al. (South Korea) [Case series] | 3 | 61 (F) | Right-breast IDC with ipsilateral axillary nodal metastases | 1st dose of Vaxzevria | Left arm | 22 | Ipsilateral | Left axilla | NR | NR | NR | Patient’s preference | Left axilla | US-guided 14-gauge gun biopsy | Benign hyperplasia |
| 75 (F) | Right-breast IDC without axillary nodal metastases | 2nd dose of Pfizer-BioNTech | NR | 19 | NR | Left axilla | NR | Maximum cortical thickness of 5.38 mm | CT/MRI: mean length/width ratio less than 1.5 | NR | Left axilla | NR | Reactive hyperplasia | ||
| 71 (F) | Right-breast IDC without axillary nodal metastases | 1st dose of Vaxzevria | Left arm | 14 | Ipsilateral | Left axilla | NR | Smooth and diffuse enlargement and borderline maximum cortical thickness of 3 mm | CT: length/width ratio less than 1.5 and interval change in maximum cortical thickening greater than 2 times compared with previous scans | NR | Left axilla | US-guided biopsy | Benign hyperplasia | ||
| Ozutemiz et al. (USA) [Case series] | 1 | 46 (F) | Left-breast IDC | 1st dose of Pfizer-BioNTech | Left arm | 15 | Ipsilateral | Left axilla | 20 × 12 mm | NR | PET/CT: supraclavicular lymph nodes; multiple enlarged hypermetabolic lymph nodes in left axilla, largest 20 × 12 mm | Patient’s preference | Left axilla and supraclavicular | US-guided core needle biopsy | Benign and reactive; no evidence of breast cancer metastases |
| Robinson et al. (USA) [Retrospective] | 1 | NR (F) | Breast cancer | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | Biopsy | Negative for malignancy |
| Prieto et al. (USA) [Case report] | 1 | 48 (F) | Melanoma | 1st dose of Moderna | Right arm | 5 | Ipsilateral | Right axilla | NR | NR | PET/CT: substantial FDG avidity in right axilla and neck | Suspicion of malignancy | Right axilla | Image-guided biopsy | Reactive lymphoid tissue; negative for metastatic melanoma |
| Trikannad et al. (NR) [Case report] | 1 | 57 (F) | Melanoma | Pfizer-BioNTech | Right arm | 21 | Ipsilateral | Mediastinum, right axilla and right neck | NR | NR | PET/CT: increased uptake | Suspicion of malignancy | Mediastinum | FNA | Non-caseating granulomas and reactive changes; no evidence of malignancy |
| Placke et al. (Germany) [Case series] | 8 | 28 (F) | Melanoma | 1st dose of Pfizer-BioNTech | Left arm | 28 | Ipsilateral in three patients | Left axilla in three patients | 16 mm | Echo-deficient, increased marginal vascularisation | NR | Suspicion of malignancy | Left axilla | Selective lymph node excision | Marked lymphofollicular hyperplasia |
| 43 (F) | Melanoma | 2nd dose of Pfizer-BioNTech | Left arm | 50 | NR | NR | NR | Suspicion of malignancy | Left axilla | Complete lymphadenectomy | Sarcoid-like reaction; no evidence of melanoma metastases | ||||
| 54 (F) | Melanoma | 2nd dose of CureVac | Left arm | 30 | NR | NR | NR | NR | NR | Sentinel lymph node excision | Metastases in 2 patients | ||||
| 58 (M) | Merkel cell carcinoma | 1st dose of Pfizer-BioNTech | Left arm | 7 | NR | NR | NR | NR | NR | Selective lymph node excision | |||||
| 77 (M) | Melanoma | 1st dose of Pfizer-BioNTech | Left arm | 11 | NR | NR | NR | NR | NR | Sentinel lymph node excision | |||||
| 91 (M) | Merkel cell carcinoma | 2nd dose of Pfizer-BioNTech | Left arm | 16 | NR | NR | NR | NR | NR | Sentinel lymph node excision | |||||
| 44 (M) | Melanoma | 1st dose of Pfizer-BioNTech | Left arm | 15 | NR | NR | NR | NR | NR | Sentinel lymph node excision | |||||
| 84 (F) | Melanoma | 2nd dose of Pfizer-BioNTech | Left arm | 12 | NR | NR | NR | NR | NR | Sentinel lymph node excision | |||||
| Pudis et al. (Spain) [Case report] | 1 | 30 (F) | Cecum-appendix NET | 2nd dose of Pfizer-BioNTech | Right arm | 40 | Ipsilateral | Right axilla and supraclavicular | NR | Normal morphology, central hilum, thin cortex | PET/CT: intense uptake in right axillary and supraclavicular region | NR | Right axilla | Surgical resection | Benign reactive changes |
| Author (Year and Country of Publication) | Type of Publication | Type of Vaccine | Key Findings |
|---|---|---|---|
| Aelami et al. (2015, Iran) | Retrospective | BCG | A total of 12/13 (92.3%) biopsies or aspirations of distant lymph nodes showed presence of granuloma and/or acid-fast bacilli. |
| Barouni et al. (2003, Brazil) | Case report | BCG | A 2-year-old male with BCG vaccination at 1 month of age developed lymphadenopathy secondary to atypical tuberculosis. Lymph node aspirates showed presence of atypical tuberculosis. |
| Biers et al. (2007, UK) | Case report | BCG | A 64-year-old male that received intravesical BCG for urothelial carcinoma developed solitary iliac lymphadenopathy. Biopsy showed granulomatous inflammation. |
| Gupta et al. (1996, India) | Retrospective | BCG | A total of 112 cases of BCG lymphadenitis with fine-needle aspiration that showed caseating granulomatous inflammation. |
| Pal et al. (2015, India) | Prospective | BCG | A total of 30 cases of BCG lymphadenitis with needle aspiration smears showing caseating granulomatous inflammation and reactive hyperplasia. |
| Wang et al. (2019, Taiwan) | Case report | BCG | A 2-year-old female with left axillary lymphadenopathy and caseating granulomatous inflammation seen on excision biopsy. |
| Dotlic et al. (2012, Croatia) | Case report | BCG and Hepatitis B | A 2-week-old male with inguinal lymphadenopathy after receiving both vaccines at birth. Excision biopsy showed caseating granulomatous inflammation and hyperimmune post-vaccinal reaction involving an atypical T cell proliferation. |
| Toy et al. (2010, Turkey) | Case report | H1N1 | A 23-year-old male with a left supraclavicular painful lymphadenopathy received H1N1 vaccination one week prior to admission. The lymph node was resected and showed post-vaccinal lymphadenitis with CD30-positive immunoblasts, as well as unusually large immunoblasts resembling Hodgkin cells. |
| Pereira et al. (2019, Portugal) | Case series | HPV 9-valent | An 11-year-old male with inferior cervical and supraclavicular lymphadenopathy that received HPV 9-valent vaccine in the previous week. He subsequently underwent biopsy of the two largest lymph nodes, that showed follicular lymphoid hyperplasia and parafollicular hyperplasia. |
| Watanabe et al. (2012, Japan) | Case report | HPV and JEV | A 14-year-old female with bilateral tender cervical lymphadenopathy who received HPV and JEV vaccine three days prior. Excision biopsy showed Kikuchi–Fujimoto disease. |
| Dorfman et al. (1966, USA) | Case report | Measles | An 11-month-old female with left inguinal lymphadenopathy that received a measles vaccine 13 days prior. The resected lymph nodes showed lymphoid hyperplasia. |
| Sumaya et al. (1976, USA) | Case report | Rubella | A 6-year-old male with painless cervical lymphadenopathy that enlarged after he was vaccinated with HPV-77 DK/5 rubella vaccine. Biopsies showed sinus histiocytosis with massive lymphadenopathy. |
| White et al. (2012, USA) | Case report | Tetanus | A 50-year-old female with left supraclavicular lymphadenopathy 72 h after she was given the booster dose of tetanus toxoid. Excision biopsy showed sheets of small-to-medium-sized atypical lymphocytes with a flow cytometry analysis interpretation of atypical T cell population co-expressing CD4 and CD8. This was initially reported as a T cell lymphoproliferative disorder. However, after a review, this was reported as ‘pseudolymphomatous’ florid proliferation of CD4 T cells in response to the vaccine. |
| Hartsock et al. (1967, USA) | Retrospective | Smallpox, cholera, typhus, tetanus, diphtheria, pertussis, Salk (polio), influenza | A total of 20 cases of post-vaccinal lymphadenitis, with 9 cases initially diagnosed as lymphoma. All 20 cases showed a diffuse, follicular, or combined diffuse and follicular hyperplasia, an increased number of reticular lymphoblasts, vascular and sinusoidal changes, and mixed inflammatory response with a variable number of eosinophils, plasma cells, and mast cells. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chua, T.H.; Takano, A. Pathological Findings in COVID-19 and Non-COVID-19 Vaccine-Associated Lymphadenopathy: A Systematic Review. J. Clin. Med. 2022, 11, 6290. https://doi.org/10.3390/jcm11216290
Chua TH, Takano A. Pathological Findings in COVID-19 and Non-COVID-19 Vaccine-Associated Lymphadenopathy: A Systematic Review. Journal of Clinical Medicine. 2022; 11(21):6290. https://doi.org/10.3390/jcm11216290
Chicago/Turabian StyleChua, Tzy Harn, and Angela Takano. 2022. "Pathological Findings in COVID-19 and Non-COVID-19 Vaccine-Associated Lymphadenopathy: A Systematic Review" Journal of Clinical Medicine 11, no. 21: 6290. https://doi.org/10.3390/jcm11216290
APA StyleChua, T. H., & Takano, A. (2022). Pathological Findings in COVID-19 and Non-COVID-19 Vaccine-Associated Lymphadenopathy: A Systematic Review. Journal of Clinical Medicine, 11(21), 6290. https://doi.org/10.3390/jcm11216290






