Lower Levels of ABO Anti-A and Anti-B of IgM, IgG and IgA Isotypes in the Serum but Not the Saliva of COVID-19 Convalescents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Convalescent COVID-19 Study Participants
2.2. Control Groups
2.3. Preparation of Saliva Samples
2.4. ABO Antibody Analysis
2.5. Statistical Analysis
3. Results
3.1. Study Participants
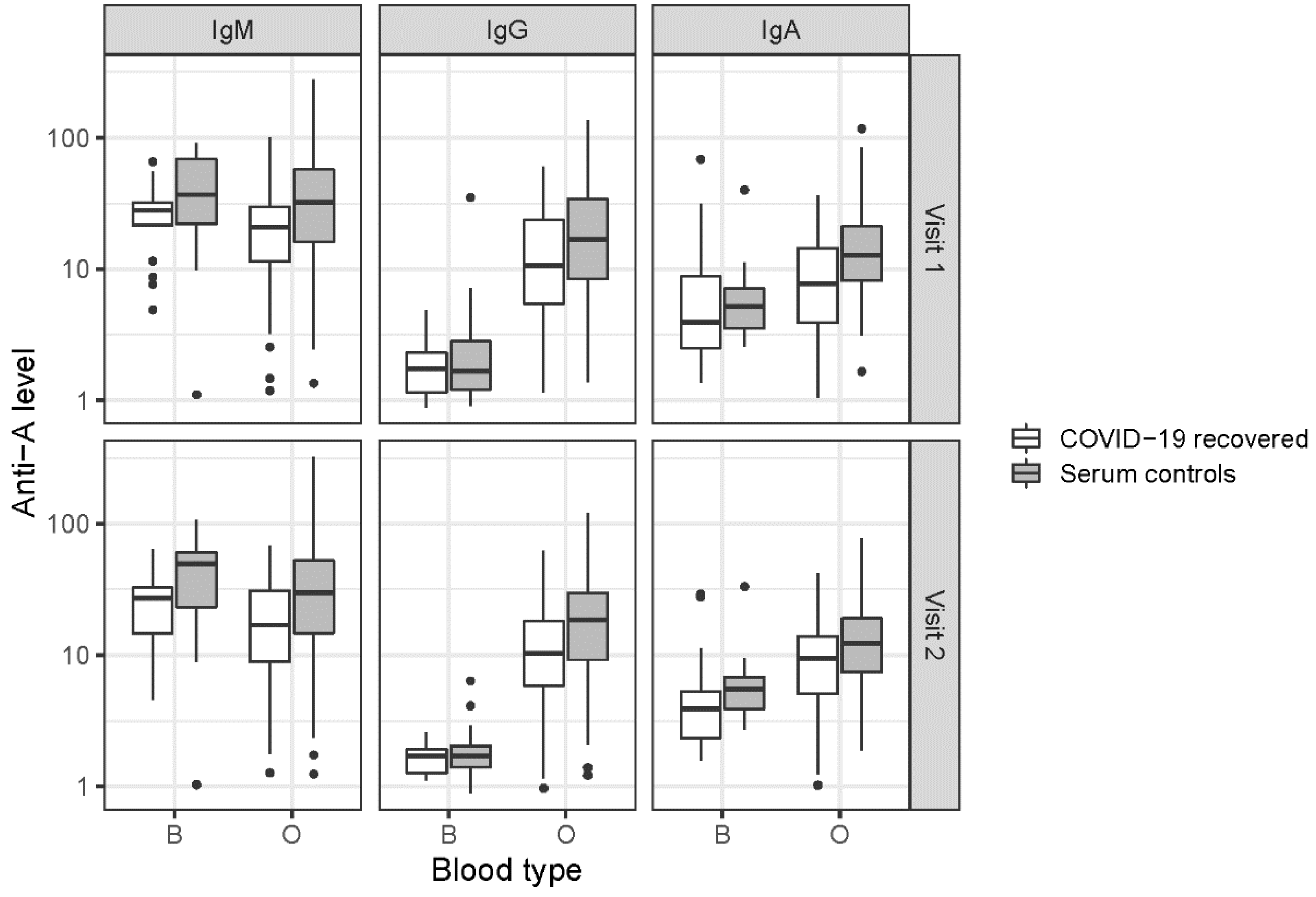
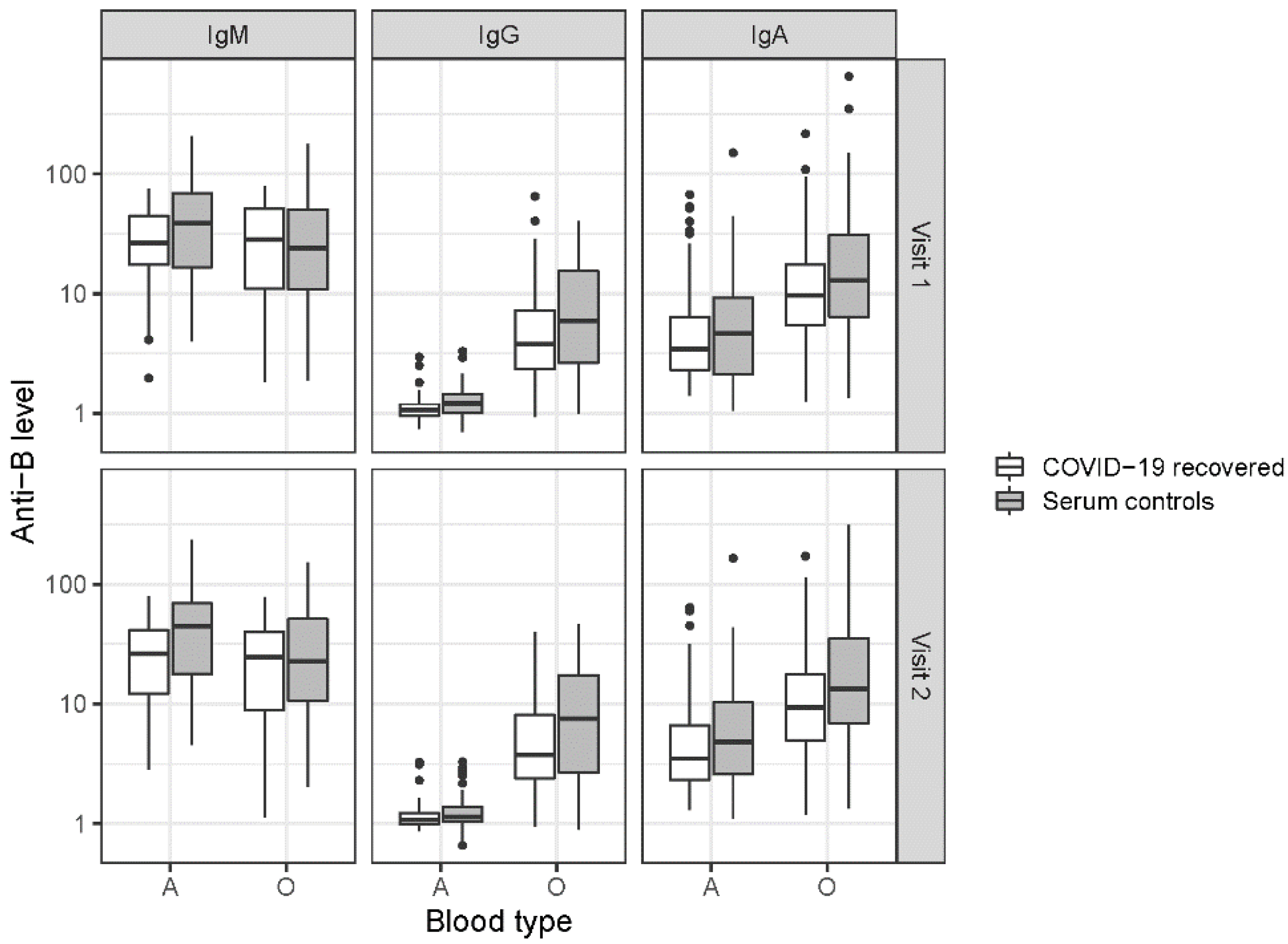
3.2. ABO Antibodies
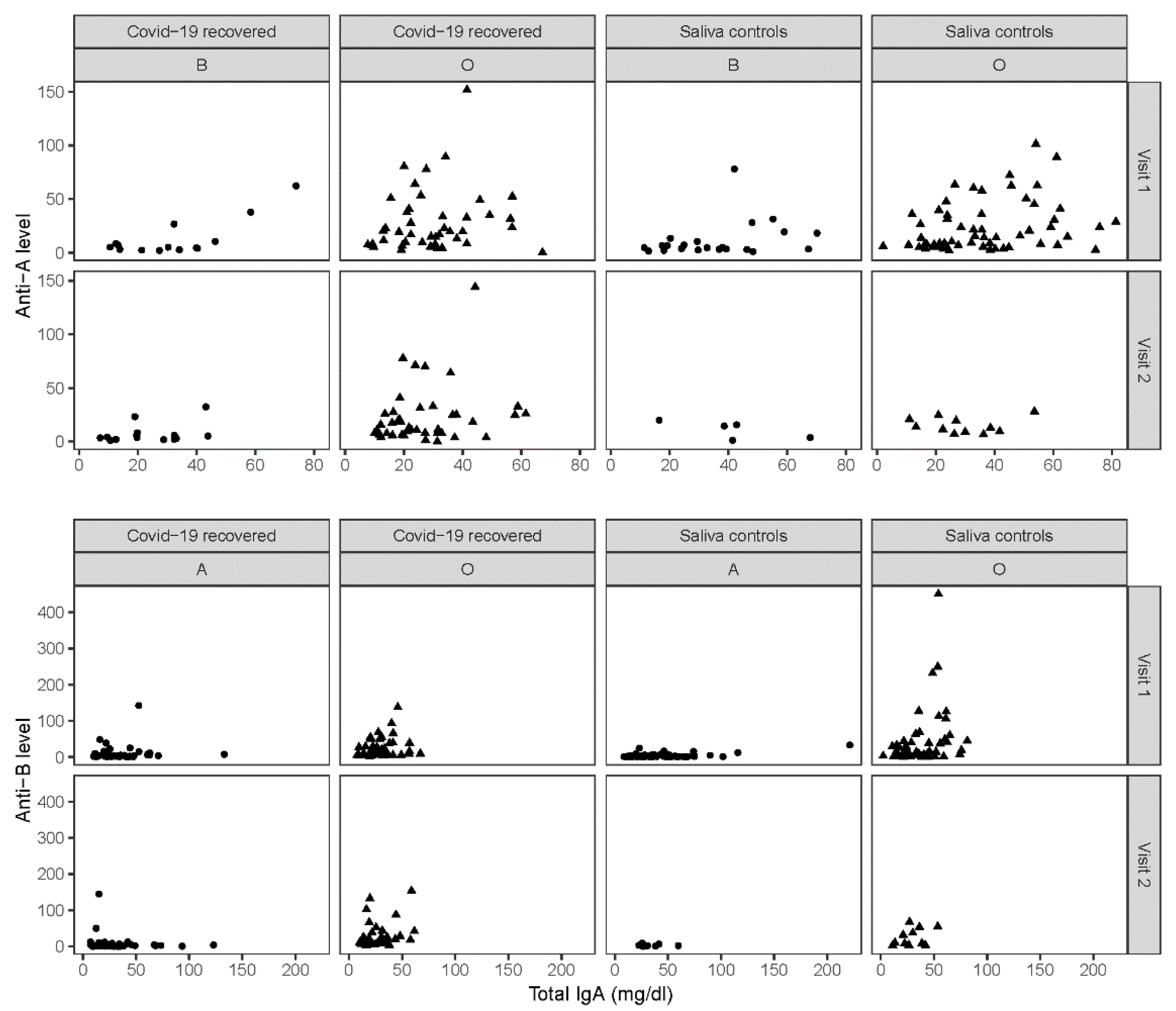
3.3. Total IgA in Saliva
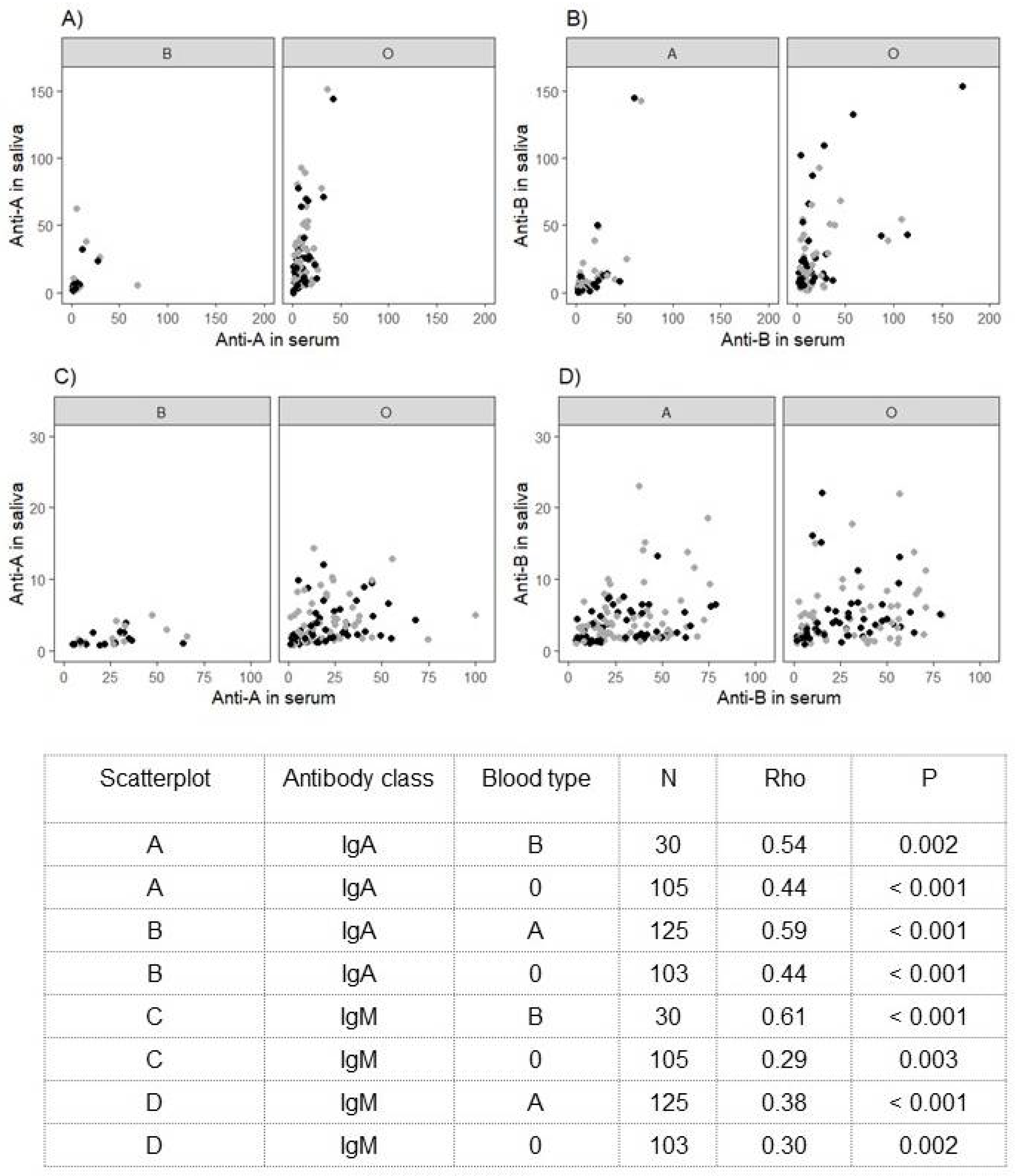
3.4. ABO Antibodies and SARS-CoV-2-Specific Antibody Concentrations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, B.B.; Gu, D.Z.; Yu, J.N.; Yang, J.; Shen, W.Q. Association between ABO blood groups and COVID-19 infection, severity and demise: A systematic review and meta-analysis. Infect. Genet. Evol. 2020, 84, 104485. [Google Scholar] [CrossRef]
- Wu, Y.; Feng, Z.; Li, P.; Yu, Q. Relationship between ABO blood group distribution and clinical characteristics in patients with COVID-19. Clin. Chim. Acta 2020, 509, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Franchini, M.; Cruciani, M.; Mengoli, C.; Marano, G.; Candura, F.; Lopez, N.; Pati, I.; Pupella, S.; De Angelis, V. ABO blood group and COVID-19: An updated systematic literature review and meta-analysis. Blood Transfus. Trasfus. Sangue 2021, 19, 317–326. [Google Scholar]
- Abdollahi, A.; Mahmoudi-Aliabadi, M.; Mehrtash, V.; Jafarzadeh, B.; Salehi, M. The Novel Coronavirus SARS-CoV-2 Vulnerability Association with ABO/Rh Blood Types. Iran. J. Pathol. 2020, 15, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Zietz, M.; Zucker, J.; Tatonetti, N.P. Associations between blood type and COVID-19 infection, intubation, and death. Nat. Commun. 2020, 11, 5761. [Google Scholar] [CrossRef] [PubMed]
- Matzhold, E.M.; Berghold, A.; Bemelmans, M.K.B.; Banfi, C.; Stelzl, E.; Kessler, H.H.; Steinmetz, I.; Krause, R.; Wurzer, H.; Schlenke, P. Lewis and ABO histo-blood types and the secretor status of patients hospitalized with COVID-19 implicate a role for ABO antibodies in susceptibility to infection with SARS-CoV-2. Transfusion 2021, 61, 2736–2745. [Google Scholar] [CrossRef] [PubMed]
- Guillon, P.; Clement, M.; Sebille, V.; Rivain, J.G.; Chou, C.F.; Ruvoen-Clouet, N.; Le Pendu, J. Inhibition of the interaction between the SARS-CoV spike protein and its cellular receptor by anti-histo-blood group antibodies. Glycobiology 2008, 18, 1085–1093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamayo-Velasco, A.; Penarrubia-Ponce, M.J.; Alvarez, F.J.; de la Fuente, I.; Perez-Gonzalez, S.; Andaluz-Ojeda, D. ABO Blood System and COVID-19 Susceptibility: Anti-A and Anti-B Antibodies Are the Key Points. Front. Med. 2022, 9, 882477. [Google Scholar] [CrossRef]
- Wu, S.C.; Arthur, C.M.; Wang, J.; Verkerke, H.; Josephson, C.D.; Kalman, D.; Roback, J.D.; Cummings, R.D.; Stowell, S.R. The SARS-CoV-2 receptor-binding domain preferentially recognizes blood group A. Blood Adv. 2021, 5, 1305–1309. [Google Scholar] [CrossRef] [PubMed]
- Deleers, M.; Breiman, A.; Daubie, V.; Maggetto, C.; Barreau, I.; Besse, T.; Clémenceau, B.; Ruvoën-Clouet, N.; Fils, J.-F.; Maillart, E.; et al. Covid-19 and blood groups: ABO antibody levels may also matter. Int. J. Infect. Dis. 2021, 104, 242–249. [Google Scholar] [CrossRef]
- Arend, P. Why blood group A individuals are at risk whereas blood group O individuals are protected from SARS-CoV-2 (COVID-19) infection: A hypothesis regarding how the virus invades the human body via ABO(H) blood group-determining carbohydrates. Immunobiology 2021, 226, 152027. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef] [Green Version]
- Wu, K.; Chen, L.; Peng, G.; Zhou, W.; Pennell, C.A.; Mansky, L.M.; Geraghty, R.; Li, F. A virus-binding hot spot on human angiotensin-converting enzyme 2 is critical for binding of two different coronaviruses. J. Virol. 2011, 85, 5331–5337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, H.G.A.D. Blood Transfusion in Clinical Medicine, 12th ed.; Wiley Blackwell: London, UK, 2014. [Google Scholar]
- Mazda, T.; Yabe, R.; NaThalang, O.; Thammavong, T.; Tadokoro, K. Differences in ABO antibody levels among blood donors: A comparison between past and present Japanese, Laotian, and Thai populations. Immunohematology 2007, 23, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Redman, M.; Malde, R.; Contreras, M. Comparison of IgM and IgG anti-A and anti-B levels in Asian, Caucasian and Negro donors in the North West Thames Region. Vox Sang. 1990, 59, 89–91. [Google Scholar] [CrossRef]
- Fung, M.G.B.J.; Hiller, C.D.; Westhoff, C.M. American Association of Blood Banks (AABB) Technical Manual, 18th ed.; American Association of Blood Banks: Washington, DC, USA, 2014. [Google Scholar]
- Khampanon, K.; Chanprakop, T.; Sriwanitchrak, P.; Setthakarn, M.; Oota, S.; Nathalang, O. The characteristics of ABO antibodies in group O Thai blood donors. J. Clin. Lab. Anal. 2012, 26, 223–226. [Google Scholar] [CrossRef]
- Berseus, O.; Boman, K.; Nessen, S.C.; Westerberg, L.A. Risks of hemolysis due to anti-A and anti-B caused by the transfusion of blood or blood components containing ABO-incompatible plasma. Transfusion 2013, 53 (Suppl. S1), 114S–123S. [Google Scholar] [CrossRef]
- Sprogoe, U.; Yazer, M.H.; Rasmussen, M.H.; Antonsen, B.; Bistrup, C.; Assing, K. Minimal variation in anti-A and -B titers among healthy volunteers over time: Implications for the use of out-of-group blood components. J. Trauma Acute Care Surg. 2017, 82 (Suppl. S1), S87–S90. [Google Scholar] [CrossRef]
- Schroeder, H.W., Jr.; Cavacini, L. Structure and function of immunoglobulins. J. Allergy Clin. Immunol. 2010, 125 (Suppl. S2), S41–S52. [Google Scholar] [CrossRef] [Green Version]
- de Sousa-Pereira, P.; Woof, J.M. IgA: Structure, Function, and Developability. Antibodies 2019, 8, 57. [Google Scholar] [CrossRef] [Green Version]
- Corthesy, B. Roundtrip ticket for secretory IgA: Role in mucosal homeostasis? J. Immunol. 2007, 178, 27–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [Green Version]
- Ma, C.; Cong, Y.; Zhang, H. COVID-19 and the Digestive System. Am. J. Gastroenterol. 2020, 115, 1003–1006. [Google Scholar] [CrossRef] [PubMed]
- de Freitas Dutra, V.; Bonet-Bub, C.; Yokoyama, A.P.H.; Achkar, R.; Machado, R.R.G.; Assuncao, M.; Candelária, G.; Soares, C.P.; Fachini, R.M.; Fontão-Wendel, R.; et al. Anti-A and SARS-CoV-2: An intriguing association. Vox Sang. 2021, 116, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Hayes, C.; Rubenstein, W.; Gibb, D.; Klapper, E.; Tanaka, J.; Pepkowitz, S. Blood group O convalescent plasma donations have significantly lower levels of SARS-CoV-2 IgG antibodies compared to blood group A donations. Transfusion 2021, 61, 2245–2249. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Y.; Huang, H.; Li, D.; Gu, D.; Lu, X.; Zhang, Z.; Liu, L.; Liu, T.; Liu, Y.; et al. Relationship between the ABO Blood Group and the COVID-19 Susceptibility. Clin. Infect. Dis. 2020, 73, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Gerard, C.; Maggipinto, G.; Minon, J.M. COVID-19 and ABO blood group: Another viewpoint. Br. J. Haematol. 2020, 190, e93–e94. [Google Scholar] [CrossRef] [PubMed]
- Breiman, A.; Ruven-Clouet, N.; Le Pendu, J. Harnessing the natural anti-glycan immune response to limit the transmission of enveloped viruses such as SARS-CoV-2. PLoS Pathog. 2020, 16, e1008556. [Google Scholar] [CrossRef] [PubMed]
- Boukhari, R.; Breiman, A.; Jazat, J.; Ruvoen-Clouet, N.; Martinez, S.; Damais-Cepitelli, A.; Le Niger, C.; Devie-Hubert, I.; Penasse, F.; Mauriere, D.; et al. ABO Blood Group Incompatibility Protects Against SARS-CoV-2 Transmission. Front. Microbiol. 2021, 12, 799519. [Google Scholar] [CrossRef]
- Kral, S.; Banfi, C.; Niedrist, T.; Sareban, N.; Guelly, C.; Kriegl, L.; Schiffmann, S.; Zurl, C.; Herrmann, M.; Steinmetz, I.; et al. Long-lasting immune response to a mild course of PCR-confirmed SARS-CoV-2 infection: A cohort study. J. Infect. 2021, 83, 607–635. [Google Scholar] [CrossRef]
- Wahrmann, M.; Schiemann, M.; Marinova, L.; Kormoczi, G.F.; Derfler, K.; Fehr, T.; Stussi, G.; Böhmig, G.A. Anti-A/B antibody depletion by semiselective versus ABO blood group-specific immunoadsorption. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2012, 27, 2122–2129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daniels, G. Human Blood Groups, 3rd ed.; Wiley-Blackwell: Oxford, UK, 2013. [Google Scholar]
- Reinhold Eckstein, R.Z. Immunhhämatologie und Klinische Transfusionsmedizin, 6th ed.; Elsevier: Urban & Fischer, München, 2010. [Google Scholar]
- Yu, H.Q.; Sun, B.Q.; Fang, Z.F.; Zhao, J.C.; Liu, X.Y.; Li, Y.M.; Sun, X.Z.; Liang, H.F.; Zhong, B.; Huang, Z.F.; et al. Distinct features of SARS-CoV-2-specific IgA response in COVID-19 patients. Eur. Respir. J. 2020, 56. [Google Scholar] [CrossRef]
- Hasan Ali, O.; Bomze, D.; Risch, L.; Brugger, S.D.; Paprotny, M.; Weber, M.; Thiel, S.; Kern, L.; Albrich, W.C.; Kohler, P.; et al. Severe Coronavirus Disease 2019 (COVID-19) is Associated With Elevated Serum Immunoglobulin (Ig) A and Antiphospholipid IgA Antibodies. Clin. Infect. Dis. 2021, 73, e2869–e2874. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Tong, X.; Chen, H.; He, R.; Lv, Q.; Yang, R.; Zhao, L.; Wang, J.; Xu, H.; Liu, C.; et al. Characteristics and serological patterns of COVID-19 convalescent plasma donors: Optimal donors and timing of donation. Transfusion 2020, 60, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Wendel, S.; Fontao-Wendel, R.; Fachini, R.; Candelaria, G.; Scuracchio, P.; Achkar, R.; Brito, M.; Reis, L.F.; Camargo, A.; Amano, M.; et al. A longitudinal study of convalescent plasma (CCP) donors and correlation of ABO group, initial neutralizing antibodies (nAb), and body mass index (BMI) with nAb and anti-nucleocapsid (NP) SARS-CoV-2 antibody kinetics: Proposals for better quality of CCP collections. Transfusion 2021, 61, 1447–1460. [Google Scholar] [PubMed]
- Mensah, A.A.; Lacy, J.; Stowe, J.; Seghezzo, G.; Sachdeva, R.; Simmons, R.; Bukasa, A.; O’Boyle, S.; Andrews, N.; Ramsay, M.; et al. Disease severity during SARS-COV-2 reinfection: A nationwide study. J. Infect. 2022, 84, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Valenti, L.; Villa, S.; Baselli, G.; Temporiti, R.; Bandera, A.; Scudeller, L.; Prati, D. Association of ABO blood group and secretor phenotype with severe COVID-19. Transfusion 2020, 60, 3067–3070. [Google Scholar] [CrossRef]
- Latz, C.A.; DeCarlo, C.; Boitano, L.; Png, C.Y.M.; Patell, R.; Conrad, M.F.; Eagleton, M.; Dua, A. Blood type and outcomes in patients with COVID-19. Ann. Hematol. 2020, 99, 2113–2118. [Google Scholar] [CrossRef]
- Breiman, A.; Ruvoen-Clouet, N.; Deleers, M.; Beauvais, T.; Jouand, N.; Rocher, J.; Bovin, N.; Labarrière, N.; El Kenz, H.; Le Pendu, J. Low Levels of Natural Anti-alpha-N-Acetylgalactosamine (Tn) Antibodies Are Associated With COVID-19. Front. Microbiol. 2021, 12, 641460. [Google Scholar] [CrossRef] [PubMed]
- Czlonkowska, A.; Korlak, J. The immune response during aging. J. Gerontol. 1979, 34, 9–14. [Google Scholar] [CrossRef]
- Wasiluk, T.; Bujno, M.; Rybinska, K.; Rogowska, A.; Zebrowska, A.; Boczkowska-Radziwon, B.; Piszcz, J.; Bolkun, L.; Radziwon, P. No increase in anti-A isohemagglutinin titer after SARS-CoV-2 infection: A retrospective cohort analysis of group O apheresis platelet donors. J. Clin. Apher. 2021, 36, 882–885. [Google Scholar] [CrossRef] [PubMed]
- Gupte, S.C.; Bhatia, H.M. Anti-A and anti-B titre response after tetanus toxoid injections in normal adults and pregnant women. Indian J. Med. Res. 1979, 70, 221–228. [Google Scholar] [PubMed]
- Voak, D. The pathogenesis of ABO haemolytic disease of the newborn. Vox Sang. 1969, 17, 481–513. [Google Scholar] [CrossRef] [PubMed]
- Storry, J.R.; Olsson, M.L. The ABO blood group system revisited: A review and update. Immunohematology 2009, 25, 48–59. [Google Scholar] [CrossRef]
- Josephson, C.D.; Mullis, N.C.; Van Demark, C.; Hillyer, C.D. Significant numbers of apheresis-derived group O platelet units have “high-titer” anti-A/A,B: Implications for transfusion policy. Transfusion 2004, 44, 805–808. [Google Scholar] [CrossRef]
- Bastos, E.P.; Castilho, L.; Bub, C.B.; Kutner, J.M. Comparison of ABO antibody titration, IgG subclasses and qualitative haemolysin test to reduce the risk of passive haemolysis associated with platelet transfusion. Transfus. Med. 2020, 30, 317–323. [Google Scholar] [CrossRef]
- Centeres for Disease Control and Prevention. Coronavirus (COVID-19). Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/assessing-risk-factors.html (accessed on 16 February 2021).
- Barbalic, M.; Dupuis, J.; Dehghan, A.; Bis, J.C.; Hoogeveen, R.C.; Schnabel, R.B.; Nambi, V.; Bretler, M.; Smith, N.L.; Peters, A.; et al. Large-scale genomic studies reveal central role of ABO in sP-selectin and sICAM-1 levels. Hum. Mol. Genet. 2010, 19, 1863–1872. [Google Scholar] [CrossRef] [Green Version]
- Chung, C.M.; Wang, R.Y.; Chen, J.W.; Fann, C.S.; Leu, H.B.; Ho, H.Y.; Ting, C.-T.; Lin, T.-H.; Sheu, S.-H.; Tsai, W.-C.; et al. A genome-wide association study identifies new loci for ACE activity: Potential implications for response to ACE inhibitor. Pharm. J. 2010, 10, 537–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, M.; Wolpin, B.; Rexrode, K.; Manson, J.E.; Rimm, E.; Hu, F.B.; Qi, L. ABO blood group and risk of coronary heart disease in two prospective cohort studies. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2314–2320. [Google Scholar] [CrossRef] [Green Version]
- Wiggins, K.L.; Smith, N.L.; Glazer, N.L.; Rosendaal, F.R.; Heckbert, S.R.; Psaty, B.M.; Rice, K.M.; Lumley, T. ABO genotype and risk of thrombotic events and hemorrhagic stroke. J. Thromb. Haemost. 2009, 7, 263–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| COVID-19 Convalescents (N = 187) | Serum Controls (N = 195) | p (Convalescents vs. Serum Controls) | Saliva Controls (N = 164) | p (Convalescents vs. Saliva Controls) | |
|---|---|---|---|---|---|
| Age Median (Q1, Q3) | 39.0 (30.0, 49.0) | 39.0 (30.0, 49.5) | 0.806 * | 34.0 (26.0, 46.0) | 0.004 * |
| Sex | 1.000 † | <0.001 † | |||
| Male | 75 (39.9%) | 78 (40.0%) | 98 (59.8%) | ||
| Female | 112 (59.9%) | 117 (60.0%) | 66 (40.2%) | ||
| ABO blood type | 0.983 † | 0.576 † | |||
| A | 82 (43.9%) | 85 (43.6%) | 66 (40.2%) | ||
| AB | 18 (9.6%) | 17 (8.7%) | 18 (11.0%) | ||
| B | 17 (9.1%) | 17 (8.7%) | 22 (13.4%) | ||
| O | 70 (37.4%) | 76 (39.0%) | 58 (35.4%) | ||
| Days between COVID-19 diagnosis and first sampling (V1) Median (Q1, Q3) | 44.0 (30.0, 61.0) | NA | NA | NA | NA |
| Days between samplings 1 and 2 Median (Q1, Q3) | 66.0 (62.0, 74.0) | 64.0 (62.0, 72.0) | 0.125 * | 56.0 (56.0, 60.0) | <0.001 * |
| COVID-19 Convalescents | Serum Controls | |||||
|---|---|---|---|---|---|---|
| Anti-A | Blood Type | Visit | N | Median (Q1, Q3) | N | Median (Q1, Q3) |
| IgM | B | 1 | 17 | 27.9 (21.6, 32.3) | 17 | 36.9 (22.1, 68.7) |
| IgM | B | 2 | 16 | 27.1 (14.7, 32.6) | 17 | 49.4 (23.3, 60.6) |
| IgM | O | 1 | 70 | 21.0 (11.4, 29.8) | 76 | 32.5 (16, 57.4) |
| IgM | O | 2 | 58 | 16.9 (8.9, 30.7) | 76 | 29.6 (14.7, 52.1) |
| IgG | B | 1 | 17 | 1.7 (1.2, 2.3) | 17 | 1.7 (1.2, 2.8) |
| IgG | B | 2 | 16 | 1.7 (1.3, 1.9) | 17 | 1.7 (1.4, 2) |
| IgG | O | 1 | 70 | 10.6 (5.5, 23.7) | 76 | 16.9 (8.4, 34.1) |
| IgG | O | 2 | 58 | 10.4 (5.8, 18.3) | 76 | 18.6 (9.2, 29.6) |
| IgA | B | 1 | 17 | 3.9 (2.5, 8.8) | 17 | 5.2 (3.5, 7.1) |
| IgA | B | 2 | 16 | 3.9 (2.4, 5.5) | 17 | 5.5 (3.9, 6.8) |
| IgA | O | 1 | 70 | 7.7 (3.9, 14.4) | 76 | 12.7 (8.2, 21.3) |
| IgA | O | 2 | 58 | 9.5 (5.1, 14) | 76 | 12.3 (7.4, 19) |
| Anti-B | ||||||
| IgM | A | 1 | 82 | 26.5 (17.5, 44.3) | 85 | 38.8 (16.5, 69.3) |
| IgM | A | 2 | 63 | 26.2 (12.2, 41.2) | 85 | 44.6 (17.8, 69) |
| IgM | O | 1 | 70 | 28.3 (11, 51.4) | 76 | 24.1 (10.8, 50.5) |
| IgM | O | 2 | 58 | 24.5 (8.9, 40) | 76 | 22.5 (10.6, 51.2) |
| IgG | A | 1 | 82 | 1.1 (0.9, 1.2) | 85 | 1.2 (1, 1.4) |
| IgG | A | 2 | 63 | 1.1 (1, 1.2) | 85 | 1.1 (1, 1.4) |
| IgG | O | 1 | 70 | 3.8 (2.3, 7.2) | 76 | 5.9 (2.6, 15.5) |
| IgG | O | 2 | 58 | 3.8 (2.4, 8.1) | 76 | 7.5 (2.7, 17.3) |
| IgA | A | 1 | 82 | 3.4 (2.3, 6.3) | 85 | 4.6 (2.1, 9.3) |
| IgA | A | 2 | 63 | 3.5 (2.3, 6.6) | 85 | 4.8 (2.6, 10.3) |
| IgA | O | 1 | 70 | 9.6 (5.4, 17.5) | 76 | 12.8 (6.4, 31) |
| IgA | O | 2 | 58 | 9.3 (5, 17.5) | 76 | 13.4 (6.8, 35.2) |
| Estimate | 95% CI | p | |
|---|---|---|---|
| Anti-A | |||
| Age | 0.0001 | (−0.003; 0.003) | 0.976 |
| Sex (male) | 0.03 | (−0.05; 0.11) | 0.444 |
| Time point (visit 2) | −0.02 | (−0.06; 0.02) | 0.273 |
| Antibody_class: IgM | 1.16 | (1.05; 1.28) | <0.001 |
| Antibody_class: IgA | 0.46 | (0.35; 0.58) | <0.001 |
| Group: Serum controls | 0.19 | (0.11; 0.26) | <0.001 |
| Blood type: O | 0.82 | (0.7; 0.94) | <0.001 |
| Antibody_class × blood type (IgM:O) | −0.94 | (−1.06; −0.81) | <0.001 |
| Antibody_class × blood type (IgA:O) | −0.57 | (−0.69; −0.44) | <0.001 |
| Anti-B | |||
| Age | −0.001 | (−0.003; 0.002) | 0.618 |
| Sex (male) | −0.08 | (−0.14; −0.02) | 0.011 |
| Time point (visit 2) | −0.004 | (−0.04; 0.03) | 0.786 |
| Antibody_class: IgM | 1.4 | (1.34; 1.45) | <0.001 |
| Antibody_class: IgA | 0.63 | (0.58; 0.68) | <0.001 |
| Group: Serum controls | 0.10 | (0.04; 0.16) | 0.001 |
| Blood type: O | 0.65 | (0.58; 0.73) | <0.001 |
| Antibody_class × blood type (IgM:O) | −0.8 | (−0.88; −0.73) | <0.001 |
| Antibody_class × blood type (IgA:O) | −0.27 | (−0.34; −0.19) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matzhold, E.M.; Körmöczi, G.F.; Banfi, C.; Schönbacher, M.; Drexler-Helmberg, C.; Steinmetz, I.; Berghold, A.; Schlenke, P.; Wagner, G.E.; Stoisser, A.; et al. Lower Levels of ABO Anti-A and Anti-B of IgM, IgG and IgA Isotypes in the Serum but Not the Saliva of COVID-19 Convalescents. J. Clin. Med. 2022, 11, 4513. https://doi.org/10.3390/jcm11154513
Matzhold EM, Körmöczi GF, Banfi C, Schönbacher M, Drexler-Helmberg C, Steinmetz I, Berghold A, Schlenke P, Wagner GE, Stoisser A, et al. Lower Levels of ABO Anti-A and Anti-B of IgM, IgG and IgA Isotypes in the Serum but Not the Saliva of COVID-19 Convalescents. Journal of Clinical Medicine. 2022; 11(15):4513. https://doi.org/10.3390/jcm11154513
Chicago/Turabian StyleMatzhold, Eva M., Günther F. Körmöczi, Chiara Banfi, Marlies Schönbacher, Camilla Drexler-Helmberg, Ivo Steinmetz, Andrea Berghold, Peter Schlenke, Gabriel E. Wagner, Anja Stoisser, and et al. 2022. "Lower Levels of ABO Anti-A and Anti-B of IgM, IgG and IgA Isotypes in the Serum but Not the Saliva of COVID-19 Convalescents" Journal of Clinical Medicine 11, no. 15: 4513. https://doi.org/10.3390/jcm11154513
APA StyleMatzhold, E. M., Körmöczi, G. F., Banfi, C., Schönbacher, M., Drexler-Helmberg, C., Steinmetz, I., Berghold, A., Schlenke, P., Wagner, G. E., Stoisser, A., Kleinhappl, B., Mayr, W. R., & Wagner, T. (2022). Lower Levels of ABO Anti-A and Anti-B of IgM, IgG and IgA Isotypes in the Serum but Not the Saliva of COVID-19 Convalescents. Journal of Clinical Medicine, 11(15), 4513. https://doi.org/10.3390/jcm11154513







