Assessment of Serum Urea, Creatinine and Uric Acid in Oral Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients’ Samples
2.2. Data Collection
2.3. Statistical Analysis
3. Results
3.1. Patients’ Group Characteristics
3.2. Comparison of Serum Urea, Uric Acid and Creatinine Levels between OSCC and Control Groups
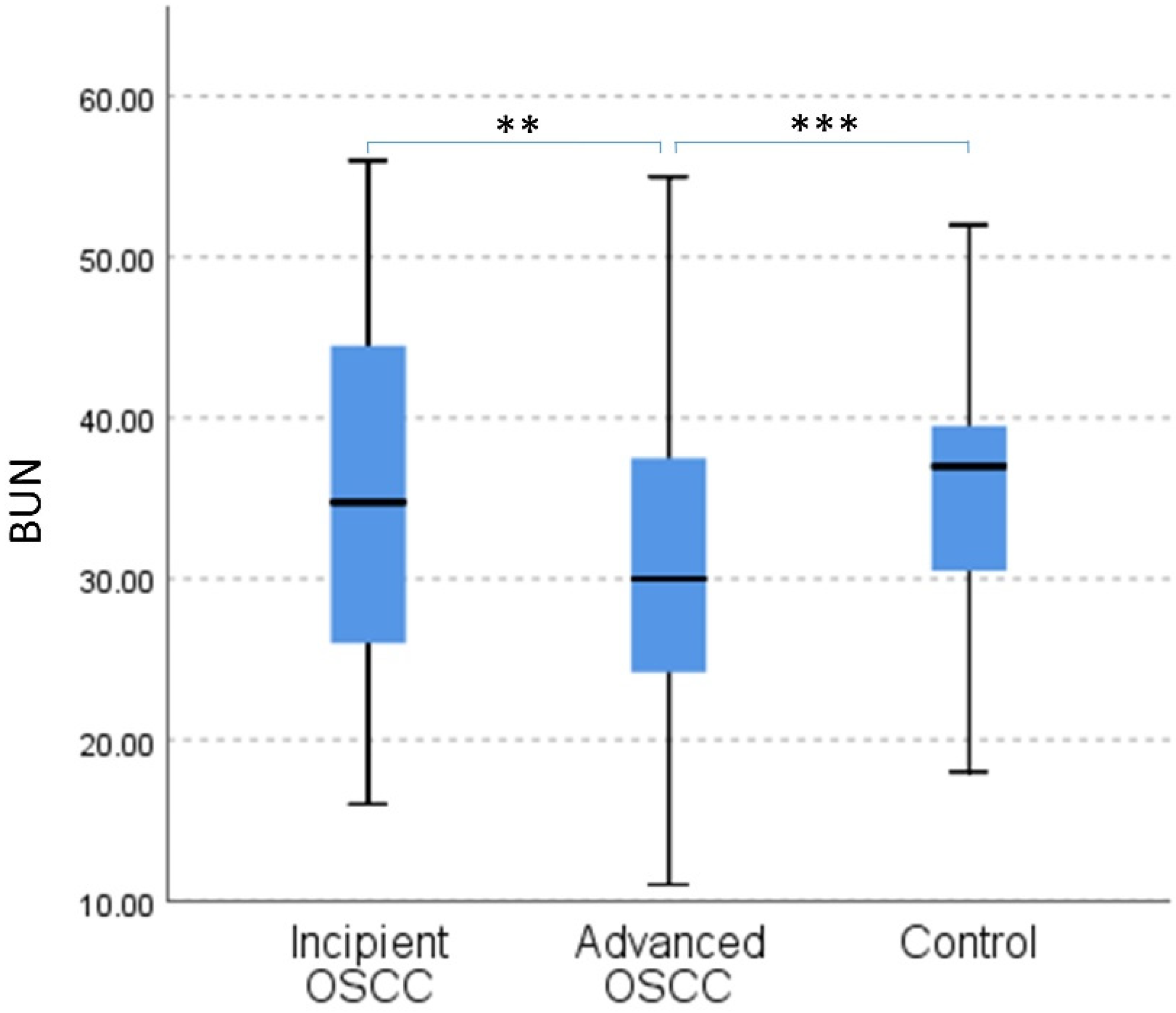
3.3. Analysis of Serum Urea in Relation to Disease Progression in OSCC
3.4. Correlation and Multivariate Analysis between Serum Urea and Clinico-Pathological Characteristics in OSCC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Estimated Number of New Cases in 2020, Worldwide, both Sexes, All Ages (excl. NMSC), International Agency for Research on Cance. Available online: https://gco.iarc.fr/today/online-analysis-table?v=2020&mode=cancer&mode_population=continents&population=900&populations=900&key=asr&sex=0&cancer=39&type=0&statistic=5&prevalence=0&population_group=0&ages_group%5B%5D=0&ages_group%5B%5D=17&group_cancer=1&i (accessed on 3 September 2021).
- Thompson-Harvey, A.; Yetukuri, M.; Ba, A.R.H.; Mph, M.C.S.; Boakye, E.A.; Varvares, M.A.; Osazuwa-Peters, N. Rising incidence of late-stage head and neck cancer in the United States. Cancer 2020, 126, 1090–1101. [Google Scholar] [CrossRef]
- Jafari, A.; Najafi, S.; Moradi, F.; Kharazifard, M.; Khami, M.R. Delay in the Diagnosis and Treatment of Oral Cancer. J. Dent. 2013, 14, 146–150. [Google Scholar]
- Meier, J.K.; Schuderer, J.G.; Zeman, F.; Klingelhöffer, C.; Hullmann, M.; Spanier, G.; Reichert, T.E.; Ettl, T. Health-related quality of life: A retrospective study on local vs. microvascular reconstruction in patients with oral cancer. BMC Oral Health 2019, 19, 62. [Google Scholar] [CrossRef]
- Caruntu, A.; Moraru, L.; Comaneanu, R.M.; Costache, R.S.; Farcasiu, T.A.; Scheau, C.; Costache, D.O.; Caruntu, C. A combination of two regional flaps for the complex reconstruction after head and neck cancer surgery as an alternative approach during the COVID-19 pandemic: A case report. Exp. Ther. Med. 2022, 23, 364. [Google Scholar] [CrossRef]
- Nocini, R.; Lippi, G.; Mattiuzzi, C. Biological and epidemiologic updates on lip and oral cavity cancers. Ann. Cancer Epidemiol. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Maso, L.D.; Panato, C.; Tavilla, A.; Guzzinati, S.; Serraino, D.; Mallone, S.; Botta, L.; Boussari, O.; Capocaccia, R.; Colonna, M.; et al. Cancer cure for 32 cancer types: Results from the EUROCARE-5 study. Int. J. Epidemiol. 2020, 49, 1517–1525. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Kim, J.H. Increasing incidence and improving survival of oral tongue squamous cell carcinoma. Sci. Rep. 2020, 10, 7877. [Google Scholar] [CrossRef]
- Stathopoulos, P.; Smith, W.P. Analysis of Survival Rates Following Primary Surgery of 178 Consecutive Patients with Oral Cancer in a Large District General Hospital. J. Maxillofac. Oral Surg. 2017, 16, 158–163. [Google Scholar] [CrossRef]
- Sayáns, M.P.; Petronacci, C.M.C.; Pouso, A.I.L.; Iruegas, E.P.; Carrión, A.B.; Peñaranda, J.M.S.; García, A.G. Comprehensive Genomic Review of TCGA Head and Neck Squamous Cell Carcinomas (HNSCC). J. Clin. Med. 2019, 8, 1896. [Google Scholar] [CrossRef]
- Caruntu, A.; Moraru, L.; Lupu, M.; Ciubotaru, D.; Dumitrescu, M.A.; Eftimie, L.; Hertzog, R.; Zurac, S.; Caruntu, C.; Voinea, O.C. Assessment of Histological Features in Squamous Cell Carcinoma Involving Head and Neck Skin and Mucosa. J. Clin. Med. 2021, 10, 2343. [Google Scholar] [CrossRef]
- Caruntu, A.; Moraru, L.; Surcel, M.; Munteanu, A.; Costache, D.O.; Tanase, C.; Constantin, C.; Scheau, C.; Neagu, M.; Caruntu, C. Persistent Changes of Peripheral Blood Lymphocyte Subsets in Patients with Oral Squamous Cell Carcinoma. Healthcare 2022, 10, 342. [Google Scholar] [CrossRef]
- Caruntu, A.; Moraru, L.; Surcel, M.; Munteanu, A.; Tanase, C.; Constantin, C.; Zurac, S.; Caruntu, C.; Neagu, M. Assessment of Immune Cell Populations in Tumor Tissue and Peripheral Blood Samples from Head and Neck Squamous Cell Carcinoma Patients. Anal. Cell. Pathol. 2021, 2021, 2328218. [Google Scholar] [CrossRef]
- Mohan, S.P.; Bhaskaran, M.K.; George, A.L.; Thirutheri, A.; Somasundaran, M.; Pavithran, A. Immunotherapy in Oral Cancer. J. Pharm. Bioallied Sci. 2019, 11, S107–S111. [Google Scholar] [CrossRef]
- Cohen, E.E.W.; Bell, R.B.; Bifulco, C.B.; Burtness, B.; Gillison, M.L.; Harrington, K.J.; Le, Q.-T.; Lee, N.Y.; Leidner, R.; Lewis, R.L.; et al. The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of squamous cell carcinoma of the head and neck (HNSCC). J. Immunother. Cancer 2019, 7, 184. [Google Scholar] [CrossRef]
- Jiang, X.; Ye, J.; Dong, Z.; Hu, S.; Xiao, M. Novel genetic alterations and their impact on target therapy response in head and neck squamous cell carcinoma. Cancer Manag. Res. 2019, 11, 1321–1336. [Google Scholar] [CrossRef]
- Caruntu, A.; Moraru, L.; Lupu, M.; Taubner, L.; Caruntu, C.; Tanase, C. The Hidden Treasures of Preoperative Blood Assessment in Oral Cancer: A Potential Source of Biomarkers. Cancers 2021, 13, 4475. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Chandel, N.S. Fundamentals of cancer metabolism. Sci. Adv. 2016, 2, e1600200. [Google Scholar] [CrossRef]
- Schiliro, C.; Firestein, B.L. Mechanisms of Metabolic Reprogramming in Cancer Cells Supporting Enhanced Growth and Proliferation. Cells 2021, 10, 1056. [Google Scholar] [CrossRef]
- Spinelli, J.B.; Yoon, H.; Ringel, A.E.; Jeanfavre, S.; Clish, C.B.; Haigis, M.C. Metabolic recycling of ammonia via glutamate dehydrogenase supports breast cancer biomass. Science 2017, 358, 941–946. [Google Scholar] [CrossRef]
- Kim, J.; Hu, Z.; Cai, L.; Li, K.; Choi, E.; Faubert, B.; Bezwada, D.; Rodriguez-Canales, J.; Villalobos, P.; Lin, Y.-F.; et al. CPS1 maintains pyrimidine pools and DNA synthesis in KRAS/LKB1-mutant lung cancer cells. Nature 2017, 546, 168–172. [Google Scholar] [CrossRef]
- Yin, J.; Ren, W.; Huang, X.; Deng, J.; Li, T.; Yin, Y. Potential Mechanisms Connecting Purine Metabolism and Cancer Therapy. Front. Immunol. 2018, 9, 1697. [Google Scholar] [CrossRef]
- Lee, J.S.; Adler, L.; Karathia, H.; Carmel, N.; Rabinovich, S.; Auslander, N.; Keshet, R.; Stettner, N.; Silberman, A.; Agemy, L.; et al. Urea Cycle Dysregulation Generates Clinically Relevant Genomic and Biochemical Signatures. Cell 2018, 174, 1559–1570.e22. [Google Scholar] [CrossRef]
- Fini, M.A.; Elias, A.; Johnson, R.J.; Wright, R.M. Contribution of uric acid to cancer risk, recurrence, and mortality. Clin. Transl. Med. 2012, 1, 16. [Google Scholar] [CrossRef]
- Kobylecki, C.J.; Afzal, S.; Nordestgaard, B.G. Plasma Urate, Cancer Incidence, and All-Cause Mortality: A Mendelian Randomization Study. Clin. Chem. 2017, 63, 1151–1160. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.; Qu, Z.; Yang, Z.; Jia, X.; Lin, Y.; He, Q.; Zhang, L.; Luo, Y. Causal Associations between Serum Urea and Cancer: A Mendelian Randomization Study. Genes 2021, 12, 498. [Google Scholar] [CrossRef]
- Bai, C.; Wang, H.; Dong, D.; Li, T.; Yu, Z.; Guo, J.; Zhou, W.; Li, D.; Yan, R.; Wang, L.; et al. Urea as a By-Product of Ammonia Metabolism Can Be a Potential Serum Biomarker of Hepatocellular Carcinoma. Front. Cell Dev. Biol. 2021, 9, 677. [Google Scholar] [CrossRef]
- Schwameis, R.; Postl, M.; Bekos, C.; Hefler, L.; Reinthaller, A.; Seebacher, V.; Grimm, C.; Polterauer, S.; Helmy-Bader, S. Prognostic value of serum creatine level in patients with vulvar cancer. Sci. Rep. 2019, 9, 11129. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, Q.; Ruan, G.-T.; Tang, M.; Zhang, X.; Song, M.-M.; Zhang, X.-W.; Zhang, K.-P.; Ge, Y.-Z.; Shi, H.-P. Association between Serum Creatinine Concentrations and Overall Survival in Patients with Colorectal Cancer: A Multi-Center Cohort Study. Front. Oncol. 2021, 11, 4110. [Google Scholar] [CrossRef]
- LaFleur, J.; Hefler-Frischmuth, K.; Grimm, C.; Schwameis, R.; Gensthaler, L.; Reiser, E.; Hefler, L.A. Prognostic Value of Serum Creatinine Levels in Patients with Epithelial Ovarian Cancer. Anticancer Res. 2018, 38, 5127–5130. [Google Scholar] [CrossRef]
- Dasarathy, S.; Mookerjee, R.; Rackayova, V.; Thrane, V.R.; Vairappan, B.; Ott, P.; Rose, C.F. Ammonia toxicity: From head to toe? Metab. Brain Dis. 2016, 32, 529–538. [Google Scholar] [CrossRef]
- Kornberg, H. Krebs and his trinity of cycles. Nat. Rev. Mol. Cell Biol. 2000, 1, 225–228. [Google Scholar] [CrossRef]
- Morris, S.M. Regulation of enzymes of the urea cycle and arginine metabolism. Annu. Rev. Nutr. 2002, 22, 87–105. [Google Scholar] [CrossRef]
- Hajaj, E.; Sciacovelli, M.; Frezza, C.; Erez, A. The context-specific roles of urea cycle enzymes in tumorigenesis. Mol. Cell 2021, 81, 3749–3759. [Google Scholar] [CrossRef]
- Wang, H.; Ran, J.; Jiang, T. Urea. In Urea Transporters; Yang, B., Sands, J.M., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 7–29. [Google Scholar] [CrossRef]
- Keshet, R.; Szlosarek, P.; Carracedo, A.; Erez, A. Rewiring urea cycle metabolism in cancer to support anabolism. Nat. Cancer 2018, 18, 634–645. [Google Scholar] [CrossRef]
- Lee, Y.-Y.; Li, C.-F.; Lin, C.-Y.; Lee, S.-W.; Sheu, M.-J.; Lin, L.-C.; Chen, T.-J.; Wu, T.-F.; Hsing, C.-H. Overexpression of CPS1 is an independent negative prognosticator in rectal cancers receiving concurrent chemoradiotherapy. Tumor Biol. 2014, 35, 11097–11105. [Google Scholar] [CrossRef]
- Ohshima, M.; Sugahara, K.; Kasahara, K.; Katakura, A. Metabolomic analysis of the saliva of Japanese patients with oral squamous cell carcinoma. Oncol. Rep. 2017, 37, 2727–2734. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, Y.; Chen, Z.; Guo, X.; Lv, Y. Age- and sex-specific reference intervals for blood urea nitrogen in Chinese general population. Sci. Rep. 2021, 11, 10058. [Google Scholar] [CrossRef]
- Musch, W.; Verfaillie, L.; Decaux, G. Age-Related Increase in Plasma Urea Level and Decrease in Fractional Urea Excretion: Clinical Application in the Syndrome of Inappropriate Secretion of Antidiuretic Hormone. Clin. J. Am. Soc. Nephrol. 2006, 1, 909–914. [Google Scholar] [CrossRef]
- Bowker, L.K.; Briggs, R.S.; Gallagher, P.J.; Robertson, D.R. Raised blood urea in the elderly: A clinical and pathological study. Postgrad. Med. J. 1992, 68, 174–179. [Google Scholar] [CrossRef][Green Version]
- Wu, C.-C.; Wang, H.-E.; Liu, Y.-C.; Zheng, C.-M.; Chu, P.; Lu, K.-C.; Chu, C.-M.; Chang, Y.-T. Sleeping, Smoking, and Kidney Diseases: Evidence from the NHANES 2017–2018. Front. Med. 2021, 1671. [Google Scholar] [CrossRef]
- Xu, T.; Holzapfel, C.; Dong, X.; Bader, E.; Yu, Z.; Prehn, C.; Perstorfer, K.; Jaremek, M.; Roemisch-Margl, W.; Rathmann, W.; et al. Effects of smoking and smoking cessation on human serum metabolite profile: Results from the KORA cohort study. BMC Med. 2013, 11, 60. [Google Scholar] [CrossRef]
- Kesteloot, H.E.; Joossens, J.V. Relationship between dietary protein intake and serum urea, uric acid and creatinine, and 24-hour urinary creatinine excretion: The BIRNH Study. J. Am. Coll. Nutr. 1993, 12, 42–46. [Google Scholar] [CrossRef]
- Peter, F.; Wittekindt, C.; Finkensieper, M.; Kiehntopf, M.; Guntinas-Lichius, O. Prognostic impact of pretherapeutic laboratory values in head and neck cancer patients. J. Cancer Res. Clin. Oncol. 2013, 139, 171–178. [Google Scholar] [CrossRef]
- De Oliveira Faria, S.; Howell, D.; Kulcsar, M.A.V.; Eluf-Neto, J. Nutritional outcomes in head and neck cancer patients: Is intensive nutritional care worth it? Cancer Treat. Res. Commun. 2020, 25, 100233. [Google Scholar] [CrossRef]
- Bassett, M.R.; Dobie, R.A. Patterns of Nutritional Deficiency in Head and Neck Cancer. Otolaryngol. Neck Surg. 1983, 91, 119–125. [Google Scholar] [CrossRef]
- Bron, L.; Jandus, C.; Andrejevic-Blant, S.; Speiser, D.E.; Monnier, P.; Romero, P.; Rivals, J.-P. Prognostic value of arginase-II expression and regulatory T-cell infiltration in head and neck squamous cell carcinoma. Int. J. Cancer 2013, 132, E85–E93. [Google Scholar] [CrossRef]
- Gannon, P.O.; Godin-Ethier, J.; Hassler, M.; Delvoye, N.; Aversa, M.; Poisson, A.O.; Peant, B.; Alam Fahmy, M.; Saad, F.; Lapointe, R.; et al. Androgen-Regulated Expression of Arginase 1, Arginase 2 and Interleukin-8 in Human Prostate Cancer. PLoS ONE 2010, 5, e12107. [Google Scholar] [CrossRef]
- Czystowska-Kuzmicz, M.; Sosnowska, A.; Nowis, D.; Ramji, K.; Szajnik, M.; Chlebowska-Tuz, J.; Wolinska, E.; Gaj, P.; Grazul, M.; Pilch, Z.; et al. Small extracellular vesicles containing arginase-1 suppress T-cell responses and promote tumor growth in ovarian carcinoma. Nat. Commun. 2019, 10, 3000. [Google Scholar] [CrossRef]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef]
- Lomelino, C.L.; Andring, J.T.; McKenna, R.; Kilberg, M.S. Asparagine synthetase: Function, structure, and role in disease. J. Biol. Chem. 2017, 292, 19952–19958. [Google Scholar] [CrossRef]
- Mew, N.A.; Pappa, M.B.; Gropman, A.L. Urea Cycle Disorders. In Rosenberg’s Molecular and Genetic Basis of Neurological and Psychiatric Disease; Academic Press: Cambridge, MA, USA, 2015; pp. 633–647. [Google Scholar] [CrossRef]
- Yao, S.; Nguyen, T.-V.; Rolfe, A.; Agrawal, A.A.; Ke, J.; Peng, S.; Colombo, F.; Yu, S.; Bouchard, P.; Wu, J.; et al. Small Molecule Inhibition of CPS1 Activity through an Allosteric Pocket. Cell Chem. Biol. 2020, 27, 259–268.e5. [Google Scholar] [CrossRef]
- Crump, N.T.; Hadjinicolaou, A.V.; Xia, M.; Walsby-Tickle, J.; Gileadi, U.; Chen, J.-L.; Setshedi, M.; Olsen, L.R.; Lau, I.-J.; Godfrey, L.; et al. Chromatin accessibility governs the differential response of cancer and T cells to arginine starvation. Cell Rep. 2021, 35, 109101. [Google Scholar] [CrossRef]
- Weis-Banke, S.E.; Hübbe, M.L.; Holmström, M.O.; Jørgensen, M.A.; Bendtsen, S.K.; Martinenaite, E.; Carretta, M.; Svane, I.M.; Ødum, N.; Pedersen, A.W.; et al. The metabolic enzyme arginase-2 is a potential target for novel immune modulatory vaccines. OncoImmunology 2020, 9, 1771142. [Google Scholar] [CrossRef]
| Total Patients (145) | No. | % | |
|---|---|---|---|
| Age | Mean ± SD | 63.32 ± 11.83 (ranging 33–92) | |
| Gender | Males | 114 | 79 |
| Females | 31 | 21 | |
| Smoking status | Yes | 97 | 67 |
| No | 40 | 28 | |
| Missing data | 8 | 6 | |
| Alcohol abuse | Yes | 70 | 48 |
| No | 67 | 46 | |
| Missing data | 8 | 6 | |
| TNM staging | 1 | 19 | 13 |
| 2 | 43 | 30 | |
| 3 | 28 | 19 | |
| 4 | 55 | 38 | |
| Disease status | Incipient (TNM 1–2) | 62 | 43 |
| Advanced (TNM 3–4) | 83 | 57 | |
| Primary tumor dimensions | Small (T stages 1–2) | 89 | 61 |
| Large (T stages 3–4) | 56 | 39 | |
| Lymph node metastasis * | Positive neck | 61 | 42 |
| Negative neck | 60 | 41 | |
| Histological differentiation degree | Well-differentiated | 35 | 24 |
| Moderately differentiated | 80 | 55 | |
| Poorly differentiated | 30 | 21 | |
| Disease related survival | Alive | 115 | 79 |
| Deceased secondarily to disease progression | 30 | 21 | |
| Serum Urea | Creatinine | Uric Acid | ||||
|---|---|---|---|---|---|---|
| OSCC Group | Control Group | OSCC Group | Control Group | OSCC Group | Control Group | |
| No subjects | 145 | 80 | 145 | 80 | 116 # | 72 |
| Mean/Median (mg/dL) | 32.00 | 33.00 | 0.7600 | 0.7750 | 5.267 | 5.338 |
| SD/IQR | 15.55 | 10.25 | 0.2350 | 0.2125 | 1.361 | 1.193 |
| p value | 0.0344 ^ | 0.2090 ^ | 0.7190 * | |||
| Parameter | Mean | SD | Correl. Coeff. | pValue | |
|---|---|---|---|---|---|
| Age | 0.409 | 0.0000 | |||
| Gender | Males | 32.67 | 10.14 | 0.075 | 0.3714 |
| Females | 34.52 | 9.932 | |||
| Smoking | Smokers | 30.98 | 9.689 | −0.300 | 0.0004 |
| Non-smokers | 37.35 | 9.016 | |||
| Alcohol abuse | Confirmed | 31.19 | 9.083 | −0.156 | 0.0679 |
| No alcohol abuse | 34.56 | 10.48 | |||
| T stage | Small tumors (T1–2) | 33.49 | 10.4 | −0.056 | 0.5061 |
| Large tumors (T3–4) | 32.39 | 9.628 | |||
| Lymph node invasion * | Positive nodes | 30.24 | 8.797 | −0.244 | 0.0070 |
| Negative nodes | 35.16 | 10.63 | |||
| Histological differentiation degree | Well-differentiated | 34.03 | 9.738 | −0.035 | 0.6793 |
| Moderately differentiated | 32.65 | 10.28 | |||
| Poorly differentiated | 33.06 | 10.23 | |||
| Disease related prognosis | Alive | 33.51 | 9.771 | −0.088 | 0.2932 |
| Deceased secondarily to disease | 31.36 | 11.24 |
| Model | β | Std. Error | p Value | |
|---|---|---|---|---|
| 1 | Age | 0.295 | 0.086 | 0.001 |
| Gender | −1.742 | 2.339 | 0.458 | |
| Smoking status | −4.624 | 2.238 | 0.041 | |
| Alcohol abuse | −0.549 | 2.122 | 0.796 | |
| Histological differentiation | 0.171 | 1.311 | 0.897 | |
| T stage | 0.166 | 0.893 | 0.853 | |
| Lymph node invasion | −1.831 | 0.910 | 0.047 | |
| Disease related prognosis | 0.287 | 2.153 | 0.894 | |
| 2 | Age | 0.288 | 0.082 | 0.001 |
| Smoking status | −4.510 | 1.888 | 0.019 | |
| Lymph node invasion | −1.702 | 0.779 | 0.031 | |
| Dependent variable: serum urea | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caruntu, A.; Moraru, L.; Ciubotaru, D.A.; Tanase, C.; Scheau, C.; Caruntu, C. Assessment of Serum Urea, Creatinine and Uric Acid in Oral Cancer. J. Clin. Med. 2022, 11, 3459. https://doi.org/10.3390/jcm11123459
Caruntu A, Moraru L, Ciubotaru DA, Tanase C, Scheau C, Caruntu C. Assessment of Serum Urea, Creatinine and Uric Acid in Oral Cancer. Journal of Clinical Medicine. 2022; 11(12):3459. https://doi.org/10.3390/jcm11123459
Chicago/Turabian StyleCaruntu, Ana, Liliana Moraru, Diana Alina Ciubotaru, Cristiana Tanase, Cristian Scheau, and Constantin Caruntu. 2022. "Assessment of Serum Urea, Creatinine and Uric Acid in Oral Cancer" Journal of Clinical Medicine 11, no. 12: 3459. https://doi.org/10.3390/jcm11123459
APA StyleCaruntu, A., Moraru, L., Ciubotaru, D. A., Tanase, C., Scheau, C., & Caruntu, C. (2022). Assessment of Serum Urea, Creatinine and Uric Acid in Oral Cancer. Journal of Clinical Medicine, 11(12), 3459. https://doi.org/10.3390/jcm11123459









