The Interactive Effects of Post-Traumatic Stress Symptoms and Breathlessness on Fatigue Severity in Post-COVID-19 Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Outcome Measures
2.3. Statistical Analysis
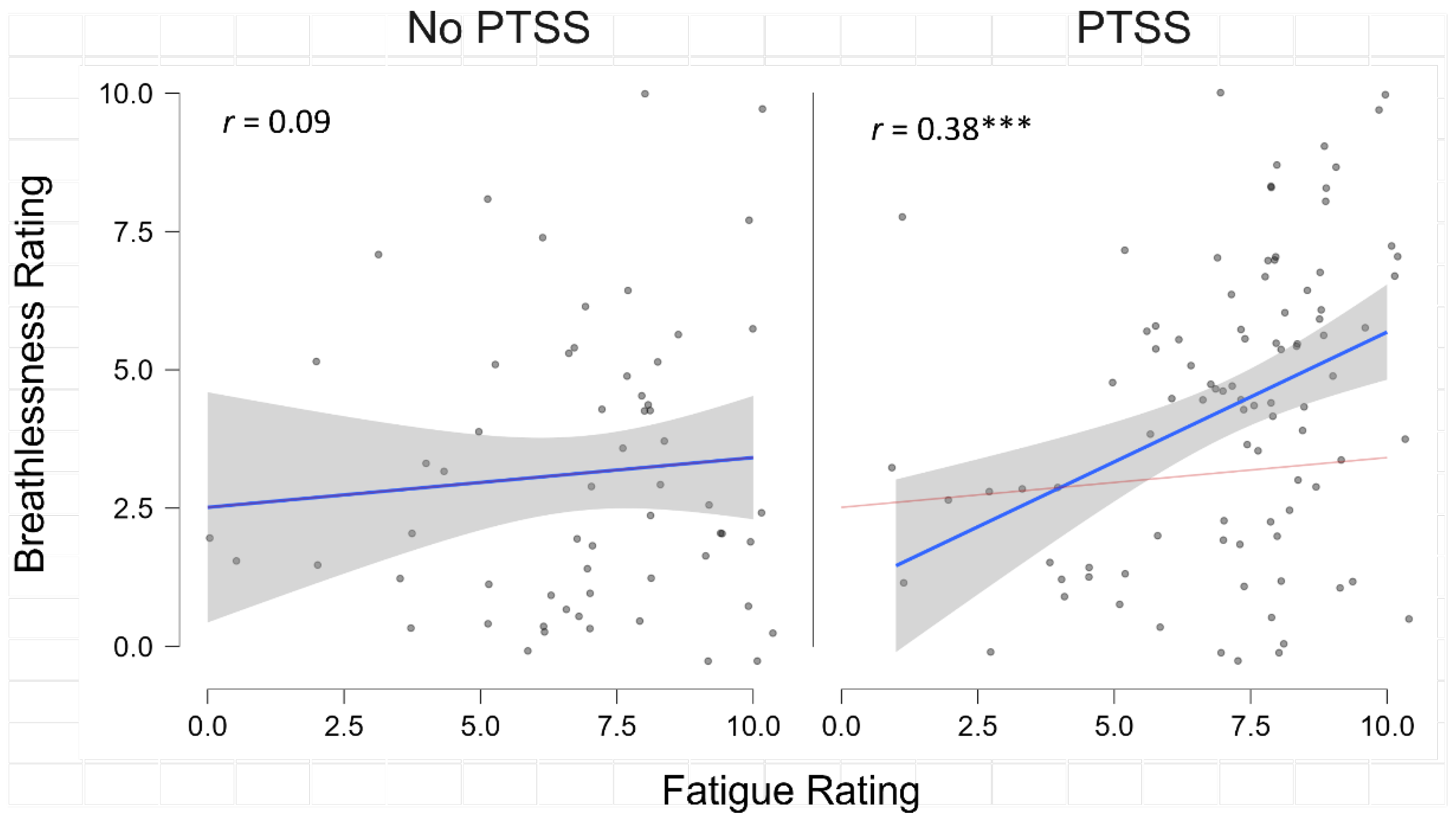
3. Results
3.1. Pre-COVID HRQoL
3.2. Post-COVID-19 Syndrome Symptom Impact on Fatigue Severity
3.3. Post-Course Symptom Improvement
4. Discussion
Limitations
5. Conclusions
- In conclusion, it is possible that improving mental health comorbidities associated with PCS may not only improve mental health and quality of life among COVID-19 survivors, but may also help to reduce some of the physical symptoms associated with PCS.
- The present study demonstrates that interactions between breathlessness and PTSS may contribute to the persistent fatigue observed in PCS and that improvements in PTSS may lead to improvements in reported fatigue. This suggests that PTSS may be an important therapeutic target for multidisciplinary rehabilitation in recovery from PCS.
- The results from the present study highlight the need to move beyond the dichotomy of mental and physical heath and move towards a framework where we consider the interplay between mental and physical health. The treatment models for PCS currently being developed nationally are in a unique position to progress this further without the traditional barriers of commissioning, which often separate the two.
5.1. Clinical Implementations
5.2. Future Directions
- It will be important to investigate to what extent PTSS is a barrier to effective PCS rehabilitation, including self-help, group and individual treatment to develop more helpful clinical guidelines and to establish the cut-off for PTSS in PCS.
- More emphasis is needed on understanding patient characteristics from a biopsychosocial perspective, in order to better triage and assign to effective treatment modalities. Ideally, this should move beyond symptoms and the absence of symptoms, and include meaningful measures of function and coping amongst people living with PCS.
- Finding cost effective treatment for PCS needs to be a priority for a number of reasons; including the disproportionally higher prevalence of PCS in HCP’s, putting healthcare services under further strain due to high levels of sickness absence and further risk of burnout.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nehme, M.; Braillard, O.; Chappuis, F.; Courvoisier, D.S.; Guessous, I. Prevalence of Symptoms More Than Seven Months After Diagnosis of Symptomatic COVID-19 in an Outpatient Setting. Ann. Intern. Med. 2021, 174, 1252–1260. [Google Scholar] [CrossRef] [PubMed]
- Pavli, A.; Theodoridou, M.; Maltezou, H.C. Post-COVID Syndrome: Incidence, Clinical Spectrum, and Challenges for Primary Healthcare Professionals. Arch. Med. Res. 2021, 52, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Malik, P.; Patel, K.; Pinto, C.; Jaiswal, R.; Tirupathi, R.; Pillai, S.; Patel, U. Post-acute COVID-19 syndrome (PCS) and health-related quality of life (HRQoL)—A systematic review and meta-analysis. J. Med. Virol. 2022, 94, 253–262. [Google Scholar] [CrossRef]
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. eClinicalMedicine 2021, 38, 101019. [Google Scholar] [CrossRef] [PubMed]
- Jennings, G.; Monaghan, A.; Xue, F.; Mockler, D.; Romero-Ortuño, R. A Systematic Review of Persistent Symptoms and Residual Abnormal Functioning following Acute COVID-19: Ongoing Symptomatic Phase vs. Post-COVID-19 Syndrome. J. Clin. Med. 2021, 10, 5913. [Google Scholar] [CrossRef]
- Twomey, R.; DeMars, J.; Franklin, K.; Culos-Reed, S.N.; Weatherald, J.; Wrightson, J.G. Chronic Fatigue and Postexertional Malaise in People Living with Long COVID: An Observational Study. Phys. Ther. 2022, 102, pzac005. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.H.-B.; Wing, Y.-K.; Yu, M.W.-M.; Leung, C.-M.; Ma, R.C.W.; Kong, A.P.S.; So, W.Y.; Fong, S.Y.-Y.; Lam, S.-P. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: Long-term follow-up. Arch. Intern. Med. 2009, 169, 2142–2147. [Google Scholar] [CrossRef] [PubMed]
- Hulme, K.; Hudson, J.L.; Rojczyk, P.; Little, P.; Moss-Morris, R. Biopsychosocial risk factors of persistent fatigue after acute infection: A systematic review to inform interventions. J. Psychosom. Res. 2017, 99, 120–129. [Google Scholar] [CrossRef]
- Sandler, C.X.; Wyller, V.B.B.; Moss-Morris, R.; Buchwald, D.; Crawley, E.; Hautvast, J.; Katz, B.Z.; Knoop, H.; Little, P.; Taylor, R.; et al. Long COVID and Post-infective Fatigue Syndrome: A Review. Open Forum Infect. Dis. 2021, 8, ofab440. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.R.; Jason, L.A. Chronic fatigue, abuse-related traumatization, and psychiatric disorders in a community-based sample. Soc. Sci. Med. 2002, 55, 247–256. [Google Scholar] [CrossRef]
- Lerdal, A.; Lee, K.A.; Rokne, B.; Knudsen, Ø., Jr.; Wahl, A.K.; Dahl, A.A. A population-based study of associations between current posttraumatic stress symptoms and current fatigue. J. Trauma. Stress 2010, 23, 606–614. [Google Scholar] [CrossRef]
- Tedstone, J.E.; Tarrier, N. Posttraumatic stress disorder following medical illness and treatment. Clin. Psychol. Rev. 2003, 23, 409–448. [Google Scholar] [CrossRef]
- Tanriverdi, F.; Karaca, Z.; Unluhizarci, K.; Kelestimur, F. The hypothalamo–pituitary–adrenal axis in chronic fatigue syndrome and fibromyalgia syndrome. Stress 2007, 10, 13–25. [Google Scholar] [CrossRef] [PubMed]
- de Kloet, C.S.; Vermetten, E.; Geuze, E.; Kavelaars, A.; Heijnen, C.J.; Westenberg, H.G.M. Assessment of HPA-axis function in posttraumatic stress disorder: Pharmacological and non-pharmacological challenge tests, a review. J. Psychiatr. Res. 2006, 40, 550–567. [Google Scholar] [CrossRef] [PubMed]
- Yehuda, R. Biology of posttraumatic stress disorder. J. Clin. Psychiatry 2000, 61, 14–21. [Google Scholar]
- Afari, N.; Buchwald, D. Chronic Fatigue Syndrome: A Review. Am. J. Psychiatry 2003, 160, 221–236. [Google Scholar] [CrossRef] [PubMed]
- Simani, L.; Ramezani, M.; Darazam, I.A.; Sagharichi, M.; Aalipour, M.A.; Ghorbani, F.; Pakdaman, H. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. J. Neurovirol. 2021, 27, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Houben-Wilke, S.; Goërtz, Y.M.J.; Delbressine, J.M.; Vaes, A.W.; Meys, R.; Machado, F.V.C.; van Herck, M.; Burtin, C.; Posthuma, R.; Franssen, F.M.E.; et al. The Impact of Long COVID-19 on Mental Health: Observational 6-Month Follow-Up Study. JMIR Ment. Health 2022, 9, e33704. [Google Scholar] [CrossRef]
- Mak, I.W.C.; Chu, C.M.; Pan, P.C.; You, M.G.C.; Chan, V.L. Long-term psychiatric morbidities among SARS survivors. Gen. Hosp. Psychiatry 2009, 31, 318–326. [Google Scholar] [CrossRef]
- Lee, S.H.; Shin, H.-S.; Park, H.Y.; Kim, J.L.; Lee, J.J.; Lee, H.; Won, S.-D.; Han, W. Depression as a mediator of chronic fatigue and post-traumatic stress symptoms in Middle East respiratory syndrome survivors. Psychiatry Investig. 2019, 16, 59. [Google Scholar] [CrossRef] [PubMed]
- Kaseda, E.T.; Levine, A.J. Post-traumatic stress disorder: A differential diagnostic consideration for COVID-19 survivors. Clin. Neuropsychol. 2020, 34, 1498–1514. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.; Biswas, P.; Ghosh, R.; Chatterjee, S.; Dubey, M.J.; Chatterjee, S.; Lahiri, D.; Lavie, C.J. Psychosocial impact of COVID-19. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.M.; Sricharoenchai, T.; Raparla, S.; Schneck, K.W.; Bienvenu, O.J.; Needham, D.M. Posttraumatic stress disorder in critical illness survivors: A metaanalysis. Crit. Care Med. 2015, 43, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Schou, T.M.; Joca, S.; Wegener, G.; Bay-Richter, C. Psychiatric and neuropsychiatric sequelae of COVID-19—A systematic review. Brain. Behav. Immun. 2021, 97, 328–348. [Google Scholar] [CrossRef]
- National Institute for Health and Clinical Excellence. COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19. Available online: https://www.nice.org.uk/guidance/ng188 (accessed on 1 October 2022).
- Newton-John, T.; Menzies, R.; Chambers, S.; Menzies, R. Psychological Distress Associated with COVID-19: Estimations of Threat and the Relationship with Death Anxiety. SSRN, 2020; ppcovidwho-4630preprint. [Google Scholar] [CrossRef]
- Chaix, B.; Delamon, G.; Guillemassé, A.; Brouard, B.; Bibault, J.-E. Psychological distress during the COVID-19 pandemic in France: A national assessment of at-risk populations. Gen. Psychiatry 2020, 33. [Google Scholar] [CrossRef]
- Horn, M.; Wathelet, M.; Fovet, T.; Amad, A.; Vuotto, F.; Faure, K.; Astier, T.; Noël, H.; Duhem, S.; Vaiva, G.; et al. Is COVID-19 associated with posttraumatic stress disorder? J. Clin. Psychiatry 2020, 82, 9886. [Google Scholar] [CrossRef]
- Matalon, N.; Dorman-Ilan, S.; Hasson-Ohayon, I.; Hertz-Palmor, N.; Shani, S.; Basel, D.; Gross, R.; Chen, W.; Abramovich, A.; Afek, A.; et al. Trajectories of post-traumatic stress symptoms, anxiety, and depression in hospitalized COVID-19 patients: A one-month follow-up. J. Psychosom. Res. 2021, 143, 110399. [Google Scholar] [CrossRef]
- Ismael, F.; Bizario, J.C.S.; Battagin, T.; Zaramella, B.; Leal, F.E.; Torales, J.; Ventriglio, A.; Marziali, M.E.; Martins, S.S.; Castaldelli-Maia, J.M. Post-infection depressive, anxiety and post-traumatic stress symptoms: A prospective cohort study in patients with mild COVID-19. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 111, 110341. [Google Scholar] [CrossRef]
- Einvik, G.; Dammen, T.; Ghanima, W.; Heir, T.; Stavem, K. Prevalence and risk factors for post-traumatic stress in hospitalized and non-hospitalized COVID-19 patients. Int. J. Environ. Res. Public Health 2021, 18, 2079. [Google Scholar] [CrossRef]
- Giannopoulou, I.; Efstathiou, V.; Triantafyllou, G.; Korkoliakou, P.; Douzenis, A. Adding stress to the stressed: Senior high school students’ mental health amidst the COVID-19 nationwide lockdown in Greece. Psychiatry Res. 2021, 295, 113560. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Scoiety. Dyspnea. Am. J. Respir. Crit. Care Med. 1999, 159, 321–340. [Google Scholar] [CrossRef] [PubMed]
- Başoğlu, M. Effective management of breathlessness: A review of potential human rights issues. Eur. Respir. J. 2017, 49, 1602099. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.C.; Banzett, R.B.; Adams, L.; Mckay, L.; Frackowiak, R.S.J.; Corfield, D.R. BOLD fMRI identifies limbic, paralimbic, and cerebellar activation during air hunger. J. Neurophysiol. 2002, 88, 1500–1511. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, C.; Koch, B.; Grabe, H.J.; Ewert, R.; Barnow, S.; Felix, S.B.; Ittermann, T.; Obst, A.; Völzke, H.; Gläser, S.; et al. Association of airflow limitation with trauma exposure and post-traumatic stress disorder. Eur. Respir. J. 2011, 37, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.; Spathis, A.; Clow, A.; Fallon, M.; Booth, S. The biological impact of living with chronic breathlessness—A role for the hypothalamic–pituitary–adrenal axis? Med. Hypotheses 2014, 83, 232–237. [Google Scholar] [CrossRef]
- Kapella, M.C.; Larson, J.L.; Patel, M.K.; Covey, M.K.; Berry, J.K. Subjective fatigue, influencing variables, and consequences in chronic obstructive pulmonary disease. Nurs. Res. 2006, 55, 10–17. [Google Scholar] [CrossRef]
- Fredriksson-Larsson, U.; Alsén, P.; Karlson, B.W.; Brink, E. Fatigue two months after myocardial infarction and its relationships with other concurrent symptoms, sleep quality and coping strategies. J. Clin. Nurs. 2015, 24, 2192–2200. [Google Scholar] [CrossRef]
- Teixeira, P.J.Z.; Porto, L.; Kristensen, C.H.; Santos, A.H.; Menna-Barreto, S.S.; Do Prado-Lima, P.A.S. Post-traumatic stress symptoms and exacerbations in COPD patients. COPD J. Chronic Obstr. Pulm. Dis. 2015, 12, 90–95. [Google Scholar] [CrossRef]
- Harenwall, S.; Heywood-Everett, S.; Henderson, R.; Godsell, S.; Jordan, S.; Moore, A.; Philpot, U.; Shepherd, K.; Smith, J.; Bland, A.R. Post-Covid-19 Syndrome: Improvements in Health-Related Quality of Life Following Psychology-Led Interdisciplinary Virtual Rehabilitation. J. Prim. Care Community Health 2021, 12, 21501319211067674. [Google Scholar] [CrossRef]
- O’Connor, R.J.; Preston, N.; Parkin, A.; Makower, S.; Ross, D.; Gee, J.; Halpin, S.J.; Horton, M.; Sivan, M. The COVID-19 Yorkshire Rehabilitation Scale (C19-YRS): Application and psychometric analysis in a post-COVID-19 syndrome cohort. J. Med. Virol. 2021, 94, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, R.; Fletcher, A.; Gore, S.; Jones, D.; Spiegelhalter, D.; Cox, D. Quality of life measures in health care. I: Applications and issues in assessment. Br. Med. J. 1992, 305, 1074–1077. [Google Scholar] [CrossRef] [PubMed]
- Dempster, A.P.; Laird, N.M.; Rubin, D.B. Maximum likelihood from incomplete data via the EM algorithm. J. R. Stat. Soc. Ser. B 1977, 39, 1–22. [Google Scholar]
- Held, L.; Ott, M. On p-values and Bayes factors. Annu. Rev. Stat. Its Appl. 2018, 5, 419–593. [Google Scholar] [CrossRef]
- Wagenmakers, E.-J.; Marsman, M.; Jamil, T.; Ly, A.; Verhagen, J.; Love, J.; Selker, R.; Gronau, Q.F.; Šmíra, M.; Epskamp, S.; et al. Bayesian inference for psychology. Part I: Theoretical advantages and practical ramifications. Psychon. Bull. Rev. 2018, 25, 35–57. [Google Scholar] [CrossRef]
- Gaber, T.A.-Z.K.; Ashish, A.; Unsworth, A. Persistent post-covid symptoms in healthcare workers. Occup. Med. 2021, 71, 144–146. [Google Scholar] [CrossRef]
- Kang, L.; Li, Y.; Hu, S.; Chen, M.; Yang, C.; Yang, B.X.; Wang, Y.; Hu, J.; Lai, J.; Ma, X.; et al. The mental health of medical workers in Wuhan, China dealing with the 2019 novel coronavirus. Lancet Psychiatry 2020, 7, e14. [Google Scholar] [CrossRef]
- Leo, C.G.; Sabina, S.; Tumolo, M.R.; Bodini, A.; Ponzini, G.; Sabato, E.; Mincarone, P. Burnout Among Healthcare Workers in the COVID 19 Era: A Review of the Existing Literature. Front. Public Health 2021, 9, 750529. [Google Scholar] [CrossRef]
- Gupta, S.; Sahoo, S. Pandemic and mental health of the front-line healthcare workers: A review and implications in the Indian context amidst COVID-19. Gen. Psychiatry 2020, 33, e100284. [Google Scholar] [CrossRef]
- de Pablo, G.S.; Vaquerizo-Serrano, J.; Catalan, A.; Arango, C.; Moreno, C.; Ferre, F.; Shin, J.I.; Sullivan, S.; Brondino, N.; Solmi, M.; et al. Impact of coronavirus syndromes on physical and mental health of health care workers: Systematic review and meta-analysis. J. Affect. Disord. 2020, 275, 48–57. [Google Scholar] [CrossRef]
- Rossi, R.; Socci, V.; Pacitti, F.; Di Lorenzo, G.; Di Marco, A.; Siracusano, A.; Rossi, A. Mental Health Outcomes Among Frontline and Second-Line Health Care Workers During the Coronavirus Disease 2019 (COVID-19) Pandemic in Italy. JAMA Netw. Open 2020, 3, e2010185. [Google Scholar] [CrossRef] [PubMed]
- Sahebi, A.; Nejati-Zarnaqi, B.; Moayedi, S.; Yousefi, K.; Torres, M.; Golitaleb, M. The prevalence of anxiety and depression among healthcare workers during the COVID-19 pandemic: An umbrella review of meta-analyses. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 107, 110247. [Google Scholar] [CrossRef]
- Marvaldi, M.; Mallet, J.; Dubertret, C.; Moro, M.R.; Guessoum, S.B. Anxiety, depression, trauma-related, and sleep disorders among healthcare workers during the COVID-19 pandemic: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2021, 126, 252–264. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, B.I.; Catts, S.V. Trauma, PTSD, and physical health: An epidemiological study of Australian Vietnam veterans. J. Psychosom. Res. 2008, 64, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Dobie, D.J.; Kivlahan, D.R.; Maynard, C.; Bush, K.R.; Davis, T.M.; Bradley, K.A. Posttraumatic Stress Disorder in Female Veterans: Association with Self-reported Health Problems and Functional Impairment. Arch. Intern. Med. 2004, 164, 394–400. [Google Scholar] [CrossRef]
- Bower, J.E.; Radin, A.; Kuhlman, K.R. Psychoneuroimmunology in the time of COVID-19: Why neuro-immune interactions matter for mental and physical health. Behav. Res. Ther. 2022, 154, 104104. [Google Scholar] [CrossRef]
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J.; et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784. [Google Scholar] [CrossRef]
- Lazzari, C.; Shoka, A.; Nusair, A.; Rabottini, M. Psychiatry in time of COVID-19 pandemic. Psychiatr. Danub. 2020, 32, 229–235. [Google Scholar] [CrossRef]
- Gruet, M. Fatigue in Chronic Respiratory Diseases: Theoretical Framework and Implications for Real-Life Performance and Rehabilitation. Front. Physiol. 2018, 9, 1285. [Google Scholar] [CrossRef]
- Daines, L.; Zheng, B.; Pfeffer, P.; Hurst, J.R.; Sheikh, A. A clinical review of long-COVID with a focus on the respiratory system. Curr. Opin. Pulm. Med. 2022, 28, 174–179. [Google Scholar] [CrossRef]
- Hylton, H.; Long, A.; Francis, C.; Taylor, R.R.; Ricketts, W.M.; Singh, R.; Pfeffer, P.E. Real-world use of the Breathing Pattern Assessment Tool in assessment of breathlessness post-COVID-19. Clin. Med. 2022, 22, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Hentsch, L.; Stancu, P.; Allali, G.; Lövblad, K.-O.; Lobrinus, J.A.; Cocetta, S.; Pautex, S.; Uginet, M.; Serratrice, J.; Coen, M. Decrease in pain perception during acute SARS-CoV-2 infection: A case series. Pain 2022, 163, 1019–1022. [Google Scholar] [CrossRef] [PubMed]
- van der Kolk, B.A. The psychobiology and psychopharmacology of PTSD. Hum. Psychopharmacol. Clin. Exp. 2001, 16, S49–S64. [Google Scholar] [CrossRef]
- Stoeckel, M.C.; Esser, R.W.; Gamer, M.; von Leupoldt, A. Breathlessness amplifies amygdala responses during affective processing. Psychophysiology 2018, 55, e13092. [Google Scholar] [CrossRef] [PubMed]
- von Leupoldt, A.; Riedel, F.; Dahme, B. The impact of emotions on the perception of dyspnea in pediatric asthma. Psychophysiology 2006, 43, 641–644. [Google Scholar] [CrossRef]
- Weidenfeld, J.; Ovadia, H. The Role of the Amygdala in Regulating the Hypothalamic-Pituitary-Adrenal Axis. In The Amygdala—Where Emotions Shape Perception, Learning and Memories; Ferry, B., Ed.; IntechOpen: London, UK, 2017; pp. 173–186. [Google Scholar]
- Tomas, C.; Newton, J.; Watson, S. A review of hypothalamic-pituitary-adrenal axis function in chronic fatigue syndrome. Int. Sch. Res. Not. 2013, 2013, 784520. [Google Scholar] [CrossRef]
- James, J.; Harris, Y.T.; Kronish, I.M.; Wisnivesky, J.P.; Lin, J.J. Exploratory study of impact of cancer-related posttraumatic stress symptoms on diabetes self-management among cancer survivors. Psycho.-Oncol. 2018, 27, 648–653. [Google Scholar] [CrossRef]
- Ehlers, A.; Clark, D.M.; Hackmann, A.; McManus, F.; Fennell, M.; Herbert, C.; Mayou, R. A Randomized Controlled Trial of Cognitive Therapy, a Self-help Booklet, and Repeated Assessments as Early Interventions for Posttraumatic Stress Disorder. Arch. Gen. Psychiatry 2003, 60, 1024–1032. [Google Scholar] [CrossRef]
- Scholes, C.; Turpin, G.; Mason, S. A randomised controlled trial to assess the effectiveness of providing self-help information to people with symptoms of acute stress disorder following a traumatic injury. Behav. Res. Ther. 2007, 45, 2527–2536. [Google Scholar] [CrossRef]
- National Institute for Health and Clinical Excellence. Post-Traumatic Stress Disorder. Available online: https://www.nice.org.uk/guidance/ng116 (accessed on 1 October 2022).
| Model | Predictors | β | t | p | BF10 |
|---|---|---|---|---|---|
| 1 | pre-COVID HRQoL | −0.23 | −2.93 | 0.004 ** | 3.43 |
| R2 = 0.07 | age | −0.04 | −0.54 | 0.593 | 0.39 |
| symptom duration | 0.00 | 0.04 | 0.966 | 0.35 | |
| hospital admittance | −0.15 | −1.90 | 0.060 | 1.06 | |
| 2 | pre-COVID HRQoL | −0.23 | −2.90 | 0.004 ** | 7.64 |
| R2 = 0.147 | age | −0.04 | −0.46 | 0.647 | 0.82 |
| symptom duration | 0.01 | 0.12 | 0.906 | 0.76 | |
| hospital admittance | −0.21 | −2.64 | 0.009 ** | 4.82 | |
| PTSS | 0.15 | 1.72 | 0.088 | 1.80 | |
| breathlessness | 0.20 | 2.51 | 0.013 * | 11.39 | |
| 3 | pre-COVID HRQoL | −0.23 | −2.99 | 0.003 ** | 21.60 |
| R2 = 0.180 | age | −0.01 | −0.07 | 0.942 | 0.64 |
| symptom duration | 0.03 | 0.36 | 0.716 | 0.65 | |
| hospital admittance | −0.24 | −2.96 | 0.004 ** | 16.96 | |
| PTSS | −0.13 | −0.95 | 0.346 | 0.99 | |
| breathlessness | 0.05 | 0.48 | 0.633 | 0.85 | |
| PTSS * breathlessness | 0.41 | 2.44 | 0.016 * | 20.97 |
| Outcome Measure | Pre-Course Rating | Post-Course Rating |
|---|---|---|
| HRQoL | 0.55 (0.21) | 0.61 (0.24) * |
| Fatigue (0–10) | 7.08 (2.34) | 6.47 (2.40) *** |
| PTSS (0–4) | 0.72 (0.08) | 0.68 (0.09) * |
| Breathlessness (0–10) | 3.96 (2.88) | 3.19 (2.25) |
| Model | Predictors | β | t | p | BF10 |
|---|---|---|---|---|---|
| 1 | pre-COVID HRQoL | −0.33 | −3.01 | 0.003 ** | 6.54 |
| R2 = 0.12 | hospital admittance | −0.21 | −1.95 | 0.055 | 2.04 |
| 2 | pre-COVID HRQoL | −0.40 | −3,56 | <0.001 *** | 12.23 |
| R2 = 0.19 | hospital admittance | −0.21 | −2.00 | 0.053 | 2.60 |
| PTSS change | 0.24 | 2.12 | 0.037 * | 3.57 | |
| Breathlessness change | 0.06 | 0.58 | 0.562 | 1.00 | |
| 3 | pre-COVID HRQoL | −0.39 | −3.51 | <0.001 *** | 8.92 |
| R2 = 0.20 | hospital admittance | −0.20 | −1.89 | 0.063 | 2.15 |
| PTSS change | 0.23 | 2.06 | 0.043 * | 2.55 | |
| Breathlessness change | 0.07 | 0.65 | 0.520 | 0.80 | |
| PTSS*breathlessness change | −0.10 | −0.91 | 0.364 | 0.85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harenwall, S.; Heywood-Everett, S.; Henderson, R.; Smith, J.; McEnery, R.; Bland, A.R. The Interactive Effects of Post-Traumatic Stress Symptoms and Breathlessness on Fatigue Severity in Post-COVID-19 Syndrome. J. Clin. Med. 2022, 11, 6214. https://doi.org/10.3390/jcm11206214
Harenwall S, Heywood-Everett S, Henderson R, Smith J, McEnery R, Bland AR. The Interactive Effects of Post-Traumatic Stress Symptoms and Breathlessness on Fatigue Severity in Post-COVID-19 Syndrome. Journal of Clinical Medicine. 2022; 11(20):6214. https://doi.org/10.3390/jcm11206214
Chicago/Turabian StyleHarenwall, Sari, Suzanne Heywood-Everett, Rebecca Henderson, Joanne Smith, Rachel McEnery, and Amy R. Bland. 2022. "The Interactive Effects of Post-Traumatic Stress Symptoms and Breathlessness on Fatigue Severity in Post-COVID-19 Syndrome" Journal of Clinical Medicine 11, no. 20: 6214. https://doi.org/10.3390/jcm11206214
APA StyleHarenwall, S., Heywood-Everett, S., Henderson, R., Smith, J., McEnery, R., & Bland, A. R. (2022). The Interactive Effects of Post-Traumatic Stress Symptoms and Breathlessness on Fatigue Severity in Post-COVID-19 Syndrome. Journal of Clinical Medicine, 11(20), 6214. https://doi.org/10.3390/jcm11206214






