Is There Still a Place for Reconstructive Surgery in Distal Tubal Disease?
Abstract
1. Introduction
2. Etiopathogenesis and Morphology of Distal Tubal Disease
3. Treatment Strategy and Decision-Making Process
4. Operative Techniques in Reconstructive Distal Tubal Surgery
5. Reproductive Outcomes
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Gebeh, A.; Metwally, M. Surgical management of tubal disease and infertility. Obstet. Gynaecol. Reprod. Med. 2019, 29, 123–128. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Role of tubal surgery in the era of assisted reproductive technology: A committee opinion. Fertil. Steril. 2021, 115, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Honore, G.M.; Holden, A.E.; Schenken, R.S. Pathophysiology and management of proximal tubal blockage. Fertil. Steril. 1999, 5, 785–795. [Google Scholar] [CrossRef]
- Ng, K.Y.B.; Cheong, Y. Hydrosalpinx—Salpingostomy, salpingectomy or tubal occlusion. Best Pract. Res. Clin. Obs. Gynaecol. 2019, 59, 41–47. [Google Scholar] [CrossRef]
- Chua, S.J.; Akande, V.A.; Mol, B.W.J. Surgery for tubal infertility. Cochrane Database Syst. Rev. 2017, 2017, CD006415. [Google Scholar] [CrossRef]
- Tamblyn, J.; Jeve, Y. Surgical management of tubal disease and infertility. Obstet. Gynaecol. Reprod. Med. 2022, 32, 7–13. [Google Scholar] [CrossRef]
- Puttemans, P.J.; Brosens, I.A. Salpingectomy improves in-vitro fertilization outcome in patients with a hydrosalpinx: Blind victimization of the fallopian tube? Hum. Reprod. 1996, 11, 2079–2081. [Google Scholar] [CrossRef]
- Rezvani, M.; Shaaban, A. Fallopian Tube Disease in the Nonpregnant Patient. RadioGraphics 2011, 31, 527–548. [Google Scholar] [CrossRef]
- Jenkins, S.; Olive, D.L.; Haney, A.F. Endometriosis: Pathogenetic implications of the anatomic distribution. Obstet. Gynecol. 1986, 67, 335–338. [Google Scholar]
- Kim, M.Y.; Rha, S.E.; Oh, S.N.; Jung, S.E.; Lee, Y.J.; Kim, Y.S.; Byun, J.Y.; Lee, A.; Kim, M.R. MR Imaging findings of hydrosalpinx: A com-prehensive review. Radiographics 2009, 29, 495–507. [Google Scholar] [CrossRef]
- Katz, E.; Akman, M.A.; Damewood, M.D.; García, J.E. Deleterious effect of the presence of hydrosalpinx on implantation and pregnancy rates with in vitro fertilization. Fertil. Steril. 1996, 66, 122–125. [Google Scholar] [CrossRef]
- Andersen, A.N.; Yue, Z.; Meng, F.J.; Petersen, K. Low implantation rate after in-vitro fertilization in patients with hydrosalpinges diagnosed by ultrasonography. Hum. Reprod. 1994, 9, 1935–1938. [Google Scholar] [CrossRef] [PubMed]
- Strandell, A.; Waldenström, U.; Nilsson, L.; Hamberger, L. Hydrosalpinx reduces in-vitro fertilization/embryo transfer pregnancy rates. Hum. Reprod. 1994, 9, 861–863. [Google Scholar] [CrossRef] [PubMed]
- Blazar, A.S.; Hogan, J.W.; Seifer, D.B.; Frishman, G.N.; Wheeler, C.A.; Haning, R.V. The impact of hydrosalpinx on successful preg-nancy in tubal factor infertility treated by in vitro fertilization. Fertil. Steril. 1997, 67, 517–520. [Google Scholar] [CrossRef]
- Lass, A. What effect does hydrosalpinx have on assisted reproduction? What is the preferred treatment for hydrosalpines? The ovary’s perspective. Hum. Reprod. 1999, 14, 1674e7. [Google Scholar] [CrossRef]
- Benjaminov, O.; Atri, M. Sonography of the Abnormal Fallopian Tube. Am. J. Roentgenol. 2004, 183, 737–742. [Google Scholar] [CrossRef]
- Hulka, J.F.; Omran, K.; Berger, G.S. Classification of adnexal adhesions: A proposal and evaluation of its prognostic value. Fertil. Steril. 1978, 30, 661–665. [Google Scholar] [CrossRef]
- Hulka, J.F. Adnexal adhesions: A prognostic staging and classification system based on a five-year survey of fertility surgery results at Chapel Hill, North Carolina. Am. J. Obstet. Gynecol. 1982, 144, 141–148. [Google Scholar] [CrossRef]
- Mage, G.; Pouly, J.L.; de Jolinière, J.B.; Chabrand, S.; Riouallon, A.; Bruhat, M.A. A preoperative classification to predict the intrau-terine and ectopic pregnancy rates after distal tubal microsurgery. Fertil. Steril. 1986, 46, 807–810. [Google Scholar] [CrossRef]
- Wu, C.H.; Gocial, B. A pelvic scoring system for infertility surgery. Int. J. Fertil. 1988, 33, 341–346. [Google Scholar]
- The American Fertility Society. Classifications of adnexal adhesions, distal tubal occlusion, tubal occlusion secondary to tubal ligation, tubal pregnancies, Mullerian anomalies and intrauterine adhesions. Fertil. Steril. 1988, 49, 944–951. [Google Scholar] [CrossRef]
- Allen, S.; Feste, J.R. Pelvic disease classifications. Fertil. Steril. 1989, 51, 199–201. [Google Scholar] [CrossRef]
- Winston, R.M.L.; Margara, R.A. Microsurgical salpingostomy is not an obsolete procedure. BJOG Int. J. Obstet. Gynaecol. 1991, 98, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Boer-Meisel, M.E.; te Velde, E.R.; Habbema, J.D.; Kardaun, J.W. Predicting the pregnancy outcome in patients treated for hydrosal-pinx: A prospective study. Fertil. Steril. 1986, 45, 23–29. [Google Scholar] [CrossRef]
- Rutherford, A.J.; Jenkins, J. Hull and Rutherford classification of infertility. Hum. Fertil. 2002, 5, S41–S45. [Google Scholar] [CrossRef]
- Akande, V.A.; Cahill, D.J.; Wardle, P.G.; Rutherford, A.J.; Jenkins, J. The predictive value of the ‘Hull & Rutherford’ classification for tubal damage. BJOG Int. J. Obstet. Gynaecol. 2004, 111, 1236–1241. [Google Scholar] [CrossRef]
- Zou, S.-E.; Jin, Y.; Ko, Y.-L.; Zhu, J. A new classification system for pregnancy prognosis of tubal factor infertility. Int. J. Clin. Exp. Med. 2014, 7, 1410–1416. [Google Scholar]
- Guzick, D.S.; Silliman, N.P.; Adamson, G.D.; Buttram VCJr Canis, M.; Malinak, L.R.; Schenken, R.S. Prediction of pregnancy in infer-tile women based on the American Society for Reproductive Medicine’s revised classification of endometriosis. Fertil. Steril. 1997, 67, 822–829. [Google Scholar] [CrossRef]
- Gomel, V.; Wang, I. Laparoscopic surgery for infertility therapy. Curr. Opin. Obstet. Gynecol. 1994, 6, 141–148. [Google Scholar] [CrossRef]
- Kasia, J.M.; Ngowa, J.D.; Mimboe, Y.S.; Toukam, M.; Ngassam, A.; Noa, C.C.; Belinga, E.; Medou, A. Laparoscopic Fimbrioplasty and Neosalpingostomy in Female Infertility: A Review of 402 Cases at the Gynecological Endoscopic Surgery and Human Re-productive Teaching Hospital in Yaoundé-Cameroon. J. Reprod. Infertil. 2016, 17, 104–109. [Google Scholar]
- Audebert, A.; Pouly, J.L.; Bonifacie, B.; Yazbeck, C. Laparoscopic surgery for distal tubal occlusions: Lessons learned from a histor-ical series of 434 cases. Fertil. Steril. 2014, 102, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Gomel, V. Salpingostomy by Microsurgery. Fertil. Steril. 1978, 29, 380–387. [Google Scholar] [CrossRef]
- DeCherney, A.H.; Kase, N. A comparison of treatment for bilateral fimhrial occlusion. Fertil. Steril. 1981, 35, 162–166. [Google Scholar] [CrossRef]
- Wallach, E.E.; Manara, L.R.; Eisenberg, E. Experience with 143 cases of tubal surgery. Fertil. Steril. 1983, 39, 609–617. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Optimal evaluation of the infertile female. Fertil. Steril. 2006, 86, S264–S267. [Google Scholar] [CrossRef] [PubMed]
- Bontis, J.N.; Theodoridis, T.D. Laparoscopic Management of Hydrosalpinx. Ann. N. Y. Acad. Sci. 2006, 1092, 199–210. [Google Scholar] [CrossRef]
- Infertility Workup for the Women’s Health Specialist: ACOG Committee Opinion, Number 781. Obstet. Gynecol. 2019, 133, e377–e384. [CrossRef]
- Ahmad, G.; Watson, A.J.S.; Metwally, M. Laparoscopy or laparotomy for distal tubal surgery? A meta-analysis. Hum. Fertil. 2007, 10, 43–47. [Google Scholar] [CrossRef]
- Azziz, R. Microsurgery alone or with INTERCEED Absorbable Adhesion Barrier for pelvic sidewall adhesion re-formation. The INTERCEED (TC7) Adhesion Barrier Study Group II. Surg. Gynecol. Obstet. 1993, 177, 135–139. [Google Scholar]
- Becker, J.M.; Dayton, M.T.; Fazio, V.W.; Beck, D.E.; Stryker, S.J.; Wexner, S.D.; Wolff, B.G.; Roberts, P.L.; Smith, L.E.; Sweeney, S.A.; et al. Prevention of postoperative abdominal adhesions by a sodium hyaluronate-based bioresorbable membrane: A prospective, randomized, double-blind multicenter study. J. Am. Coll. Surg. 1996, 183, 297–306. [Google Scholar]
- Brown, C.B.; Luciano, A.A.; Martin, D.; Peers, E.; Scrimgeour, A.; Dizerega, G.S.; Adept Adhesion Reduction Study Group. Adept (icodextrin 4% solution) reduces adhesions after laparoscopic surgery for adhesiolysis: A double-blind, randomized, controlled study. Fertil. Steril. 2007, 88, 1413–1426. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.P. Reduction of de novo postsurgical adhesions by intraoperative precoating with Sepracoat (HAL-C) solution: A prospective, randomized, blinded, placebo-controlled multicenter study. The Sepracoat Adhesion Study Group. Fertil. Steril. 1998, 69, 1067–1074. [Google Scholar] [CrossRef]
- Franklin, R.R. Reduction of ovarian adhesions by the use of interceed. Ovarian Adhes. Study Group Obstet. Gynecol. 1995, 86, 335–340. [Google Scholar] [CrossRef]
- Mais, V.; Bracco, G.; Litta, P.; Gargiulo, T.; Melis, G. Reduction of postoperative adhesions with an auto-crosslinked hyaluronan gel in gynaecological laparoscopic surgery: A blinded, controlled, randomized, multicentre study. Hum. Reprod. 2006, 21, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Sekiba, K. Use of Interceed(TC7) absorbable adhesion barrier to reduce postoperative adhesion reformation in infertility and endometriosis surgery. The Obstetrics and Gynecology Adhesion Prevention Committee. Obstet. Gynecol. 1992, 79, 518–522. [Google Scholar]
- Takeuchi, H.; Kitade, M.; Kikuchi, I.; Shimanuki, H.; Kumakiri, J.; Kinoshita, K. Adhesion-prevention effects of fibrin sealants after laparoscopic myomectomy as determined by second-look laparoscopy: A prospective, randomized, controlled study. J. Reprod. Med. 2005, 50, 571–577. [Google Scholar] [PubMed]
- Fletcher, N.M.; Awonuga, A.O.; Neubauer, B.R.; Abusamaan, M.S.; Saed, M.G.; Diamond, M.P.; Saed, G.M. Shifting anaerobic to aerobic metabolism stimulates apoptosis through modulation of redox balance: Potential intervention in the pathogenesis of postop-erative adhesions. Fertil. Steril. 2015, 104, 1022–1029. [Google Scholar] [CrossRef]
- Fortin, C.N.; Saed, G.M.; Diamond, M. Predisposing factors to post-operative adhesion development. Hum. Reprod. Updat. 2015, 21, 536–551. [Google Scholar] [CrossRef]
- Goldberg, J.M.; Falcone, T.; Diamond, M.P. Current controversies in tubal disease, endometriosis, and pelvic adhesion. Fertil. Steril. 2019, 112, 417–425. [Google Scholar] [CrossRef]
- Robertson, D.; Lefebvre, G.; Clinical Practice Gynaecology Committee. Adhesion prevention in gynaecological surgery. J. Obs. Gynaecol Can. 2010, 32, 598–602. [Google Scholar] [CrossRef]
- Abuzeid, O.M.; Raju, R.; Hebert, J.; Ashraf, M.; Abuzeid, M.I. A Modified Technique of Temporary Suspension of the Ovary to the Anterior Abdominal Wall. J. Minim. Invasive Gynecol. 2018, 25, 26–27. [Google Scholar] [CrossRef] [PubMed]
- Giampaolino, P.; Della Corte, L.; Saccone, G.; Vitagliano, A.; Bifulco, G.; Calagna, G.; Carugno, J.; Di Spiezio Sardo, A. Role of Ovari-an Suspension in Preventing Postsurgical Ovarian Adhesions in Patients with Stage III-IV Pelvic Endometriosis: A Systematic Review. J. Minim. Invasive Gynecol. 2019, 26, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Hoo, W.L.; Stavroulis, A.; Pateman, K.; Saridogan, E.; Cutner, A.; Pandis, G.; Tong, E.N.; Jurkovic, D. Does ovarian suspension follow-ing laparoscopic surgery for endometriosis reduce postoperative adhesions? RCT. Hum Reprod. 2014, 29, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Heylen, S.; Brosens, I.; Puttemans, P. Clinical value and cumulative pregnancy rates following rigid salpingoscopy during laparoscopy for infertility. Hum. Reprod. 1995, 10, 2913–2916. [Google Scholar] [CrossRef] [PubMed]
- Marana, R.; Rizzi, M.; Muzii, L.; Catalano, G.F.; Caruana, P.; Mancuso, S. Correlation between the American Fertility Society classi-fications of adnexal adhesions and distal tubal occlusion, salpingoscopy, and reproductive outcome in tubal surgery. Fertil. Steril. 1995, 64, 924–929. [Google Scholar] [CrossRef]
- Marana, R.; Catalano, G.; Muzii, L.; Caruana, P.; Margutti, F.; Mancuso, S. The prognostic role of salpingoscopy in laparoscopic tubal surgery. Hum. Reprod. 1999, 14, 2991–2995. [Google Scholar] [CrossRef][Green Version]
- Marana, R.; Catalano, G.F.; Muzii, L. Salpingoscopy. Curr. Opin. Obs. Gynecol. 2003, 15, 333–336. [Google Scholar] [CrossRef]
- Nian, L.; Yang, D.H.; Zhang, J.; Zhao, H.; Zhu, C.F.; Dong, M.F.; Ai, Y. Analysis of the Clinical Efficacy of Laparoscopy and Hyster-oscopy in the Treatment of Tubal-Factor Infertility. Front. Med. 2021, 8, 712222. [Google Scholar] [CrossRef]
- Tran, D.K. Can open tubal microsurgery still be helpful in tubal infertility treatment? Gynecol. Surg. 2010, 7, 385–400. [Google Scholar] [CrossRef]
- Chu, J.; Harb, H.M.; Gallos, I.D.; Dhillon, R.; Al-Rshoud, F.M.; Robinson, L.; Coomarasamy, A. Salpingostomy in the treatment of hy-drosalpinx: A systematic review and meta-analysis. Hum. Reprod. 2015, 30, 1882e95. [Google Scholar] [CrossRef]
- Nackley, A.C.; Muasher, S.J. The significance of hydrosalpinx in in vitro fertilization. Fertil. Steril. 1998, 69, 373–384. [Google Scholar] [CrossRef]
- Ponomarev, V.V.; Zhuyko, A.A.; Artyushkov, V.V.; Bashirov, E.V.; Vengerenko, M.E. Our experience in laparoscopic treatment of tubo—Peritoneal infertility. Gynecol. Surg. 2009, 6, S149–S150. [Google Scholar]
- Chandra, A.; Copen, C.E.; Stephen, E.H. Infertility service use in the United States: Data from the National Survey of Family Growth, 1982–2010. Natl. Health Stat. Rep. 2014, 73, 1–21. [Google Scholar]
- Society for Assisted Reproductive Technology. Clinic Summary Report. Available online: https://www.sartcorsonline.com/rptCSR_PublicMultYear.aspx?Clin-icPKID=0 (accessed on 15 April 2022).
- Sharma, V.; Allgar, V.; Rajkhowa, M. Factors influencing the cumulative conception rate and discontinuation of in vitro fertilization treatment for infertility. Fertil. Steril. 2002, 78, 40–46. [Google Scholar] [CrossRef]
- Rajkhowa, M.; McConnell, A.; Thomas, G. Reasons for discontinuation of IVF treatment: A questionnaire study. Hum. Reprod. 2005, 21, 358–363. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Prevention and treatment of moderate and severe ovarian hyper-stimulation syndrome: A guideline. Fertil. Steril. 2016, 106, 1634–1647. [Google Scholar] [CrossRef]
- Hansen, M.; Bower, C.; Milne, E.; de Klerk, N.; Kurinczuk, J. Assisted reproductive technologies and the risk of birth defects—A systematic review. Hum. Reprod. 2005, 20, 328–338. [Google Scholar] [CrossRef]
- McDonald, S.D.; Murphy, K.; Beyene, J.; Ohlsson, A. Perinatel outcomes of singleton pregnancies achieved by in vitro fertilization: A systematic review and meta-analysis. J. Obs. Gynaecol. Can. 2005, 27, 449–459. [Google Scholar] [CrossRef]
- Jackson, R.A.; Gibson, K.A.; Wu, Y.W.; Croughan, M.S. Perinatal outcomes in singletons following in vitro fertilization: A me-ta-analysis. Obs. Gynecol. 2004, 103, 551–563. [Google Scholar] [CrossRef]
- Källén, B.; Finnström, O.; Lindam, A.; Nilsson, E.; Nygren, K.G.; Otterblad, P.O. Congenital malformations in infants born after in vitro fertilization in Sweden. Birth Defects Res. A. Clin. Mol. Teratol. 2010, 88, 137–143. [Google Scholar]
- El-Chaar, D.; Yang, Q.; Gao, J.; Bottomley, J.; Leader, A.; Wen, S.W.; Walker, M. Risk of birth defects increased in pregnancies con-ceived by assisted human reproduction. Fertil. Steril. 2009, 92, 1557–1561. [Google Scholar] [CrossRef] [PubMed]
- Giorgione, V.; Parazzini, F.; Fesslova, V.; Cipriani, S.; Candiani, M.; Inversetti, A.; Sigismondi, C.; Tiberio, F.; Cavoretto, P. Congenital heart defects in IVF/ICSI pregnancy: Systematic review and meta-analysis. Ultrasound Obs. Gynecol. 2018, 51, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Boulet, S.L.; Kirby, R.S.; Reefhuis, J.; Zhang, Y.; Sunderam, S.; Cohen, B.; Bernson, D.; Copeland, G.; Bailey, M.; Jamieson, D.; et al. Assisted Reproductive Technology and Birth Defects Among Liveborn Infants in Florida, Massachusetts, and Michigan, 2000–2010. JAMA Pediatr. 2016, 170, e154934. [Google Scholar] [CrossRef] [PubMed]
- Uk, A.; Collardeau-Frachon, S.; Scanvion, Q.; Michon, L.; Amar, E. Assisted Reproductive Technologies and imprinting disorders: Results of a study from a French congenital malformations registry. Eur. J. Med. Genet. 2018, 61, 518–523. [Google Scholar] [CrossRef]
- Abrão, M.S.; Muzii, L.; Marana, R. Anatomical causes of female infertility and their management. Int. J. Gynecol. Obstet. 2013, 123 (Suppl. 2), S18–S24. [Google Scholar] [CrossRef]
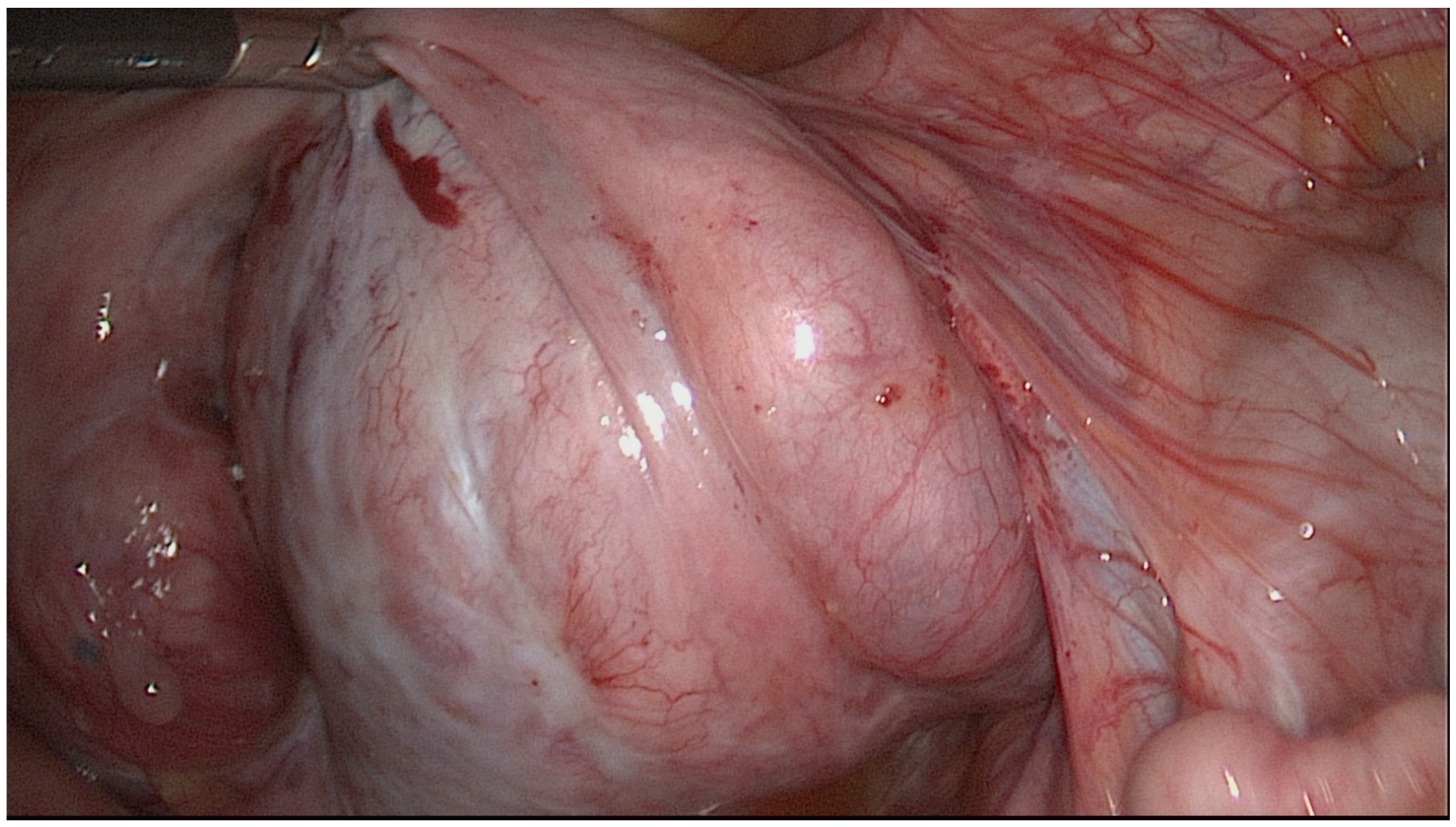
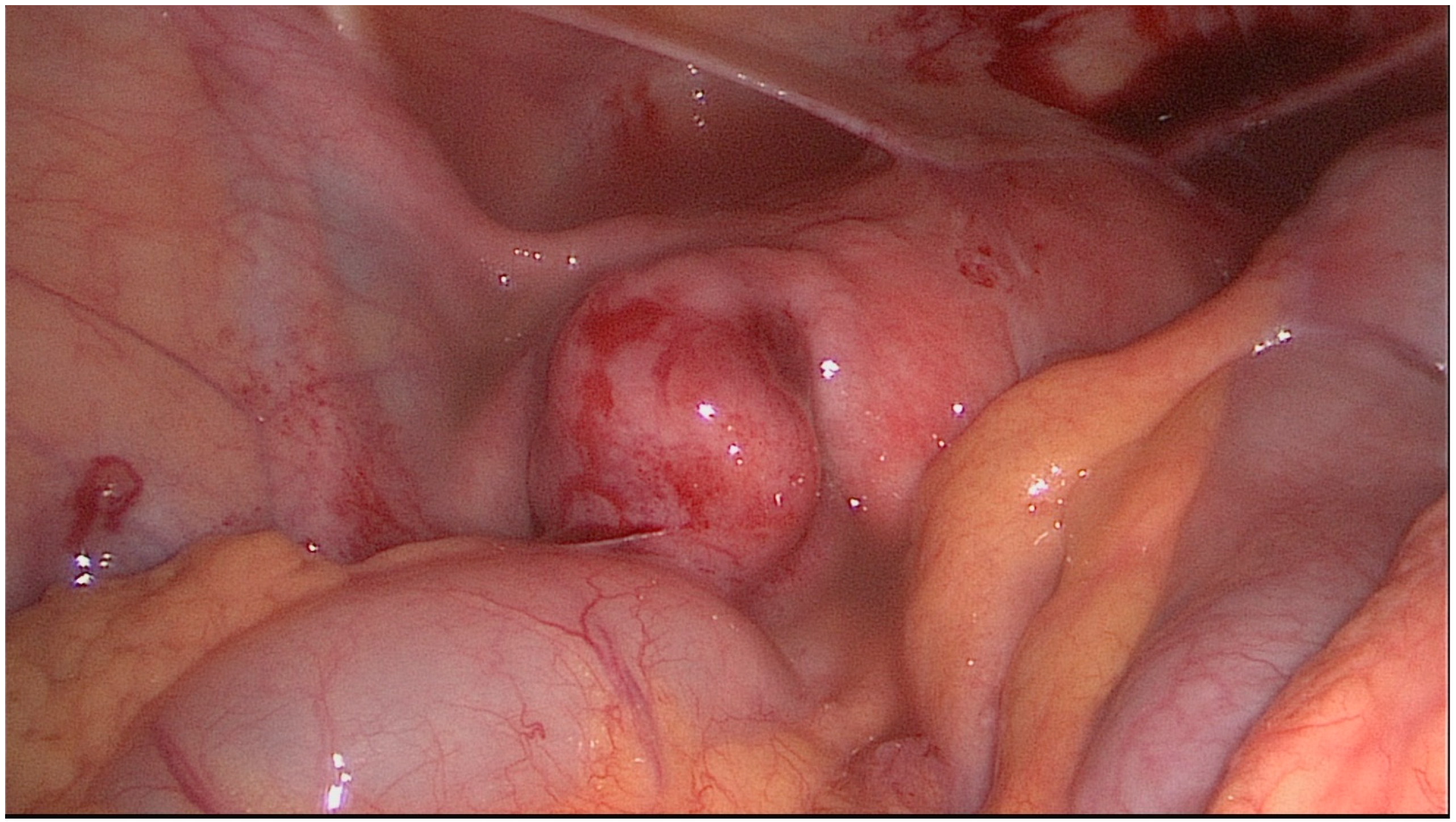
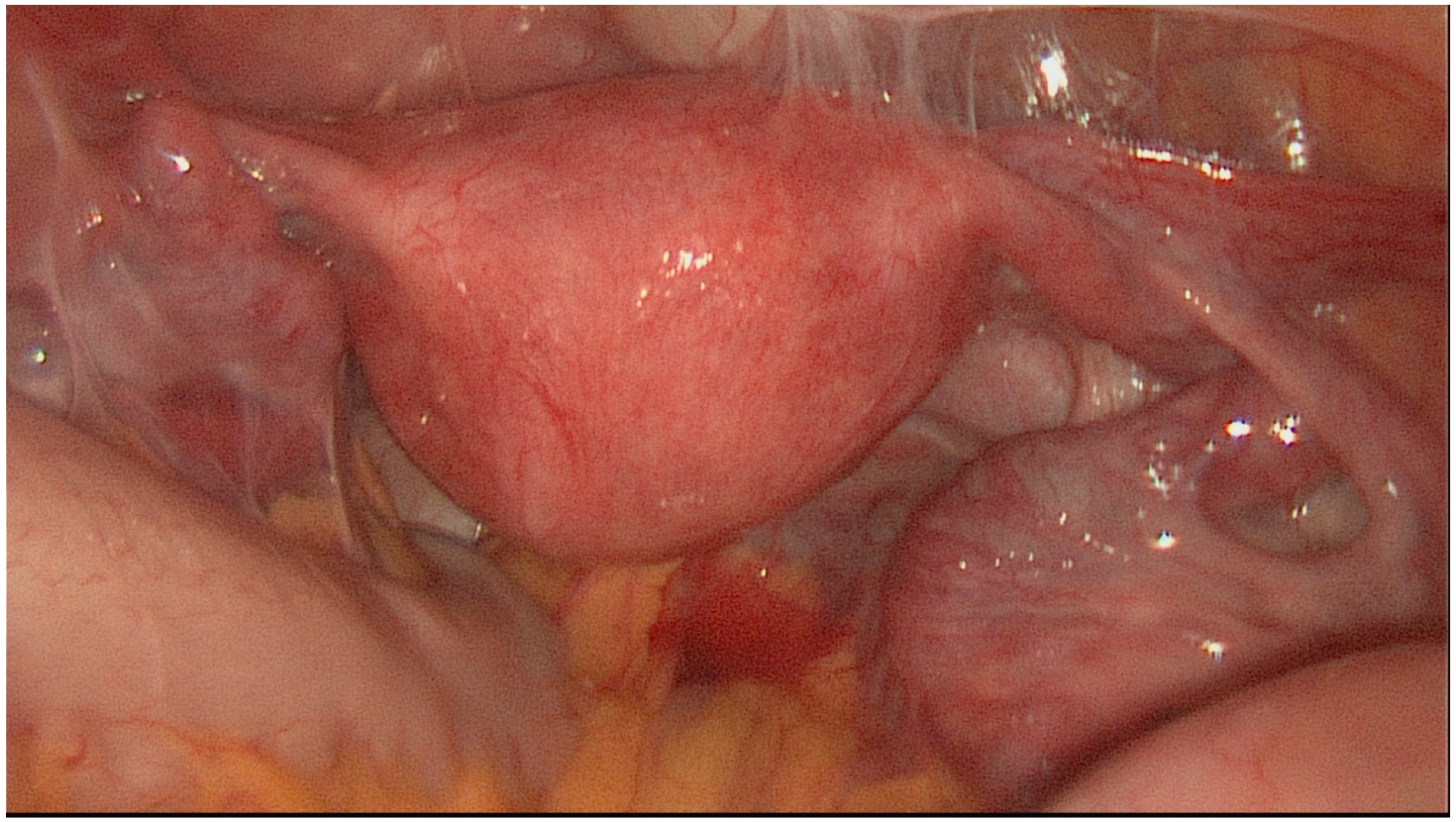
| Stage I | Thin-walled hydrosalpinx with little or no fibrosis. Mucosa thrown into folds with no flattened areas. Adhesions (if present) flimsy and limited to the ampulla and ovary only. Ovary present and mainly free. |
| Stage II | Thick-walled hydrosalpinx with good mucosa. Mucosa flattened with attenuated or few folds but thin-walled areas. Mucosal fold markedly adherent in lumen. Fibrous thick adhesions involving tube and/or ovary. Ovary present and mainly free. |
| Stage III | Combination of thick-walled hydrosalpinx with marked mucosal damage or thick fibrous adhesions. Clean hydrosalpinx with thin wall but with nodularity of patent isthmus. Ovary incarcerated against pelvic side wall or absent on that side. |
| Stage IV | Tubo-ovarian mass or fibrous, adherent hydrosalpinx with incarcerated ovary and/or isthmic damage. |
| Minor disease/Grade I |
| Tubal fibrosis absent even if occluded (proximally) Tubal distension absent even if occluded (distally) Mucosal appearances favourable Adhesions (peritubal–ovarian) flimsy |
| Intermediate disease/Grade II |
| Unilateral severe tubal damage With or without contralateral minor disease “Limited” dense adhesions of tubes and/or ovaries |
| Severe disease/Grade III |
| Bilateral severe tubal damage Tubal fibrosis extensive Tubal distension >1.5 cm Mucosal appearance abnormal Bipolar occlusion “Extensive” dense adhesions |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obrzut, B.; Obrzut, M. Is There Still a Place for Reconstructive Surgery in Distal Tubal Disease? J. Clin. Med. 2022, 11, 3278. https://doi.org/10.3390/jcm11123278
Obrzut B, Obrzut M. Is There Still a Place for Reconstructive Surgery in Distal Tubal Disease? Journal of Clinical Medicine. 2022; 11(12):3278. https://doi.org/10.3390/jcm11123278
Chicago/Turabian StyleObrzut, Bogdan, and Marzanna Obrzut. 2022. "Is There Still a Place for Reconstructive Surgery in Distal Tubal Disease?" Journal of Clinical Medicine 11, no. 12: 3278. https://doi.org/10.3390/jcm11123278
APA StyleObrzut, B., & Obrzut, M. (2022). Is There Still a Place for Reconstructive Surgery in Distal Tubal Disease? Journal of Clinical Medicine, 11(12), 3278. https://doi.org/10.3390/jcm11123278







