Effect of Intragastric Botulinum Type A Injection Combined with a Low-Calorie High-Protein Diet in Adults with Overweight or Obesity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Endoscopic Procedures of IGBI
2.2. Definition and Assessment of Fatty Liver
2.3. Measures
2.4. End Points and Assessments
2.5. Safety Assessments
2.6. Statistical Analysis
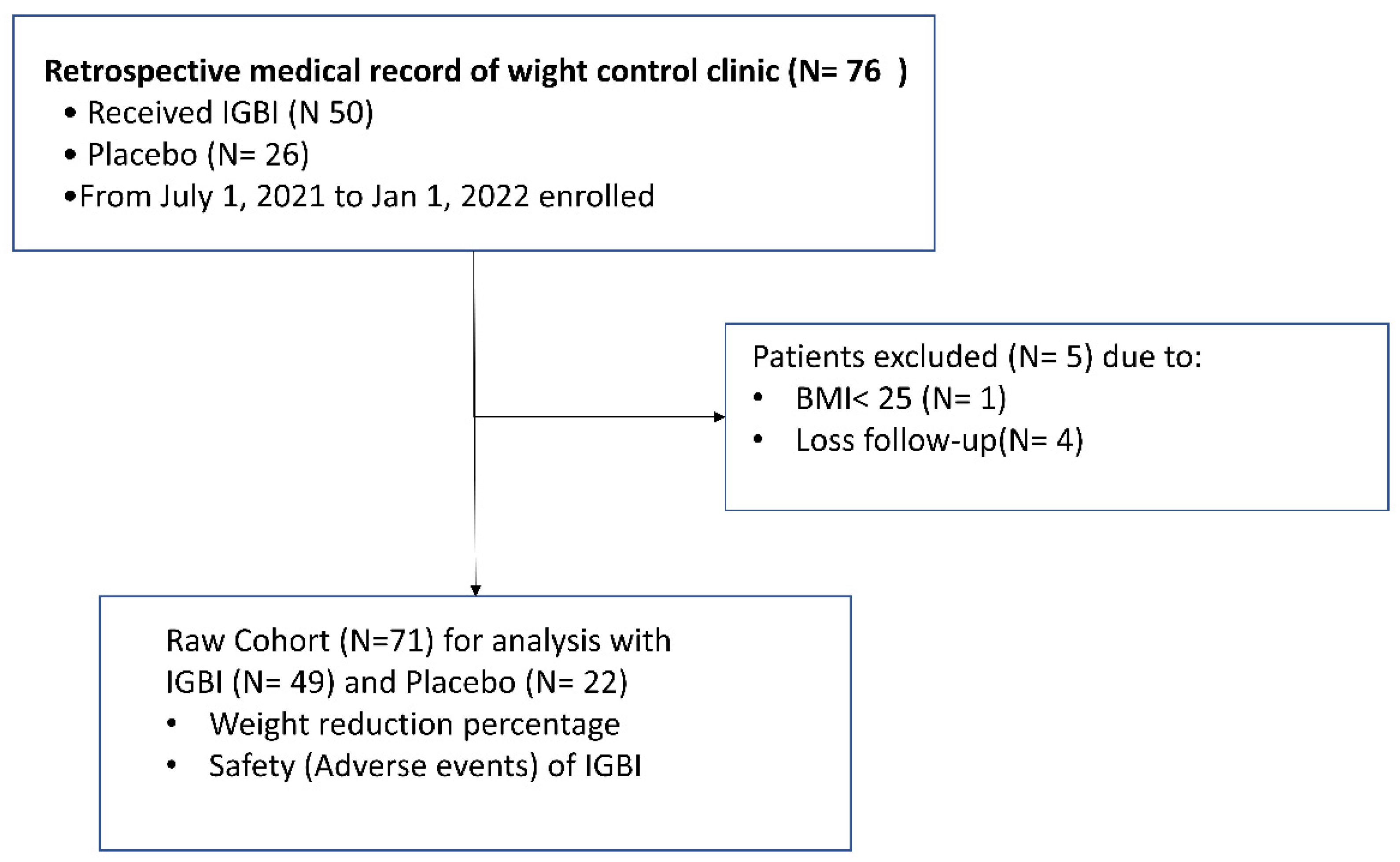
3. Results
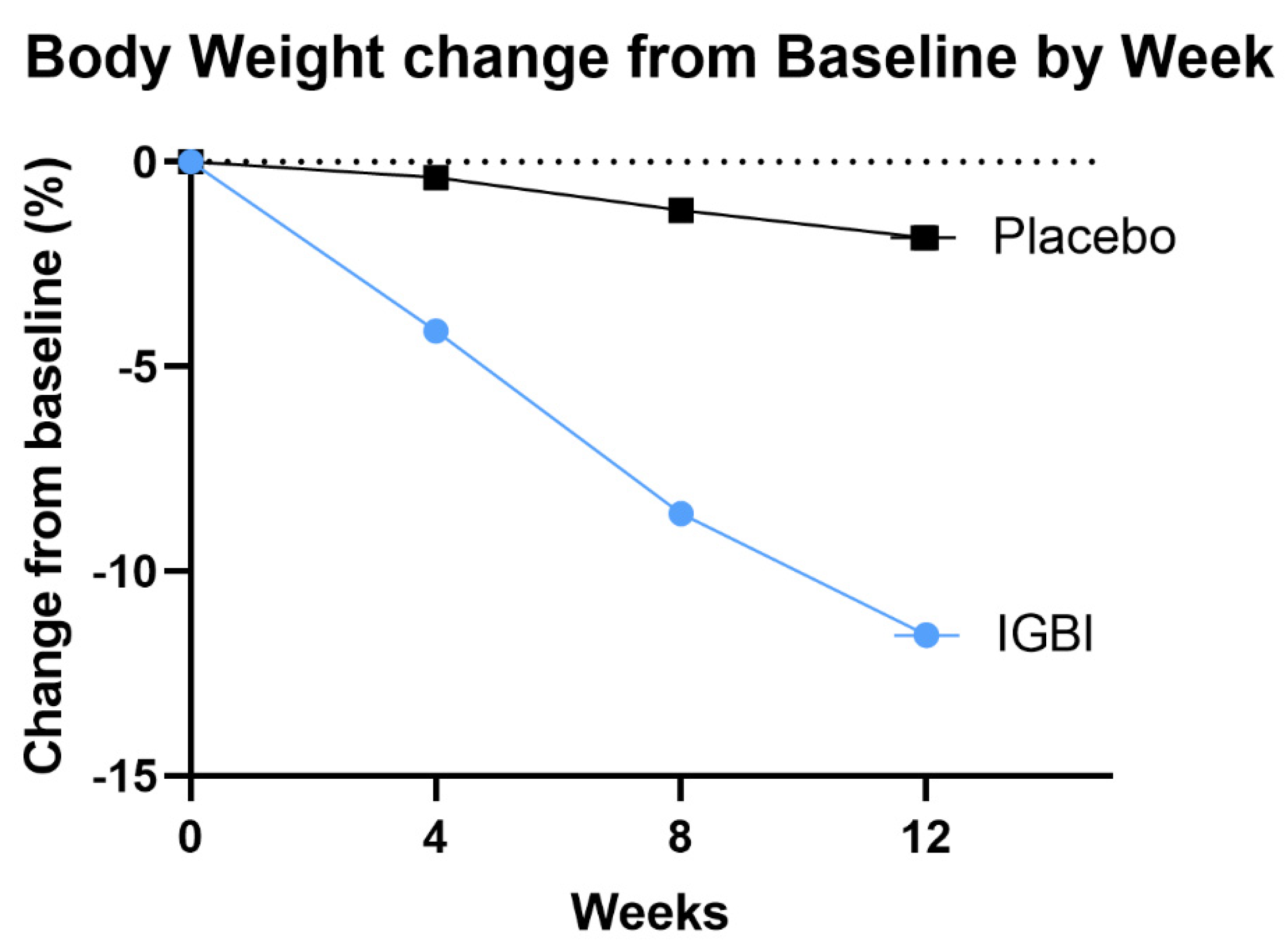
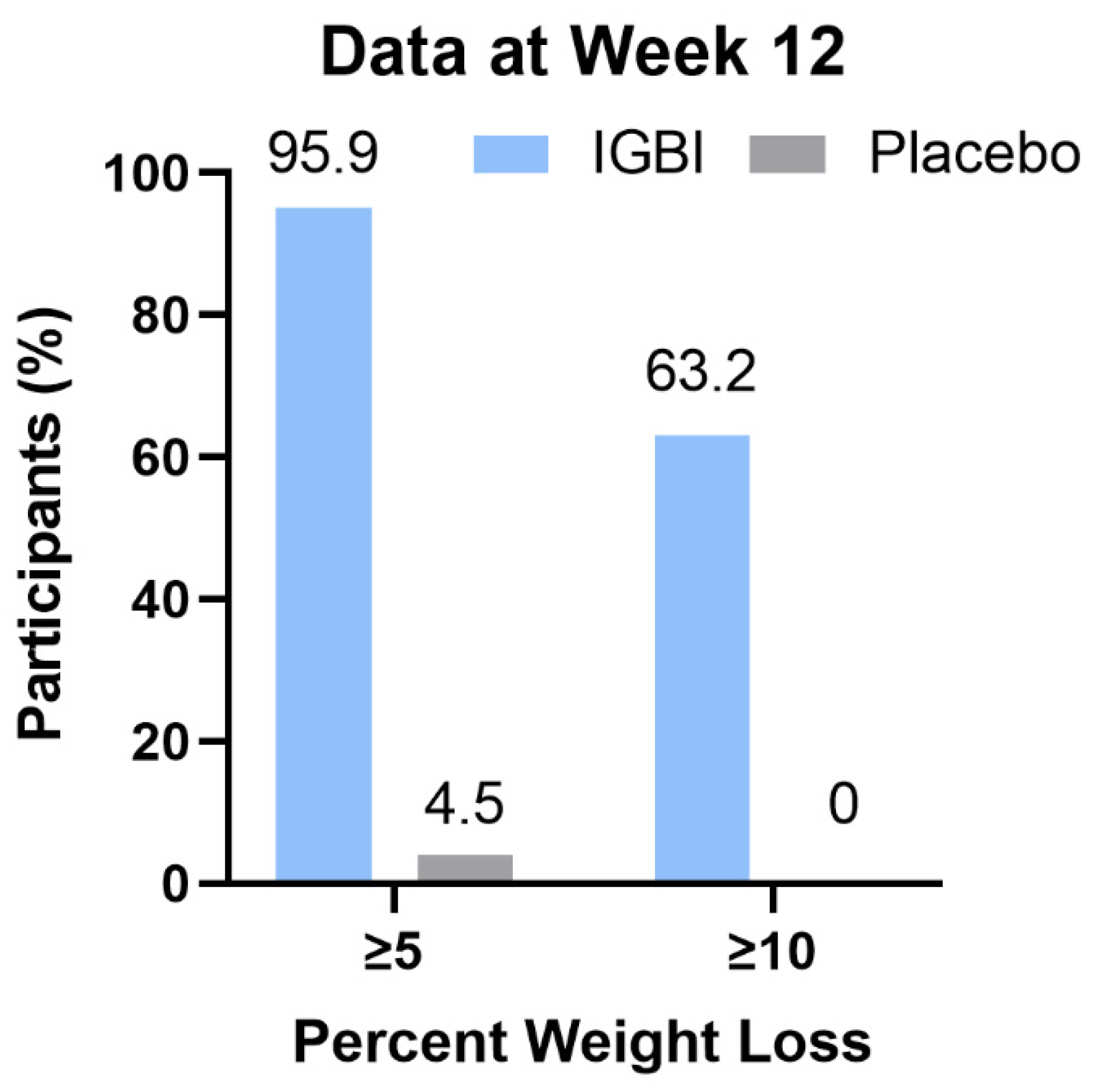
Change in Body Weight as End-Point Assessment
4. Safety Assessment
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kolahi, A.-A.; Moghisi, A.; Ekhtiari, Y.S. Socio-demographic determinants of obesity indexes in Iran: Findings from a nationwide STEPS survey. Health Promot. Perspect. 2018, 8, 187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, T.; Yang, W.; Chen, C.-S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA 2014, 311, 806–814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavie, C.J.; Sanchis-Gomar, F.; Henry, B.M.; Lippi, G. COVID-19 and obesity: Links and risks. Expert Rev. Endocrinol. Metab. 2020, 15, 215–216. [Google Scholar] [CrossRef] [PubMed]
- Sattar, N.; McInnes, I.B.; McMurray, J.J. Obesity is a risk factor for severe COVID-19 infection: Multiple potential mechanisms. Circulation 2020, 142, 4–6. [Google Scholar] [CrossRef]
- Hsu, P.-K.; Wu, L.-S.; Yen, H.-H.; Huang, H.P.; Chen, Y.-Y.; Su, P.-Y.; Su, W.-W. Attenuation imaging with ultrasound as a novel evaluation method for liver steatosis. J. Clin. Med. 2021, 10, 965. [Google Scholar] [CrossRef]
- Hsu, P.-K.; Wu, L.-S.; Su, W.-W.; Su, P.-Y.; Chen, Y.-Y.; Hsu, Y.-C.; Yen, H.-H.; Wu, C.-L. Comparing the controlled attenuation parameter using FibroScan and attenuation imaging with ultrasound as a novel measurement for liver steatosis. PLoS ONE 2021, 16, e0254892. [Google Scholar] [CrossRef]
- Members, E.P.; Jensen, M.D.; Ryan, D.H.; Donato, K.A.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M. Executive summary: Guidelines (2013) for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Obesity Society published by the Obesity Society and American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Based on a systematic review from the The Obesity Expert Panel, 2013. Obesity 2014, 22, S5–S39. [Google Scholar]
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 2014, 129, S139–S140. [Google Scholar] [CrossRef] [Green Version]
- Wadden, T.A.; Butryn, M.L.; Byrne, K.J. Efficacy of lifestyle modification for long-term weight control. Obes. Res. 2004, 12, 151S–162S. [Google Scholar] [CrossRef]
- de Zwaan, M. Binge eating disorder and obesity. Int. J. Obes. 2001, 25, S51–S55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozsoy, Z.; Demir, E. Which bariatric procedure is the most popular in the world? A bibliometric comparison. Obes. Surg. 2018, 28, 2339–2352. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, A.; Czernichow, S.; Hertig, A.; Ledoux, S.; Poghosyan, T.; Quilliot, D.; Le Gall, M.; Bado, A.; Joly, F. Prevention and treatment of nutritional complications after bariatric surgery. Lancet Gastroenterol. Hepatol. 2021, 6, 238–251. [Google Scholar] [CrossRef]
- Park, J.-S.; Zheng, H.-M.; Kim, J.-M.; Kim, C.S.; Jeong, S.; Lee, D.H. The effect of intragastric administration of botulinum toxin type A on reducing adiposity in a rat model of obesity using micro-CT and histological examinations. Gut Liver 2017, 11, 798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Topazian, M.; Camilleri, M.; La Mora-Levy, D.; Enders, F.B.; Foxx-Orenstein, A.E.; Levy, M.J.; Nehra, V.; Talley, N.J. Endoscopic ultrasound-guided gastric botulinum toxin injections in obese subjects: A pilot study. Obes. Surg. 2008, 18, 401–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elshakh, H.; El-Ejji, K.; Taheri, S. The role of endoscopic intra-gastric botulinum toxin-A for obesity treatment. Obes. Surg. 2017, 27, 2471–2478. [Google Scholar] [CrossRef] [PubMed]
- Dasarathy, S.; Dasarathy, J.; Khiyami, A.; Joseph, R.; Lopez, R.; McCullough, A.J. Validity of real time ultrasound in the diagnosis of hepatic steatosis: A prospective study. J. Hepatol. 2009, 51, 1061–1067. [Google Scholar] [CrossRef]
- Ting, P.T.; Freiman, A. The story of Clostridium botulinum: From food poisoning to Botox. Clin. Med. 2004, 4, 258. [Google Scholar] [CrossRef]
- Ezzeddine, D.; Jit, R.; Katz, N.; Gopalswamy, N.; Bhutani, M.S. Pyloric injection of botulinum toxin for treatment of diabetic gastroparesis. Gastrointest. Endosc. 2002, 55, 920–923. [Google Scholar] [CrossRef]
- França, K.; Kumar, A.; Fioranelli, M.; Lotti, T.; Tirant, M.; Roccia, M.G. The history of Botulinum toxin: From poison to beauty. Wien. Med. Wochenschr. 2017, 167, 46–48. [Google Scholar] [CrossRef]
- Small, R. Botulinum toxin injection for facial wrinkles. Am. Fam. Physician 2014, 90, 168–175. [Google Scholar] [PubMed]
- Liu, W.; Jin, Y.; Wilde, P.J.; Hou, Y.; Wang, Y.; Han, J. Mechanisms, physiology, and recent research progress of gastric emptying. Crit. Rev. Food Sci. Nutr. 2021, 61, 2742–2755. [Google Scholar] [CrossRef] [PubMed]
- Janssen, P.; Vanden Berghe, P.; Verschueren, S.; Lehmann, A.; Depoortere, I.; Tack, J. The role of gastric motility in the control of food intake. Aliment. Pharmacol. Ther. 2011, 33, 880–894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shulkes, A.; Allen, R.; Hardy, K. Meal stimulated gastrin and pancreatic polypeptide levels before and after partial gastric transection for morbid obesity. Aust. N. Z. J. Med. 1982, 12, 27–30. [Google Scholar] [CrossRef]
- Nunez-Salces, M.; Li, H.; Feinle-Bisset, C.; Young, R.L.; Page, A.J. The regulation of gastric ghrelin secretion. Acta Physiol. 2021, 231, e13588. [Google Scholar] [CrossRef]
- Bang, C.S.; Baik, G.H.; Shin, I.S.; Kim, J.B.; Suk, K.T.; Yoon, J.H.; Kim, Y.S.; Kim, D.J. Effect of intragastric injection of botulinum toxin A for the treatment of obesity: A meta-analysis and meta-regression. Gastrointest. Endosc. 2015, 81, 1141–1149.e7. [Google Scholar] [CrossRef]
- Jones, O.M.; Brading, A.F.; Mortensen, N.J. Mechanism of action of botulinum toxin on the internal anal sphincter. Br. J. Surg. 2004, 91, 224–228. [Google Scholar] [CrossRef]
- Bigalke, H.; Habermann, E. Blockade by tetanus and botulinum A toxin of postganglionic cholinergic nerve endings in the myenteric plexus. Naunyn-Schmiedeberg’s Arch. Pharmacol. 1980, 312, 255–263. [Google Scholar] [CrossRef]
- James, A.N.; Ryan, J.P.; Parkman, H.P. Inhibitory effects of botulinum toxin on pyloric and antral smooth muscle. Am. J. Physiol.-Gastrointest. Liver Physiol. 2003, 285, G291–G297. [Google Scholar] [CrossRef] [Green Version]
- Gaetano, D.; Spada, P.L. Botulinum toxin injected in the gastric wall reduces body weight and food intake in rats. Aliment. Pharmacol. Ther. 2000, 14, 829–834. [Google Scholar]
- Rollnik, J.D.; Meier, P.N.; Manns, M.P.; Gke, M. Antral injections of botulinum a toxin for the treatment of obesity. Ann. Intern. Med. 2003, 138, 359–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gui, D.; Mingrone, G.; Valenza, V.; Spada, P.; Mutignani, M.; Runfola, M.; Scarfone, A.; Di Mugno, M.; Panunzi, S. Effect of botulinum toxin antral injection on gastric emptying and weight reduction in obese patients: A pilot study. Aliment. Pharmacol. Ther. 2006, 23, 675–680. [Google Scholar] [CrossRef] [PubMed]
- de Moura, E.G.H.; Ribeiro, I.B.; Frazão, M.S.V.; Mestieri, L.H.M.; de Moura, D.T.H.; Dal Bó, C.M.R.; Brunaldi, V.O.; de Moura, E.T.H.; Nunes, G.C.; Bustamante, F.A.C. EUS-guided intragastric injection of botulinum toxin A in the preoperative treatment of super-obese patients: A randomized clinical trial. Obes. Surg. 2019, 29, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, F.; Brunaldi, V.O.; Bernardo, W.M.; de Moura, D.T.H.; de Moura, E.T.H.; Galvão, M.; Santo, M.A.; de Moura, E.G.H. Obesity treatment with botulinum toxin-A is not effective: A systematic review and meta-analysis. Obes. Surg. 2017, 27, 2716–2723. [Google Scholar] [CrossRef]
- Chang, P.-C.; Jhou, H.-J.; Chen, P.-H.; Huang, C.-K.; Chiang, H.-H.; Chen, K.-H.; Chang, T.-W. Intragastric Botulinum Toxin A Injection Is an Effective Obesity Therapy for Patients with BMI > 40 kg/m2: A Systematic Review and Meta-analysis. Obes. Surg. 2020, 30, 4081–4090. [Google Scholar] [CrossRef]
- Mittermair, R.; Keller, C.; Geibel, J. Intragastric injection of botulinum toxin A for the treatment of obesity. Obes. Surg. 2007, 17, 732–736. [Google Scholar] [CrossRef]
- Foschi, D.; Corsi, F.; Lazzaroni, M.; Sangaletti, O.; Riva, P.; La Tartara, G.; Bevilacqua, M.; Osio, M.; Alciati, A.; Bianchi Porro, G. Treatment of morbid obesity by intraparietogastric administration of botulinum toxin: A randomized, double-blind, controlled study. Int. J. Obes. 2007, 31, 707–712. [Google Scholar] [CrossRef] [Green Version]
- Ferhatoglu, M.F.; Kartal, A.; Filiz, A.I.; Kebudi, A. The positive effects of a calorie-restricting high-protein diet combined with intragastric botulinum toxin type A application among morbidly obese patients: A prospective, observational analysis of eighty-seven grade 2 obese patients. Obes. Surg. 2020, 30, 3472–3479. [Google Scholar] [CrossRef]
- Yen, Y.A.; Wang, C.C.; Sung, W.W.; Fang, K.C.; Huang, S.M.; Lin, C.C.; Tsai, M.C.; Yang, T.W. Intragastric Injection of Botulinum Toxin A for Weight Loss: A Systematic Review and Meta-analysis of Randomized Controlled Trials. J. Gastroenterol. Hepatol. 2022. [Google Scholar] [CrossRef]
- Kaya, B.; Esen Bulut, N.; Fersahoglu, M. Is Intragastric Botulinum Toxin A Injection Effective in Obesity Treatment? Surg. Res. Pract. 2020, 2419491. [Google Scholar] [CrossRef]
| Characteristic | IGBI (N = 49) | Placebo (N = 22) | p-Value |
|---|---|---|---|
| Age (years) | 39.8 (37.1–42.4) | 49.7 (43.1–56.3) | 0.007 |
| Female sex, n (%) | 37 (76) | 16 (72) | 0.80 |
| Weight (kg) | 82.1 (77.2–87.1) | 77.3 (70.7–83.8) | 0.25 |
| BMI (kg/m2) | 29.3 (26.2–33.3) | 28.0 (26.8–30.8) | 0.63 |
| Overweight, n (%) | 27 (55%) | 12 (54%) | 0.66 |
| Obesity, n (%) | 22 (45%) | 10 (46%) | 0.66 |
| Fatty liver stage, n (%) | 0.25 | ||
| Mild | 9 (18%) | 3 (13%) | |
| Moderate | 9 (18%) | 5 (22%) | |
| Severe | 27 (55%) | 14 (63.6%) | |
| HBA1c (%) | 5.5 (5.4–5.6) | 5.5 (5.3–5.7) | 0.49 |
| Cholesterol (mg/dL) | |||
| Total | 164.1 (155.4–172.7) | 186.0 (164.1–207.9) | 0.005 |
| HDL cholesterol | 43.5 (37.6–49.4) | 51.8 (43.2–60.5) | 0.11 |
| LDL cholesterol | 100.1 (92.7–107.5) | 134.2 (113.8–154.5) | 0.003 |
| Triglycerides (mg/dL) | 112.9 (82.8–143.0) | 158.6 (132.0–185.2) | 0.02 |
| Adverse Event | IGBI (49) | Placebo (22) |
|---|---|---|
| Time to any adverse event, weeks | 1 (1–2) | 1 (1) |
| Any adverse event, no (%) | 20 (40.8) | 2 |
| Serious adverse events, no (%) | 0(0) | 0 |
| Reported adverse evet, no. (%) | ||
| Nausea | 2 (4.0) | 0 |
| Vomiting | 0(0) | 0 |
| Diarrhea | 1 (2.0) | 0 |
| Constipation | 12 (24.4) | 2 (9.0) |
| Nasopharyngitis | 2 (4.0) | 0 |
| Urinary disorder | 1 (2.0) | 0 |
| Abdominal pain | 1 (2.0) | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, P.-K.; Wu, C.-L.; Yang, Y.-H.; Wei, J.C.-C. Effect of Intragastric Botulinum Type A Injection Combined with a Low-Calorie High-Protein Diet in Adults with Overweight or Obesity. J. Clin. Med. 2022, 11, 3325. https://doi.org/10.3390/jcm11123325
Hsu P-K, Wu C-L, Yang Y-H, Wei JC-C. Effect of Intragastric Botulinum Type A Injection Combined with a Low-Calorie High-Protein Diet in Adults with Overweight or Obesity. Journal of Clinical Medicine. 2022; 11(12):3325. https://doi.org/10.3390/jcm11123325
Chicago/Turabian StyleHsu, Po-Ke, Chia-Lin Wu, Yu-Hsuan Yang, and James Cheng-Chung Wei. 2022. "Effect of Intragastric Botulinum Type A Injection Combined with a Low-Calorie High-Protein Diet in Adults with Overweight or Obesity" Journal of Clinical Medicine 11, no. 12: 3325. https://doi.org/10.3390/jcm11123325
APA StyleHsu, P.-K., Wu, C.-L., Yang, Y.-H., & Wei, J. C.-C. (2022). Effect of Intragastric Botulinum Type A Injection Combined with a Low-Calorie High-Protein Diet in Adults with Overweight or Obesity. Journal of Clinical Medicine, 11(12), 3325. https://doi.org/10.3390/jcm11123325








