Mesenchymal Stem Cells Profile in Adult Atopic Dermatitis and Effect of IL4-IL13 Inflammatory Pathway Inhibition In Vivo: Prospective Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design of the Study
2.2. Patients’ Population
2.3. Skin Samples
2.4. Isolation, Cell Culture and Characterization of MSCs
2.5. IL4 and IL13 Expression by ICC and IIF
2.6. ELISA Test for Evaluation of Cytokines Levels in Supernatant
2.7. Statistical Analysis
3. Results
3.1. Clinical Features
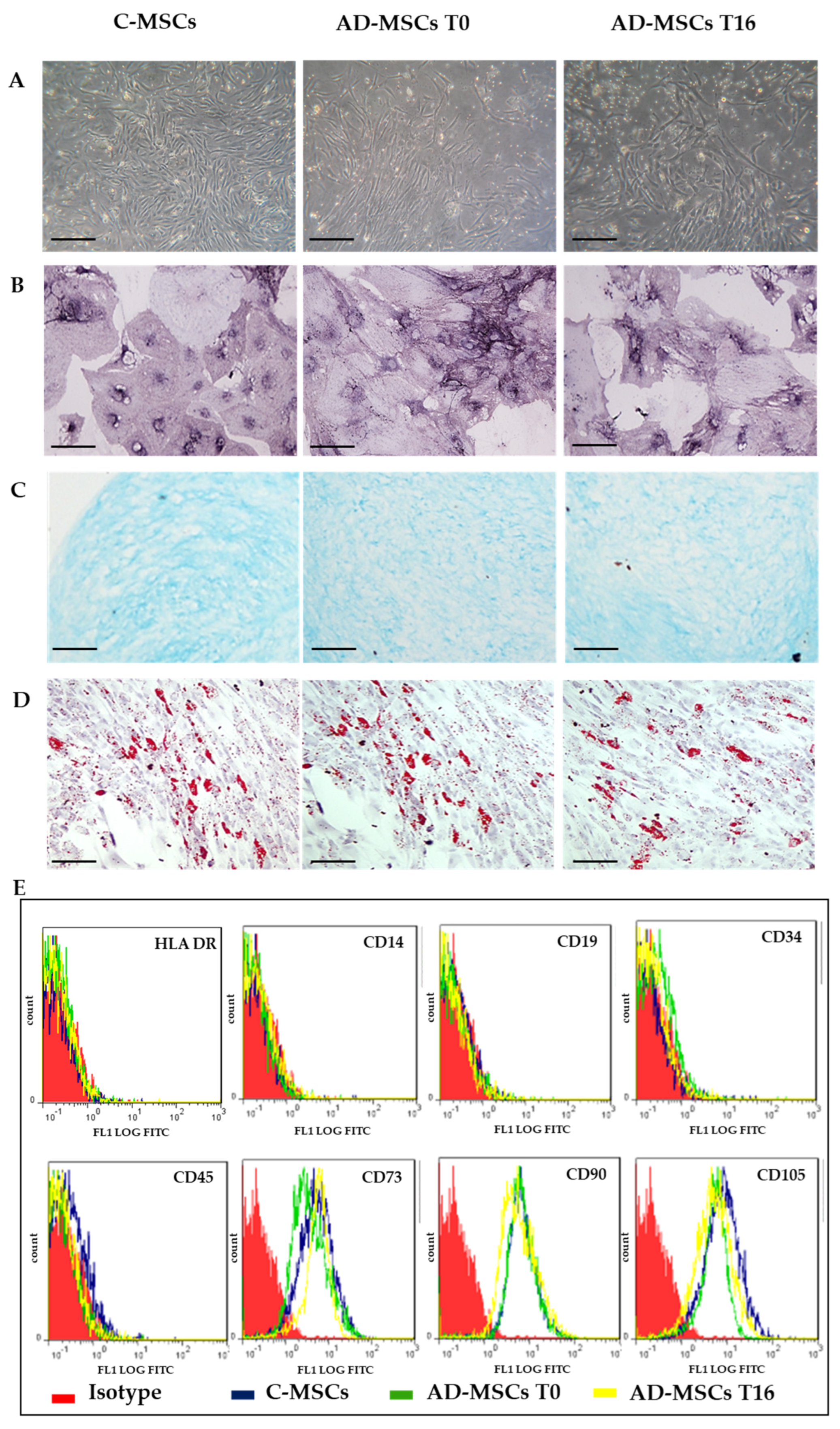
3.2. Isolation, Culture and Characterization of MSCs
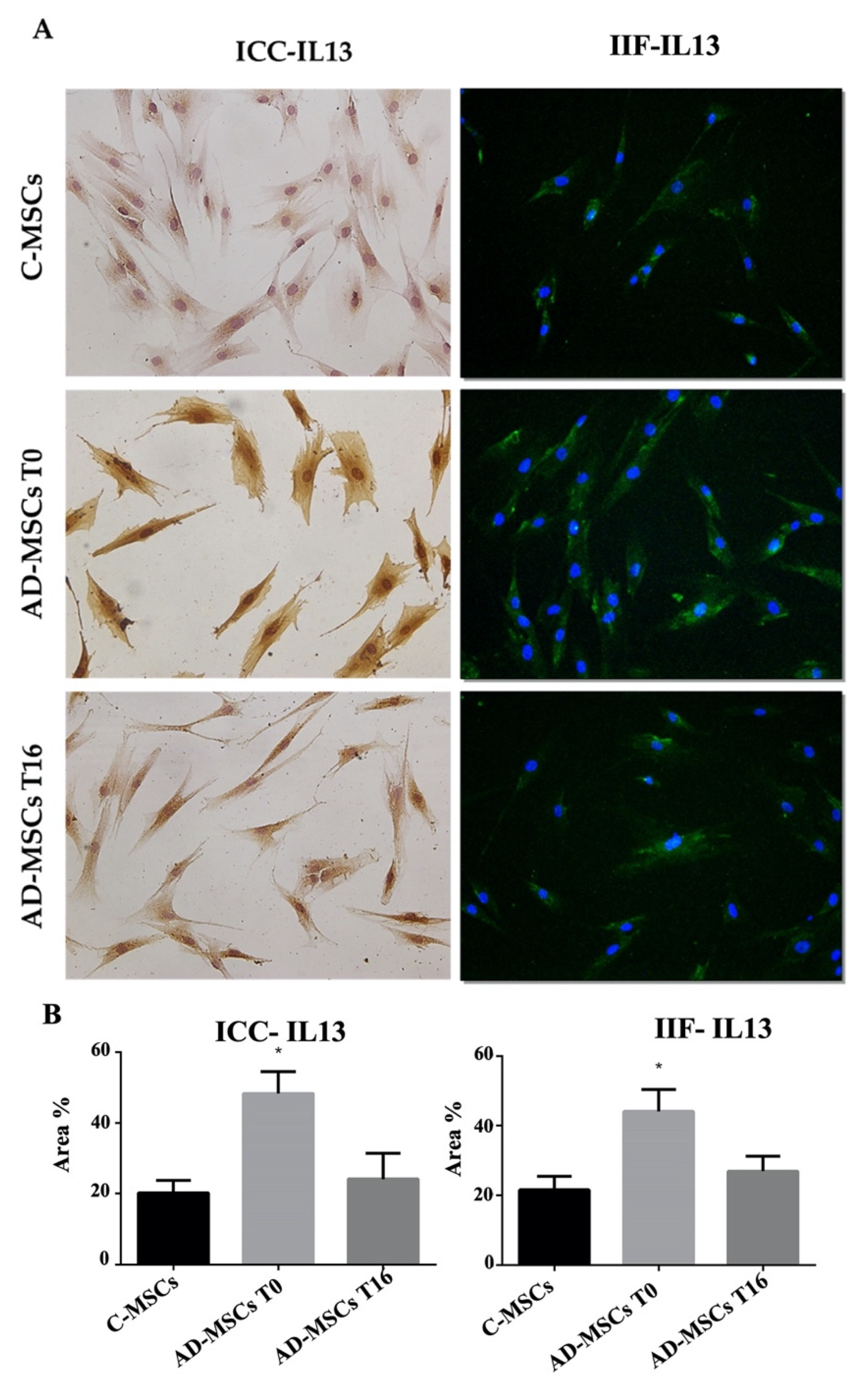
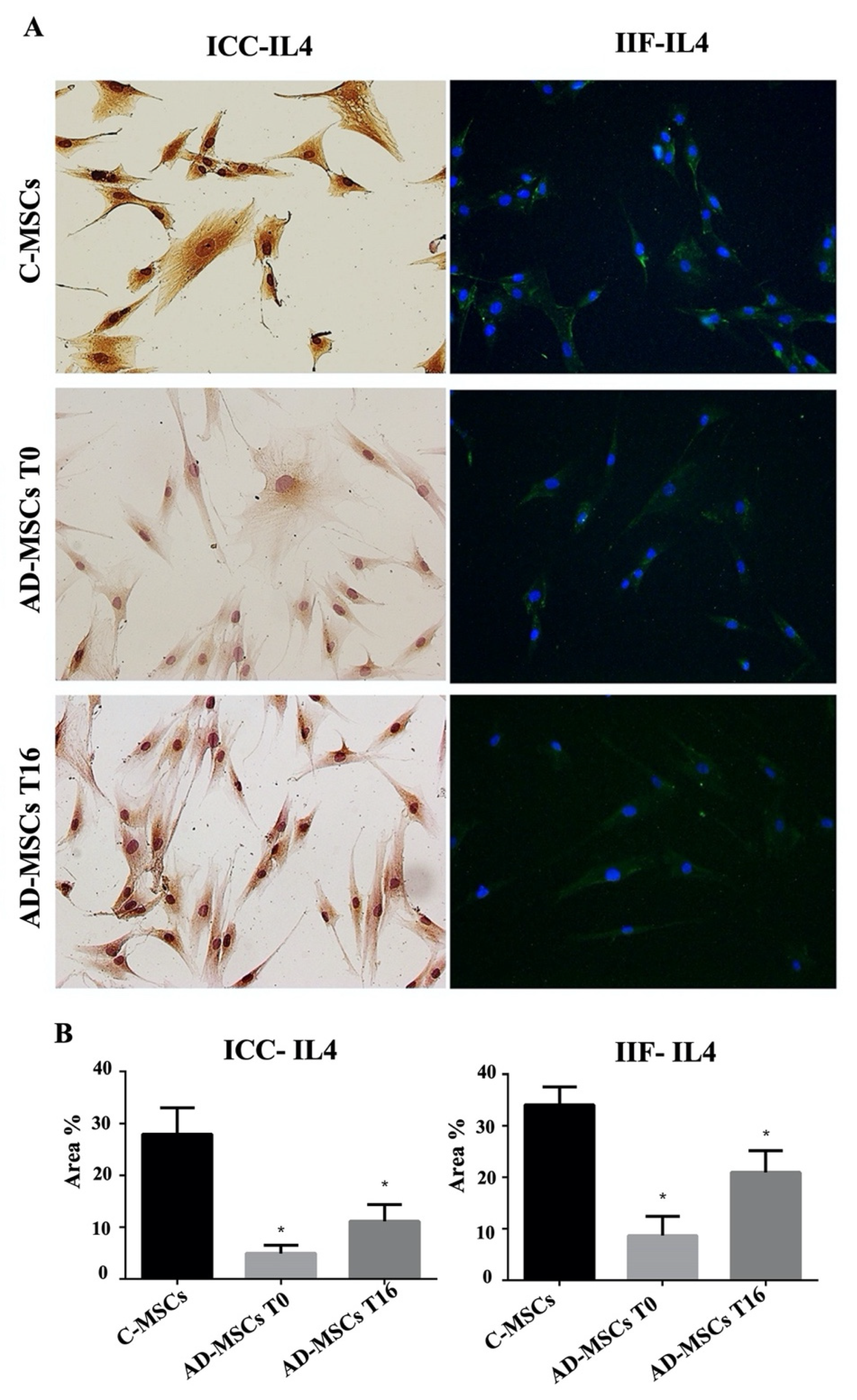
3.3. IL4 and IL13 Expression by ICC and IF in C-MSCs and AD-MSCs at T0 and T16
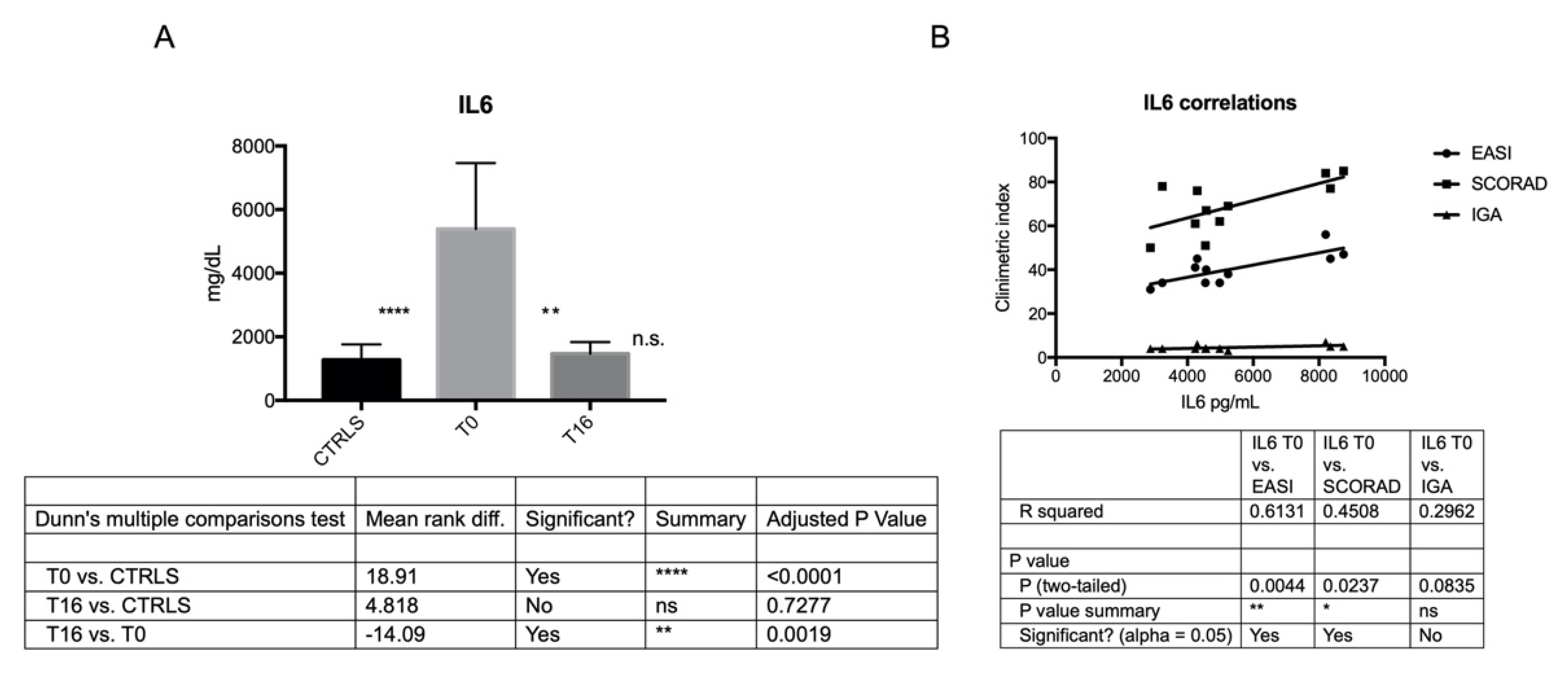
3.4. Expression Profiles of Th1, Th2, Th17 Cytokines by ELISA in C-MSCs and AD-MSCs at T0 and T16
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fölster-Holst, R. Management of atopic dermatitis: Are there differences between children and adults? J. Eur. Acad. Dermatol. Venereol. 2014, 28, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Mortz, C.G.; Andersen, K.E.; Dellgren, C.D.; Barington, T.; Bindslev-Jensen, C. Atopic dermatitis from adolescence to adulthood in the TOACS cohort: Prevalence, persistence and comorbidities. Allergy 2015, 70, 836–845. [Google Scholar] [CrossRef]
- Bannister, M.J.; Freeman, S. Adult-onset atopic dermatitis. Australas. J. Dermatol. 2000, 41, 225–228. [Google Scholar] [CrossRef]
- Shaw, T.E.; Currie, G.P.; Koudelka, C.W.; Simpson, E.L. Eczema prevalence in the United States: Data from the 2003 national Survey of Children’s Health. J. Investig. Dermatol. 2011, 131, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Barbarot, S.; Auziere, S.; Gadkari, A.; Girolomoni, G.; Puig, L.; Simpson, E.L.; Margolis, D.J.; De Bruin-Weller, M.; Eckert, L. Epidemiology of atopic dermatitis in adults: Results from an international survey. Allergy 2018, 73, 1284–1293. [Google Scholar] [CrossRef]
- De Benedetto, A.; Kubo, A.; Beck, L.A. Skin barrier disruption: A requirement for allergen sensitization? J. Investig. Dermatol. 2012, 132, 949–963. [Google Scholar] [CrossRef] [PubMed]
- Visscher, M.O.; Adam, R.; Brink, S.; Odio, M. Newborn infant skin: Physiology, development, and care. Clin. Dermatol. 2015, 33, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, J.M. Evolving concepts of pathogenesis in atopic dermatitis and other eczemas. J. Investig. Dermatol. 2009, 129, 320–322. [Google Scholar] [CrossRef]
- Kondo, H.; Ichikawa, Y.; Imokawa, G. Percutaneous sensiti- zation with allergens through barrier-disrupted skin elicits a Th2-dominant cytokine response. Eur. J. Immunol. 1998, 28, 769–779. [Google Scholar] [CrossRef]
- Correa da Rosa, J.; Malajian, D.; Shemer, A.; Rozenblit, M.; Dhingra, N.; Czarnowicki, T.; Khattri, S.; Ungar, B.; Finney, R.; Xu, H.; et al. Patients with atopic dermatitis have attenuated and distinct con- tact hypersensitivity responses to common allergens in skin. J. Allergy Clin. Immunol. 2015, 135, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Paller, A.S. Latest approaches to treating atopic dermatitis. Chem. Immunol. Allergy 2012, 96, 132–140. [Google Scholar] [PubMed]
- Batista, D.I.S.; Perez, L.; Orfali, R.L.; Zaniboni, M.C.; Samorano, L.P.; Pereira, N.V.; Sotto, M.N.; Ishizaki, A.S.; Oliveira, L.M.S.; Sato, M.N.; et al. Profile of skin barrier proteins (filaggrin, claudins 1 and 4) and Th1/Th2/Th17 cytokines in adults with atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Orciani, M.; Campanati, A.; Salvolini, E.; Lucarini, G.; Di Benedetto, G.; Offidani, A.; Di Primio, R. The mesenchymal stem cell profile in psoriasis. Br. J. Dermatol. 2011, 165, 585–592. [Google Scholar] [CrossRef]
- Orciani, M.; Campanati, A.; Caffarini, M.; Ganzetti, G.; Consales, V.; Lucarini, G.; Offidani, A.; Di Primio, R. T helper (Th)1, Th17 and Th2 imbalance in mesenchymal stem cells of adult patients with atopic dermatitis: At the origin of the problem. Br. J. Dermatol. 2017, 176, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Yang, T.; Wu, Z.; Zhong, J.; Huang, Y.; Huang, T.; Zheng, E. Differentiation of T-helper cells in distinct phases of atopic dermatitis involves Th1/Th2 and Th17/Treg. Eur. J. Inflamm. 2017, 15, 46–52. [Google Scholar] [CrossRef]
- Patruno, C.; Amerio, P.; Chiricozzi, A.; Costanzo, A.; Cristaudo, A.; Cusano, F.; Foti, C.; Girolomoni, G.; Guarneri, F.; Naldi, L.; et al. Optimizing a clinical guidance for diagnosis of atopic dermatitis in adults: Joint recommendations of the Italian Society of Dermatology and Venereology (SIDeMaST), Italian Association of Hospital Dermatologists (ADOI), and Italian Society of Allergological, Occupational and Environmental Dermatology (SIDAPA). G. Ital. Dermatol. Venereol. 2020, 155, 1–7. [Google Scholar]
- Hanifin, J.M.; Thurston, M.; Omoto, M.; Cherill, R.; Tofte, S.J.; Graeber, M. The eczema area and severity index (EASI): Assessment of reliability in atopic dermatitis. Easi Evaluator Group. Exp. Dermatol. 2001, 10, 11–18. [Google Scholar] [CrossRef]
- Kunz, B.; Oranje, A.; Labrèze, L.; Stalder, J.-F.; Ring, J.; Taïeb, A. Clinical validation and guidelines for the SCORAD index: Consensus report of the European Task Force on Atopic Dermatitis. Dermatology 1997, 195, 10–19. [Google Scholar] [CrossRef]
- Rehal, B.; Armstrong, A.W. Health outcome measures in atopic dermatitis: A systematic review of trends in disease severity and quality-of-life instruments 1985–2010. PLoS ONE 2011, 6, e17520. [Google Scholar] [CrossRef]
- Campanati, A.; Orciani, M.; Consales, V.; Lazzarini, R.; Ganzetti, G.; di Benedetto, G.; di Primio, R.; Offidani, A. Characterization and profiling of immunomodulatory genes in resident mesenchymal stem cells reflect the Th1-Th17/Th2 imbalance of psoriasis. Arch. Dermatol. Res. 2014, 306, 915–920. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Rueden, C.T.; Schindelin, J.; Hiner, M.C.; Dezonia, B.E.; Walter, A.E.; Arena, E.T.; Eliceiri, K.W. ImageJ2: ImageJ for the next generation of scientific image data. BMC Bioinform. 2017, 18, 529. [Google Scholar] [CrossRef] [PubMed]
- Campanati, A.; Orciani, M.; Lazzarini, R.; Ganzetti, G.; Consales, V.; Sorgentoni, G.; Di Primio, R.; Offidani, A. TNF-α inhibitors reduce the pathological Th1–Th17/Th2 imbalance in cutaneous mesenchymal stem cells of psoriasis patients. Exp. Dermatol. 2017, 26, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, J.D.; Suárez-Fariñas, M.; Dhingra, N.; Cardinale, I.; Li, X.; Kostic, A.; Ming, J.E.; Radin, A.R.; Krueger, J.G.; Graham, N.; et al. Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J. Allergy Clin. Immunol. 2014, 134, 1293–1300. [Google Scholar] [CrossRef]
- Ariens, L.F.; Bakker, D.S.; Van Der Schaft, J.; Garritsen, F.M.; Thijs, J.L.; De Bruin-Weller, M.S. Dupilumab in atopic dermatitis: Rationale, latest evidence and place in therapy. Ther. Adv. Chronic Dis. 2018, 9, 159–170. [Google Scholar] [CrossRef]
- Klonowska, J.; Gleń, J.; Nowicki, R.J.; Trzeciak, M. New cytokines in the pathogenesis of atopic dermatitis—New therapeutic targets. Int. J. Mol. Sci. 2018, 19, 3086. [Google Scholar] [CrossRef]
- Gittler, J.K.; Shemer, A.; Suárez-Fariñas, M.; Fuentes-Duculan, J.; Gulewicz, K.J.; Wang, C.Q.F.; Mitsui, H.; Cardinale, I.; de Guzman Strong, C.; Krueger, J.G.; et al. Progressive activation of T(H)2/T(H)22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis. J. Allergy Clin. Immunol. 2012, 130, 1344–1354. [Google Scholar] [CrossRef]
- Chaudhary, S.K.; Singh, S.K.; Kumari, P.; Kanwal, S.; Soman, S.P.; Choudhury, S.; Garg, S.K. Alterations in circulating concentrations of IL-17, IL-31 and total IgE in dogs with atopic dermatitits. Vet. Dermatol. 2019, 30, 383-e114. [Google Scholar] [CrossRef]
- Nutten, S. Atopic demartitis: Global epidemiology and risk factors. Ann. Nutr. Metab. 2015, 66 (Suppl. 1), 8–18. [Google Scholar] [CrossRef]
- Miossec, P.; Kolls, J.K. Targeting IL-17 and TH17 cells in chronic Inflammation. Nat. Rev. Drug Discov. 2012, 11, 763–776. [Google Scholar] [CrossRef]
- Sidburry, R.; Khorsand, K. Evolving concepts in atopic dermatitis. Curr. Allergy Asthma Rep. 2017, 17, 42. [Google Scholar] [CrossRef] [PubMed]
- Campanati, A.; Orciani, M.; Sorgentoni, G.; Consales, V.; Offidani, A.; Di Primio, R. Pathogenetic Characteristics of Mesenchymal Stem Cells in Hidradenitis Suppurativa. JAMA Dermatol. 2018, 154, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Campanati, A.; Consales, V.; Orciani, M.; Giuliodori, K.; Ganzetti, G.; Bobyr, I.; Sorgentoni, G.; Di Primio, R.; Offidani, A. Role of mesenchymal stem cells in the pathogenesis of psoriasis: Current perspectives. Psoriasis 2017, 7, 73–85. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Campanati, A.; Orciani, M.; Sorgentoni, G.; Consales, V.; Mattioli Belmonte, M.; Di Primio, R.; Offidani, A. Indirect co-cultures of healthy mesenchymal stem cells restore the physiological phenotypical profile of psoriatic mesenchymal stem cells. Clin. Exp. Immunol. 2018, 193, 234–240. [Google Scholar] [CrossRef]
- Campanati, A.; Orciani, M.; Ganzetti, G.; Consales, V.; Di Primio, R.; Offidani, A. The effect of etanercept on vascular endothelial growth factor production by cutaneous mesenchymal stem cells from patients with psoriasis. J. Int. Med. Res. 2016, 44 (Suppl. 1), 6–9. [Google Scholar] [CrossRef]
- Campanati, A.; Orciani, M.; Gorbi, S.; Regoli, F.; Di Primio, R.; Offidani, A. Effect of biologic therapies targeting tumour necrosis factor-α on cutaneous mesenchymal stem cells in psoriasis. Br. J. Dermatol. 2012, 167, 68–76. [Google Scholar] [CrossRef]
- Campanati, A.; Caffarini, M.; Diotallevi, F.; Radi, G.; Lucarini, G.; Di Vincenzo, M.; Orciani, M.; Offidani, A. The efficacy of in vivo administration of Apremilast on mesenchymal stem cells derived from psoriatic patients. Inflamm. Res. 2020, 70, 79–87. [Google Scholar] [CrossRef]
- Campione, E.; Lanna, C.; Diluvio, L.; Cannizzaro, M.V.; Grelli, S.; Galluzzo, M.; Talamonti, M.; Annicchiarico-Petruzzelli, M.; Mancini, M.; Melino, G.; et al. Skin immunity and its dysregulation in vitiligo, atopic dermatitis, hidradenitis supurativa and vitiligo. Cell Cycle 2020, 19, 257–267. [Google Scholar] [CrossRef]
- Gharagozlou, M.; Farhadi, E.; Khaledi, M.; Behniafard, N.; Sotoudeh, S.; Salari, R.; Darabi, B.; Fathi, S.M.; Mahmoudi, M.; Aghamohammadi, A.; et al. Association between the interleukin 6 genotype at position-174 and atopic dermatitis. J. Investig. Allergol. Clin. Immunol. 2013, 23, 89–93. [Google Scholar]
- Raap, U.; Weißmantel, S.; Gehring, M.; Eisenberg, A.M.; Kapp, A.; Fölster-Holst, R. IL-31 significantly correlates with disease activity and Th2 cytokine levels in children with atopic dermatitis. Pediatr. Allergy Immunol. 2012, 23, 285–288. [Google Scholar] [CrossRef]
- Ungar, B.; Garcet, S.; Gonzalez, J.; Dhingra, N.; Da Rosa, J.C.; Shemer, A.; Krueger, J.G.; Suarez-Farinas, M.; Guttman-Yassky, E. An integrated model of atopic dermatitis biomarkers highlights the systemic nature of the disease. J. Investig. Dermatol. 2017, 137, 603–613. [Google Scholar] [CrossRef] [PubMed]
- Szegedi, K.; Lutter, R.; Bos, J.D.; Luiten, R.M.; Kezic, S.; Middelkamp-Hup, M.A. Cytokine profiles in intersti- tial fluid from chronic atopic dermatitis skin. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 2136–2144. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T. Interleukin-13: Targeting an underestimated cytokine in atopic dermatitis. Allergy 2020, 75, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Tazawa, T.; Sugiura, H.; Sugiura, Y.; Uehara, M. Relative importance of IL-4 and IL-13 in lesional skin of atopic dermatitis. Arch. Dermatol. Res. 2004, 295, 459–464. [Google Scholar] [CrossRef]
- Tsoi, L.C.; Rodriguez, E.; Degenhardt, F.; Baurecht, H.; Wehkamp, U.; Volks, N.; Szymczak, S.; Swindell, W.R.; Sarkar, M.K.; Raja, K.; et al. Atopic dermatitis is an IL-13–dominant disease with greater molecular heterogeneity compared to psoriasis. J. Investig. Dermatol. 2019, 139, 1480–1489. [Google Scholar] [CrossRef]
- Koppes, S.A.; Brans, R.; Ljubojevic Hadzavdic, S.; Frings-Dresen, M.H.; Rustemeyer, T.; Kezic, S. Stratum Corneum Tape Stripping: Monitoring of Inflammatory Mediators in Atopic Dermatitis Patients Using Topical Therapy. Int. Arch. Allergy Immunol. 2016, 170, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Bao, K.; Reinhardt, R.L. The differential expression of IL-4 and IL-13 and its impact on type-2 immunity. Cytokine 2015, 75, 25–37. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Olesen, C.M.; Pavel, A.B.; Clausen, M.-L.; Wu, J.; Estrada, Y.; Zhang, N.; Agner, T.; Guttman-Yassky, E. Tape-Strip Proteomic Profiling of Atopic Dermatitis on Dupilumab Identifies Minimally Invasive Biomarkers. Front. Immunol. 2020, 11, 1768. [Google Scholar] [CrossRef] [PubMed]
- Toshitani, A.; Ansel, J.C.; Chan, S.C.; Li, S.-H.; Hanifin, J.M. Increased interleukin 6 production by T cells derived from patients with atopic dermatitis. J. Investig. Dermatol. 1993, 100, 299–304. [Google Scholar] [CrossRef]
- Diehl, S.; Rincon, M. The two faces of IL-6 on Th1/Th2 differentiation. Mol. Immunol. 2002, 39, 531–536. [Google Scholar] [CrossRef]
- Howell, M.D.; Parker, M.L.; Mustelin, T.; Ranade, K. Past, present, and future for biologic intervention in atopic dermatitis. Allergy 2015, 70, 887–896. [Google Scholar] [CrossRef]
- Saeki, H.; Kabashima, K.; Tokura, Y.; Murata, Y.; Shiraishi, A.; Tamamura, R.; Randazzo, B.; Imanaka, K. Efficacy and safety of Ustekinumab in Japanese patients with severe atopic dermatitis: A randomised, double-blind, placebo-controlled, phase 2 study. Br. J. Dermatol. 2017, 177, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Sobolev, V.V.; Denisova, E.V.; Chebysheva, S.N.; Geppe, N.A.; Korsunskaya, I.M. IL-6 Gene Expression as a Marker of Pathological State in Psoriasis and Psoriatic Arthritis. Bull. Exp. Biol. Med. 2022, 173, 77–80. [Google Scholar] [CrossRef]
- Navarini, A.; French, L.; Hofbauer, G. Interrupting IL-6–receptor signaling improves atopic dermatitis but associates with bacterial superinfection. J. Allergy Clin. Immunol. 2011, 128, 1128–1130. [Google Scholar] [CrossRef] [PubMed]
- Chatrath, S.; Lei, D.; Yousaf, M.; Chavda, R.; Gabriel, S.; Silverberg, J.I. Longitudinal course and predictors of depressive symptoms in atopic dermatitis. J. Am. Acad. Dermatol. 2022; in press. [Google Scholar] [CrossRef]
- Bardazzi, F.; Magnano, M.; Campanati, A.; Loconsole, F.; Carpentieri, A.; Potenza, C.; Bernardini, N.; Lernia, V.; Carrera, C.; Raone, B.; et al. Biologic Therapies in HIV-infected Patients with Psoriasis: An Italian Experience. Acta Derm. Venereol. 2017, 97, 989–990. [Google Scholar] [CrossRef]
- Marek-Józefowicz, L.; Jaracz, M.; Borkowska, A. Affective temperament, depressive symptoms and interleukins in patients with psoriasis. Postepy Dermatol. Alergol. 2021, 38, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Daltro, S.R.T.; Meira, C.S.; Santos, I.P.; Dos Santos, R.R.; Soares, M.B.P. Mesenchymal Stem Cells and Atopic Dermatitis: A Review. Front. Cell Dev. Biol. 2020, 8, 326. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Lee, J.H.; Roh, K.-H.; Jun, H.J.; Kang, K.-S.; Kim, T.-Y. Clinical trial of human umbilical cord blood-derived stem cells for the treatment of moderate-to-severe atopic dermatitis: Phase I/IIa studies. Stem Cells 2017, 35, 248–255. [Google Scholar] [CrossRef] [PubMed]
| Patients (n = 11) | EASI T0 | SCORAD T0 | PGA T0 | EASI T16 | SCORAD T16 | PGA T16 |
|---|---|---|---|---|---|---|
| 1 | 45 | 76 | 6 | 11 | 20 | 2 |
| 2 | 56 | 84 | 6 | 13 | 22 | 2 |
| 3 | 34 | 62 | 4 | 8 | 15 | 2 |
| 4 | 33 | 51 | 4 | 7 | 13 | 2 |
| 5 | 45 | 77 | 5 | 11 | 20 | 2 |
| 6 | 40 | 67 | 4 | 10 | 17 | 2 |
| 7 | 41 | 61 | 4 | 10 | 16 | 2 |
| 8 | 47 | 85 | 5 | 11 | 21 | 2 |
| 9 | 31 | 50 | 4 | 7 | 12 | 2 |
| 10 | 34 | 78 | 4 | 9 | 23 | 3 |
| 11 | 38 | 69 | 3 | 11 | 26 | 2 |
| EASI T0 Mean Value ± SD | EASI T16 Mean Value ± SD | p < 0.0001 |
| 41.79 ± 2.38 | 9.81 ± 1.8 | |
| SCORAD T0 Mean Value ± SD | SCORAD T16 Mean Value ± SD | p < 0.0001 |
| 69.09 ± 11.15 | 18.64 ± 1.32 | |
| IGA T0 Mean Value ± SD | IGA T16 Mean Value ± SD | p < 0.0001 |
| 4.45 ± 0.93 | 2.09 ± 0.30 |
| IL | MSCs-AD T0 Mean Value ± SD | MSCs-AD T16 Mean Value ± SD | C-MSCs Mean Value ± SD | p |
|---|---|---|---|---|
| IL2 | 48.98 ± 6.73 | 65.75 ± 13.83 | 35.56 ± 8.54 | T0 vs. Controls p = 0.0523 |
| T0 vs. T16 p = 0.1299 | ||||
| IL4 | 7.14 ± 0.89 | 9.53 ± 1.31 | 20.71 ± 1.92 | T0 vs. Controls p < 0.0001 |
| T0 vs. T16 p = 0.0777 | ||||
| IL5 | 4.40 ± 0.81 | 3.65 ± 0.55 | 3.64 ± 0.38 | T0 vs. Controls p = 0.1088 |
| T0 vs. T16 p = 0.1213 | ||||
| IL6 | 5387.03 ± 2073 | 1469.01 ± 364.9 | 1277.02 ± 483.1 | T0 vs. Controls p < 0.0001 |
| T0 vs. T16 p = 0.0019 | ||||
| IL10 | 29.80 ± 5.82 | 31.55 ± 6.45 | 34.66 ± 6.11 | T0 vs. Controls p = 0.1399 |
| T0 vs. T16 p > 0.9999 | ||||
| IL12 | 3.32 ± 3.12 | 6.17 ± 3.48 | 26.38 ± 3.48 | T0 vs. Controls p = 0.1399 |
| T0 vs. T16 p > 0.9999 | ||||
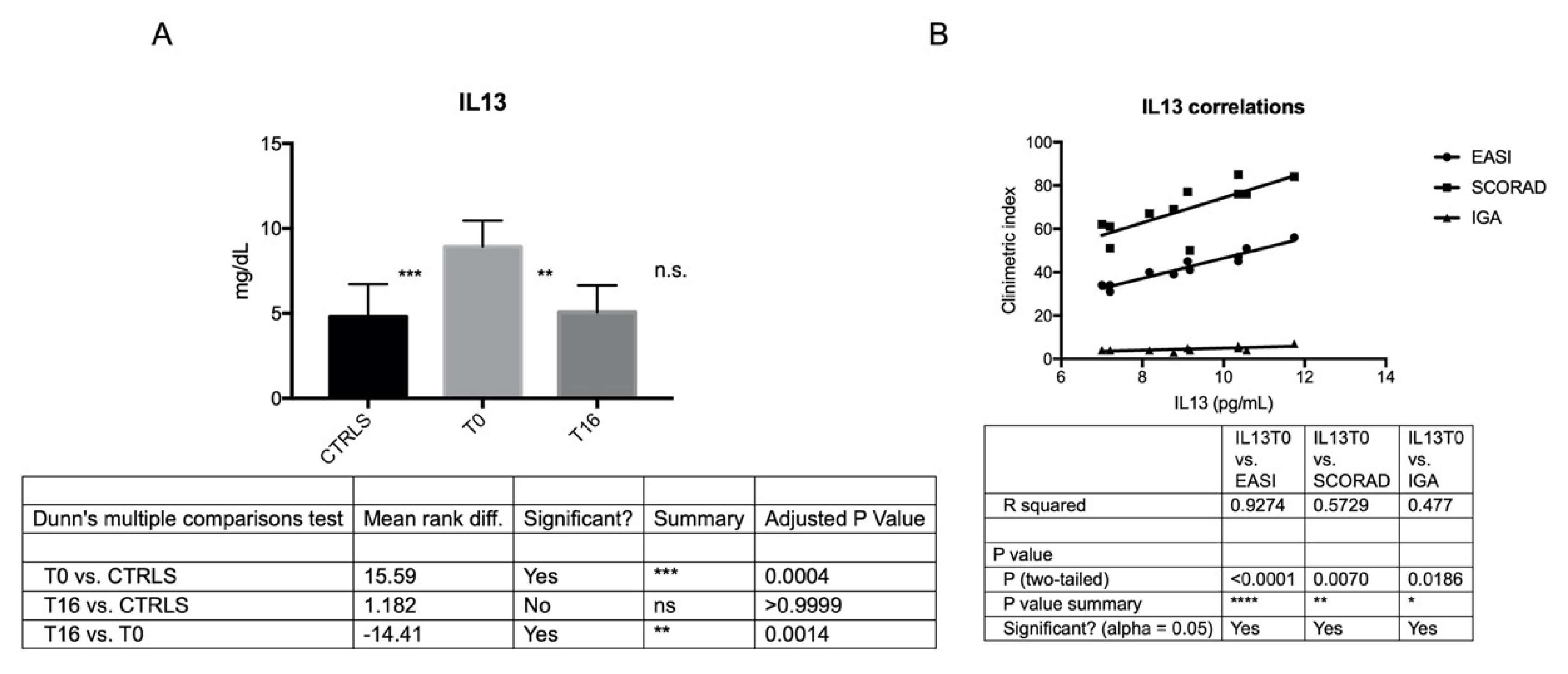
| IL13 | 8.937 ± 1.51 | 5.0 ± 1.5 | 4.807 ± 1.91 | T0 vs. Controls p = 0.0004 |
| T0 vs. T16 p = 0.0014 | ||||
| IL17A | 5.90 ± 0.86 | 6.13 ± 1.19 | 5.31 ± 0.89 | T0 vs. Controls p = 0.3628 |
| T0 vs. T16 p > 0.9999 | ||||
| IFNγ | 6.19 ± 0.92 | 5.13 ± 0.88 | 4.81 ± 1.44 | T0 vs. Controls p = 0.1170 |
| T0 vs. T16 p = 0.0808 | ||||
| TNFα | 81.46 ± 19.48 | 66.60 ± 6.55 | 62.89 ± 11.52 | T0 vs. Controls p = 0.0639 |
| T0 vs. T16 p = 0.4878 | ||||
| G-CSF | 94.07 ± 80.53 | 68.53 ± 38.44 | 30.93 ± 3.25 | T0 vs. Controls p = 0.6532 |
| T0 vs. T16 p > 0.9999 | ||||
| TGFβ | 843.09 ± 170.40 | 641.10 ± 155.70 | 759.5 ± 302.70 | T0 vs. Controls p = 0.0807 |
| T0 vs. T16 p > 0.9999 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campanati, A.; Orciani, M.; Marani, A.; Di Vincenzo, M.; Magi, S.; Gregoriou, S.; Diotallevi, F.; Martina, E.; Radi, G.; Offidani, A. Mesenchymal Stem Cells Profile in Adult Atopic Dermatitis and Effect of IL4-IL13 Inflammatory Pathway Inhibition In Vivo: Prospective Case-Control Study. J. Clin. Med. 2022, 11, 4759. https://doi.org/10.3390/jcm11164759
Campanati A, Orciani M, Marani A, Di Vincenzo M, Magi S, Gregoriou S, Diotallevi F, Martina E, Radi G, Offidani A. Mesenchymal Stem Cells Profile in Adult Atopic Dermatitis and Effect of IL4-IL13 Inflammatory Pathway Inhibition In Vivo: Prospective Case-Control Study. Journal of Clinical Medicine. 2022; 11(16):4759. https://doi.org/10.3390/jcm11164759
Chicago/Turabian StyleCampanati, Anna, Monia Orciani, Andrea Marani, Mariangela Di Vincenzo, Simona Magi, Stamatios Gregoriou, Federico Diotallevi, Emanuela Martina, Giulia Radi, and Annamaria Offidani. 2022. "Mesenchymal Stem Cells Profile in Adult Atopic Dermatitis and Effect of IL4-IL13 Inflammatory Pathway Inhibition In Vivo: Prospective Case-Control Study" Journal of Clinical Medicine 11, no. 16: 4759. https://doi.org/10.3390/jcm11164759
APA StyleCampanati, A., Orciani, M., Marani, A., Di Vincenzo, M., Magi, S., Gregoriou, S., Diotallevi, F., Martina, E., Radi, G., & Offidani, A. (2022). Mesenchymal Stem Cells Profile in Adult Atopic Dermatitis and Effect of IL4-IL13 Inflammatory Pathway Inhibition In Vivo: Prospective Case-Control Study. Journal of Clinical Medicine, 11(16), 4759. https://doi.org/10.3390/jcm11164759








