Obesity Impact on SARS-CoV-2 Infection: Pros and Cons “Obesity Paradox”—A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Literature Search
2.2. Inclusion Criteria
- Studies published between January 2020 and December 2021
- Studies published in English
- Studies measuring obesity by BMI
- Studies that must have been longitudinal studies (retrospective or prospective or clinical trial)
- Studies that examined the association between obesity and mortality, specifically in SARS-CoV-2 infection
- Studies published as abstracts, letters to the editor, book chapters
- Studies regarding animal model
- Systematic reviews and meta-analysis
- Studies on paediatric patients or pregnant women.
3. Results
4. Discussion
4.1. Severe COVID-19 in Higher Obesity
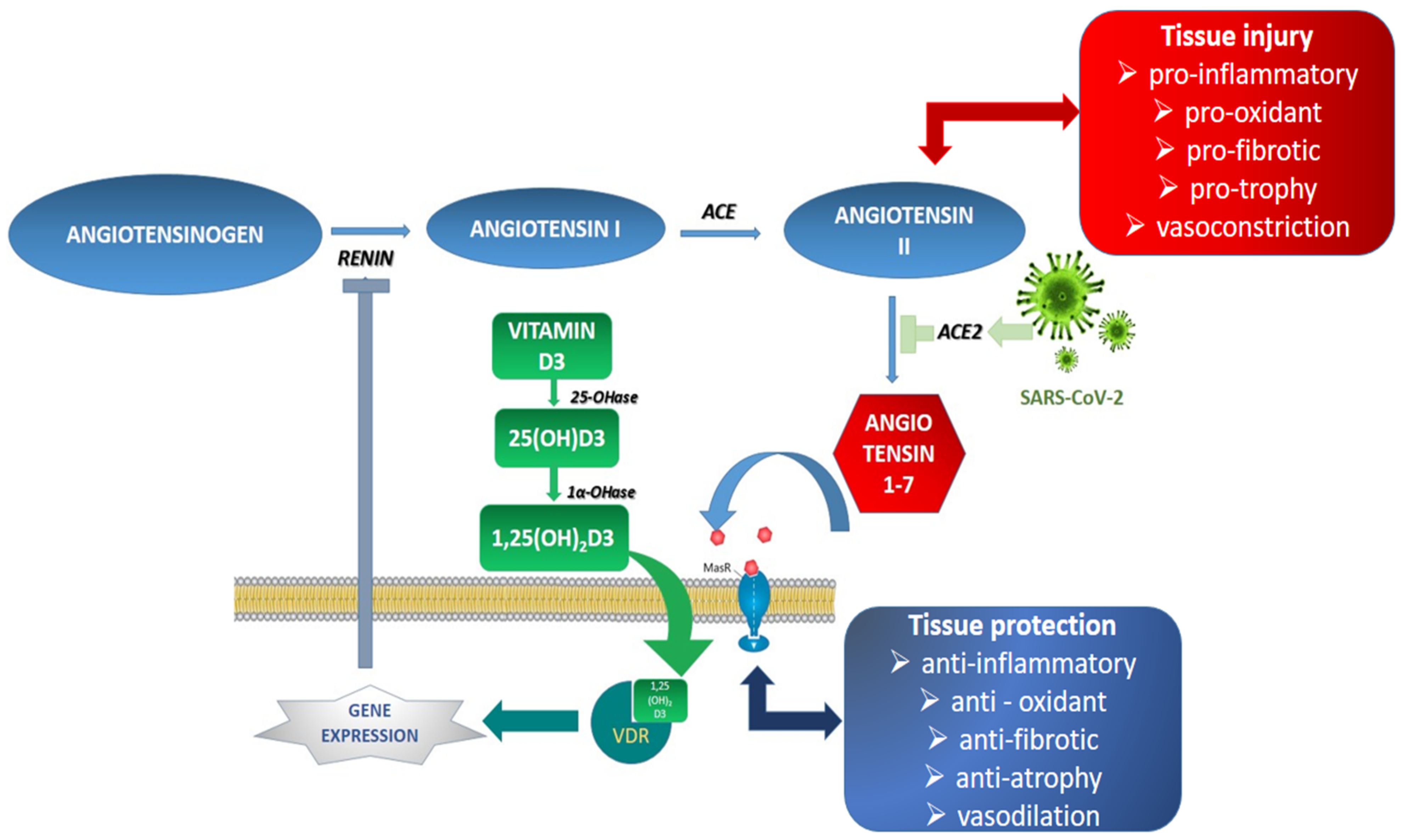
4.2. The Role of ACE2 Receptors in COVID 19 Infection
4.3. The Implication of Vitamin D in COVID-19 Obese Patients
4.4. Limitations and Strengths of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Naming the Coronavirus Disease (COVID-19) and the Virus That Causes It. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it (accessed on 16 January 2022).
- Surveillance Case Definitions for Human Infection with Novel Coronavirus (NCoV) Interim Guidance Surveillance Objectives of Surveillance the Following People Should Be Investigated and Tested for NCoV Infection Case Definitions for Surveillance. Available online: https://www.who.int/publications/m/item/surveillance-case-definitions-for-human-infection-withnovel-coronavirus-(ncov) (accessed on 16 January 2022).
- Wong, G.; Bi, Y.H.; Wang, Q.H.; Chen, X.W.; Zhang, Z.G.; Yao, Y.G. Zoonotic Origins of Human Coronavirus 2019 (HCoV-19/SARS-CoV-2): Why is This Work Important? Zool. Res. 2020, 41, 213. [Google Scholar] [CrossRef] [PubMed]
- Hadfield, J.; Megill, C.; Bell, S.M.; Huddleston, J.; Potter, B.; Callender, C.; Sagulenko, P.; Bedford, T.; Neher, R.A. NextStrain: Real-Time Tracking of Pathogen Evolution. Bioinformatics 2018, 34, 4121–4123. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.Y.; Zhao, R.; Gao, L.J.; Gao, X.F.; Wang, D.P.; Cao, J.M. SARS-CoV-2: Structure, Biology, and Structure-Based Therapeutics Development. Front. Cell. Infect. Microbiol. 2020, 10, 724. [Google Scholar] [CrossRef]
- Cevik, M.; Kuppalli, K.; Kindrachuk, J.; Peiris, M. Virology, Transmission, and Pathogenesis of SARS-CoV-2. BMJ 2020, 371, m3862. [Google Scholar] [CrossRef] [PubMed]
- LSBio. SARS-CoV-2 and COVID-19 Pathogenesis: A Review. Available online: https://www.lsbio.com/media/whitepapers/sars-cov-2-and-covid-19-pathogenesis-a-review (accessed on 17 April 2021).
- Coronavirus. Available online: https://www.who.int/health-topics/coronavirus#tab=tab_3 (accessed on 9 May 2021).
- Jordan, R.E.; Adab, P.; Cheng, K.K. COVID-19: Risk Factors for Severe Disease and Death. BMJ 2020, 368, m1198. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, Q.; Chi, J.; Dong, B.; Lv, W.; Shen, L.; Wang, Y. Comorbidities and the Risk of Severe or Fatal Outcomes Associated with Coronavirus Disease 2019: A Systematic Review and Meta-Analysis. Int. J. Infect. Dis. 2020, 99, 47–56. [Google Scholar] [CrossRef]
- De Siqueira, J.V.V.; Almeida, L.G.; Zica, B.O.; Brum, I.B.; Barceló, A.; de Siqueira Galil, A.G. Impact of Obesity on Hospitalizations and Mortality, Due to COVID-19: A Systematic Review. Obes. Res. Clin. Pract. 2020, 14, 398–403. [Google Scholar] [CrossRef]
- Yang, J.; Hu, J.; Zhu, C. Obesity Aggravates COVID-19: A Systematic Review and Meta-Analysis. J. Med. Virol. 2021, 93, 257–261. [Google Scholar] [CrossRef]
- Rusu, A.; Todea, D.; Rosca, L.; Nita, C.; Bala, C. The Development of a Sleep Apnea Screening Program in Romanian Type 2 Diabetic Patients: A Pilot Study. Acta Diabetol. 2012, 49, 105–109. [Google Scholar] [CrossRef]
- Rusu, A.; Nita, C.; Todea, D.; Rosca, L.; Bala, C.; Hancu, N. Correlation of the Daytime Sleepiness with Respiratory Sleep Parameters in Patients with Sleep Apnea and Type 2 Diabetes. Acta Endocrinol. 2011, 7, 163–171. [Google Scholar] [CrossRef]
- Simonnet, A.; Chetboun, M.; Poissy, J.; Raverdy, V.; Noulette, J.; Duhamel, A.; Labreuche, J.; Mathieu, D.; Pattou, F.; Jourdain, M.; et al. High Prevalence of Obesity in Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) Requiring Invasive Mechanical Ventilation. Obesity 2020, 28, 1195–1199. [Google Scholar] [CrossRef] [PubMed]
- Lavie, C.J.; Coursin, D.B.; Long, M.T. The Obesity Paradox in Infections and Implications for COVID-19. Mayo Clin. Proc. 2021, 96, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Naderi, N.; Kleine, C.E.; Park, C.; Hsiung, J.T.; Soohoo, M.; Tantisattamo, E.; Streja, E.; Kalantar-Zadeh, K.; Moradi, H. Obesity Paradox in Advanced Kidney Disease: From Bedside to the Bench. Prog. Cardiovasc. Dis. 2018, 61, 168–181. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Wang, X.; Wang, Y.; Xing, G.; Liu, S. “Obesity Paradox” in Acute Respiratory Distress Syndrome: Asystematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0163677. [Google Scholar] [CrossRef]
- Horwich, T.B.; Fonarow, G.C.; Clark, A.L. Obesity and the Obesity Paradox in Heart Failure. Prog. Cardiovasc. Dis. 2018, 61, 151–156. [Google Scholar] [CrossRef]
- Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 9 May 2021).
- Cai, S.H.; Liao, W.; Chen, S.W.; Liu, L.L.; Liu, S.Y.; Zheng, Z.D. Association between Obesity and Clinical Prognosis in Patients Infected with SARS-CoV-2. Infect. Dis. Poverty 2020, 9, 80. [Google Scholar] [CrossRef]
- CDC. Adult Obesity Facts: Overweight & Obesity. Available online: https://www.cdc.gov/obesity/data/adult.html (accessed on 15 January 2022).
- Statista. Obesity rates in Brazil. Available online: https://www.statista.com/statistics/781305/share-adult-people-obese-brazil/ (accessed on 15 January 2022).
- WHO/Europe. Obesity—Data and Statistics. Available online: https://www.euro.who.int/en/health-topics/noncommunicable-diseases/obesity/data-and-statistics (accessed on 15 January 2022).
- Chen, Y.; Peng, Q.; Yang, Y.; Zheng, S.; Wang, Y.; Lu, W. The Prevalence and Increasing Trends of Overweight, General Obesity, and Abdominal Obesity among Chinese Adults: A Repeated Cross-Sectional Study. BMC Public Health 2019, 19, 1293. [Google Scholar] [CrossRef]
- Mu, L.; Liu, J.; Zhou, G.; Wu, C.; Chen, B.; Lu, Y.; Lu, J.; Yan, X.; Zhu, Z.; Nasir, K.; et al. Obesity Prevalence and Risks Among Chinese Adults: Findings From the China PEACE Million Persons Project, 2014–2018. Circ. Cardiovasc. Qual. Outcomes 2021, 14, e007292. [Google Scholar] [CrossRef]
- Wikipedia. Obesity in China. Available online: https://en.wikipedia.org/wiki/Obesity_in_China (accessed on 15 January 2022).
- Krams, I.A.; Jõers, P.; Luoto, S.; Trakimas, G.; Lietuvietis, V.; Krams, R.; Kaminska, I.; Rantala, M.J.; Krama, T.; Hadjichristodoulou, C. The Obesity Paradox Predicts the Second Wave of COVID-19 to Be Severe in Western Countries. Int. J. Environ. Res. Public Health 2021, 18, 1029. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, Y.; Huang, Y.M.; Wang, M.; Ling, W.; Sui, Y.; Zhao, H.L. Obesity in Patients with COVID-19: A Systematic Review and Meta-Analysis. Metabolism 2020, 113, 154378. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Cummings, M.J.; Baldwin, M.R.; Abrams, D.; Jacobson, S.D.; Meyer, B.J.; Balough, E.M.; Aaron, J.G.; Claassen, J.; Rabbani, L.R.E.; Hastie, J.; et al. Epidemiology, Clinical Course, and Outcomes of Critically Ill Adults with COVID-19 in New York City: A Prospective Cohort Study. Lancet 2020, 395, 1763–1770. [Google Scholar] [CrossRef]
- Busetto, L.; Bettini, S.; Fabris, R.; Serra, R.; Dal Pra, C.; Maffei, P.; Rossato, M.; Fioretto, P.; Vettor, R. Obesity and COVID-19: An Italian Snapshot. Obesity 2020, 28, 1600–1605. [Google Scholar] [CrossRef] [PubMed]
- Goyal, P.; Ringel, J.B.; Rajan, M.; Choi, J.J.; Pinheiro, L.C.; Li, H.A.; Wehmeyer, G.T.; Alshak, M.N.; Jabri, A.; Schenck, E.J.; et al. Obesity and COVID-19 in New York City: A Retrospective Cohort Study. Ann. Intern. Med. 2020, 173, 855–858. [Google Scholar] [CrossRef] [PubMed]
- Biscarini, S.; Colaneri, M.; Ludovisi, S.; Seminari, E.; Pieri, T.C.; Valsecchi, P.; Gallazzi, I.; Giusti, E.; Cammà, C.; Zuccaro, V.; et al. The Obesity Paradox: Analysis from the SMAtteo COVID-19 REgistry (SMACORE) Cohort. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Dana, R.; Bannay, A.; Bourst, P.; Ziegler, C.; Losser, M.R.; Gibot, S.; Levy, B.; Audibert, G.; Ziegler, O. Obesity and Mortality in Critically Ill COVID-19 Patients with Respiratory Failure. Int. J. Obes. 2021, 45, 2028–2037. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.S.; Roslin, M.; Wang, J.J.; Kane, J.; Hirsch, J.S.; Kim, E.J. BMI as a Risk Factor for Clinical Outcomes in Patients Hospitalized with COVID-19 in New York. Obesity 2021, 29, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Gillet, S.A.; Brown, M.I.; Zu, Y.; Wilson, S.M.; Ahmed, S.J.; Tirumalasetty, S.; Lovre, D.; Krousel-Wood, M.; Denson, J.L.; et al. Clinical Characteristics and Outcomes in Women and Men Hospitalized for Coronavirus Disease 2019 in New Orleans. Biol. Sex Differ. 2021, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.; Garg, S.; O’Halloran, A.; Whitaker, M.; Pham, H.; Anderson, E.J.; Armistead, I.; Bennett, N.M.; Billing, L.; Como-Sabetti, K.; et al. Risk Factors for Intensive Care Unit Admission and In-Hospital Mortality Among Hospitalized Adults Identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET). Clin. Infect. Dis. 2021, 72, e206–e214. [Google Scholar] [CrossRef]
- Mankowski, N.; Al-Qurayshi, Z.; Souza, S.; Campbell, B.; Beighley, A.; Denson, J.; Mauldin, B.; Bojanowski, C.; Friedlander, P.; Zifodya, J. The Effect of Race, Socioeconomic Status, and Comorbidity on Patients Afflicted with COVID 19: A Local Perspective. Ann. Epidemiol. 2021, 64, 83. [Google Scholar] [CrossRef]
- Halasz, G.; Leoni, M.L.G.; Villani, G.Q.; Nolli, M.; Villani, M. Obesity, Overweight and Survival in Critically Ill Patients with SARS-CoV-2 Pneumonia: Is There an Obesity Paradox? Preliminary Results from Italy. Eur. J. Prev. Cardiol. 2020, 28, e15–e17. [Google Scholar] [CrossRef] [PubMed]
- Tartof, S.Y.; Qian, L.; Hong, V.; Wei, R.; Nadjafi, R.F.; Fischer, H.; Li, Z.; Shaw, S.F.; Caparosa, S.L.; Nau, C.L.; et al. Obesity and Mortality Among Patients Diagnosed With COVID-19: Results from an Integrated Health Care Organization. Ann. Intern. Med. 2020, 173, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Bello-Chavolla, O.Y.; Bahena-López, J.P.; Antonio-Villa, N.E.; Vargas-Vázquez, A.; González-Díaz, A.; Márquez-Salinas, A.; Fermín-Martínez, C.A.; Naveja, J.J.; Aguilar-Salinas, C.A. Predicting Mortality Due to SARS-CoV-2: A Mechanistic Score Relating Obesity and Diabetes to COVID-19 Outcomes in Mexico. J. Clin. Endocrinol. Metab. 2020, 105, 2752–2761. [Google Scholar] [CrossRef] [PubMed]
- Arjun, S.; Farraj, K.; Yeroushalmi, K.; Desai, J.; Paz, S.G.; Im, J.; Castillo, A.; Patel, U.; Rammohan, R.; Mustacchia, P.; et al. Evaluating Obesity Paradox in COVID 19: Insights from a Safety-Net Hospital in New York. Chest 2020, 158, A302. [Google Scholar] [CrossRef]
- Czernichow, S.; Beeker, N.; Rives-Lange, C.; Guerot, E.; Diehl, J.L.; Katsahian, S.; Hulot, J.S.; Poghosyan, T.; Carette, C.; Jannot, A.S. Obesity Doubles Mortality in Patients Hospitalized for Severe Acute Respiratory Syndrome Coronavirus 2 in Paris Hospitals, France: A Cohort Study on 5,795 Patients. Obesity 2020, 28, 2282–2289. [Google Scholar] [CrossRef]
- Abumayyaleh, M.; Núñez Gil, I.J.; El-Battrawy, I.; Estrada, V.; Becerra-Muñoz, V.M.; Aparisi, A.; Fernández-Rozas, I.; Feltes, G.; Arroyo-Espliguero, R.; Trabattoni, D.; et al. Does There Exist an Obesity Paradox in COVID-19? Insights of the International HOPE-COVID-19-Registry. Obes. Res. Clin. Pract. 2021, 15, 275–280. [Google Scholar] [CrossRef]
- Schavemaker, R.; Schultz, M.; Lagrand, W.; van Slobbe-Bijlsma, E.; Serpa Neto, A.; Paulus, F. Associations of Body Mass Index with Ventilation Management and Clinical Outcomes in Invasively Ventilated Patients with ARDS Related to COVID-19—Insights from the PRoVENT-COVID Study. J. Clin. Med. 2021, 10, 1176. [Google Scholar] [CrossRef]
- Wolf, M.; Alladina, J.; Navarrete-Welton, A.; Shoults, B.; Brait, K.; Ziehr, D.; Malhotra, A.; Hardin, C.C.; Hibbert, K.A. Obesity and Critical Illness in COVID-19: Respiratory Pathophysiology. Obesity 2021, 29, 870–878. [Google Scholar] [CrossRef]
- Carneiro, R.A.V.D.; Hillesheim, D.; Hallal, A.L.C. Correlation of Overweight Condition and Obesity with Mortality by COVID-19 in Brazil’s State Capitals. Arch. Endocrinol. Metab. 2021, 65, 386–391. [Google Scholar] [CrossRef]
- Foulkes, A.S.; Selvaggi, C.; Shinnick, D.; Lumish, H.; Kim, E.; Cao, T.; Thaweethai, T.; Qian, J.; Lu, F.; Yan, J.; et al. Understanding the Link Between Obesity and Severe COVID-19 Outcomes: Causal Mediation by Systemic Inflammatory Response. J. Clin. Endocrinol. Metab. 2022, 107, E698–E707. [Google Scholar] [CrossRef]
- Motaib, I.; Zbiri, S.; Elamari, S.; Dini, N.; Chadli, A.; El Kettani, C. Obesity and Disease Severity Among Patients With COVID-19. Cureus 2021, 13, e13165. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, G.; Samson, R.; Nedunchezian, S.H.; Srivastav, S.; Dixit, N.; Le Jemtel, T.H. COVID 19 In-Hospital Mortality, Body Mass Index and Obesity Related Conditions. J. Diabetes Complicat. 2021, 35, 108054. [Google Scholar] [CrossRef] [PubMed]
- Yates, T.; Razieh, C.; Zaccardi, F.; Rowlands, A.V.; Seidu, S.; Davies, M.J.; Khunti, K. Obesity, Walking Pace and Risk of Severe COVID-19 and Mortality: Analysis of UK Biobank. Int. J. Obes. 2021, 45, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Kaeuffer, C.; Le Hyaric, C.; Fabacher, T.; Mootien, J.; Dervieux, B.; Ruch, Y.; Hugerot, A.; Zhu, Y.J.; Pointurier, V.; Clere-Jehl, R.; et al. Clinical Characteristics and Risk Factors Associated with Severe COVID-19: Prospective Analysis of 1,045 Hospitalised Cases in North-Eastern France, March 2020. Eurosurveillance 2020, 25, 2000895. [Google Scholar] [CrossRef]
- Dixon, A.E.; Peters, U. The Effect of Obesity on Lung Function. Expert Rev. Respir. Med. 2018, 12, 755–767. [Google Scholar] [CrossRef]
- Cercato, C.; Fonseca, F.A. Cardiovascular Risk and Obesity. Diabetol. Metab. Syndr. 2019, 11, 74. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Gomar, F.; Lavie, C.J.; Mehra, M.R.; Henry, B.M.; Lippi, G. Obesity and Outcomes in COVID-19: When an Epidemic and Pandemic Collide. Mayo Clin. Proc. 2020, 95, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Zălar, D.-M.; Pop, C.; Buzdugan, E.; Todea, D.; Mogoșan, C.I. The atherosclerosis-inflammation relationship-a pathophysiological approach. Farmacia 2019, 67, 6. [Google Scholar] [CrossRef]
- Hanna, R.; Dalvi, S.; Benedicenti, S.; Amaroli, A.; Sălăgean, T.; Pop, I.D.; Todea, D.; Bordea, I.R. Photobiomodulation Therapy in Oral Mucositis and Potentially Malignant Oral Lesions: A Therapy Towards the Future. Cancers 2020, 12, 1949. [Google Scholar] [CrossRef]
- Radescu, O.D.; Albu, S.; Baciut, M.; Bran, S.; Coman, A.C.; Bechir, E.S.; Pacurar, M.; Todea, D.A. Results in the Treatment with Twin Block Polymeric Appliance of the Retrognathic Mandible in Sleep Apnea Patients. Mater. Plast. 2017, 54, 473–476. [Google Scholar] [CrossRef]
- Yanai, H. Significant Correlations of SARS-CoV-2 Infection with Prevalence of Overweight/Obesity and Mean Body Mass Index in the SARS-CoV-2 Endemic Countries. Cardiol. Res. 2020, 11, 412. [Google Scholar] [CrossRef] [PubMed]
- Ghoneim, S.; Butt, M.U.; Hamid, O.; Shah, A.; Asaad, I. The Incidence of COVID-19 in Patients with Metabolic Syndrome and Non-Alcoholic Steatohepatitis: A Population-Based Study. Metab. Open 2020, 8, 100057. [Google Scholar] [CrossRef] [PubMed]
- Scalsky, R.J.; Chen, Y.-J.; Desai, K.; O’Connell, J.R.; Perry, J.A.; Hong, C.C. Baseline Cardiometabolic Profiles and SARS-CoV-2 Infection in the UK Biobank. medRxiv 2020. [Google Scholar] [CrossRef]
- Hariyanto, T.I.; Kurniawan, A. Dyslipidemia Is Associated with Severe Coronavirus Disease 2019 (COVID-19) Infection. Diabetes Metab. Syndr. 2020, 14, 1463–1465. [Google Scholar] [CrossRef]
- Leong, A.; Cole, J.; Brenner, L.N.; Meigs, J.B.; Florez, J.C.; Mercader, J.M. Cardiometabolic Risk Factors for COVID-19 Susceptibility and Severity: A Mendelian Randomization Analysis. medRxiv 2020. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.-N.; Luo, J.; Yu, H.; Wang, Y.-W.; Hu, Y.-H.; Liu, D.; Liang, B.-M.; Liang, Z.-A. Can Body Mass Index Predict Clinical Outcomes for Patients with Acute Lung Injury/Acute Respiratory Distress Syndrome? A Meta-Analysis. Crit. Care 2017, 21, 36. [Google Scholar] [CrossRef]
- Chittal, P.; Babu, A.S.; Lavie, C.J. Obesity Paradox: Does Fat Alter Outcomes in Chronic Obstructive Pulmonary Disease? COPD J. Chronic Obstr. Pulm. Dis. 2015, 12, 14–18. [Google Scholar] [CrossRef]
- Keller, K.; Hobohm, L.; Münzel, T.; Ostad, M.A.; Espinola-Klein, C.; Lavie, C.J.; Konstantinides, S.; Lankeit, M. Survival Benefit of Obese Patients with Pulmonary Embolism. Mayo Clin. Proc. 2019, 94, 1960–1973. [Google Scholar] [CrossRef]
- Arbel, Y.; Fialkoff, C.; Kerner, A.; Kerner, M. Can Reduction in Infection and Mortality Rates from Coronavirus Be Explained by an Obesity Survival Paradox? An Analysis at the US Statewide Level. Int. J. Obes. 2020, 44, 2339–2342. [Google Scholar] [CrossRef]
- Hibbert, K.; Rice, M.; Malhotra, A. Obesity and ARDS. Chest 2012, 142, 785–790. [Google Scholar] [CrossRef]
- Wiedemann, H.P.; Clinic, C.; Wheeler, A.P.; Bernard, G.R.; University, V.; Taylor Thompson, B.; Hayden, D.; Hite, D. Comparison of Two Fluid-Management Strategies in Acute Lung Injury. N. Engl. J. Med. 2006, 354, 2564–2575. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Bustamante, A.; Repine, J.E. Adipose-Lung Cell Crosstalk in the Obesity-ARDS Paradox. J. Pulmon. Resp. Med. 2013, 3, 144. [Google Scholar] [CrossRef]
- Bladbjerg, E.M.; Stolberg, C.R.; Juhl, C.B. Effects of Obesity Surgery on Blood Coagulation and Fibrinolysis: A Literature Review. Thromb. Haemost. 2020, 120, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Parameswaran, K.; Todd, D.C.; Soth, M. Altered Respiratory Physiology in Obesity. Can. Respir. J. 2006, 13, 203–210. [Google Scholar] [CrossRef] [PubMed]
- McCormick, J.P.; Connaughton, J.; McDonnell, N. Anticoagulation in the Obese Patient with COVID-19-Associated Venous Thromboembolism. BMJ Case Rep. 2021, 14, 242675. [Google Scholar] [CrossRef]
- Drakos, P.; Volteas, P.; Naeem, Z.; Asencio, A.A.; Cleri, N.A.; Alkadaa, L.N.; Oganov, A.; Gammel, T.; Saadon, J.R.; Bannazadeh, M.; et al. Aggressive Anticoagulation May Decrease Mortality in Obese Critically Ill COVID-19 Patients. Obes. Surg. 2022, 32, 391–397. [Google Scholar] [CrossRef]
- Petrakis, D.; Margină, D.; Tsarouhas, K.; Tekos, F.; Stan, M.; Nikitovic, D.; Kouretas, D.; Spandidos, D.A.; Tsatsakis, A. Obesity—A Risk Factor for Increased COVID-19 Prevalence, Severity and Lethality (Review). Mol. Med. Rep. 2020, 22, 9–19. [Google Scholar] [CrossRef]
- Vieillard-Baron, A.; Rabiller, A.; Chergui, K.; Peyrouset, O.; Page, B.; Beauchet, A.; Jardin, F. Prone Position Improves Mechanics and Alveolar Ventilation in Acute Respiratory Distress Syndrome. Intensive Care Med. 2005, 31, 220–226. [Google Scholar] [CrossRef]
- Coman, A.C.; Todea, D.A.; Popa, F.; Radu, T.; Cadar, O.; Borzan, C. Multilateral Characterization of Masks and Tubes Surfaces in Contact with Respiratory System through Ventilation. J. Optoelectron. Adv. Mater. 2015, 17, 1563–1571. [Google Scholar]
- Ni, Y.N.; Yu, H.; Xu, H.; Li, W.-J.; Liang, B.-M.; Yang, L.; Liang, Z.A. High Visceral Adipose Tissue to Subcutaneous Adipose Tissue Ratio as a Predictor of Mortality in Acute Respiratory Distress Syndrome. Am. J. Med. Sci. 2019, 357, 213–222. [Google Scholar] [CrossRef]
- Battisti, S.; Pedone, C.; Napoli, N.; Russo, E.; Agnoletti, V.; Nigra, S.G.; Dengo, C.; Mughetti, M.; Conte, C.; Pozzilli, P.; et al. Computed Tomography Highlights Increased Visceral Adiposity Associated with Critical Illness in COVID-19. Diabetes Care 2020, 43, e129–e130. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Caruso, D.; Tuccinardi, D.; Risi, R.; Zerunian, M.; Polici, M.; Pucciarelli, F.; Tarallo, M.; Strigari, L.; Manfrini, S.; et al. Visceral Fat Shows the Strongest Association with the Need of Intensive Care in Patients with COVID-19. Metabolism 2020, 111, 154319. [Google Scholar] [CrossRef] [PubMed]
- Oga, E.A.; Eseyin, O.R. The Obesity Paradox and Heart Failure: A Systematic Review of a Decade of Evidence. J. Obes. 2016, 2016, 9040248. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.M.; Macedo-de la Concha, L.E.; Pantoja-Meléndez, C.A. Low-Grade Inflammation and Its Relation to Obesity and Chronic Degenerative Diseases. Rev. Méd. Hosp. Gen. México 2017, 80, 101–105. [Google Scholar] [CrossRef]
- Stapleton, R.D.; Dixon, A.E.; Parsons, P.E.; Ware, L.B.; Suratt, B.T. The Association between BMI and Plasma Cytokine Levels in Patients with Acute Lung Injury. Chest 2010, 138, 568–577. [Google Scholar] [CrossRef]
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of Obesity and Visceral Adiposity with Serum Concentrations of CRP, TNF-Alpha and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef]
- Kim, C.S.; Park, H.S.; Kawada, T.; Kim, J.H.; Lim, D.; Hubbard, N.E.; Kwon, B.S.; Erickson, K.L.; Yu, R. Circulating Levels of MCP-1 and IL-8 Are Elevated in Human Obese Subjects and Associated with Obesity-Related Parameters. Int. J. Obes. 2006, 30, 1347–1355. [Google Scholar] [CrossRef]
- Budin, C.E.; Alexescu, T.G.; Bordea, I.R.; Gherginescu, M.C.; Aluas, M.; Grigorescu, B.L.; Biro, L.; Buzoianu, A.D.; Nemes, R.M.; Tantu, M.M.; et al. Nicotine Addiction: Objective in Educational Programs for Smoking Prevention in Young People. Rev. Chim. 2019, 70, 2168–2172. [Google Scholar] [CrossRef]
- Lakkis, J.I.; Weir, M.R. Obesity and Kidney Disease. Prog. Cardiovasc. Dis. 2018, 61, 157–167. [Google Scholar] [CrossRef]
- Bornstein, S.R.; Dalan, R.; Hopkins, D.; Mingrone, G.; Boehm, B.O. Endocrine and Metabolic Link to Coronavirus Infection. Nat. Rev. Endocrinol. 2020, 16, 297–298. [Google Scholar] [CrossRef]
- Jia, X.; Yin, C.; Lu, S.; Chen, Y.; Liu, Q.; Bai, J.; Lu, Y. Two Things about COVID-19 Might Need Attention. Preprints 2020, 2020020315. [Google Scholar] [CrossRef]
- Wu, J.; Deng, W.; Li, S.; Yang, X. Advances in Research on ACE2 as a Receptor for 2019-NCoV. Cell. Mol. Life Sci. 2021, 78, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Lodigiani, C.; Iapichino, G.; Carenzo, L.; Cecconi, M.; Ferrazzi, P.; Sebastian, T.; Kucher, N.; Studt, J.D.; Sacco, C.; Alexia, B.; et al. Venous and Arterial Thromboembolic Complications in COVID-19 Patients Admitted to an Academic Hospital in Milan, Italy. Thromb. Res. 2020, 191, 9–14. [Google Scholar] [CrossRef]
- Moustafa, J.S.E.-S.; Jackson, A.U.; Brotman, S.M.; Guan, L.; Villicaña, S.; Roberts, A.L.; Zito, A.; Bonnycastle, L.; Erdos, M.R.; Narisu, N.; et al. ACE2 Expression in Adipose Tissue Is Associated with COVID-19 Cardio-Metabolic Risk Factors and Cell Type Composition. medRxiv 2020. [Google Scholar] [CrossRef]
- De Smet, D.; De Smet, K.; Herroelen, P.; Gryspeerdt, S.; Martens, G.A. Serum 25(OH)D Level on Hospital Admission Associated With COVID-19 Stage and Mortality. Am. J. Clin. Pathol. 2021, 155, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Quesada-Gomez, J.M.; Entrenas-Castillo, M.; Bouillon, R. Vitamin D Receptor Stimulation to Reduce Acute Respiratory Distress Syndrome (ARDS) in Patients with Coronavirus SARS-CoV-2 Infections: Revised Ms SBMB 2020_166. J. Steroid Biochem. Mol. Biol. 2020, 202, 105719. [Google Scholar] [CrossRef]
- Campi, I.; Gennari, L.; Merlotti, D.; Mingiano, C.; Frosali, A.; Giovanelli, L.; Torlasco, C.; Pengo, M.F.; Heilbron, F.; Soranna, D.; et al. Vitamin D and COVID-19 Severity and Related Mortality: A Prospective Study in Italy. BMC Infect. Dis. 2021, 21, 566. [Google Scholar] [CrossRef]
- Audibert, G.; Bannay, A.; Ziegler, O. Obesity Paradox in ICU? A Topic of Discussion, Not a Key Issue! Int. J. Obes. 2022, 46, 1248–1249. [Google Scholar] [CrossRef]
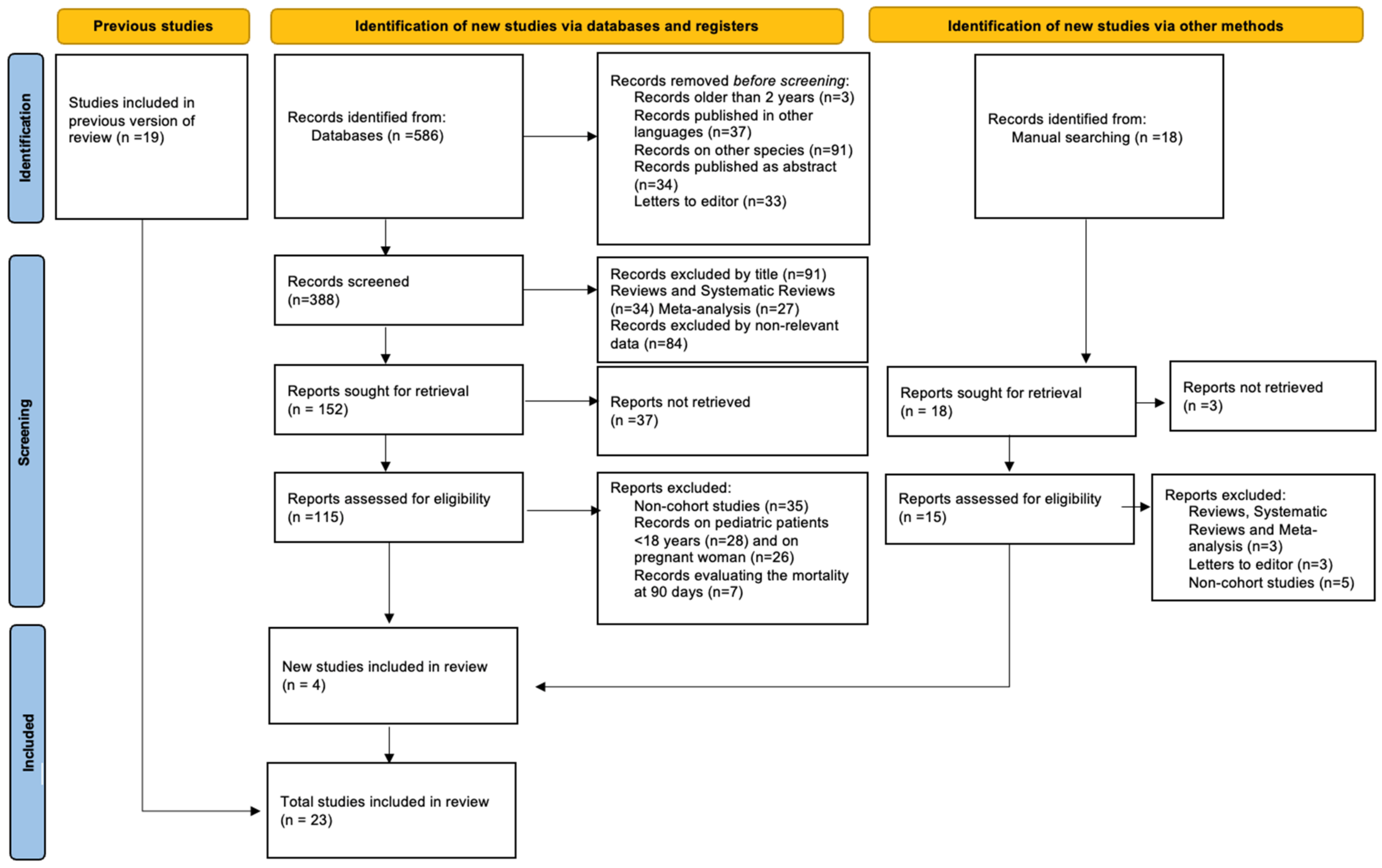

| No. | Authors | Study design No. of Participants Country | Diagnosis of COVID-19 | Measure of Obesity | Outcomes | Results/Conclusions |
|---|---|---|---|---|---|---|
| 1. | Cummings et al. [31] | Prospective observational cohort study (n = 257) in New York-USA | RT-PCR | Obesity (BMI > 30 kg/m2) Severe obesity (BMI > 40 kg/m2) | Mortality-in hospital | The results of the study did not identify morbid obesity with a BMI ≥ 40 as an independent risk factor for mortality in COVID-19 disease. |
| 2. | Busetto et al. [32] | Retrospective cohort study (n = 92) in Italy | RT-PCR | Normal weight (<25 kg/m2) Overweight (from 25 to <30 kg/m2) Obesity (≥30 kg/m2) | ICU admission IMV | A protective effect of obesity (obesity paradox) or other factors not related to BMI can explain the lack of worsening of the severity of the disease. |
| 3. | Goyal et al. [33] | Retrospective cohort study (n = 1687) in New York- USA | RT-PCR | Underweight (<18.5 kg/m2) Normal (18.5 to 24.9 kg/m2) Overweight (25.0 to 29.9 kg/m2) Mild to moderate obesity (30.0 to 39.9 kg/m2) Morbid obesity (≥40.0 kg/m2) | In-hospital mortality | The study concluded that obesity was not an independent risk factor for in-hospital mortality, providing insights regarding a plausible obesity paradox in COVID-19. |
| 4. | Biscarini et al. [34] | Retrospective cohort study (n = 427) in Italy | RT-PCR | Obesity (BMI ≥ 30 kg/m2) | ICU admission Mortality in ICU Mortality | The obesity does not interfere with survival rate or hospitalization length. |
| 5. | Dana et al. [35] | Prospective study (n = 226) in France | RT-PCR | Underweight (<18.5 kg/m2) Normal (18.5 to 24.9 kg/m2) Overweight (25.0 to 29.9 kg/m2) Mild to moderate obesity (30.0 to 39.9 kg/m2) Morbid obesity (≥40.0 kg/m2) | In-hospital mortality | Interestingly, the mortality rate was lower in those with moderate obesity and overweight compared to those with normal weight and severe obesity, challenging the paradox of obesity. |
| 6. | Kaeuffer et al. [32] | Prospective study (n = 1045) in France | RT-PCR | Normal weight (<25 kg/m2) Overweight (from 25 to <30 kg/m2) Obesity (≥30 kg/m2) | In-hospital morality | It has been demonstrated that the factors associated with an increased risk of death were the age, male sex, and immunosuppression and not the obesity. |
| 7. | Kim et al. [36] | Retrospective study (n = 10,861) in New York USA | RT-PCR | Underweight (<18.5 kg/m2) normal weight (18.5 to 24.9 kg/m2) Overweight (25.0 to 29.9 kg/m2) Class I (30.0 to 34.9 kg/m2) Class II (35 to 39.9 kg/m2) Class III (≥40.0 kg/m2) | IMV In-hospital morality | Once intubated there are no statistical differences in death rate between obese patients and normal weight individuals. |
| 8. | Yoshida et al. [37] | Retrospective study (n = 776) in New Orleans-USA | RT-PCR | Morbid obesity (BMI ≥ 40 kg/m2) | ICU admission IMV Mortality | No association between obesity and death was found in the non-Black group of patients. |
| 9. | Kim et al. [38] | Multi-site, geographically retrospective study (n = 2491) in USA | RT-PCR | Obesity BMI ≥ 30 kg/m2 Severe obesity BMI ≥ 40 kg/m2 | ICU admission In-hospital morality | Despite the higher prevalent of obesity in the study, there was found only an increased risk for ICU admission, but not for death. |
| 10. | Mankowski et al. [39] | Retrospective study (n = 309) in New Orleans-USA | RT-PCR | Obesity (BMI ≥ 30 kg/m2) | IMV In-Hospital Mortality | Even though obese patients required more invasive mechanical ventilation, there was no difference in risk of in-hospital mortality. |
| No. | Authors | Study Design No. of Participants Country | Diagnosis of COVID-19 | Measure of Obesity | Outcomes | Results/Conclusions |
|---|---|---|---|---|---|---|
| 1. | Halasz et al. [40] | Retrospective cohort study (n = 242) in Italy | RT-PCR | Underweight (<18.5 kg/m2) Normal weight (18.5–25 kg/m2) Overweight (25–30 kg/m2) Obese class I (30–35 kg/m2) Obese class II (35–40 kg/m2) Obese class III (>40 kg/m2) | Mortality | Severe obesity is associated with a greater mortality rate in individuals that were invasively ventilated, the study not being able to validate the theory of the obesity paradox. |
| 2. | Tartof et al. [41] | Retrospective cohort study (n = 6916) in California | RT-PCR | Underweight (less than 18.5 kg/m2) Normal (18.5 to 24 kg/m2) Overweight (25 to 29 kg/m2) Obese class I (30 to 34 kg/m2) Obese class II (35 to 39 kg/m2) Obese class III or extreme obesity (>40 kg/m2) | In-hospital morality | There is a relationship between BMI and death, as BMI increases, the risk for death also increases, with more than 4 times for the highest BMI. |
| 3. | Bello-Chavolla et al. [42] | Retrospective study (n = 51,633) in Mexico | RT-PCR | N/A | In-hospital morality | Obesity increases the risk of bad outcomes in COVID-19 disease, including mortality. |
| 4. | Arjun et al. [43] | Retrospective study (n = 142) in USA | RT-PCR | Nonobese (BMI < 30 kg/m2) Obese (BMI >30 kg/m2) | ICU admission Mortality-in hospital | The study did not support the theory of obesity paradox in COVID-19. |
| 5. | Czernichow et al. [44] | Prospective study (n = 5795) in France | RT-PCR | Underweight (<18.5 kg/m2) Normal weight (18.5–25 kg/m2) Overweight (25–30 kg/m2) Obese class I (30–35 kg/m2) Obese class II (35–40 kg/m2) Obese class III (>40 kg/m2) | Mortality | The study showed that mortality rate was higher in those with obesity. |
| 6. | Abumayyaleh et al. [45] | Retrospective cohort study (n = 3635) | RT-PCR | Obese patients (BMI > 30 kg/m2) | In-hospital morality | It was pointed out the absence of evidence for the obesity paradox in COVID-19 patients. |
| 7. | Schavemaker et al. [46] | Prospective study (n = 1122) Netherland | RT-PCR | Normal weight (18.5–24.9 kg/m2) Overweight (25–29.9 kg/m2) Obese (>30 kg/m2) | ICU In-hospital morality | The study was not able to validate the obesity survival paradox in COVID-19 infected patients. |
| 8. | Wolf et al. [47] | Retrospective study (n = 277) in Boston-USA | RT-PCR | Without obesity (BMI ≤ 29.9 kg/m2) Obesity class 1 (30 to 34.9 kg/m2) Obesity class 2 (35 to 39.9 kg/m2) Obesity class 3 (≥40 kg/m2) | ICU admission Survival | The obesity was not significant associated with clinical outcomes, the study not being able to demonstrate an obesity survival paradox in COVID-19 infected patients. |
| 9. | Carneiro RAVD et al. [48] | Retrospective study conducted in Brazil | RT-PCR | Overweight (BMI ≥ 25 kg/m2) Obesity (BMI ≥ 30 kg/m2) | Mortality | There is a positive corelation between obesity and overall mortality. |
| 10. | Foulkes et al. [49] | Retrospective study conducted in USA (n = 3828) | RT-PCR | Obesity (BMI >30 kg/m2) | IMV Mortality | Obesity increases the systemic inflammation response COVID-19 patients and leads to severe outcomes. |
| 11. | Motaib et al. [50] | Retrospective study in Morocco (n = 107) | RT-PCR | Obesity (BMI ≥ 30 kg/m2) | ICU admission | Obesity is independently associated with an increased rate of ICU admission. |
| 12. | Sidhu et al. [51] | Retrospective study (n = 425) in New Orleans, USA | RT-PCR | Obesity (BMI >30 kg/m2) Severe obesity (≥35 kg/m2 and < 40 kg/ m2) Morbid obesity (>40.0 kg/m2) | Mortality | COVID-19 obese patients with at least one obesity related condition have an increased risk of death. |
| 13. | Yates et al. [52] | Retrospective study (n = 412,596) in UK | RT-PCR | Normal weight (18.5 to 24.9 kg/m2) Overweight (25.0 to 29.9 kg/m2) Obesity (BMI ≥ 30 kg/m2) | Mortality | Obesity is associate with a higher rate of Mortality |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vulturar, D.-M.; Crivii, C.-B.; Orăsan, O.H.; Palade, E.; Buzoianu, A.-D.; Zehan, I.G.; Todea, D.A. Obesity Impact on SARS-CoV-2 Infection: Pros and Cons “Obesity Paradox”—A Systematic Review. J. Clin. Med. 2022, 11, 3844. https://doi.org/10.3390/jcm11133844
Vulturar D-M, Crivii C-B, Orăsan OH, Palade E, Buzoianu A-D, Zehan IG, Todea DA. Obesity Impact on SARS-CoV-2 Infection: Pros and Cons “Obesity Paradox”—A Systematic Review. Journal of Clinical Medicine. 2022; 11(13):3844. https://doi.org/10.3390/jcm11133844
Chicago/Turabian StyleVulturar, Damiana-Maria, Carmen-Bianca Crivii, Olga Hilda Orăsan, Emanuel Palade, Anca-Dana Buzoianu, Iulia Georgiana Zehan, and Doina Adina Todea. 2022. "Obesity Impact on SARS-CoV-2 Infection: Pros and Cons “Obesity Paradox”—A Systematic Review" Journal of Clinical Medicine 11, no. 13: 3844. https://doi.org/10.3390/jcm11133844
APA StyleVulturar, D.-M., Crivii, C.-B., Orăsan, O. H., Palade, E., Buzoianu, A.-D., Zehan, I. G., & Todea, D. A. (2022). Obesity Impact on SARS-CoV-2 Infection: Pros and Cons “Obesity Paradox”—A Systematic Review. Journal of Clinical Medicine, 11(13), 3844. https://doi.org/10.3390/jcm11133844






