Prophylactic Hydroxyurea Treatment Is Associated with Improved Cerebral Hemodynamics as a Surrogate Marker of Stroke Risk in Sickle Cell Disease: A Retrospective Comparative Analysis
Abstract
:1. Introduction
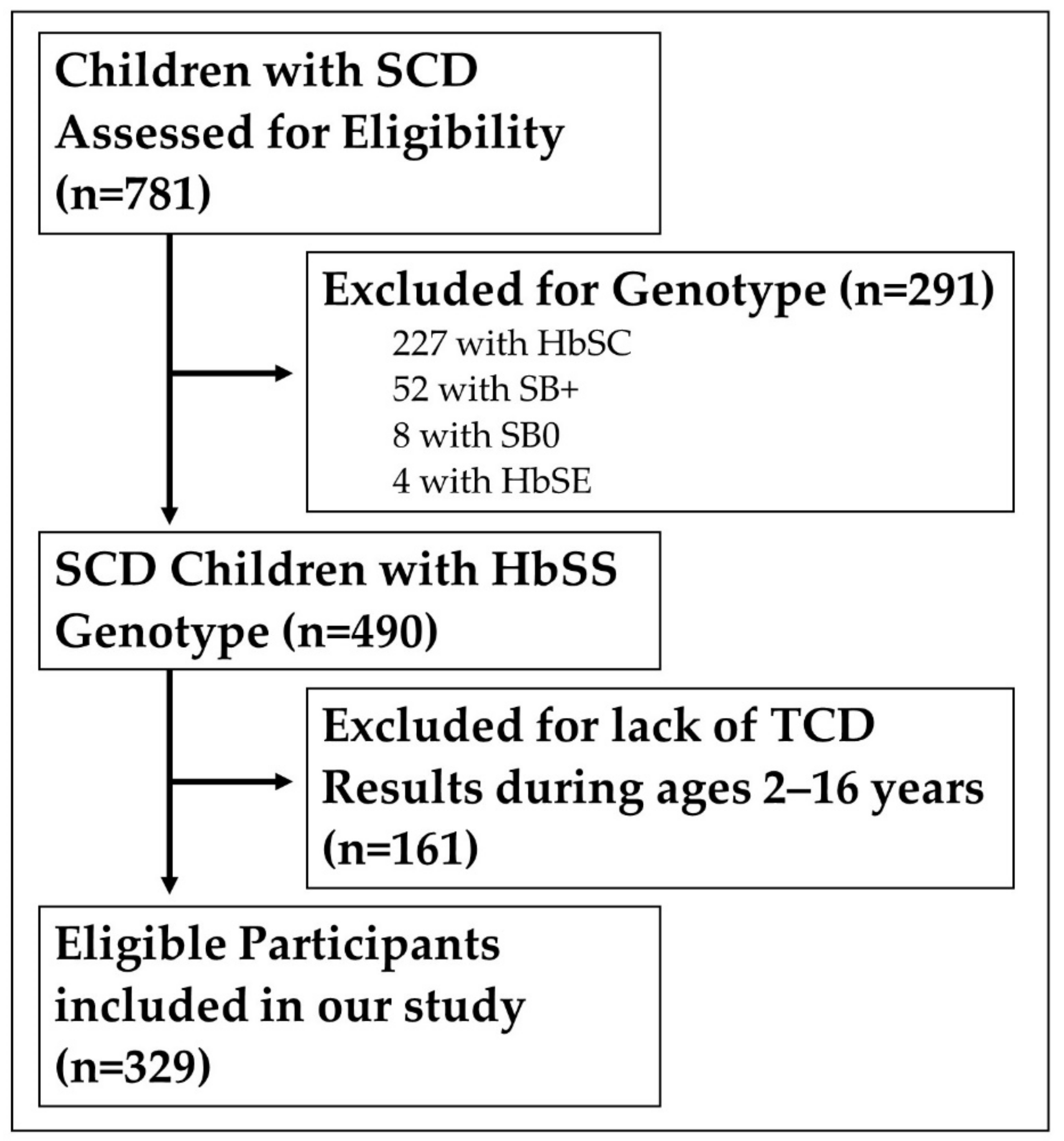
2. Materials and Methods
2.1. Study Type: We Conducted a Retrospective Cohort Chart Review
2.2. Data Analysis
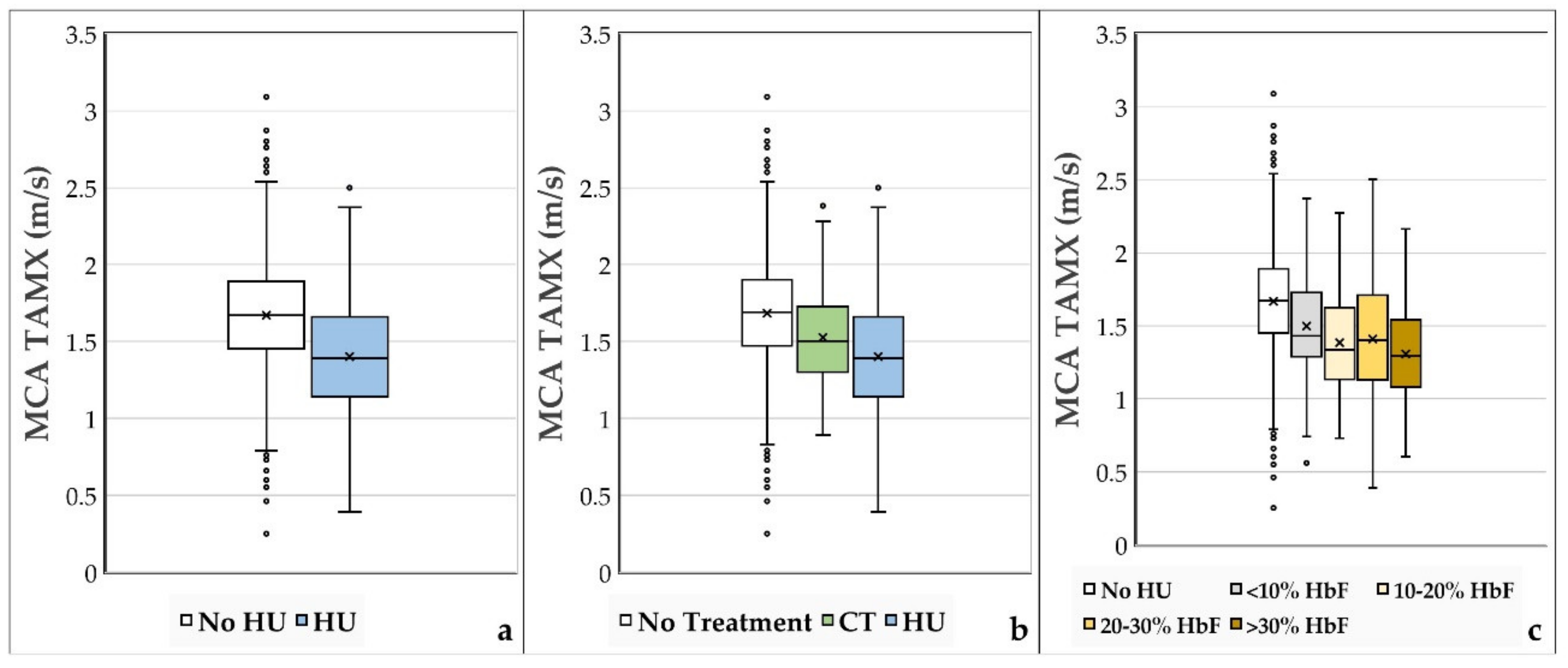
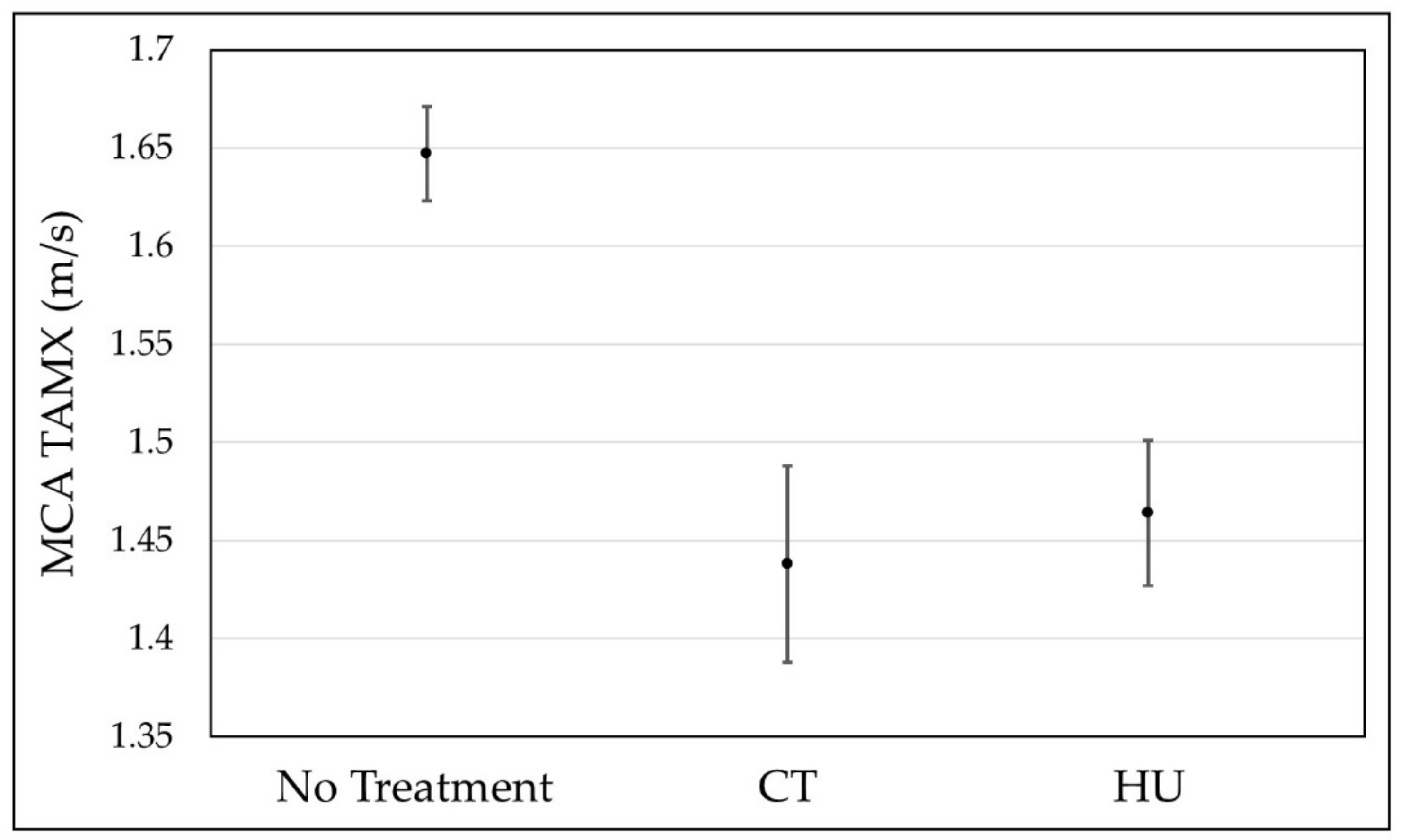
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CDC. Data & Statistics on Sickle Cell Disease|CDC. Available online: https://www.cdc.gov/ncbddd/sicklecell/data.html (accessed on 14 December 2020).
- Jordan, L.C.; Casella, J.F.; Debaun, M.R. Prospects for primary stroke prevention in children with sickle cell anaemia. Br. J. Haematol. 2012, 157, 14–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, R.J.; McKie, V.C.; Carl, E.M.; Nichols, F.T.; Perry, R.; Brock, K.; McKie, K.; Figueroa, R.; Litaker, M.; Weiner, S.; et al. Long-term stroke risk in children with sickle cell disease screened with transcranial Doppler. Ann. Neurol. 1997, 42, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.J.; Brambilla, D.J.; Granger, S.; Vichinsky, E.; Abboud, M.R.; Pegelow, C.H.; Woods, G.; Rohde, E.M.; Nichols, F.T.; Jones, A.; et al. Stroke and conversion to high risk in children screened with transcranial Doppler ultrasound during the STOP study. Blood 2004, 103, 3689–3694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarnaik, S. Periodic transfusions for sickle cell anemia and CNS Infarction. Arch. Pediatr. Adolesc. Med. 1979, 133, 1254. [Google Scholar] [CrossRef] [PubMed]
- Russell, M.O.; Goldberg, H.I.; Reis, L.; Friedman, S.; Slater, R.; Reivich, M.; Schwartz, E. Transfusion therapy for cerebrovascular abnormalities in sickle cell disease. J. Pediatr. 1976, 88, 382–387. [Google Scholar] [CrossRef]
- Adams, R.J.; Mckie, V.C.; Brambilla, D.; Carl, E.; Gallagher, D.; Nichols, F.T.; Roach, S.; Abboud, M.; Berman, B.; Driscoll, C.; et al. Stroke Prevention Trial in Sickle Cell Anemia. Control. Clin. Trials 1998, 19, 110–129. [Google Scholar] [CrossRef]
- National Heart, Lung, and Blood Institute; National Library of Medicine (U.S.). Clinical Alert: Periodic Transfusions Lower Stroke Risk in Children with Sickle Cell Anemia. 1997. Available online: https://www.nlm.nih.gov/databases/alerts/sickle97.html (accessed on 19 December 2020).
- Ohene-Frempong, K.; Weiner, S.J.; Sleeper, L.A.; Miller, S.T.; Embury, S.; Moohr, J.M.; Wethers, D.L.; Pegelow, C.H.; Gill, F.M.; The Cooperative Study of Sickle Cell Disease. Cerebrovascular accidents in sickle cell disease: Rates and risk factors. Blood 1998, 91, 288–294. [Google Scholar] [PubMed]
- Bernaudin, F.; Verlhac, S.; Arnaud, C.; Kamdem, A.; Chevret, S.; Hau, I.; Coïc, L.; Leveillé, E.; Lemarchand, E.; Lesprit, E.; et al. Impact of early transcranial Doppler screening and intensive therapy on cerebral vasculopathy outcome in a newborn sickle cell anemia cohort. Blood 2011, 117, 1130–1140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, R.J.; Brambilla, D. Optimizing Primary Stroke Prevention in Sickle Cell Anemia (STOP 2) Trial Investigators. Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. N. Engl. J. Med. 2005, 353, 2769–2778. [Google Scholar] [PubMed]
- Agrawal, R.K.; Patel, R.K.; Shah, V.; Nainiwal, L.; Trivedi, B. Hydroxyurea in Sickle Cell Disease: Drug Review. Indian J. Hematol. Blood Transfus. 2013, 30, 91–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gulbis, B.; Haberman, D.; Dufour, D.; Christophe, C.; Vermylen, C.; Kagambega, F.; Corazza, F.; Devalck, C.; Dresse, M.-F.; Hunninck, K.; et al. Hydroxyurea for sickle cell disease in children and for prevention of cerebrovascular events: The Belgian experience. Blood 2005, 105, 2685–2690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kratovil, T.; Bulas, D.; Driscoll, M.C.; Speller-Brown, B.; McCarter, R.; Minniti, C.P. Hydroxyurea therapy lowers TCD velocities in children with sickle cell disease. Pediatr. Blood Cancer 2006, 47, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, S.A.; Schultz, W.H.; Burgett, S.; Mortier, N.A.; Ware, R.E. Hydroxyurea therapy lowers transcranial Doppler flow velocities in children with sickle cell anemia. Blood 2007, 110, 1043–1047. [Google Scholar] [CrossRef] [PubMed]
- Lagunju, I.; Brown, B.J.; Oyinlade, A.O.; Asinobi, A.; Ibeh, J.; Esione, A.; Sodeinde, O.O. Annual stroke incidence in Nigerian children with sickle cell disease and elevated TCD velocities treated with hydroxyurea. Pediatr. Blood Cancer 2019, 66, e27252. [Google Scholar] [CrossRef] [PubMed]
- Rankine-Mullings, A.; Reid, M.E.; Soares, D.; Taylor-Bryan, C.; Wisdom-Phipps, M.; Aldred, K.; Latham, T.; Knight-Madden, J.; King, L.; Badaloo, A.; et al. Hydroxyurea therapy to prevent incident stroke among children with sickle cell anaemia in Jamaica: The extend trial. Blood 2019, 134 (Suppl. 1), 2269. [Google Scholar] [CrossRef]
- Ware, R.E.; Davis, B.R.; Schultz, W.H.; Brown, R.C.; Aygun, B.; Sarnaik, S.; Odame, I.; Fuh, B.; George, A.; Owen, W.; et al. Hydroxycarbamide versus chronic transfusion for maintenance of transcranial doppler flow velocities in children with sickle cell anaemia-TCD with Transfusions Changing to Hydroxyurea (TWiTCH): A multicentre, open-label, phase 3, non-inferiority trial. Lancet 2016, 387, 661–670. [Google Scholar] [CrossRef] [Green Version]
- Abdullahi, S.U.; Jibir, B.W.; Bello-Manga, H.; Gambo, S.; Inuwa, H.; Tijjani, A.G.; Idris, N.; Galadanci, A.; Hikima, M.S.; Galadanci, N.; et al. Hydroxyurea for primary stroke prevention in children with sickle cell anaemia in Nigeria (SPRING): A double-blind, multicentre, randomised, phase 3 trial. Lancet Haematol. 2022, 9, 26–37. [Google Scholar] [CrossRef]
- Weisman, J.K.; Diamond, C.E.; Kappa, S.; Nickel, R.S. Transcranial Doppler Screening Adherence among Children with Sickle Cell Anemia Seen in the Emergency Department. J. Pediatr. 2020, 217, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.C.; Ware, R.E.; Miller, S.T.; Iyer, R.V.; Casella, J.F.; Minniti, C.P.; Rana, S.; Thornburg, C.D.; Rogers, Z.R.; Kalpatthi, R.V.; et al. Hydroxycarbamide in very young children with sickle-cell anaemia: A multicentre, randomized, controlled trial (BABY HUG). Lancet 2011, 377, 1663–1672. [Google Scholar] [CrossRef] [Green Version]
| Hydroxyurea (n = 111; 34%) | No Hydroxyurea (n = 218; 66%) | |
|---|---|---|
| Sex | ||
| Female | 54 (49%) | 119 (55%) |
| Male | 57 (51%) | 99 (45%) |
| Age at start of HU, years | 7.16 +/− 4.11 | N/A |
| Duration of HU Treatment at time of TCD, years | 2.53 +/− 2.15 | N/A |
| Age at time of TCD, years | 8.75 +/− 3.65 | 7.16 +/− 3.61 |
| HU Dosage (mg/kg/day) | 24.7 +/− 5.3 | N/A |
| MCA TAMX (m/s) | 1.40 +/− 0.34 | 1.67 +/− 0.33 |
| History of prior CT therapy | 20 (18%) | 92 (42%) |
| Abnormal TAMX (>2 m/s) during study, requiring transfusion | 5 (4%) | 34 (16%) |
| No Treatment | CT | Hydroxyurea | Total | |
|---|---|---|---|---|
| #Patients | 167 | 70 | 92 | 329 |
| Number of TCDs | 1094 | 124 | 252 | 1470 |
| Mean Number of TCDs/Patient | 6.55 | 1.77 | 2.74 | 4.47 |
| Mean Age at TCD | 6.92 | 9.67 | 8.75 | 7.47 |
| Mean MCA TAMX | 1.69 | 1.54 | 1.37 | 1.62 |
| Mean TAMX | SE | DF | Lower 95% | Upper 95% | |
|---|---|---|---|---|---|
| No Treatment | 1.648 | 0.012 | 689 | 1.624 | 1.672 |
| CT | 1.437 | 0.025 | 2590 | 1.388 | 1.487 |
| HU | 1.464 | 0.019 | 1681 | 1.427 | 1.501 |
| Treatment | Normal (<1.7 m/s) MCA TAMX | Conditional (1.7–2.0 m/s) MCA TAMX | Abnormal (>2.0 m/s) MCA TAMX | Total |
|---|---|---|---|---|
| HU | 408 | 79 | 17 | 504 |
| CT | 172 | 54 | 22 | 248 |
| No Treatment | 1099 | 749 | 340 | 2188 |
| Total | 1679 | 882 | 379 | 2940 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peine, B.R.; Callaghan, M.U.; Callaghan, J.H.; Glaros, A.K. Prophylactic Hydroxyurea Treatment Is Associated with Improved Cerebral Hemodynamics as a Surrogate Marker of Stroke Risk in Sickle Cell Disease: A Retrospective Comparative Analysis. J. Clin. Med. 2022, 11, 3491. https://doi.org/10.3390/jcm11123491
Peine BR, Callaghan MU, Callaghan JH, Glaros AK. Prophylactic Hydroxyurea Treatment Is Associated with Improved Cerebral Hemodynamics as a Surrogate Marker of Stroke Risk in Sickle Cell Disease: A Retrospective Comparative Analysis. Journal of Clinical Medicine. 2022; 11(12):3491. https://doi.org/10.3390/jcm11123491
Chicago/Turabian StylePeine, Brian R., Michael U. Callaghan, Joseph H. Callaghan, and Alexander K. Glaros. 2022. "Prophylactic Hydroxyurea Treatment Is Associated with Improved Cerebral Hemodynamics as a Surrogate Marker of Stroke Risk in Sickle Cell Disease: A Retrospective Comparative Analysis" Journal of Clinical Medicine 11, no. 12: 3491. https://doi.org/10.3390/jcm11123491
APA StylePeine, B. R., Callaghan, M. U., Callaghan, J. H., & Glaros, A. K. (2022). Prophylactic Hydroxyurea Treatment Is Associated with Improved Cerebral Hemodynamics as a Surrogate Marker of Stroke Risk in Sickle Cell Disease: A Retrospective Comparative Analysis. Journal of Clinical Medicine, 11(12), 3491. https://doi.org/10.3390/jcm11123491







