Clinical Outcomes of Individuals with COVID-19 and Tuberculosis during the Pre-Vaccination Period of the Pandemic: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Study Selection and Data Extraction
2.3. Synthesis of Results
2.4. Quality Assessment
3. Results
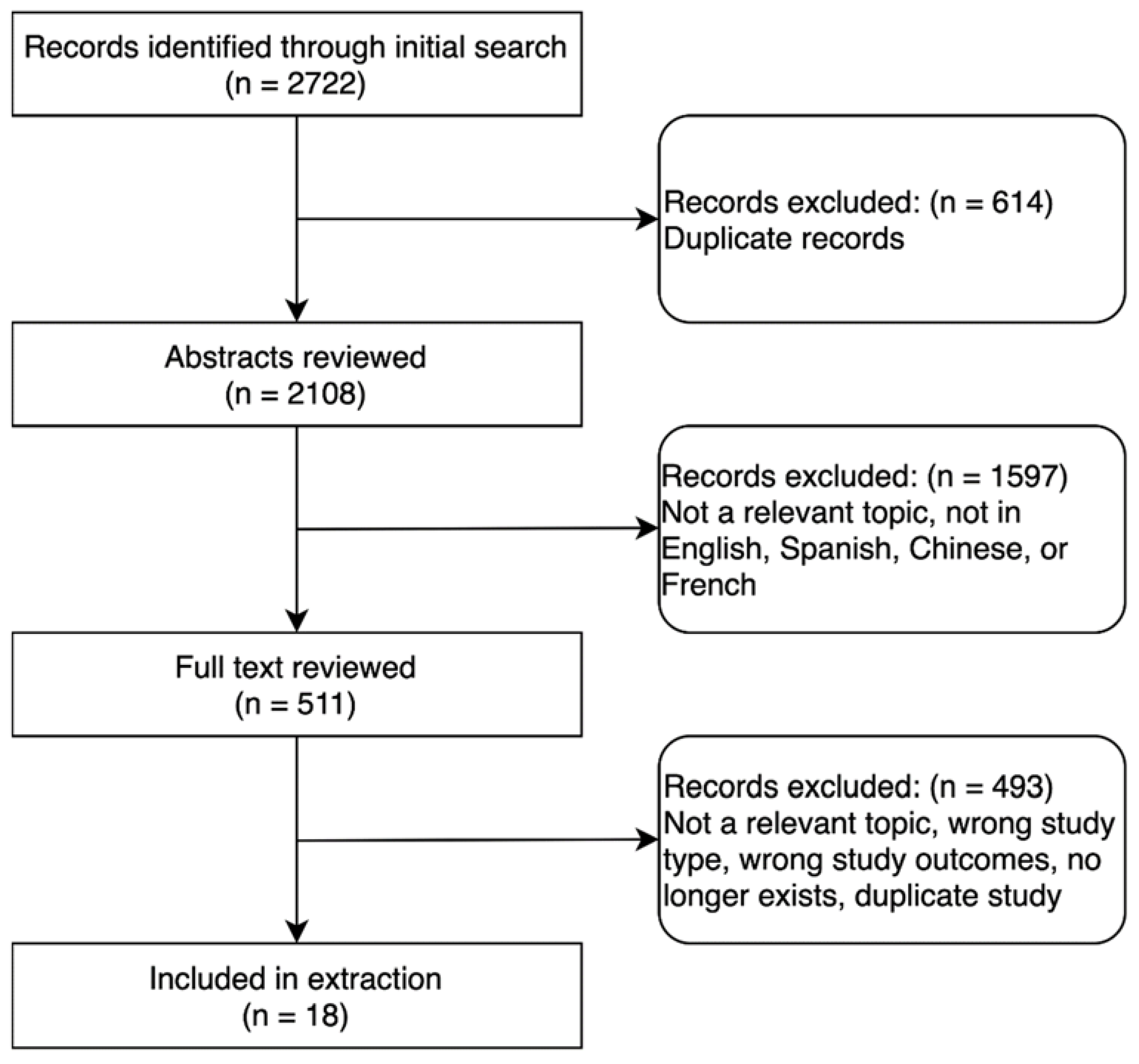
3.1. Study Selection
3.2. Mortality
3.3. Other Adverse Outcomes Related to COVID-19
3.4. COVID-19 as a Risk Factor for Adverse TB Treatment Outcomes
3.5. Quality Assessment
4. Discussion
4.1. TB as a Risk Factor for Mortality Related to COVID-19
4.2. TB as a Risk Factor for Adverse Outcomes Related to COVID-19
4.3. COVID-19 as a Risk Factor for Adverse TB Treatment Outcomes
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Echeverría, G.; Espinoza, W.; De Waard, J. How TB and COVID-19 compare: An opportunity to integrate both control programmes. Int. J. Tuberc. Lung Dis. 2020, 24, 971–974. [Google Scholar] [CrossRef] [PubMed]
- Hopewell, P.C.; Reichman, L.B.; Castro, K.G. Parallels and Mutual Lessons in Tuberculosis and COVID-19 Transmission, Prevention, and Control. Emerg. Infect. Dis. 2021, 27, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Visca, D.; Ong, C.; Tiberi, S.; Centis, R.; D’Ambrosio, L.; Chen, B.; Mueller, J.; Duarte, R.; Dalcolmo, M.; Sotgiu, G.; et al. Tuberculosis and COVID-19 interaction: A review of biological, clinical and public health effects. Pulmonology 2021, 27, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Drobniewski, F.; Keshavjee, S. COVID-19 and tuberculosis-a global tale of hubris and lessons unlearned? Front. Med. 2021, 8, 799640. [Google Scholar] [CrossRef]
- WHO. WHO Global Lists of High Burden Countries for TB, Multidrug/Rifampicin Resistant TB (MDR/RR-TB) and TB/HIV, 2021–2025; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Johns Hopkins Coronavirus Resource Center. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Available online: https://coronavirus.jhu.edu/map.html (accessed on 21 March 2022).
- Cox, V.; Wilkinson, L.; Grimsrud, A.; Hughes, J.; Reuter, A.; Conradie, F.; Nel, J.; Boyles, T. Critical changes to services for TB patients during the COVID-19 pandemic. Int. J. Tuberc. Lung Dis. 2020, 24, 542–544. [Google Scholar] [CrossRef]
- Pang, Y.; Liu, Y.; Du, J.; Gao, J.; Li, L. Impact of COVID-19 on tuberculosis control in China. Int. J. Tuberc. Lung Dis. 2020, 24, 545–547. [Google Scholar] [CrossRef]
- Amimo, F.; Lambert, B.; Magit, A. What does the COVID-19 pandemic mean for HIV, tuberculosis, and malaria control? Trop. Med. Health 2020, 48, 1–4. [Google Scholar] [CrossRef]
- Yadav, S.R.; Kumar, R.; Gupta, N.; Ish, P.; Chakrabarti, S.; Kumar, A. COVID-19: Avoiding a second tragedy in a tuberculosis burdened country. Monaldi Arch. Chest Dis. 2020, 90. [Google Scholar] [CrossRef]
- Hinton, D.M. Pfizer-BioNTech COVID-19 Vaccine EUA Letter of Authorization Reissued 05-10-2021. 2021. Available online: https://www.fda.gov/media/144412/download (accessed on 10 May 2021).
- Tagoe, E.T.; Sheikh, N.; Morton, A.; Nonvignon, J.; Sarker, A.R.; Williams, L.; Megiddo, I. COVID-19 Vaccination in Lower-Middle Income Countries: National Stakeholder Views on Challenges, Barriers, and Potential Solutions. Front. Public Health 2021, 9, 709127. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Ottawa Hospital Research Institute. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 21 March 2022).
- Boulle, A.; Davies, M.-A.; Hussey, H.; Ismail, M.; Morden, E.; Vundle, Z.; Zewighental, V.; Mahomed, H.; Paleker, M.; Pienaar, D.; et al. Risk factors for COVID-19 death in a population cohort study from the Western Cape Province, South Africa. Clin. Infect Dis. 2020, 29, ciaa1198. [Google Scholar]
- Pillay-van Wyk, V.; Bradshaw, D.; Groenewald, P.; Seocharan, I.; Manda, S.; Roomaney, R.A.; Awotiwon, O.; Nkwenika, T.; Gray, G.; Buthelezi, S.S.; et al. COVID deaths in South Africa: 99 days since South Africa’s first death. S. Afr. Med. J. 2020, 110, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Torun, Ş.; Özkaya, Ş.; Şen, N.; Kanat, F.; Karaman, I.; Yosunkaya, Ş.; Dikiş, Ö.Ş.; Asan, A.; Eroğlu, S.A.; Atal, S.S.; et al. The Characteristics of Health Care Workers with COVID-19 and Relationship Between COVID-19 Mortality and BCG/Tuberculosis History: A multi-center study. medRxiv 2020. [Google Scholar] [CrossRef]
- Sun, Y.; Dong, Y.; Wang, L.; Xie, H.; Li, B.; Chang, C.; Wang, F.-S. Characteristics and prognostic factors of disease severity in patients with COVID-19: The Beijing experience. J. Autoimmun. 2020, 112, 102473. [Google Scholar] [CrossRef] [PubMed]
- van der Zalm, M.M.; Lishman, J.; Verhagen, L.M.; Redfern, A.; Smit, L.; Barday, M.; Ruttens, D.; da Costa, A.; van Jaarsveld, S.; Itana, J.; et al. Clinical Experience With Severe Acute Respiratory Syndrome Coronavirus 2-Related Illness in Children: Hospital Experience in Cape Town, South Africa. Clin. Infect Dis. 2021, 72, e938–e944. [Google Scholar] [CrossRef]
- Sy, K.T.L.; Haw, N.J.L.; Uy, J. Previous and active tuberculosis increases risk of death and prolongs recovery in patients with COVID-19. Infect. Dis. 2020, 52, 902–907. [Google Scholar] [CrossRef]
- Jassat, W.; Cohen, C.; Masha, M.; Goldstein, S.; Kufa, T.; Savulescu, D.; Walaza, S.; Bam, J.L.; Davies, M.A.; Prozesky, H.W.; et al. COVID-19 in-hospital mortality in South Africa: The intersection of communicable and non-communicable chronic diseases in a high HIV prevalence setting. medRxiv 2020. [Google Scholar] [CrossRef]
- Gajbhiye, R.K.; Mahajan, N.N.; Kamath, N.; Bahirat, S.; Patokar, G.; Bhurke, A.V.; Modi, D.N.; Mahale, S.D. Clinical presentations, pregnancy complications, and maternal outcomes in pregnant women with COVID-19 and tuberculosis: A retrospective cohort study. Int. J. Gynaecol. Obstet. 2021, 153, 176–179. [Google Scholar] [CrossRef]
- Gupta, N.; Ish, P.; Gupta, A.; Malhotra, N.; Caminero, J.A.; Singla, R.; Kumar, R.; Yadav, S.R.; Dev, N.; Agrawal, S.; et al. A profile of a retrospective cohort of 22 patients with COVID-19 and active/treated tuberculosis. Eur. Respir. J. 2020, 56, 2003408. [Google Scholar] [CrossRef]
- Fisman, D.N.; Greer, A.L.; Hillmer, M.; Tuite, A.R. Derivation and Validation of Clinical Prediction Rules for COVID-19 Mortality in Ontario, Canada. Open Forum Infect. Dis. 2020, 7, ofaa463. [Google Scholar] [CrossRef]
- Demkina, A.E.; Morozov, S.P.; Vladzymyrskyy, A.V.; Kljashtorny, V.G.; Guseva, O.I.; Pugachev, P.S.; Artemova, O.R.; Reshetnikov, R.V.; Gombolevskiy, V.A.; Ryabinina, M.N. Risk factors for outcomes of COVID-19 patients: An observational study of 795 572 patients in Russia. medRxiv 2020. [Google Scholar] [CrossRef]
- Kuwari, H.M.A.; Rahim, H.F.A.; Raddad, L.J.A.; Abou-Samra, A.B.; Kanaani, Z.A.; Khal, A.A.; Kuwari, E.A.; Marri, S.A.; Masalmani, M.A.; Romaihi, H.E.A.; et al. Characterization of the SARS-CoV-2 outbreak in the State of Qatar, February 28-April 18, 2020. medRxiv 2020. [Google Scholar] [CrossRef]
- Chen, M.; Tu, C.; Tan, C.; Zheng, X.; Wang, X.; Wu, J.; Huang, Y.; Wang, Z.; Yan, Y.; Li, Z.; et al. Key to successful treatment of COVID-19: Accurate identification of severe risks and early intervention of disease progression. medRxiv 2020. [Google Scholar] [CrossRef]
- Lei, Y.; Huang, X.; SiLang, B.; Lan, Y.; Lu, J.; Zeng, F. Clinical features of imported cases of coronavirus disease 2019 in Tibetan patients in the Plateau area. medRxiv 2020. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Fleming, J.; Yu, Y.; Gu, Y.; Liu, C.; Fan, L.; Wang, X.; Cheng, M.; Bi, L.; et al. Active or latent tuberculosis increases susceptibility to COVID-19 and disease severity. medRxiv 2020. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, L.; Chen, Y.; Wu, Q.; Chen, G.; Shen, X.; Wang, Q.; Yan, Y.; Yu, Y.; Zhong, Y.; et al. Outcomes of novel coronavirus disease 2019 (COVID-19) infection in 107 patients with cancer from Wuhan, China. Cancer 2020, 126, 4023–4031. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, X.; Fu, Z.; Luo, M.; Zhang, Z.; Zhang, K.; He, Y.; Wan, D.; Zhang, L.; Yan, X.; et al. Potential factors for prediction of disease severity of COVID-19 patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Bi, Q.; Hong, C.; Meng, J.; Wu, Z.; Zhou, P.; Ye, C.; Sun, B.; Kucirka, L.M.; Azman, A.S.; Wang, T.; et al. Characterizing clinical progression of COVID-19 among patients in Shenzhen, China: An observational cohort study. medRxiv 2020. [Google Scholar] [CrossRef]
- Martínez Orozco, J.A.; Sánchez Tinajero, Á.; Becerril Vargas, E.; Delgado Cueva, A.I.; Reséndiz Escobar, H.; Vázquez Alcocer, E.; Narváez Díaz, L.A.; Ruiz Santillán, D.P. COVID-19 and tuberculosis coinfection in a 51-year-old taxi driver in Mexico City. Am. J. Case Rep. 2020, 21, e927628. [Google Scholar] [CrossRef]
- Musso, M.; Di Gennaro, F.; Gualano, G.; Mosti, S.; Cerva, C.; Fard, S.N.; Libertone, R.; Di Bari, V.; Cristofaro, M.; Tonnarini, R.; et al. Concurrent cavitary pulmonary tuberculosis and COVID-19 pneumonia with in vitro immune cell anergy. Infection 2021, 49, 1061–1064. [Google Scholar] [CrossRef]
- Goel, J.; Yadav, S.; Sharma, S.; Sherawat, R.K. COVID-19 Pneumonia with Delayed Viral Clearance in a Patient with Active Drug-resistant Pulmonary Tuberculosis. Indian J. Crit. Care Med. 2020, 24, 1132–1134. [Google Scholar] [CrossRef]
- Cao, B.; Wei, M.; Du, Y.; Xiao, K.; Li, Q.; Lu, W.; Huang, Y.; Wu, G.; Huang, F. Coronavirus Disease 2019 with Comorbid Pulmonary Tuberculosis: A Case Report. Iran. Red Crescent Med. J. 2020, 22. [Google Scholar] [CrossRef]
- Luciani, M.; Bentivegna, E.; Spuntarelli, V.; Lamberti, P.A.; Guerritore, L.; Chiappino, D.; Nalli, G.; Proietta, M.; Del Porto, F.; Martelletti, P.; et al. Coinfection of Tuberculosis Pneumonia and COVID-19 in a Patient Vaccinated with Bacille Calmette-Guérin (BCG): Case Report. SN Compr. Clin. Med. 2020, 2, 2419–2422. [Google Scholar] [CrossRef] [PubMed]
- Gadelha Farias, L.A.B.; Gomes Moreira, A.L.; Austregésilo Corrêa, E.; Landim de Oliveira Lima, C.A.; Lopes, I.M.P.; de Holanda, P.E.L.; Nunes, F.R.; Pires Neto, R.D.J. Case report: Coronavirus disease and pulmonary tuberculosis in patients with human immunodeficiency virus: Report of two cases. Am. J. Trop. Med. Hyg. 2020, 103, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Gbenga, T.A.; Oloyede, T.; Ibrahim, O.R.I.; Sanda, A.; Suleiman, B.M. Pulmonary tuberculosis in coronavirus disease-19 patients: A report of two cases from Nigeria. Open Access Maced. J. Med. Sci. 2020, 8, 272–275. [Google Scholar] [CrossRef]
- Yadav, S.; Rawal, G. The case of pulmonary tuberculosis with COVID-19 in an Indian male-a first of its type case ever reported from South Asia. Pan. Afr. Med. J. 2020, 36, 374. [Google Scholar] [CrossRef]
- Yousaf, Z.; Khan, A.A.; Chaudhary, H.A.; Mushtaq, K.; Parengal, J.; Aboukamar, M.; Khan, M.U.; Mohamed, M.F.H. Cavitary pulmonary tuberculosis with COVID-19 coinfection. IDCases 2020, 22, e00973. [Google Scholar] [CrossRef] [PubMed]
- AlKhateeb, M.H.; Aziz, A.; Eltahir, M.; Elzouki, A. Bilateral foot-drop secondary to axonal neuropathy in a tuberculosis patient with co-infection of COVID-19: A case report. Cureus 2020, 12, e11734. [Google Scholar] [CrossRef] [PubMed]
- Ata, F.; Yousaf, Q.; Veliyankodan Parambil, J.; Parengal, J.; Mohamedali, M.G.; Yousaf, Z. A 28-year-old man from India with SARS-CoV-2 and pulmonary tuberculosis co-infection with central nervous system involvement. Am. J. Case Rep. 2020, 21, e926034. [Google Scholar] [CrossRef]
- Essajee, F.; Solomons, R.; Goussard, P.; Van Toorn, R. Child with tuberculous meningitis and COVID-19 coinfection complicated by extensive cerebral sinus venous thrombosis. BMJ Case Rep. 2020, 13, e238597. [Google Scholar] [CrossRef]
- Can Sarınoğlu, R.; Sili, U.; Eryuksel, E.; Olgun Yildizeli, S.; Cimsit, C.; Karahasan Yagci, A. Tuberculosis and COVID-19: An overlapping situation during pandemic. J. Infect. Dev. Ctries. 2020, 14, 721–725. [Google Scholar] [CrossRef]
- Arjun, S.; Patel, D.; Sanivarapu, R.; Iqbal, J.; Anjum, F. Case report of severe pulmonary fibrosis as a sequelae of COVID-19 infection. Chest 2020, 158, A433–A434. [Google Scholar] [CrossRef]
- Rivas, N.; Espinoza, M.; Loban, A.; Luque, O.; Jurado, J.; Henry-Hurtado, N.; Goodridge, A. Case report: COVID-19 recovery from triple infection with mycobacterium tuberculosis, HIV, and SARS-CoV-2. Am. J. Trop. Med. Hyg. 2020, 103, 1597–1599. [Google Scholar] [CrossRef] [PubMed]
- Vilbrun, S.C.; Mathurin, L.; Pape, J.W.; Fitzgerald, D.; Walsh, K.F. Case report: Multidrug-resistant tuberculosis and COVID-19 coinfection in Port-au-Prince, Haiti. Am. J. Trop. Med. Hyg. 2020, 103, 1986–1988. [Google Scholar] [CrossRef] [PubMed]
- Stochino, C.; Villa, S.; Zucchi, P.; Parravicini, P.; Gori, A.; Raviglione, M.C. Clinical characteristics of COVID-19 and active tuberculosis co-infection in an Italian reference hospital. Eur. Respir. J. 2020, 56, 2001708. [Google Scholar] [CrossRef]
- Migliori, G.B.; Thong, P.M.; Alffenaar, J.-W.; Denholm, J.; Tadolini, M.; Alyaquobi, F.; Blanc, F.-X.; Buonsenso, D.; Cho, J.-G.; Codecasa, L.R.; et al. Gauging the impact of the COVID-19 pandemic on tuberculosis services: A global study. Eur. Respir. J. 2021, 58, 2101786. [Google Scholar] [CrossRef]
- Botly, L.; Martin-Rhee, M.; Kasiban, A.; Swartz, R.H.; Mulvagh, S.L.; Lindsay, M.P.; Goia, C.; Smith, E.E.; Hill, M.D.; Field, T.; et al. COVID-19 Pandemic: Global Impact and Potential Implications for Cardiovascular Disease in Canada. CJC Open 2020, 2, 265–272. [Google Scholar] [CrossRef]
- McQuaid, C.F.; Vassall, A.; Cohen, T.; Fiekert, K.; White, R.G. The impact of COVID-19 on TB: A review of the data. Int. J. Tuberc. Lung Dis. 2021, 25, 436–446. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, M.; Chen, Y.; Shi, S.; Geng, J.; Tian, J. Association between tuberculosis and COVID-19 severity and mortality: A rapid systematic review and meta–analysis. J. Med Virol. 2020, 93, 194–196. [Google Scholar] [CrossRef]
- Ergönül, Ö.; Akyol, M.; Tanrıöver, C.; Tiemeier, H.; Petersen, E.; Petrosillo, N.; Gönen, M. National case fatality rates of the COVID-19 pandemic. Clin. Microbiol. Infect. 2020, 27, 118–124. [Google Scholar] [CrossRef]
- Raham, T.F. TB prevalence influence on COVID-19 mortality. medRxiv 2020. [Google Scholar] [CrossRef]
- Sala, G.; Chakraborti, R.; Ota, A.; Miyakawa, T. Association of BCG vaccination policy and tuberculosis burden with incidence and mortality of COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Al-Momen, H.; Raham, T.F.; Daher, A.M. Tuberculosis versus COVID-19 mortality: A new evidence. Open Access Maced. J. Med. Sci. 2020, 8, 179–183. [Google Scholar] [CrossRef]
- Fine, P.E. Variation in protection by BCG: Implications of and for heterologous immunity. Lancet 1995, 346, 1339–1345. [Google Scholar] [CrossRef]
- Fu, W.; Ho, P.-C.; Liu, C.-L.; Tzeng, K.-T.; Nayeem, N.; Moore, J.S.; Wang, L.-S.; Chou, S.-Y. Reconcile the debate over protective effects of BCG vaccine against COVID-19. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hamiel, U.; Kozer, E.; Youngster, I. SARS-CoV-2 rates in BCG-vaccinated and unvaccinated young adults. JAMA 2020, 323, 2340–2341. [Google Scholar] [CrossRef] [PubMed]
- de Chaisemartin, C.; de Chaisemartin, L. Bacille Calmette-Guérin vaccination in infancy does not protect against coronavirus disease 2019 (COVID-19): Evidence from a natural experiment in Sweden. Clin. Infect. Dis. 2021, 72, e501–e505. [Google Scholar] [CrossRef] [PubMed]
- Corticosteroids for COVID-19. World Health Organization. Available online: https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-Corticosteroids-2020.1 (accessed on 21 March 2022).
- Shankar-Hari, M.; Vale, C.L.; Godolphin, P.J.; Fisher, D.; Higgins, J.P.T.; Spiga, F.; Savović, J.; Tierney, J.; Baron, G.; Benbenishty, J.S.; et al. Association between administration of IL-6 antagonists and mortality among patients hospitalized for COVID-19: A meta-analysis. JAMA 2021, 326, 499–518. [Google Scholar]
- Hsu, D.; Irfan, M.; Jabeen, K.; Iqbal, N.; Hasan, R.; Migliori, G.B.; Zumla, A.; Visca, D.; Centis, R.; Tiberi, S. Post tuberculosis treatment infectious complications. Int. J. Infect. Dis. 2020, 92, S41–S45. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.G.; Muhammad, Z.S.; Shahzad, T.; Yaseen, A.; Irfan, M. Post tuberculosis sequelae in patients treated for tuberculosis: An observational study at a tertiary care center of a high TB burden country. Tuberculosis 2018, 52, PA2745. [Google Scholar] [CrossRef]
- Kim, H.Y.; Song, K.-S.; Goo, J.M.; Lee, J.S.; Lee, K.S.; Lim, T.-H. Thoracic Sequelae and Complications of Tuberculosis. RadioGraphics 2001, 21, 839–858. [Google Scholar] [CrossRef]
- Meghji, J.; Lesosky, M.; Joekes, E.; Banda, P.; Rylance, J.; Gordon, S.; Jacob, J.; Zonderland, H.; MacPherson, P.; Corbett, E.L.; et al. Patient outcomes associated with post-tuberculosis lung damage in Malawi: A prospective cohort study. Thorax 2020, 75, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Tuberculosis (TB) Health Disparities. Centers for Disease Control and Prevention. U.S. Department of Health and Human Services. 2020. Available online: https://www.cdc.gov/tb/topic/populations/healthdisparities/default.htm (accessed on 16 August 2021).
- WHO. A Human Rights Approach to TB: Stop TB Guidelines for Social Mobilization; Report No. WHO/CDS/STB/2001.9; World Health Organization: Geneva, Switzerland, 2001; Available online: https://www.who.int/hhr/information/A%20Human%20Rights%20Approach%20to%20Tuberculosis.pdf (accessed on 21 September 2022).
- Reddy, K.S. Pandemic lessons from India. BMJ 2021, 373, n1196. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Tu, L.; Yang, Y.; Hu, P.; Wang, R.; Hu, Q.; Cao, F.; Jiang, T.; Sun, J.; Xu, G.; et al. Management and Treatment of COVID-19: The Chinese Experience. Can. J. Cardiol. 2020, 36, 915–930. [Google Scholar] [CrossRef]
- Tamuzi, J.L.; Ayele, B.T.; Shumba, C.S.; Adetokunboh, O.O.; Uwimana-Nicol, J.; Haile, Z.T.; Inugu, J.; Nyasulu, P.S. Implications of COVID-19 in high burden countries for HIV/TB: A systematic review of evidence. BMC Infect. Dis. 2020, 20, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Koupaei, M.; Naimi, A.; Moafi, N.; Mohammadi, P.; Tabatabaei, F.S.; Ghazizadeh, S.; Heidary, M.; Khoshnood, S. Clinical characteristics, diagnosis, treatment, and mortality rate of TB/COVID-19 coinfected patients: A systematic review. Front. Med. 2021, 8, 740593. [Google Scholar] [CrossRef]
- Aggarwal, A.N.; Agarwal, R.; Dhooria, S.; Prasad, K.T.; Sehgal, I.S.; Muthu, V. Active pulmonary tuberculosis and coronavirus disease 2019: A systematic review and meta-analysis. Bhatt GC, editor. PLoS ONE 2021, 16, e0259006. [Google Scholar] [CrossRef]
- Global Tuberculosis Report 2021. Geneva: World Health Organization; 2021. Licence: CC BY-NC-SA 3.0 IGO. Available online: https://www.who.int/publications/i/item/9789240037021 (accessed on 21 September 2022).
- Subbaraman, R.; Nathavitharana, R.R.; Mayer, K.H.; Satyanarayana, S.; Chadha, V.K.; Arinaminpathy, N.; Pai, M. Constructing care cascades for active tuberculosis: A strategy for program monitoring and identifying gaps in quality of care. PLOS Med. 2019, 16, e1002754. [Google Scholar] [CrossRef]
- Subbaraman, R.; Jhaveri, T.; Nathavitharana, R.R. Closing gaps in the tuberculosis care cascade: An action-oriented research agenda. J. Clin. Tuberc. Other Mycobact. Dis. 2020, 19, 100144. [Google Scholar] [CrossRef]
- Global Tuberculosis Report 2020. Geneva: World Health Organization; 2020. Licence: CC BY-NC-SA 3.0 IGO. Available online: https://www.who.int/publications/i/item/9789240013131 (accessed on 21 September 2022).
- Fei, H.; Yinyin, X.; Hui, C.; Ni, W.; Xin, D.; Wei, C.; Tao, L.; Shitong, H.; Miaomiao, S.; Mingting, C.; et al. The impact of the COVID-19 epidemic on tuberculosis control in China. Lancet Reg. Health West. Pac. 2020, 3, 100032. [Google Scholar] [CrossRef]
- Liu, Q.; Lu, P.; Shen, Y.; Li, C.; Wang, J.; Zhu, L.; Lu, W.; Martinez, L. Collateral Impact of the Coronavirus Disease 2019 (COVID-19) Pandemic on Tuberculosis Control in Jiangsu Province, China. Clin. Infect. Dis. 2020, 73, 542–544. [Google Scholar] [CrossRef]
- Mohr-Holland, E.; Hacking, D.; Daniels, J.; Scott, V.; Mudaly, V.; Furin, J.; Pfaff, C.; Reuter, A. Diagnosis patterns for rifampicin-resistant TB after onset of COVID-19. Int. J. Tuberc. Lung Dis. 2021, 25, 772–775. [Google Scholar] [CrossRef] [PubMed]
- Geric, C.; Saroufim, M.; Landsman, D.; Richard, J.; Benedetti, A.; Batt, J.; Brode, S.K.; Ahmad Khan, F. Impact of COVID-19 on tuberculosis prevention and treatment in Canada: A multicentre analysis of 10,833 patients. J. Infect. Dis. 2021, 225, jiab608. [Google Scholar] [CrossRef] [PubMed]
- Global Tuberculosis Report 2021: COVID-19 and TB. Available online: https://www.who.int/publications/digital/global-tuberculosis-report-2021/covid-19 (accessed on 30 July 2022).
- Axfors, C.; Schmitt, A.M.; Janiaud, P.; Van’t Hooft, J.; Abd-Elsalam, S.; Abdo, E.; Abella, B.S.; Akram, J.; Amaravadi, R.K.; Angus, D.C.; et al. Mortality outcomes with hydroxychloroquine and chloroquine in COVID-19 from an international collaborative meta-analysis of randomized trials. Nat. Commun. 2021, 12, 2349. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 Clinical Management: Living Guidance, 25 January 2021. World Health Organization. Available online: https://apps.who.int/iris/handle/10665/338882 (accessed on 21 September 2022).
- Bertolini, A.; Van De Peppel, I.P.; Bodewes, F.A.; Moshage, H.; Fantin, A.; Farinati, F.; Fiorotto, R.; Jonker, J.W.; Strazzabosco, M.; Verkade, H.J.; et al. Abnormal Liver Function Tests in Patients With COVID-19: Relevance and Potential Pathogenesis. Hepatology 2020, 72, 1864–1872. [Google Scholar] [CrossRef]
- Johansson, M.A.; Reich, N.G.; Meyers, L.A.; Lipsitch, M. Preprints: An underutilized mechanism to accelerate outbreak science. PLoS Med. 2018, 15, e1002549. [Google Scholar] [CrossRef]
- Belbasis, L.; Bellou, V. Introduction to Epidemiological Studies. In Genetic Epidemiology: Methods and Protocols; Evangelou, E., Ed.; Springer: New York, NY, USA, 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Fraser, N.; Brierley, L.; Dey, G.; Polka, J.K.; Pálfy, M.; Nanni, F.; Coates, J.A. The evolving role of preprints in the dissemination of COVID-19 research and their impact on the science communication landscape. PLoS Biol. 2021, 19, e3000959. [Google Scholar]
- The Impact of COVID-19 on HIV, TB and Malaria Services and Systems for Health. Geneva: The Global Fund to Fight AIDS, Tuberculosis, and Malaria. 2021. Available online: https://www.theglobalfund.org/media/10776/covid-19_2020-disruption-impact_report_en.pdf (accessed on 21 September 2022).
| Study No. (Reference No.) | First Author (Publication Date) | Location | Study Population | Sample Size | Study Design | Study Endpoints |
|---|---|---|---|---|---|---|
| 1 [14] | Boulle (August 2020) | Western Cape, South Africa | Persons ≥ 20 years old from the public-sector Western Cape Provincial Health Data Center who were not known to have died before 1 March 2020, and with follow-up through 9 June 2020 | 22,308 | Retrospective cohort | COVID-19 mortality |
| 2 [15] | Pillay-van Wyk (October 2020) | South Africa | Persons reported to the National Department of Health as having died from COVID-19, 28 March 2020–3 July 2020 | 3088 | Retrospective cohort | COVID-19 mortality |
| 3 [16] | Torun (October 2020) | Turkey | Healthcare workers with confirmed COVID-19 from 3 hospitals who responded to an online questionnaire | 465 | Retrospective cohort | COVID-19 hospitalization and radiological pneumonia |
| 4 [17] | Sun (April 2020) | Beijing, China | Persons hospitalized with confirmed, non-imported COVID-19 | 63 | Retrospective cohort | COVID-19 disease severity |
| 5 [18] | van der Zalm (June 2021) | Cape Town, South Africa | Children 0–13 years old with confirmed COVID-19 presenting to Tygerberg Hospital, 17 April 2020–24 July 2020 | 159 | Retrospective cohort | COVID-19 hospitalization |
| 6 [19] | Sy (July 2020) | Philippines | Persons with COVID-19 reported by 17 May 2020 and followed until 17 June 2020 | 12,513 | Prospective cohort | COVID-19 mortality, recovery from COVID-19 |
| 7 [20] | Jassat (December 2020) | South Africa | Persons with confirmed COVID-19 reported in national surveillance system (DATCOV) for COVID-19 hospitalization, 5 March 2020–11 August 2020 | 41,877 | Retrospective cohort | COVID-19 related in-hospital mortality |
| 8 [21] | Gajbhiye (February 2021) | Mumbai, India | Pregnant and postpartum women with COVID-19 admitted to BYL Nair Hospital, April 2020–September 2020 | 18 | Retrospective cohort | Maternal outcomes, including COVID-19 mortality |
| 9 [22] | Gupta (October 2020) | New Delhi, India | Persons admitted to Safdarjung Hospital with COVID-19 and current or prior treated TB, 1 February 2020–14 June 2020 | 22 | Retrospective cohort | COVID-19 mortality, ICU, IMV |
| 10 [23] | Fisman (September 2020) | Ontario, Canada | Persons with confirmed COVID-19, 23 January 2020–15 May 2020 | 21,992 | Retrospective cohort | COVID-19 mortality |
| 11 [24] | Demkina (November 2020) | Russia | Persons hospitalized with and treated for COVID-19 reported in the Federal Register of COVID-19 Patients, 26 March 2020–3 June 2020 | 541,377 | Retrospective cohort | Length of COVID-19 hospitalization, mortality, ICU transfer, IMV |
| 12 [25] | Kuwari (July 2020) | Qatar | Persons with confirmed COVID-19 reported to the Ministry of Public Health, 28 February 2020–18 April 2020 | 5685 | Retrospective cohort | COVID-19 disease severity |
| 13 [26] | Meizhu Chen (April 2020) | Zhuhai, China | Persons hospitalized with COVID-19 at Fifth Affiliated Hospital of Sun Yat-sen University, 17 January 2020–10 March 2020 | 97 | Retrospective cohort | COVID-19 disease severity |
| 14 [27] | Lei (March 2020) | Daofu, Sichuan, China | Persons from Tibet hospitalized with confirmed COVID-19 at Daofu People’s Hospital, 4 January 2020–28 February 2020, and followed until 5 March 2020 | 67 | Prospective cohort | COVID-19 symptoms |
| 15 [28] | Yu Chen (March 2020) | Shenyang, China | Persons hospitalized with confirmed COVID-19 at 4 hospitals, 26 January 2020–15 February 2020 | 36 | Case control | COVID-19 disease severity |
| 16 [29] | Hongyan Zhang (September 2020) | Wuhan, Hubei, China | Persons with cancer and COVID-19 treated at 5 hospitals, 5 January 2020–March 18 2020 | 107 | Retrospective cohort | COVID-19 mortality and severity |
| 17 [30] | Huizheng Zhang (March 2020) | Chongqing, China | Persons hospitalized with COVID-19 at Chongqing Public Health Medical Center, 11 February 2020–28 February 2020 | 43 | Retrospective cohort | COVID-19 disease severity |
| 18 [31] | Bi (May 2020) | Shenzhen, China | Persons hospitalized with confirmed COVID-19 at Shenzhen Third People’s Hospital, 11 January 2020–10 March 2020, and followed until 7 April 2020 | 420 | Prospective cohort | COVID-19 disease severity |
| Study No. (Reference No.) | Exposure | COVID-19-Related Deaths/ Exposed (%) | COVID-19-Related Deaths/ Unexposed (%) | Unadjusted Estimate (95% CI) | Adjusted Estimate (95% CI) |
|---|---|---|---|---|---|
| 1 [14] | Boulle, August 2020 (South Africa) | ||||
| 1a. Population: All persons in the public sector | |||||
| Prior TB | 87/1785 (4.9%) | 512/20,180 (2.5%) | None | 1.79 (1.42–2.24) a,h | |
| None | 1.81 (1.44–2.28) a,i | ||||
| None | 1.51 (1.18–1.93) a,b | ||||
| Current TB | 26/343 (7.6%) | 512/20,180 (2.5%) | None | 2.79 (1.88–4.13) a,h | |
| None | 3.29 (2.21–4.88) a,i | ||||
| None | 2.70 (1.81–4.04) a,b | ||||
| 1b. Population: Persons in the public sector diagnosed with COVID-19 before 06/01/2020 | |||||
| Prior TB | 74/1254 (5.9%) | 414/13,744 (3.0%) | None | 1.55 (1.19–2.02) a | |
| Current TB | 22/235 (9.4%) | 414/13,744 (3.0%) | None | 1.62 (1.04–2.51) a | |
| 1c. Population: Persons hospitalized in the public sector diagnosed with COVID-19 before 06/01/2020 | |||||
| Prior TB | 77/321 (24.0%) | 448/2509 (17.9%) | None | 1.40 (1.08–1.82) a | |
| Current TB | 25/148 (16.9%) | 448/2509 (17.9%) | None | 1.09 (0.72–1.65) a | |
| 2 [15] | Pillay-van Wyk, October 2020 (South Africa) | ||||
| Population: All persons diagnosed with COVID-19 and current TB reported to the National Department of Health as having died from COVID-19 | |||||
| Age | None | None | None | ||
| <50 | 37/476 (7.8%) | ||||
| 50–69 | 33/1270 (2.6%) | ||||
| >=70 | 10/704 (1.4%) | ||||
| Sex | None | None | None | ||
| Male | 50/1259 (4.0%) | ||||
| Female | 30/1198 (2.5%) | ||||
| Location | None | None | None | ||
| Western Cape | 62/1587 (3.9%) | ||||
| Eastern Cape | 8/406 (2.0%) | ||||
| Gauteng | 6/312 (1.9%) | ||||
| Other provinces | 4/152 (2.6%) | ||||
| 6 [19] | Sy, July 2020 (Philippines) | ||||
| Current and prior TB—full cohort | 25/106 (23.6%) | 46/424 (10.8%) | NA | 2.17 (1.40–3.37) c,f | |
| Current and prior TB—hospitalized patients | 18/66 (27.3%) | 32/264 (12.1%) | NA | 2.25 (1.35–3.75) c,f | |
| 7 [20] | Jassat, December 2020 (South Africa) | ||||
| No history of TB | 6469/34,464 (18.8%) | Reference | Reference | ||
| Prior TB | 202/741 (27.3%) | 1.10 (0.90–1.30) d | 1.30 g | ||
| Current TB | 59/238 (24.8%) | 1.60 (1.20–2.20) d | 2.00 g | ||
| Current and prior TB | 92/346 (26.6%) | 1.10 (0.90–1.40) d | 2.20 g | ||
| 8 [21] | Gajbhiye, February 2020 (India) | ||||
| Current TB | 1/6 (16.7%) | 5.57 (0.26–119.53) f | |||
| Prior (treated) TB | 0/12 (0.0%) | Reference | |||
| 9 [22] | Gupta, October 2020 (India) | ||||
| Current TB | 3/13 (23.1%) | 0.69 (0.18–2.69) f | |||
| Prior (treated) TB | 3/9 (33.3%) | Reference | |||
| 10 [23] | Fisman, September 2020 (Canada) | ||||
| TB cases (timing not specified) | NR/52 | 0.88 (0.21–3.70) d | |||
| 11 [24] | Demkina, November 2020 (Russia) | ||||
| TB cases (timing not specified) | NR/324 | NR/541,053 | 1.74 (1.11–2.71) a,j | ||
| 16 [29] | Hongyan Zhang, March 2020 (China) | ||||
| TB cases (timing not specified) | 0/3 (0.0%) | 23/104 (22.1%) | None | None | |
| Study No. (Reference No.) | Exposure | Events/ Exposed (%) | Events/ Unexposed (%) | Unadjusted Estimate (95% CI) | Adjusted Estimate (95% CI) |
|---|---|---|---|---|---|
| Endpoint: Hospitalization | |||||
| 3 [16] | Torun, October 2020 (Turkey) | ||||
| TB treatment history | 4/4 (100.0%) | 58/132 (43.9%) | None | None | |
| Positive PPD | 39/96 (40.6%) | 178/369 (48.2%) | 0.84 (0.65–1.10) a | None | |
| Radiological pneumonia | 116/138 (84.1%) | 69/263 (26.2%) | 3.20 (2.58–3.97) a | None | |
| 5 [18] | van der Zalm, June 2021 (South Africa) | ||||
| Children with current pulmonary TB vs. none | 2/2 (100.0%) | 60/157 (38.2%) | None | None | |
| 6 [19] | Sy, July 2020 (Philippines) | ||||
| Current and prior TB vs. none e | 67/106 (63.2%) | 236/424 (55.7%) | None | 1.20 (1.04–1.38) b | |
| 7 [20] | Jassat, December 2020 (South Africa) | ||||
| Prior TB vs. none | 341/741 (46%) | 2403/34,184 (7.0%) | 4.0 (3.3–4.9) c | None | |
| Current TB vs. none | 80/238 (33.6%) | 2403/34,184 (7.0%) | 7.70 (5.40–10.90) c | None | |
| Current and prior TB vs. none | 212/346 (61.3%) | 2403/34,184 (7.0%) | 8.80 (6.90–11.30) c | None | |
| Endpoint: Severity | |||||
| 8 [21] | Gajbhiye, 2021 (India) | ||||
| Current TB (diagnosed in course of COVID-19 hospitalization vs. prior to hospitalization) | 1/6 (33.3%) | None | None | None | |
| 12 [25] | Kuwari, 2020 (Qatar) | ||||
| TB (timing not specified) | 3/13 (23.1%) | 291/5449 (5.3%) | 2.55 (0.94–6.90) a | None | |
| 13 [26] | Meizhu Chen, 2020 (China) | ||||
| Prior TB vs. none | 2/2 (100.0%) | 24/95 (25.3%) | None | None | |
| 15 [28] | Yu Chen, 2020 (China) | ||||
| Current active TB | 3/3 (100%) | 2/23 (8.7%) | None | None | |
| Current latent TB | 0/2 (0.0%) | 2/23 (8.7%) | None | None | |
| Prior TB | 4/8 (50.0%) | 2/23 (8.7%) | 5.25 (1.18–23.28) a | None | |
| Current and prior TB | 7/13 (77.8%) | 2/23 (8/7%) | 6.19 (1.43–26.90) a | None | |
| 16 [29] | Hongyan Zhang, 2020 (China) | ||||
| TB (timing not specified) vs. none | 0/3 (0.0%) | 56/104 (53.9%) | None | None | |
| 17 [30] | Huizheng Zhang, 2020 (China) | ||||
| TB (timing not specified) vs. none | 0/1 (0.0%) | 14/42 (33.3%) | None | None | |
| 18 [31] | Bi, 2020 (China) | ||||
| Prior TB vs. none | 1/9 (11.11%) | 92/411 (22.4%) | 0.50 (0.08–3.20) a | None | |
| Endpoint: Admitted to the intensive care unit | |||||
| 9 [22] | Gupta, 2020 (India) | ||||
| Prior (treated) vs. current active TB patients | 4/13 (30.8%) | 3/9 (33.3%) | 0.92 (0.27–3.17) a | None | |
| 11 [24] | Demkina, November, 2020 (Russia) | ||||
| TB (timing not specified) | Not reported | Not reported | Not reported | 0.27 (0.11–0.70) c | |
| Endpoint: Received invasive mechanical ventilation | |||||
| 9 [22] | Gupta, 2020 (India) | ||||
| Prior (treated) vs. current active TB patients | 3/13 (23.1%) | 3/9 (33.3%) | 0.69 (0.17–2.69) a | None | |
| 11 [24] | Demkina, November, 2020 (Russia) | ||||
| TB (timing not specified) | Not reported | Not reported | 0.80 (0.33–1.94) | None | |
| Endpoint: Radiologic pneumonia | |||||
| 3 [16] | Torun, October 2020 (Turkey) | ||||
| TB treatment history | 3/4 (75.0%) | 40/111 (36.0%) | 2.08 (1.12–3.86) a | None | |
| Positive PPD | 30/88 (34.1%) | 108/313 (34.5%) | 0.99 (0.71–1.37) a | None | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jhaveri, T.A.; Fung, C.; LaHood, A.N.; Lindeborg, A.; Zeng, C.; Rahman, R.; Bain, P.A.; Velásquez, G.E.; Mitnick, C.D. Clinical Outcomes of Individuals with COVID-19 and Tuberculosis during the Pre-Vaccination Period of the Pandemic: A Systematic Review. J. Clin. Med. 2022, 11, 5656. https://doi.org/10.3390/jcm11195656
Jhaveri TA, Fung C, LaHood AN, Lindeborg A, Zeng C, Rahman R, Bain PA, Velásquez GE, Mitnick CD. Clinical Outcomes of Individuals with COVID-19 and Tuberculosis during the Pre-Vaccination Period of the Pandemic: A Systematic Review. Journal of Clinical Medicine. 2022; 11(19):5656. https://doi.org/10.3390/jcm11195656
Chicago/Turabian StyleJhaveri, Tulip A., Celia Fung, Allison N. LaHood, Andrew Lindeborg, Chengbo Zeng, Rifat Rahman, Paul A. Bain, Gustavo E. Velásquez, and Carole D. Mitnick. 2022. "Clinical Outcomes of Individuals with COVID-19 and Tuberculosis during the Pre-Vaccination Period of the Pandemic: A Systematic Review" Journal of Clinical Medicine 11, no. 19: 5656. https://doi.org/10.3390/jcm11195656
APA StyleJhaveri, T. A., Fung, C., LaHood, A. N., Lindeborg, A., Zeng, C., Rahman, R., Bain, P. A., Velásquez, G. E., & Mitnick, C. D. (2022). Clinical Outcomes of Individuals with COVID-19 and Tuberculosis during the Pre-Vaccination Period of the Pandemic: A Systematic Review. Journal of Clinical Medicine, 11(19), 5656. https://doi.org/10.3390/jcm11195656






