An Update on the Management of Acute High-Risk Pulmonary Embolism
Abstract
:1. Introduction
2. Methods
3. Risk Stratification and Early Diagnosis of High-Risk Pulmonary Embolism
3.1. Risk Stratification
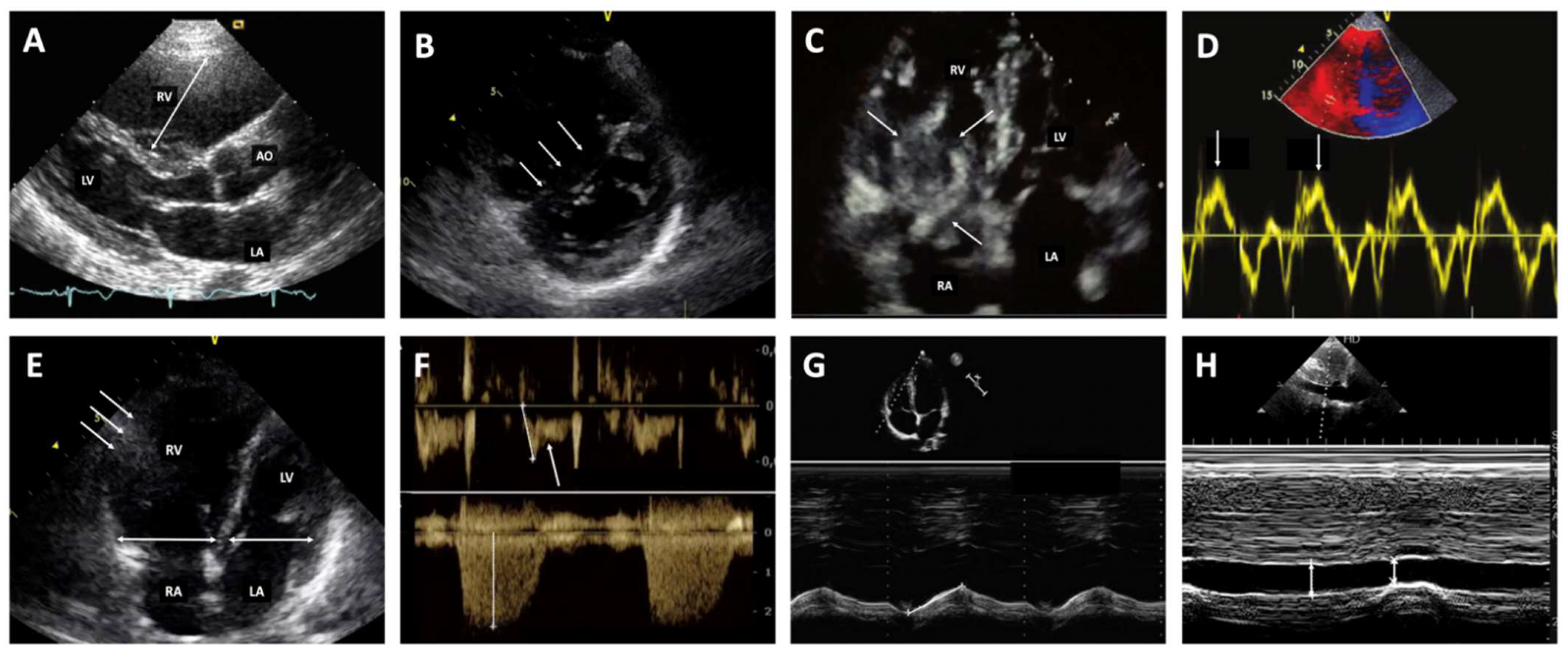
3.2. Diagnostic Strategy
4. Multidisciplinary Response Team
5. Emergency Management of Acute High-Risk Pulmonary Embolism
5.1. Anticoagulant Therapy
5.2. Hemodynamic and Respiratory Support
5.2.1. Oxygen Therapy
5.2.2. Modest Fluid Loading
5.2.3. Vasopressors or Inotropes
5.2.4. Cardio-Pulmonary Resuscitation and Extra-Corporeal Membrane Oxygenation
5.3. Reperfusion Therapies
5.3.1. Full-Dose Systemic Thrombolysis
5.3.2. Half-Dose Systemic Thrombolysis
5.3.3. Catheter-Directed Therapy
5.3.4. Surgical Embolectomy
6. Special Situations
6.1. High-Risk PE Associated with Pregnancy
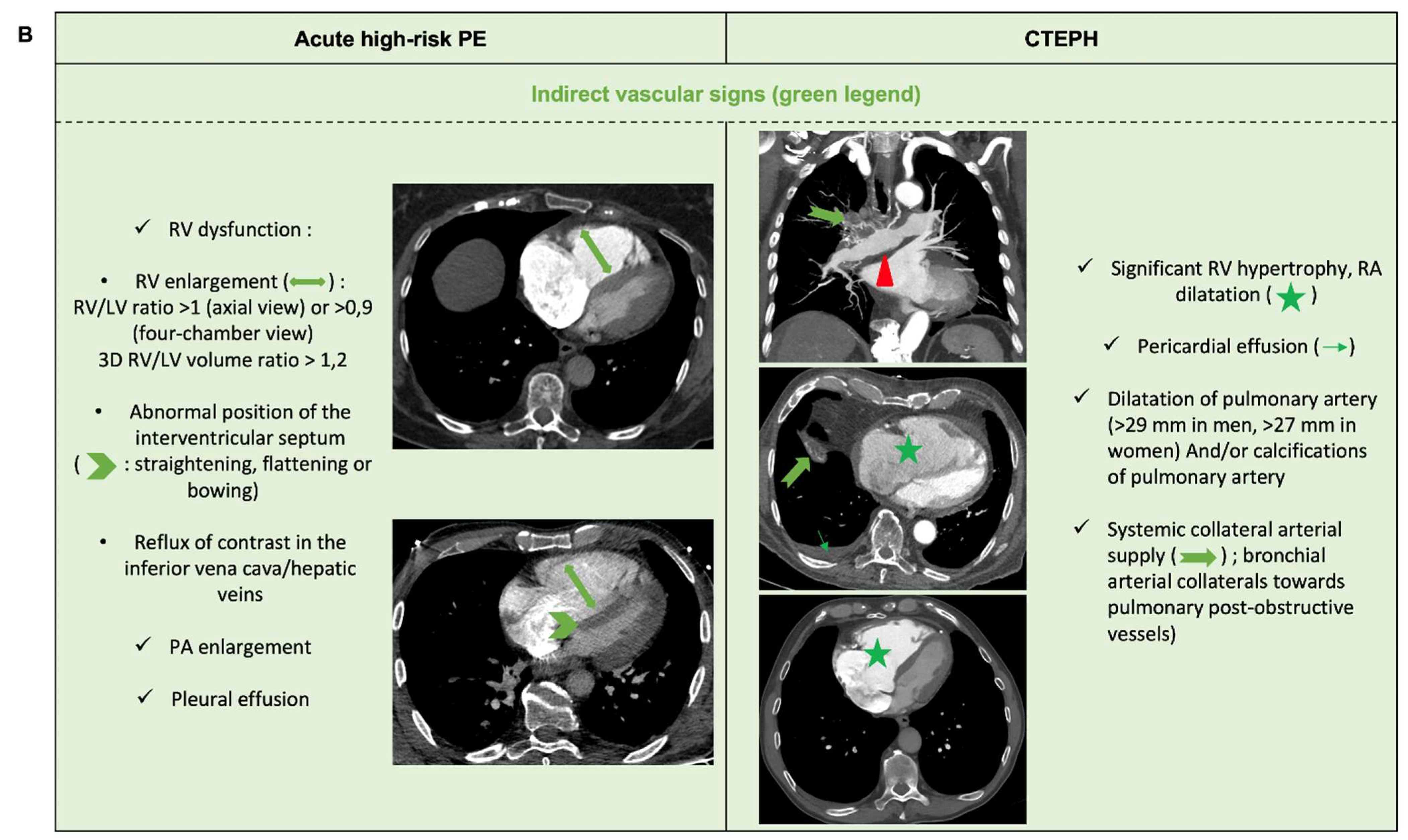
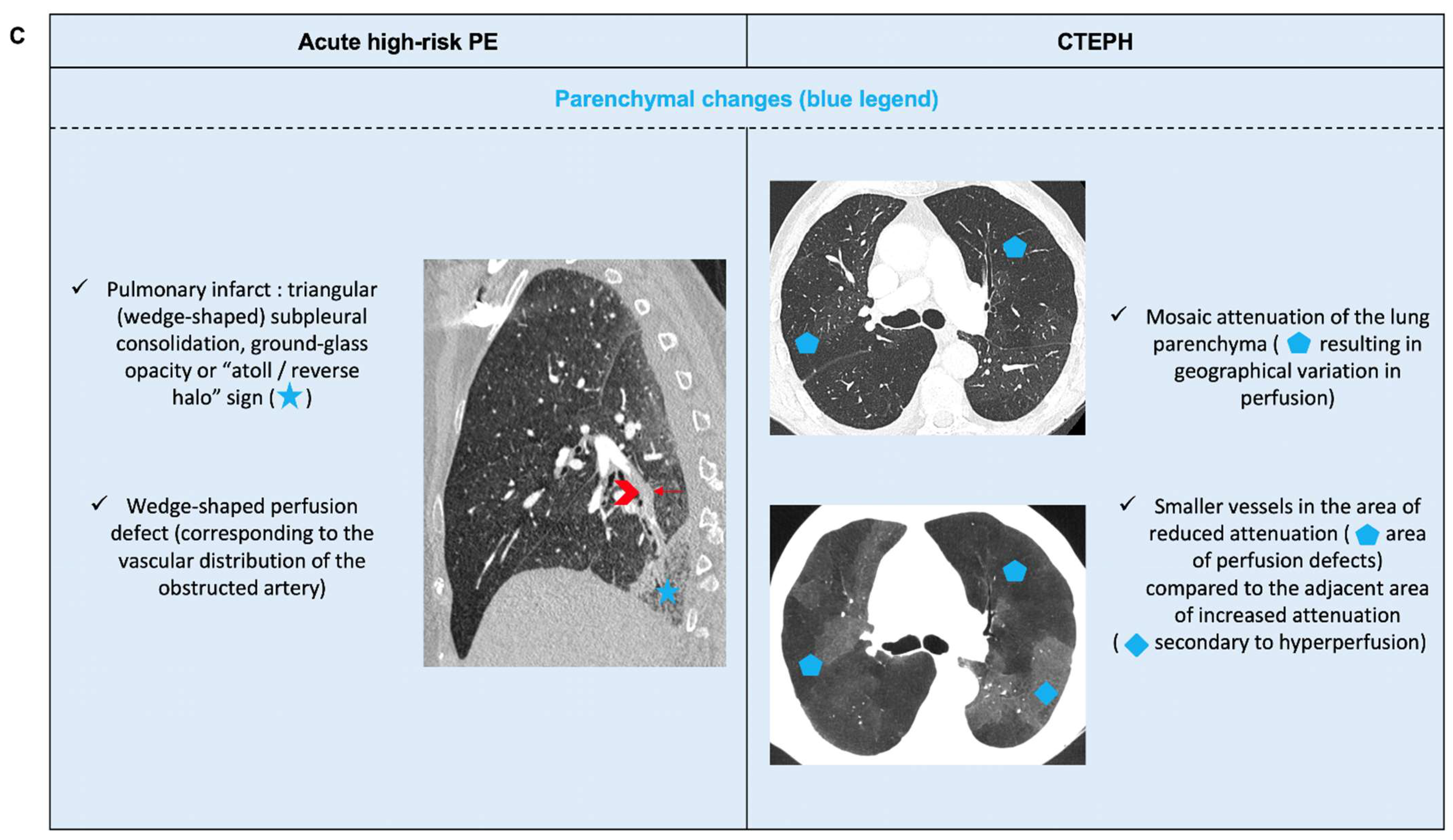
6.2. Acute PE with Underlying Chronic Thrombo-Embolic Pulmonary Hypertension
7. Current Controversies Warranting Further Research
7.1. Risk Stratification
7.2. Pulmonary Reperfusion Strategy
7.2.1. Failure of Systemic Thrombolysis
7.2.2. Right-Heart Thrombus
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wendelboe, A.M.; Raskob, G.E. Global Burden of Thrombosis: Epidemiologic Aspects. Circ. Res. 2016, 118, 1340–1347. [Google Scholar] [CrossRef] [PubMed]
- Goldhaber, S.Z.; Bounameaux, H. Pulmonary embolism and deep vein thrombosis. Lancet 2012, 379, 1835–1846. [Google Scholar] [CrossRef]
- Konstantinides, S.V.; Meyer, G.; Becattini, C.; Bueno, H.; Geersing, G.J.; Harjola, V.P.; Huisman, M.V.; Humbert, M.; Jennings, C.S.; Jimenez, D.; et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur. Heart J. 2020, 41, 543–603. [Google Scholar] [CrossRef] [PubMed]
- Stevens, S.M.; Woller, S.C.; Kreuziger, L.B.; Bounameaux, H.; Doerschug, K.; Geersing, G.J.; Huisman, M.V.; Kearon, C.; King, C.S.; Knighton, A.J.; et al. Antithrombotic Therapy for VTE Disease: Second Update of the CHEST Guideline and Expert Panel Report. Chest 2021, 160, e545–e608. [Google Scholar] [CrossRef] [PubMed]
- Becattini, C.; Agnelli, G.; Lankeit, M.; Masotti, L.; Pruszczyk, P.; Casazza, F.; Vanni, S.; Nitti, C.; Kamphuisen, P.; Vedovati, M.C.; et al. Acute pulmonary embolism: Mortality prediction by the 2014 European Society of Cardiology risk stratification model. Eur. Respir. J. 2016, 48, 780–786. [Google Scholar] [CrossRef]
- Casazza, F.; Becattini, C.; Bongarzoni, A.; Cuccia, C.; Roncon, L.; Favretto, G.; Zonzin, P.; Pignataro, L.; Agnelli, G. Clinical features and short term outcomes of patients with acute pulmonary embolism. The Italian Pulmonary Embolism Registry (IPER). Thromb. Res. 2012, 130, 847–852. [Google Scholar] [CrossRef]
- Kasper, W.; Konstantinides, S.; Geibel, A.; Olschewski, M.; Heinrich, F.; Grosser, K.D.; Rauber, K.; Iversen, S.; Redecker, M.; Kienast, J. Management strategies and determinants of outcome in acute major pulmonary embolism: Results of a multicenter registry. J. Am. Coll. Cardiol. 1997, 30, 1165–1171. [Google Scholar] [CrossRef]
- Keller, K.; Hobohm, L.; Ebner, M.; Kresoja, K.P.; Munzel, T.; Konstantinides, S.V.; Lankeit, M. Trends in thrombolytic treatment and outcomes of acute pulmonary embolism in Germany. Eur. Heart J. 2020, 41, 522–529. [Google Scholar] [CrossRef]
- Ebner, M.; Kresoja, K.P.; Keller, K.; Hobohm, L.; Rogge, N.I.J.; Hasenfuss, G.; Pieske, B.; Konstantinides, S.V.; Lankeit, M. Temporal trends in management and outcome of pulmonary embolism: A single-centre experience. Clin. Res. Cardiol. 2020, 109, 67–77. [Google Scholar] [CrossRef]
- Jimenez, D.; Aujesky, D.; Moores, L.; Gomez, V.; Lobo, J.L.; Uresandi, F.; Otero, R.; Monreal, M.; Muriel, A.; Yusen, R.D.; et al. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch. Intern. Med. 2010, 170, 1383–1389. [Google Scholar] [CrossRef]
- Aujesky, D.; Obrosky, D.S.; Stone, R.A.; Auble, T.E.; Perrier, A.; Cornuz, J.; Roy, P.M.; Fine, M.J. Derivation and validation of a prognostic model for pulmonary embolism. Am. J. Respir. Crit. Care Med. 2005, 172, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Meinel, F.G.; Nance, J.W., Jr.; Schoepf, U.J.; Hoffmann, V.S.; Thierfelder, K.M.; Costello, P.; Goldhaber, S.Z.; Bamberg, F. Predictive Value of Computed Tomography in Acute Pulmonary Embolism: Systematic Review and Meta-analysis. Am. J. Med. 2015, 128, 747–759.e2. [Google Scholar] [CrossRef] [PubMed]
- Becattini, C.; Vedovati, M.C.; Agnelli, G. Prognostic value of troponins in acute pulmonary embolism: A meta-analysis. Circulation 2007, 116, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Mos, I.C.; Huisman, M.V. Brain-type natriuretic peptide levels in the prediction of adverse outcome in patients with pulmonary embolism: A systematic review and meta-analysis. Am. J. Respir. Crit. Care Med. 2008, 178, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.; Rahko, P.S.; Blauwet, L.A.; Canaday, B.; Finstuen, J.A.; Foster, M.C.; Horton, K.; Ogunyankin, K.O.; Palma, R.A.; Velazquez, E.J. Guidelines for Performing a Comprehensive Transthoracic Echocardiographic Examination in Adults: Recommendations from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2019, 32, 1–64. [Google Scholar] [CrossRef] [PubMed]
- Crawford, F.; Andras, A.; Welch, K.; Sheares, K.; Keeling, D.; Chappell, F.M. D-dimer test for excluding the diagnosis of pulmonary embolism. Cochrane Database Syst. Rev. 2016, 2016, CD010864. [Google Scholar] [CrossRef]
- Provias, T.; Dudzinski, D.M.; Jaff, M.R.; Rosenfield, K.; Channick, R.; Baker, J.; Weinberg, I.; Donaldson, C.; Narayan, R.; Rassi, A.N.; et al. The Massachusetts General Hospital Pulmonary Embolism Response Team (MGH PERT): Creation of a multidisciplinary program to improve care of patients with massive and submassive pulmonary embolism. Hosp. Pract. 2014, 42, 31–37. [Google Scholar] [CrossRef]
- Chaudhury, P.; Gadre, S.K.; Schneider, E.; Renapurkar, R.D.; Gomes, M.; Haddadin, I.; Heresi, G.A.; Tong, M.Z.; Bartholomew, J.R. Impact of Multidisciplinary Pulmonary Embolism Response Team Availability on Management and Outcomes. Am. J. Cardiol. 2019, 124, 1465–1469. [Google Scholar] [CrossRef]
- Rosovsky, R.; Chang, Y.; Rosenfield, K.; Channick, R.; Jaff, M.R.; Weinberg, I.; Sundt, T.; Witkin, A.; Rodriguez-Lopez, J.; Parry, B.A.; et al. Changes in treatment and outcomes after creation of a pulmonary embolism response team (PERT), a 10-year analysis. J. Thromb. Thrombolysis 2019, 47, 31–40. [Google Scholar] [CrossRef]
- Jimenez, D.; Rodriguez, C.; Leon, F.; Jara-Palomares, L.; Lopez-Reyes, R.; Ruiz-Artacho, P.; Elias, T.; Otero, R.; Garcia-Ortega, A.; Rivas-Guerrero, A.; et al. Randomised controlled trial of a prognostic assessment and management pathway to reduce the length of hospital stay in normotensive patients with acute pulmonary embolism. Eur. Respir. J. 2021, 59, 2100412. [Google Scholar] [CrossRef]
- Robertson, L.; Jones, L.E. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database Syst. Rev. 2017, 2, CD001100. [Google Scholar] [CrossRef] [PubMed]
- Martel, N.; Lee, J.; Wells, P.S. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: A meta-analysis. Blood 2005, 106, 2710–2715. [Google Scholar] [CrossRef] [PubMed]
- van Es, N.; Coppens, M.; Schulman, S.; Middeldorp, S.; Buller, H.R. Direct oral anticoagulants compared with vitamin K antagonists for acute venous thromboembolism: Evidence from phase 3 trials. Blood 2014, 124, 1968–1975. [Google Scholar] [CrossRef] [PubMed]
- Meyer, G.; Vicaut, E.; Danays, T.; Agnelli, G.; Becattini, C.; Beyer-Westendorf, J.; Bluhmki, E.; Bouvaist, H.; Brenner, B.; Couturaud, F.; et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N. Engl. J. Med. 2014, 370, 1402–1411. [Google Scholar] [CrossRef]
- Kline, J.A.; Nordenholz, K.E.; Courtney, D.M.; Kabrhel, C.; Jones, A.E.; Rondina, M.T.; Diercks, D.B.; Klinger, J.R.; Hernandez, J. Treatment of submassive pulmonary embolism with tenecteplase or placebo: Cardiopulmonary outcomes at 3 months: Multicenter double-blind, placebo-controlled randomized trial. J. Thromb. Haemost. 2014, 12, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Janin, S.; Meneveau, N.; Mahemuti, A.; Descotes-Genon, V.; Dutheil, J.; Chopard, R.; Seronde, M.F.; Schiele, F.; Bernard, Y.; Bassand, J.P. Safety and efficacy of fondaparinux as an adjunctive treatment to thrombolysis in patients with high and intermediate risk pulmonary embolism. J. Thromb. Thrombolysis 2009, 28, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, M.; Bay, C.; Schwartz, F.; Skrocki, L. Safe-dose thrombolysis plus rivaroxaban for moderate and severe pulmonary embolism: Drip, drug, and discharge. Clin. Cardiol. 2014, 37, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Kearon, C.; Ginsberg, J.S.; Julian, J.A.; Douketis, J.; Solymoss, S.; Ockelford, P.; Jackson, S.; Turpie, A.G.; MacKinnon, B.; Hirsh, J.; et al. Comparison of fixed-dose weight-adjusted unfractionated heparin and low-molecular-weight heparin for acute treatment of venous thromboembolism. JAMA 2006, 296, 935–942. [Google Scholar] [CrossRef]
- Buller, H.R.; Davidson, B.L.; Decousus, H.; Gallus, A.; Gent, M.; Piovella, F.; Prins, M.H.; Raskob, G.; van den Berg-Segers, A.E.; Cariou, R.; et al. Subcutaneous fondaparinux versus intravenous unfractionated heparin in the initial treatment of pulmonary embolism. N. Engl. J. Med. 2003, 349, 1695–1702. [Google Scholar] [CrossRef]
- Cuker, A.; Arepally, G.M.; Chong, B.H.; Cines, D.B.; Greinacher, A.; Gruel, Y.; Linkins, L.A.; Rodner, S.B.; Selleng, S.; Warkentin, T.E.; et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: Heparin-induced thrombocytopenia. Blood Adv. 2018, 2, 3360–3392. [Google Scholar] [CrossRef]
- Harjola, V.P.; Mebazaa, A.; Celutkiene, J.; Bettex, D.; Bueno, H.; Chioncel, O.; Crespo-Leiro, M.G.; Falk, V.; Filippatos, G.; Gibbs, S.; et al. Contemporary management of acute right ventricular failure: A statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the European Society of Cardiology. Eur. J. Heart Fail. 2016, 18, 226–241. [Google Scholar] [CrossRef] [PubMed]
- Belenkie, I.; Dani, R.; Smith, E.R.; Tyberg, J.V. Ventricular interaction during experimental acute pulmonary embolism. Circulation 1988, 78, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Mercat, A.; Diehl, J.L.; Meyer, G.; Teboul, J.L.; Sors, H. Hemodynamic effects of fluid loading in acute massive pulmonary embolism. Crit. Care Med. 1999, 27, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Ghignone, M.; Girling, L.; Prewitt, R.M. Volume expansion versus norepinephrine in treatment of a low cardiac output complicating an acute increase in right ventricular afterload in dogs. Anesthesiology 1984, 60, 132–135. [Google Scholar] [CrossRef]
- Jardin, F.; Genevray, B.; Brun-Ney, D.; Margairaz, A. Dobutamine: A hemodynamic evaluation in pulmonary embolism shock. Crit. Care Med. 1985, 13, 1009–1012. [Google Scholar] [CrossRef]
- Perkins, G.D.; Olasveengen, T.M.; Maconochie, I.; Soar, J.; Wyllie, J.; Greif, R.; Lockey, A.; Semeraro, F.; Van de Voorde, P.; Lott, C.; et al. European Resuscitation Council Guidelines for Resuscitation: 2017 update. Resuscitation 2018, 123, 43–50. [Google Scholar] [CrossRef]
- Baldetti, L.; Beneduce, A.; Cianfanelli, L.; Falasconi, G.; Pannone, L.; Moroni, F.; Venuti, A.; Sacchi, S.; Gramegna, M.; Pazzanese, V.; et al. Use of extracorporeal membrane oxygenation in high-risk acute pulmonary embolism: A systematic review and meta-analysis. Artif. Organs 2021, 45, 569–576. [Google Scholar] [CrossRef]
- Ly, B.; Arnesen, H.; Eie, H.; Hol, R. A controlled clinical trial of streptokinase and heparin in the treatment of major pulmonary embolism. Acta Med. Scand. 1978, 203, 465–470. [Google Scholar] [CrossRef]
- Jerjes-Sanchez, C.; Ramirez-Rivera, A.; de Lourdes Garcia, M.; Arriaga-Nava, R.; Valencia, S.; Rosado-Buzzo, A.; Pierzo, J.A.; Rosas, E. Streptokinase and Heparin versus Heparin Alone in Massive Pulmonary Embolism: A Randomized Controlled Trial. J. Thromb. Thrombolysis 1995, 2, 227–229. [Google Scholar] [CrossRef]
- Dotter, C.T.; Seaman, A.J.; Rösch, J.; Porter, J.M. Streptokinase and Heparin in the Treatment of Pulmonary Embolism: A Randomized Comparison. Vasc. Surg. 1979, 13, 42–52. [Google Scholar] [CrossRef]
- Urokinase pulmonary embolism trial. Phase 1 results: A cooperative study. JAMA 1970, 214, 2163–2172. [CrossRef]
- Marti, C.; John, G.; Konstantinides, S.; Combescure, C.; Sanchez, O.; Lankeit, M.; Meyer, G.; Perrier, A. Systemic thrombolytic therapy for acute pulmonary embolism: A systematic review and meta-analysis. Eur. Heart J. 2015, 36, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Z.; Yue, J.; Dong, B.R.; Wu, T.; Liu, G.J.; Hao, Q. Thrombolytic therapy for pulmonary embolism. Cochrane Database Syst. Rev. 2021, 4, CD004437. [Google Scholar] [CrossRef] [PubMed]
- Fiumara, K.; Kucher, N.; Fanikos, J.; Goldhaber, S.Z. Predictors of major hemorrhage following fibrinolysis for acute pulmonary embolism. Am. J. Cardiol. 2006, 97, 127–129. [Google Scholar] [CrossRef]
- Goldhaber, S.Z.; Visani, L.; De Rosa, M. Acute pulmonary embolism: Clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet 1999, 353, 1386–1389. [Google Scholar] [CrossRef]
- Stein, P.D.; Matta, F. Thrombolytic therapy in unstable patients with acute pulmonary embolism: Saves lives but underused. Am. J. Med. 2012, 125, 465–470. [Google Scholar] [CrossRef]
- Kiser, T.H.; Burnham, E.L.; Clark, B.; Ho, P.M.; Allen, R.R.; Moss, M.; Vandivier, R.W. Half-Dose Versus Full-Dose Alteplase for Treatment of Pulmonary Embolism. Crit. Care Med. 2018, 46, 1617–1625. [Google Scholar] [CrossRef]
- Kucher, N.; Boekstegers, P.; Muller, O.J.; Kupatt, C.; Beyer-Westendorf, J.; Heitzer, T.; Tebbe, U.; Horstkotte, J.; Muller, R.; Blessing, E.; et al. Randomized, controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk pulmonary embolism. Circulation 2014, 129, 479–486. [Google Scholar] [CrossRef]
- Chamsuddin, A.; Nazzal, L.; Kang, B.; Best, I.; Peters, G.; Panah, S.; Martin, L.; Lewis, C.; Zeinati, C.; Ho, J.W.; et al. Catheter-directed thrombolysis with the Endowave system in the treatment of acute massive pulmonary embolism: A retrospective multicenter case series. J. Vasc. Interv. Radiol. 2008, 19, 372–376. [Google Scholar] [CrossRef]
- Lin, P.H.; Annambhotla, S.; Bechara, C.F.; Athamneh, H.; Weakley, S.M.; Kobayashi, K.; Kougias, P. Comparison of percutaneous ultrasound-accelerated thrombolysis versus catheter-directed thrombolysis in patients with acute massive pulmonary embolism. Vascular 2009, 17 (Suppl. S3), 137–147. [Google Scholar] [CrossRef]
- Engelhardt, T.C.; Taylor, A.J.; Simprini, L.A.; Kucher, N. Catheter-directed ultrasound-accelerated thrombolysis for the treatment of acute pulmonary embolism. Thromb. Res. 2011, 128, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, R.J.; Kenney, H.H.; Dunfee, B.L. Thrombus resolution and hemodynamic recovery using ultrasound-accelerated thrombolysis in acute pulmonary embolism. J. Vasc. Interv. Radiol. 2013, 24, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Dumantepe, M.; Uyar, I.; Teymen, B.; Ugur, O.; Enc, Y. Improvements in pulmonary artery pressure and right ventricular function after ultrasound-accelerated catheter-directed thrombolysis for the treatment of pulmonary embolism. J. Card. Surg. 2014, 29, 455–463. [Google Scholar] [CrossRef]
- Quintana, D.; Salsamendi, J.; Fourzali, R.; Narayanan, G. Ultrasound-assisted thrombolysis in submassive and massive pulmonary embolism: Assessment of lung obstruction before and after catheter-directed therapy. Cardiovasc. Interv. Radiol. 2014, 37, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Bagla, S.; Smirniotopoulos, J.B.; van Breda, A.; Sheridan, M.J.; Sterling, K.M. Ultrasound-accelerated catheter-directed thrombolysis for acute submassive pulmonary embolism. J. Vasc. Interv. Radiol. 2015, 26, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Engelberger, R.P.; Moschovitis, A.; Fahrni, J.; Willenberg, T.; Baumann, F.; Diehm, N.; Do, D.D.; Baumgartner, I.; Kucher, N. Fixed low-dose ultrasound-assisted catheter-directed thrombolysis for intermediate and high-risk pulmonary embolism. Eur. Heart J. 2015, 36, 597–604. [Google Scholar] [CrossRef]
- McCabe, J.M.; Huang, P.H.; Riedl, L.; Eisenhauer, A.C.; Sobieszczyk, P. Usefulness and safety of ultrasound-assisted catheter-directed thrombolysis for submassive pulmonary emboli. Am. J. Cardiol. 2015, 115, 821–824. [Google Scholar] [CrossRef]
- Nykamp, M.; VandenHull, A.; Remund, T.; Santos, A.; Kelly, P.; Schultz, G.; Laurich, C. Safety and efficacy of ultrasound-accelerated catheter-directed lytic therapy in acute pulmonary embolism with and without hemodynamic instability. J. Vasc. Surg. Venous Lymphat. Disord. 2015, 3, 251–257. [Google Scholar] [CrossRef]
- Piazza, G.; Hohlfelder, B.; Jaff, M.R.; Ouriel, K.; Engelhardt, T.C.; Sterling, K.M.; Jones, N.J.; Gurley, J.C.; Bhatheja, R.; Kennedy, R.J.; et al. A Prospective, Single-Arm, Multicenter Trial of Ultrasound-Facilitated, Catheter-Directed, Low-Dose Fibrinolysis for Acute Massive and Submassive Pulmonary Embolism: The SEATTLE II Study. JACC Cardiovasc. Interv. 2015, 8, 1382–1392. [Google Scholar] [CrossRef]
- Liang, N.L.; Chaer, R.A.; Marone, L.K.; Singh, M.J.; Makaroun, M.S.; Avgerinos, E.D. Midterm outcomes of catheter-directed interventions for the treatment of acute pulmonary embolism. Vascular 2017, 25, 130–136. [Google Scholar] [CrossRef]
- Yoo, J.W.; Choi, H.C.; Lee, S.J.; Cho, Y.J.; Lee, J.D.; Kim, H.C. Comparison between systemic and catheter thrombolysis in patients with pulmonary embolism. Am. J. Emerg. Med. 2016, 34, 985–988. [Google Scholar] [CrossRef]
- Tapson, V.F.; Sterling, K.; Jones, N.; Elder, M.; Tripathy, U.; Brower, J.; Maholic, R.L.; Ross, C.B.; Natarajan, K.; Fong, P.; et al. A Randomized Trial of the Optimum Duration of Acoustic Pulse Thrombolysis Procedure in Acute Intermediate-Risk Pulmonary Embolism: The OPTALYSE PE Trial. JACC Cardiovasc. Interv. 2018, 11, 1401–1410. [Google Scholar] [CrossRef]
- Sista, A.K.; Horowitz, J.M.; Tapson, V.F.; Rosenberg, M.; Elder, M.D.; Schiro, B.J.; Dohad, S.; Amoroso, N.E.; Dexter, D.J.; Loh, C.T.; et al. Indigo Aspiration System for Treatment of Pulmonary Embolism: Results of the EXTRACT-PE Trial. JACC Cardiovasc. Interv. 2021, 14, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Tu, T.; Toma, C.; Tapson, V.F.; Adams, C.; Jaber, W.A.; Silver, M.; Khandhar, S.; Amin, R.; Weinberg, M.; Engelhardt, T.; et al. A Prospective, Single-Arm, Multicenter Trial of Catheter-Directed Mechanical Thrombectomy for Intermediate-Risk Acute Pulmonary Embolism: The FLARE Study. JACC Cardiovasc. Interv. 2019, 12, 859–869. [Google Scholar] [CrossRef] [PubMed]
- Pasrija, C.; Kronfli, A.; Rouse, M.; Raithel, M.; Bittle, G.J.; Pousatis, S.; Ghoreishi, M.; Gammie, J.S.; Griffith, B.P.; Sanchez, P.G.; et al. Outcomes after surgical pulmonary embolectomy for acute submassive and massive pulmonary embolism: A single-center experience. J. Thorac. Cardiovasc. Surg. 2018, 155, 1095–1106.e2. [Google Scholar] [CrossRef] [PubMed]
- Kalra, R.; Bajaj, N.S.; Arora, P.; Arora, G.; Crosland, W.A.; McGiffin, D.C.; Ahmed, M.I. Surgical Embolectomy for Acute Pulmonary Embolism: Systematic Review and Comprehensive Meta-Analyses. Ann. Thorac. Surg. 2017, 103, 982–990. [Google Scholar] [CrossRef]
- Neely, R.C.; Byrne, J.G.; Gosev, I.; Cohn, L.H.; Javed, Q.; Rawn, J.D.; Goldhaber, S.Z.; Piazza, G.; Aranki, S.F.; Shekar, P.S.; et al. Surgical Embolectomy for Acute Massive and Submassive Pulmonary Embolism in a Series of 115 Patients. Ann. Thorac. Surg. 2015, 100, 1245–1251; discussion 1251–1242. [Google Scholar] [CrossRef]
- Leacche, M.; Unic, D.; Goldhaber, S.Z.; Rawn, J.D.; Aranki, S.F.; Couper, G.S.; Mihaljevic, T.; Rizzo, R.J.; Cohn, L.H.; Aklog, L.; et al. Modern surgical treatment of massive pulmonary embolism: Results in 47 consecutive patients after rapid diagnosis and aggressive surgical approach. J. Thorac. Cardiovasc. Surg. 2005, 129, 1018–1023. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Causes of Pregnancy-Related Deaths. Available online: https://www.cdc.gov/reproductivehealth/maternal-mortality/pregnancy-mortality-surveillance-system.htm#causes (accessed on 9 June 2022).
- Martillotti, G.; Boehlen, F.; Robert-Ebadi, H.; Jastrow, N.; Righini, M.; Blondon, M. Treatment options for severe pulmonary embolism during pregnancy and the postpartum period: A systematic review. J. Thromb. Haemost. 2017, 15, 1942–1950. [Google Scholar] [CrossRef]
- Sebastian, N.A.; Spence, A.R.; Bouhadoun, S.; Abenhaim, H.A. Extracorporeal membrane oxygenation in pregnant and postpartum patients: A systematic review. J. Matern. Fetal Neonatal Med. 2020, 1–11, online ahead of print. [Google Scholar] [CrossRef]
- Piazza, G.; Goldhaber, S.Z. Chronic thromboembolic pulmonary hypertension. N. Engl. J. Med. 2011, 364, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Pepke-Zaba, J.; Jansa, P.; Kim, N.H.; Naeije, R.; Simonneau, G. Chronic thromboembolic pulmonary hypertension: Role of medical therapy. Eur. Respir. J. 2013, 41, 985–990. [Google Scholar] [CrossRef]
- Chemla, D.; Lau, E.M.; Papelier, Y.; Attal, P.; Herve, P. Pulmonary vascular resistance and compliance relationship in pulmonary hypertension. Eur. Respir. J. 2015, 46, 1178–1189. [Google Scholar] [CrossRef] [PubMed]
- Abdelnour-Berchtold, E.; Donahoe, L.; McRae, K.; Asghar, U.; Thenganatt, J.; Moric, J.; Cypel, M.; Keshavjee, S.; Granton, J.; de Perrot, M. Central venoarterial extracorporeal membrane oxygenation as a bridge to recovery after pulmonary endarterectomy in patients with decompensated right heart failure. J. Heart Lung Transplant. 2022, 41, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Brookes, J.D.L.; Li, C.; Chung, S.T.W.; Brookes, E.M.; Williams, M.L.; McNamara, N.; Martin-Suarez, S.; Loforte, A. Pulmonary thromboendarterectomy for chronic thromboembolic pulmonary hypertension: A systematic review. Ann. Cardiothorac. Surg. 2022, 11, 68–81. [Google Scholar] [CrossRef]
- Li, W.; Yang, T.; Quan, R.L.; Chen, X.X.; An, J.; Zhao, Z.H.; Liu, Z.H.; Xiong, C.M.; He, J.G.; Gu, Q. Balloon pulmonary angioplasty reverse right ventricular remodelling and dysfunction in patients with inoperable chronic thromboembolic pulmonary hypertension: A systematic review and meta-analysis. Eur. Radiol. 2021, 31, 3898–3908. [Google Scholar] [CrossRef]
- Ebner, M.; Sentler, C.; Harjola, V.P.; Bueno, H.; Lerchbaumer, M.H.; Hasenfuss, G.; Eckardt, K.U.; Konstantinides, S.V.; Lankeit, M. Outcome of patients with different clinical presentations of high-risk pulmonary embolism. Eur. Heart J. Acute Cardiovasc. Care 2021, 10, 787–796. [Google Scholar] [CrossRef]
- Chopard, R.; Jimenez, D.; Serzian, G.; Ecarnot, F.; Falvo, N.; Kalbacher, E.; Bonnet, B.; Capellier, G.; Schiele, F.; Bertoletti, L.; et al. Renal dysfunction improves risk stratification and may call for a change in the management of intermediate- and high-risk acute pulmonary embolism: Results from a multicenter cohort study with external validation. Crit. Care 2021, 25, 57. [Google Scholar] [CrossRef]
- Chatterjee, S.; Chakraborty, A.; Weinberg, I.; Kadakia, M.; Wilensky, R.L.; Sardar, P.; Kumbhani, D.J.; Mukherjee, D.; Jaff, M.R.; Giri, J. Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: A meta-analysis. JAMA 2014, 311, 2414–2421. [Google Scholar] [CrossRef]
- Avgerinos, E.D.; Jaber, W.; Lacomis, J.; Markel, K.; McDaniel, M.; Rivera-Lebron, B.N.; Ross, C.B.; Sechrist, J.; Toma, C.; Chaer, R.; et al. Randomized Trial Comparing Standard Versus Ultrasound-Assisted Thrombolysis for Submassive Pulmonary Embolism: The SUNSET sPE Trial. JACC Cardiovasc. Interv. 2021, 14, 1364–1373. [Google Scholar] [CrossRef]
- Bloomer, T.L.; El-Hayek, G.E.; McDaniel, M.C.; Sandvall, B.C.; Liberman, H.A.; Devireddy, C.M.; Kumar, G.; Fong, P.P.; Jaber, W.A. Safety of catheter-directed thrombolysis for massive and submassive pulmonary embolism: Results of a multicenter registry and meta-analysis. Catheter. Cardiovasc. Interv. 2017, 89, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Hobohm, L.; Schmidt, F.P.; Gori, T.; Schmidtmann, I.; Barco, S.; Munzel, T.; Lankeit, M.; Konstantinides, S.V.; Keller, K. In-hospital outcomes of catheter-directed thrombolysis in patients with pulmonary embolism. Eur. Heart J. Acute Cardiovasc Care 2021, 10, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Klok, F.A.; Piazza, G.; Sharp, A.S.P.; Ainle, F.N.; Jaff, M.R.; Chauhan, N.; Patel, B.; Barco, S.; Goldhaber, S.Z.; Kucher, N.; et al. Ultrasound-facilitated, catheter-directed thrombolysis versus anticoagulation alone for acute intermediate-high-risk pulmonary embolism: Rationale and design of the HI-PEITHO study. Am. Heart J. 2022, 251, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, M.; Ni, Y.; Liang, B.; Liang, Z. Can Systemic Thrombolysis Improve Prognosis of Cardiac Arrest Patients During Cardiopulmonary Resuscitation? A Systematic Review and Meta-Analysis. J. Emerg. Med. 2019, 57, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, M.; Berger, J.; Beeston, P.; Bay, C.; Vajo, Z.; Javadpoor, S. Pulseless electrical activity in pulmonary embolism treated with thrombolysis (from the “PEAPETT” study). Am. J. Emerg. Med. 2016, 34, 1963–1967. [Google Scholar] [CrossRef]
- Corsi, F.; Lebreton, G.; Brechot, N.; Hekimian, G.; Nieszkowska, A.; Trouillet, J.L.; Luyt, C.E.; Leprince, P.; Chastre, J.; Combes, A.; et al. Life-threatening massive pulmonary embolism rescued by venoarterial-extracorporeal membrane oxygenation. Crit. Care 2017, 21, 76. [Google Scholar] [CrossRef]
- Meneveau, N.; Guillon, B.; Planquette, B.; Piton, G.; Kimmoun, A.; Gaide-Chevronnay, L.; Aissaoui, N.; Neuschwander, A.; Zogheib, E.; Dupont, H.; et al. Outcomes after extracorporeal membrane oxygenation for the treatment of high-risk pulmonary embolism: A multicentre series of 52 cases. Eur. Heart J. 2018, 39, 4196–4204. [Google Scholar] [CrossRef]
- Chopard, R.; Nielsen, P.; Ius, F.; Cebotari, S.; Ecarnot, F.; Pilichowski, H.; Schmidt, M.; Kjaergaard, B.; Sousa-Casasnovas, I.; Ghoreishi, M.; et al. Optimal reperfusion strategy in acute high-risk pulmonary embolism requiring extracorporeal membrane oxygenation support: A systematic review and meta-analysis. Eur. Respir. J. 2022, 2102977. [Google Scholar] [CrossRef] [PubMed]
- Meneveau, N.; Seronde, M.F.; Blonde, M.C.; Legalery, P.; Didier-Petit, K.; Briand, F.; Caulfield, F.; Schiele, F.; Bernard, Y.; Bassand, J.P. Management of unsuccessful thrombolysis in acute massive pulmonary embolism. Chest 2006, 129, 1043–1050. [Google Scholar] [CrossRef]
- Gulba, D.C.; Schmid, C.; Borst, H.G.; Lichtlen, P.; Dietz, R.; Luft, F.C. Medical compared with surgical treatment for massive pulmonary embolism. Lancet 1994, 343, 576–577. [Google Scholar] [CrossRef]
- Bikdeli, B.; Lobo, J.L.; Jimenez, D.; Green, P.; Fernandez-Capitan, C.; Bura-Riviere, A.; Otero, R.; DiTullio, M.R.; Galindo, S.; Ellis, M.; et al. Early Use of Echocardiography in Patients With Acute Pulmonary Embolism: Findings From the RIETE Registry. J. Am. Heart Assoc. 2018, 7, e009042. [Google Scholar] [CrossRef] [PubMed]
- Barrios, D.; Rosa-Salazar, V.; Morillo, R.; Nieto, R.; Fernandez, S.; Zamorano, J.L.; Monreal, M.; Torbicki, A.; Yusen, R.D.; Jimenez, D. Prognostic Significance of Right Heart Thrombi in Patients With Acute Symptomatic Pulmonary Embolism: Systematic Review and Meta-analysis. Chest 2017, 151, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Giri, J.; Sista, A.K.; Weinberg, I.; Kearon, C.; Kumbhani, D.J.; Desai, N.D.; Piazza, G.; Gladwin, M.T.; Chatterjee, S.; Kobayashi, T.; et al. Interventional Therapies for Acute Pulmonary Embolism: Current Status and Principles for the Development of Novel Evidence: A Scientific Statement From the American Heart Association. Circulation 2019, 140, e774–e801. [Google Scholar] [CrossRef] [PubMed]
- Athappan, G.; Sengodan, P.; Chacko, P.; Gandhi, S. Comparative efficacy of different modalities for treatment of right heart thrombi in transit: A pooled analysis. Vasc. Med. 2015, 20, 131–138. [Google Scholar] [CrossRef]
- Bikdeli, B.; Jimenez, D.; Muriel, A.; Barrios, D.; Ballaz, A.; Verhamme, P.; Monreal, M.; RIETE Investigators. Association between reperfusion therapy and outcomes in patients with acute pulmonary embolism and right heart thrombi. Eur. Respir. J. 2020, 56, 2000538. [Google Scholar] [CrossRef]

| Drug | Dosage | Mechanism of Action | Efficacy | Adverse Effects | Practical Issues |
|---|---|---|---|---|---|
| Unfractionated heparin | 80 IU/kg bolus followed by 18 IU/kg per hour by continuous infusion | Inhibitor of thrombin and factor Xa through an antithrombin-dependent mechanism | All-cause death within the first 10 days of unstratified VTE (19.0% PE): 0.0% (vs. 0.6% with LMWH) (n = 708) [28] Recurrent VTE within the first 10 days of unstratified VTE (19.0% PE): 0.3% (vs. 0.6% with LMWH) (n = 708) [28] | Major bleeding within the first 10 days of unstratified VTE (19.0% PE): 1.1% (vs. 1.4% with LMWH) (n = 708) [28] | aPTT ratio maintained between 1.5 to 2.0 per normal value. No issue with renal failure. |
| Low-molecular-weight heparin | All-cause death within the first 10 days of unstratified VTE (19.0% PE): 0.6% (vs. 0.0% with UFH) (n = 708) [28] Recurrent VTE during the initial within the first 10 days of unstratified VTE (19.0% PE): 0.6% (vs. 0.3% with UFH) (n = 708) [28] | Major bleeding within the first 10 days of unstratified VTE (19.0% PE): 1.4% (vs. 1.1% with UFH) (n = 708) [28] | To be reduced in case of renal failure. No evidence for dose adjustment based on coagulation tests. | ||
| -Enoxaparin SC | 1.0 mg/kg every 12 h or A.5 mg/kg once per day | ||||
| -Tinzaparin SC | 175 IU/kg once per day | ||||
| -Dalteparin SC | 100 IU/kg every 12 h or 200 IU/kg once per day | ||||
| -Nadroparin SC | 86 IU/kg every 12 h or 171 IU/kg once per day | ||||
| Fondaparinux | Once per day: 5 mg (body weight < 50 kg); 7.5 mg (body weight 50–100 kg); 10 mg (body weight > 100 kg) | Synthetic pentasaccharide that inhibits factor Xa | All-cause death at 3-month FU in unstratified VTE (19.0% of with PE): 0.8% (vs. 1.1% with UFH) (n = 2213) [29] Recurrent VTE within the first 7 days (19.0% of with PE): 1.3% (vs. 1.7% with UFH) (n = 2213) [29] | Major bleeding within the first 7 days in unstratified VTE (19.0% of with PE): 1.3% (vs. 1.1% with UFH) (n = 2213) [29] | Avoid in case of renal failure. No evidence for dose adjustment based on coagulation tests. |
| Argatroban (primarily in patients with suspected or confirmed HIT) | Initial: 2 mcg/kg/min IV continuous infusion over 1–3 h until steady state. Not to exceed infusion rate of 10 mcg/kg/min | Specific and reversible direct thrombin inhibitor | Thrombosis: 5.8% and 6.9% any new thrombosis at 30 days in HIT patients (vs. 15.0% and 23.0% in historical control groups) (n = 177 and n = 328) [30] | Major bleeding: between 3.1% and 5.3% at 30 days in patient with HIT (compared with between 8.2% and 8.6% in historical control groups) (n = 177 and n = 328) [30] | aPTT ratio maintained between 1.5–3 times initial baseline value. Check aPTT and adjust dose until target aPTT is achieved. To be reduced in case of renal failure Use caution in hepatic impairment. |
| Bivalirudin (primarily in patients with suspected or confirmed HIT) | Initial: 0.15–0.2 mg/kg/h IV; | Specific and reversible direct thrombin inhibitor | Thrombosis: 4.6% any new thrombosis at 30 days in patients with HIT (no comparator) (n = 461) [30] | Major bleeding: 7.6% at 30 days in patient with HIT (no comparator) (n = 461) [30] | Adjust to aPTT 1.5–2.5 times baseline value. Use caution in renal impairment: IV infusion, -Moderate (CrCl 30–59 mL/min): 1.75 mg/kg/h -Severe (CrCl < 30 mL/min): 1 mg/kg/h Hemodialysis: 0.25 mg/kg/h |
| No. of Patients | Eligibility | Severity Criteria | Thrombolysis | High-Risk PE (%) | Main Results | |
|---|---|---|---|---|---|---|
| UPET (1970) [41] | 160 | Acute PE < 5 days | Yes | Urokinase 12 h | 8.7% | Rapid improvement of RV function and pulmonary reperfusion. Death within 2 weeks: 7.3% in the urokinase group vs. 8.9% in the heparin group. Recurrent VTE within 2 weeks: 15.3% in the urokinase group vs. 18.3% in the heparin group. Severe bleeding within 2 weeks: 45.0% in the urokinase group vs. 27.0% in the heparin group [41]. |
| Ly (1978) [38] | 25 | Acute PE < 5 days | >1 lobe | Streptokinase 72 h | 100.0% | Significant improvement in the mean pulmonary angiographic score with streptokinase 10.3 ± 5.1 vs. 3.7 ± 7.2 with heparin. Clinical improvement: 80.0% in the streptokinase group vs. 33.3% in the heparin group. Death: 10.0% the streptokinase group vs. 33.3% in the heparin group Major bleeding: 40.0% in the streptokinase group vs. 33.3% in the heparin group [38]. |
| Dotter (1979) [40] | 31 | Acute PE | >1 lobe | Streptokinase 2–11 MIU 18–72 h | 6.5% | The mean angiographic score-improvement rating was 2.08 in streptokinase-treated patients and 0.86 in heparin-treated patients Mean PA pressure changes in both groups were similar (streptokinase −7.9 mmHg, heparin −6.2 mmHg) [40]. |
| Jerjes-Sanchez (1995) [39] | 8 | Acute PE < 14 days | Massive | Streptokinase 1.5 MIU/2 h | 100.0% | The mortality in the streptokinase group was 0% compared with 100% (p = 0.02) in the heparin group [39]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chopard, R.; Behr, J.; Vidoni, C.; Ecarnot, F.; Meneveau, N. An Update on the Management of Acute High-Risk Pulmonary Embolism. J. Clin. Med. 2022, 11, 4807. https://doi.org/10.3390/jcm11164807
Chopard R, Behr J, Vidoni C, Ecarnot F, Meneveau N. An Update on the Management of Acute High-Risk Pulmonary Embolism. Journal of Clinical Medicine. 2022; 11(16):4807. https://doi.org/10.3390/jcm11164807
Chicago/Turabian StyleChopard, Romain, Julien Behr, Charles Vidoni, Fiona Ecarnot, and Nicolas Meneveau. 2022. "An Update on the Management of Acute High-Risk Pulmonary Embolism" Journal of Clinical Medicine 11, no. 16: 4807. https://doi.org/10.3390/jcm11164807
APA StyleChopard, R., Behr, J., Vidoni, C., Ecarnot, F., & Meneveau, N. (2022). An Update on the Management of Acute High-Risk Pulmonary Embolism. Journal of Clinical Medicine, 11(16), 4807. https://doi.org/10.3390/jcm11164807






