In Search for the Missing Link in APECED-like Conditions: Analysis of the AIRE Gene in a Series of 48 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Molecular Studies
AIRE Gene Screening
2.3. Statistical Analysis
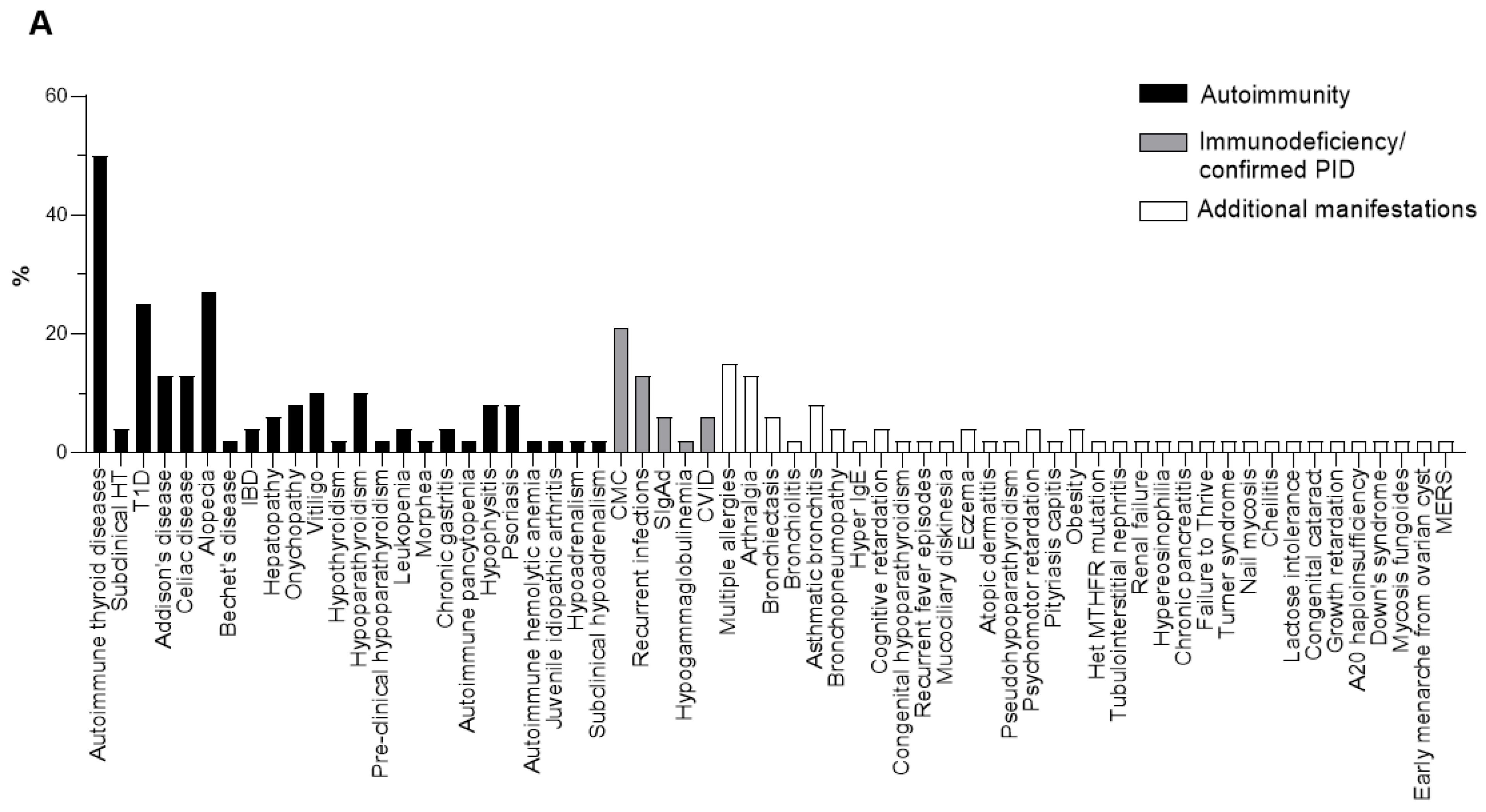
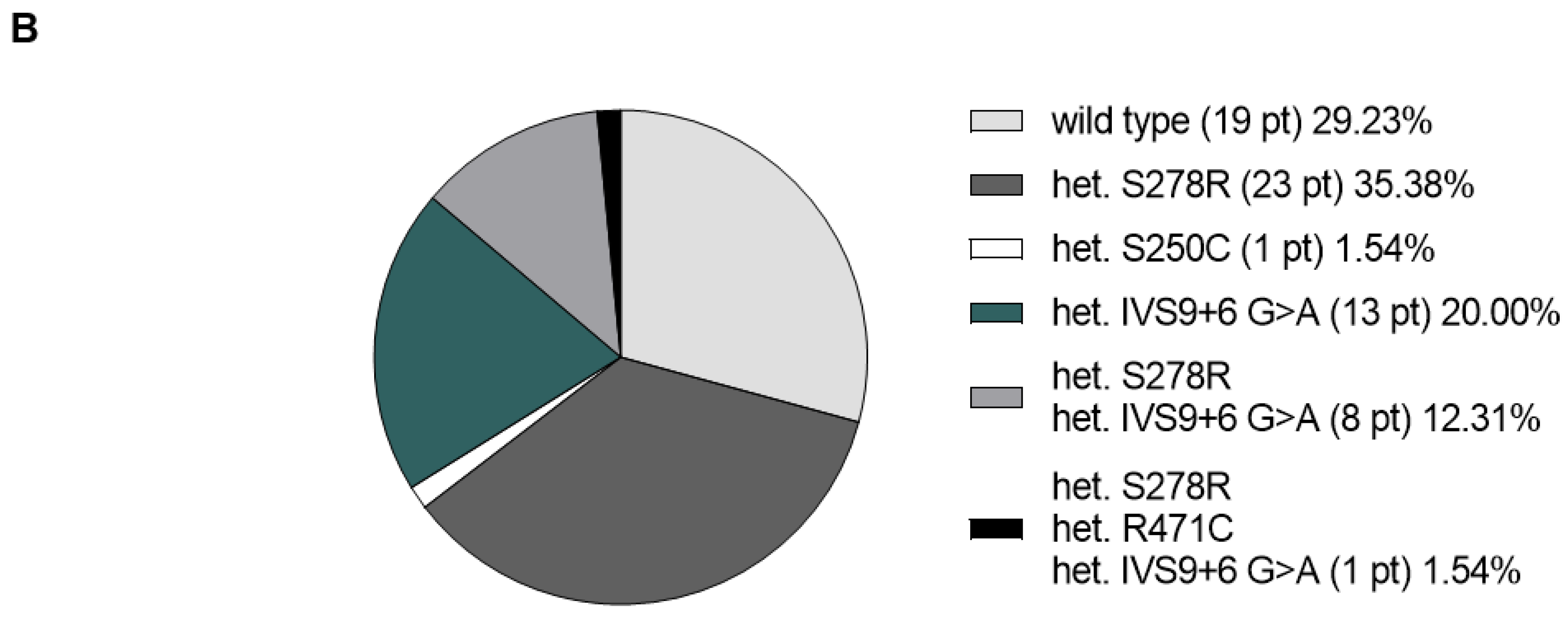
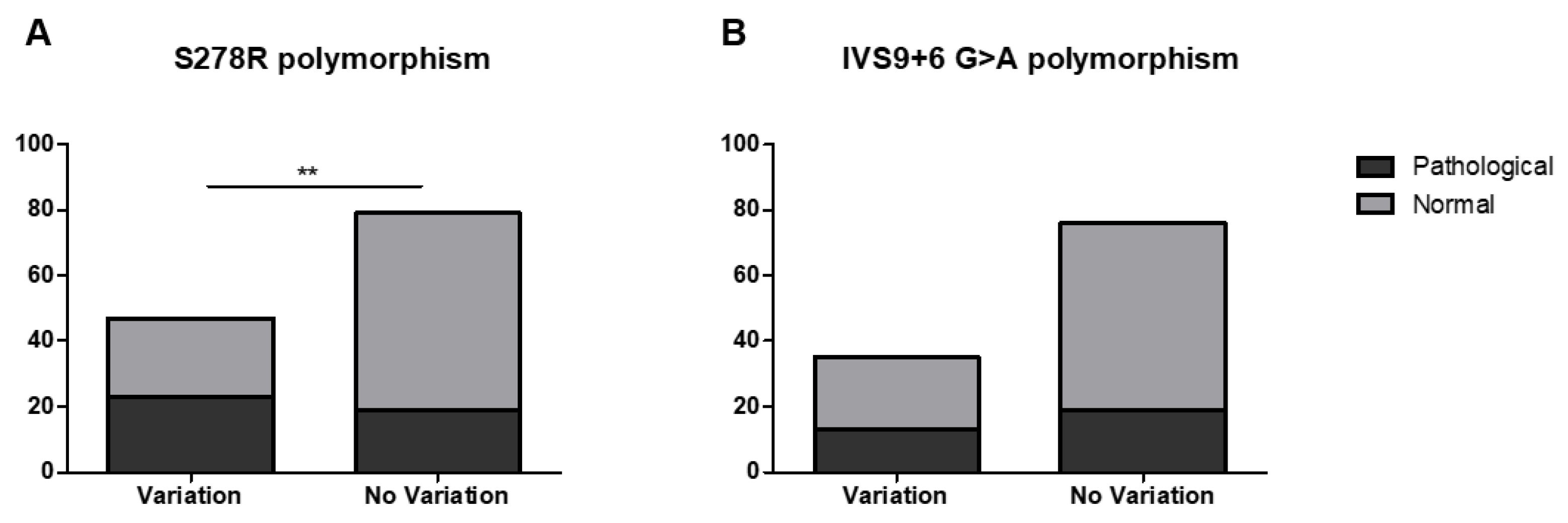
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fierabracci, A.; Milillo, A.; Locatelli, F.; Fruci, D. The putative role of endoplasmic reticulum aminopeptidases in autoimmunity: Insights from genomic-wide association studies. Autoimmun. Rev. 2012, 12, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Amaya-Uribe, L.; Rojas, M.; Azizi, G.; Anaya, J.M.; Gershwin, M.E. Primary immunodeficiency and autoimmunity: A comprehensive review. J. Autoimmun. 2019, 99, 52–72. [Google Scholar] [CrossRef]
- Husebye, E.S.; Anderson, M.S.; Kämpe, O. Autoimmune Polyendocrine Syndromes. N. Engl. J. Med. 2018, 22, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Betterle, C.; Sabbadin, C.; Scaroni, C.; Presotto, F. Autoimmune polyendocrine syndromes (APS) or multiple autoimmune syndromes (MAS) an overview. In Polyendocrine Disorders and Endocrine Neoplastic Syndromes; Colao, A.M., Jaffrain-Rea, M.L., Beckers, A., Eds.; Springer: Cham, Switzerland, 2019; pp. 1–50. [Google Scholar] [CrossRef]
- Schmidt, R.E.; Grimbacher, B.; Witte, T. Autoimmunity and primary immunodeficiency: Two sides of the same coin? Nat. Rev. Rheumatol. 2017, 19, 7–18. [Google Scholar] [CrossRef]
- Walter, E.J.; Ayala, I.A.; Milojevic, D. Autoimmunity as a continuum in primary immunodeficiency. Curr. Opin. Pediatr. 2019, 31, 851–862. [Google Scholar] [CrossRef]
- Reits, E.; Neefjes, J. HLA molecules in transplantation, autoimmunity and infection control: A comic book adventure. HLA 2022. online ahead of print. [Google Scholar] [CrossRef]
- Tobón, G.J.; Pers, J.O.; Cañas, C.A.; Rojas-Villarraga, A.; Youinou, P.; Anaya, J.M. Are autoimmune diseases predictable? Autoimmun. Rev. 2012, 11, 259–266. [Google Scholar] [CrossRef]
- Hosseini, A.; Gharibi, T.; Marofi, F.; Babaloo, Z.; Baradaran, B. CTLA-4: From mechanism to autoimmune therapy. Int. Immunopharmacol. 2020, 80, 106221. [Google Scholar] [CrossRef]
- Gough, S.C.; Walker, L.S.; Sansom, D.M. CTLA4 gene polymorphism and autoimmunity. Immunol. Rev. 2005, 204, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Kailashiya, V.; Sharma, H.B.; Kailashiya, J. Role of CTLA4 A49G polymorphism in systemic lupus erythematosus and its geographical distribution. J. Clin. Pathol. 2019, 72, 659–662. [Google Scholar] [CrossRef]
- Wing, J.B.; Tanaka, A.; Sakaguchi, S. Human FOXP3+ Regulatory T Cell Heterogeneity and Function in Autoimmunity and Cancer. Immunity 2019, 50, 302–316. [Google Scholar] [CrossRef]
- Mailer, R.K.; Mailer, R.K. IPEX as a Consequence of Alternatively Spliced FOXP3. Front. Pediatr. 2020, 8, 594375. [Google Scholar] [CrossRef]
- Dikiy, S.; Li, J.; Bai, L.; Jiang, M.; Janke, L.; Zong, X.; Hao, X.; Hoyos, B.; Wang, Z.M.; Xu, B.; et al. A distal Foxp3 enhancer enables interleukin-2 dependent thymic Treg cell lineage commitment for robust immune tolerance. Immunity 2021, 54, 931–946.e11. [Google Scholar] [CrossRef] [PubMed]
- Durães, C.; Moreira, C.S.; Alvelos, I.; Mendes, A.; Santos, L.R.; Machado, J.C.; Melo, M.; Esteves, C.; Neves, C.; Sobrinho-Simões, M.; et al. Polymorphisms in the TNFA and IL6 genes represent risk factors for autoimmune thyroid disease. PLoS ONE 2014, 9, e105492. [Google Scholar] [CrossRef]
- Abd El-Raheem, T.; Mahmoud, R.H.; Hefzy, E.M.; Masoud, M.; Ismail, R.; Aboraia, N.M.M. Tumor necrosis factor (TNF)-α- 308 G/A gene polymorphism (rs1800629) in Egyptian patients with alopecia areata and vitiligo, a laboratory and in silico analysis. PLoS ONE 2020, 15, e0240221. [Google Scholar] [CrossRef] [PubMed]
- Gianchecchi, E.; Palombi, M.; Fierabracci, A. The putative role of the C1858T polymorphism of protein tyrosine phosphatase PTPN22 gene in autoimmunity. Autoimmun. Rev. 2013, 12, 717–725. [Google Scholar] [CrossRef]
- Vang, T.; Congia, M.; Macis, M.D.; Musumeci, L.; Orrú, V.; Zavattari, P.; Nika, K.; Tautz, L.; Taskén, K.; Cucca, F.; et al. Autoimmune-associated lymphoid tyrosine phosphatase is a gain-of-function variant. Nat. Genet. 2005, 37, 1317–1319. [Google Scholar] [CrossRef]
- Fierabracci, A.; Pellegrino, M.; Frasca, F.; Kilic, S.S.; Betterle, C. APECED in Turkey: A case report and insights on genetic and phenotypic variability. Clin. Immunol. 2018, 194, 60–66. [Google Scholar] [CrossRef]
- Fierabracci, A. Recent insights into the role and molecular mechanisms of the autoimmune regulator (AIRE) gene in autoimmunity. Autoimmun. Rev. 2011, 10, 137–143. [Google Scholar] [CrossRef]
- Garelli, S.; Dalla Costa, M.; Sabbadin, C.; Barollo, S.; Rubin, B.; Scarpa, R.; Masiero, S.; Fierabracci, A.; Bizzarri, C.; Crinò, A.; et al. Autoimmune polyendocrine syndrome type 1: An Italian survey on 158 patients. J. Endocrinol. Investig. 2021, 44, 2493–2510. [Google Scholar] [CrossRef]
- Kekäläinen, E.; Tuovinen, H.; Joensuu, J.; Gylling, M.; Franssila, R.; Pöntynen, N.; Talvensaari, K.; Perheentupa, J.; Miettinen, A.; Arstila, T.P. A defect of regulatory T cells in patients with autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy. J. Immunol. 2007, 178, 1208–1215. [Google Scholar] [CrossRef]
- Fierabracci, A.; Arena, A.; Toto, F.; Gallo, N.; Puel, A.; Migaud, M.; Kumar, M.; Chengappa, K.G.; Gulati, R.; Negi, V.S.; et al. Autoimmune polyendocrine syndrome type 1 (APECED) in the Indian population: Case report and review of a series of 45 patients. J. Endocrinol. Investig. 2021, 44, 661–677. [Google Scholar] [CrossRef]
- Larosa, M.D.P.; Mackenzie, R.; Burne, P.; Garelli, S.; Barollo, S.; Masiero, S.; Rubin, B.; Chen, S.; Furmaniak, J.; Betterle, C.; et al. Assessment of autoantibodies to interferon-ω in patients with autoimmune polyendocrine syndrome type 1: Using a new immunoprecipitation assay. Clin. Chem. Lab. Med. 2017, 55, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Azizi, G.; Yazdani, R.; Rae, W.; Abolhassani, H.; Rojas, M.; Aghamohammadi, A.; Anaya, J.M. Monogenic polyautoimmunity in primary immunodeficiency diseases. Autoimmun. Rev. 2018, 17, 1028–1039. [Google Scholar] [CrossRef] [PubMed]
- Fierabracci, A. The role of heterozygous mutations of the autoimmune regulator gene (AIRE) in non-APECED autoimmunity: A comment on recent findings. Clin. Endocrinol. 2011, 74, 532–533. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Takahama, Y. Thymic epithelial cells: Working class heroes for T cell development and repertoire selection. Trends Immunol. 2012, 33, 256–263. [Google Scholar] [CrossRef]
- Sedivá, A.; Ciháková, D.; Lebl, J. Immunological findings in patients with autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy (APECED) and their family members: Are heterozygotes subclinically affected? J. Pediatr. Endocrinol. Metab. 2002, 15, 1491–1496. [Google Scholar] [CrossRef]
- Cervato, S.; Morlin, L.; Albergoni, M.P.; Masiero, S.; Greggio, N.; Meossi, C.; Chen, S.; Larosa, M.D.P.; Furmaniak, J.; Smith, B.R.; et al. AIRE gene mutations and autoantibodies to interferon omega in patients with chronic hypoparathyroidism without APECED. Clin. Endocrinol. 2010, 73, 630–636. [Google Scholar] [CrossRef]
- Tóth, B.; Wolff, A.B.S.; Halász, Z.; Tar, A.; Szüts, P.; Ilyés, I.; Erdos, M.; Szegedi, G.; Husebye, E.S.; Zeher, M.; et al. Novel sequence variation of AIRE and detection of interferon-omega antibodies in early infancy. Clin. Endocrinol. 2010, 72, 641–647. [Google Scholar] [CrossRef]
- Alimohammadi, M.; Björklund, P.; Hallgren, A.; Pöntynen, N.; Szinnai, G.; Shikama, N.; Keller, M.P.; Ekwall, O.; Kinkel, S.A.; Husebye, E.S.; et al. Autoimmune polyendocrine syndrome type 1 and NALP5, a parathyroid autoantigen. N. Engl. J. Med. 2008, 358, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Oftedal, B.E.; Wolff, A.S.; Bratland, E.; Kämpe, O.; Perheentupa, J.; Myhre, A.G.; Meager, A.; Purushothaman, R.; Ten, S.; Husebye, E.S. Radioimmunoassay for autoantibodies against interferon omega; its use in the diagnosis of autoimmune polyendocrine syndrome type I. Clin. Immunol. 2008, 129, 163–169. [Google Scholar] [CrossRef]
- Gossard, A.A.; Lindor, K.D. Autoimmune hepatitis: A review. J. Gastroenterol. 2012, 47, 498–503. [Google Scholar] [CrossRef]
- Vogel, A.; Liermann, H.; Harms, A.; Strassburg, C.P.; Manns, M.P.; Obermayer-Straub, P. Autoimmune regulator AIRE: Evidence for genetic differences between autoimmune hepatitis and hepatitis as part of the autoimmune polyglandular syndrome type 1. Hepatology 2001, 33, 1047–1052. [Google Scholar] [CrossRef] [PubMed]
- Lankisch, T.O.; Strassburg, C.P.; Debray, D.; Manns, M.P.; Jacquemin, E. Detection of autoimmune regulator gene mutations in children with type 2 autoimmune hepatitis and extrahepatic immune-mediated diseases. J. Pediatr. 2005, 146, 839–842. [Google Scholar] [CrossRef]
- Lankisch, T.O.; Mourier, O.; Sokal, E.M.; Habes, D.; Lacaille, F.; Bridoux-Henno, L.; Hermeziu, B.; Lenaerts, C.; Strassburg, C.P.; Jacquemin, E. AIRE gene analysis in children with autoimmune hepatitis type I or II. J. Pediatr. Gastroenterol. Nutr. 2009, 48, 498–500. [Google Scholar] [CrossRef] [PubMed]
- Tazi-Ahnini, R.; Cork, M.J.; Gawkrodger, D.J.; Birch, M.P.; Wengraf, D.; McDonagh, A.J.; Messenger, A.G. Role of the autoimmune regulator (AIRE) gene in alopecia areata: Strong association of a potentially functional AIRE polymorphism with alopecia universalis. Tissue Antigens 2002, 60, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.S.; Heino, M.; Peterson, P.; Mittaz, L.; Lalioti, M.D.; Betterle, C.; Cohen, A.; Seri, M.; Lerone, M.; Romeo, G.; et al. Common mutations in autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy patients of different origins. Mol. Endocrinol. 1998, 12, 1112–1119. [Google Scholar] [CrossRef]
- Faiyaz-Ul-Haque, M.; Bin-Abbas, B.; Al-Abdullatif, A.; Abdullah Abalkhail, H.; Toulimat, M.; Al-Gazlan, S.; Almutawa, A.M.; Al-Sagheir, A.; Peltekova, I.; Al-Dayel, F. Novel and recurrent mutations in the AIRE gene of autoimmune polyendocrinopathy syndrome type 1 (APS1) patients. Clin. Genet. 2009, 76, 431–440. [Google Scholar] [CrossRef]
- Pforr, J.; Blaumeiser, B.; Becker, T.; Freudenberg-Hua, Y.; Hanneken, S.; Eigelshoven, S.; Cuyt, I.; De Weert, J.; Lambert, J.; Kruse, R.; et al. Investigation of the p.Ser278Arg polymorphism of the autoimmune regulator (AIRE) gene in alopecia areata. Tissue Antigens. 2006, 68, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Ferrera, F.; Rizzi, M.; Sprecacenere, B.; Balestra, P.; Sessarego, M.; Di Carlo, A.; Filaci, G.; Gabrielli, A.; Ravazzolo, R.; Indiveri, F. AIRE gene polymorphisms in systemic sclerosis associated with autoimmune thyroiditis. Clin. Immunol. 2007, 122, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, B.; Imrie, H.; Geatch, D.R.; Perros, P.; Ball, S.G.; Baylis, P.H.; Carr, D.; Hurel, S.J.; James, R.A.; Kelly, W.F. Association analysis of the cytotoxic T lymphocyte antigen-4 (CTLA-4) and autoimmune regulator-1 (AIRE-1) genes in sporadic autoimmune Addison’s disease. J. Clin. Endocrinol. Metab. 2000, 85, 688–691. [Google Scholar] [CrossRef][Green Version]
- Nithiyananthan, R.; Heward, J.M.; Allahabadia, A.; Barnett, A.H.; Franklyn, J.A.; Gough, S.C. A heterozygous deletion of the autoimmune regulator (AIRE1) gene, autoimmune thyroid disease, and type 1 diabetes: No evidence for association. J. Clin. Endocrinol. Metab. 2000, 85, 1320–1322. [Google Scholar] [CrossRef] [PubMed]
- Török, H.P.; Tonenchi, L.; Glas, J.; Schiemann, U.; Folwaczny, C. No significant association between mutations in exons 6 and 8 of the autoimmune regulator (AIRE) gene and inflammatory bowel disease. Eur. J. Immunogenet. 2004, 31, 83–86. [Google Scholar] [CrossRef]
- Turunen, J.A.; Wessman, M.; Forsblom, C.; Kilpikari, R.; Parkkonen, M.; Pöntynen, N.; Ilmarinen, T.; Ulmanen, I.; Peltonen, L.; Groop, P.H. Association analysis of the AIRE and insulin genes in Finnish type 1 diabetic patients. Immunogenetics 2006, 58, 331–338. [Google Scholar] [CrossRef]
- Palma, A.; Gianchecchi, E.; Palombi, M.; Luciano, R.; Di Carlo, P.; Crinò, A.; Cappa, M.; Fierabracci, A. Analysis of the autoimmune regulator gene in patients with autoimmune non-APECED polyendocrinopathie. Genomics 2013, 102, 163–168. [Google Scholar] [CrossRef]
- Oftedal, B.E.; Hellesen, A.; Erichsen, M.M.; Bratland, E.; Vardi, A.; Perheentupa, J.; Kemp, E.H.; Fiskerstrand, T.; Viken, M.K.; Weetman, A.P.; et al. Dominant Mutations in the Autoimmune Regulator AIRE Are Associated with Common Organ-Specific Autoimmune Diseases. Immunity 2015, 42, 1185–1196. [Google Scholar] [CrossRef]
- Cetani, F.; Barbesino, G.; Borsari, S.; Pardi, E.; Cianferotti, A.; Pinchera, C.; Marcocci, F. A novel mutation of the autoimmune regulator gene in an Italian kindred with autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy, acting in a dominant fashion and strongly cosegregating with hypothyroid autoimmune thyroiditis. J. Clin. Endocrinol. Metab. 2001, 86, 4747–4752. [Google Scholar] [CrossRef]
- Colobran, R.; Giménez-Barcons, M.; Marín-Sánchez, A.; Porta-Pardo, E.; Pujol-Borrell, R. AIRE genetic variants and predisposition to polygenic autoimmune disease: The case of Graves’ disease and a systematic literature review. Hum. Immunol. 2016, 77, 643–651. [Google Scholar] [CrossRef]
- Laisk, T.; Lepamets, M.; Koel, M.; Abner, E.; Mägi, R.; Estonian Biobank Research Team. Genome-wide association study identifies five risk loci for pernicious anemia. Nat. Commun. 2021, 12, 3761. [Google Scholar] [CrossRef]
- Eriksson, D.; Røyrvik, E.C.; Aranda-Guillén, M.; Berger, A.H.; Landegren, N.; Artaza, H.; Hallgren, A.; Grytaas, M.A.; Ström, S.; Bratland, E.; et al. GWAS for autoimmune Addison’s disease identifies multiple risk loci and highlights AIRE in disease susceptibility. Nat. Commun. 2021, 12, 959. [Google Scholar] [CrossRef]
- Fierabracci, A. Type 1 Diabetes in Autoimmune Polyendocrinopathy-Candidiasis-Ectodermal Dystrophy Syndrome (APECED): A “Rare” Manifestation in a “Rare” Disease. Int. J. Mol. Sci. 2016, 17, 1106. [Google Scholar] [CrossRef] [PubMed]
- Odineal, D.D.; Gershwin, M.E. The Epidemiology and Clinical Manifestations of Autoimmunity in Selective IgA Deficiency. Clin. Rev. Allergy Immunol. 2020, 58, 107–133. [Google Scholar] [CrossRef]
- Bellacchio, E.; Palma, A.; Corrente, S.; Di Girolamo, F.; Kemp, H.E.; Di Matteo, G.; Comelli, L.; Carsetti, R.; Cascioli, S.; Cancrini, C.; et al. The possible implication of the S250C variant of the autoimmune regulator protein in a patient with autoimmunity and immunodeficiency: In silico analysis suggests a molecular pathogenic mechanism for the variant. Gene 2014, 549, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Picard, C.; Gaspar, H.B.; Al-Herz, W.; Bousfiha, A.; Casanova, J.L.; Chatila, T.; Crow, Y.J.; Cunningham-Rundles, C.; Etzioni, A.; Franco, J.L. International Union of Immunological Societies: 2017 Primary Immunodeficiency Diseases Committee Report on Inborn Errors of Immunity. J. Clin. Immunol. 2018, 38, 96–128. [Google Scholar] [CrossRef] [PubMed]
- Tangye, S.G.; Al-Herz, W.; Aziz Bousfiha, A.; Cunningham-Rundles, C.; Franco, J.L.; Holland, S.M.; Klein, C.; Morio, T.; Oksenhendler, E.; Picard, C.; et al. The Ever-Increasing Array of Novel Inborn Errors of Immunity: An Interim Update by the IUIS Committee. J. Clin. Immunol. 2021, 41, 666–679. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Neovius, M.; Hammarström, L. Association between IgA deficiency & other autoimmune conditions: A population-based matched cohort study. J. Clin. Immunol. 2014, 34, 444–451. [Google Scholar] [CrossRef] [PubMed]
| Patient | Gender | Age at Referral (Years) | Diseases | Auto Abs | AIRE Gene Pattern * |
|---|---|---|---|---|---|
| 1 | M | 5.42 | Multiple allergies (5), bronchiectasis (5), asthmatic bronchitis (5), hyper IgE (5), alopecia (10), CMC (10) | TgAbs, TPOAbs, IAA, IA2Abs, TRGAbs, ASCA neg; AMA, ASMA pos | het. S278R |
| 2 | F | 3.5 | Cognitive retardation (1), CMC (3), arthralgia (3), congenital hypothyroidism (3), bronchiolitis (6), Behcet’s disease (11), very early onset inflammatory bowel disease (VEO-IBD) | TgAbs, TPOAbs, ANCA, ASCA, ENA, dsDNAAbs neg; ANA pos | het. S278R |
| 3 | M | 6.19 | Selective IgA deficiency (6), Crohn’s disease (6), arthralgia (7), T1D (9), pharmacological polyallergy (12) | IAA, IA2Abs, GADA, TRGAbs, anti-adrenal Abs, ANA, ANCA, dsDNAAbs neg | het. S278R het. R471C het. IVS9+6 G>A |
| 4 | M | 3.7 | Arthralgia (1), HT (4), recurrent fever episodes (4), selective IgA deficiency (4), vitiligo (5) | IAA, IA2Abs, GADA, TRGAbs, 21OHAbs, dsDNAAbs neg; TgAbs, TPOAbs, ANA pos | het. IVS9+6 G>A |
| 5 | F | 5.91 | Bronchopneumopathy (5), arthralgia (6), combined immunodeficiency (7), hepatopathy (7), mucociliary dyskinesia (7), eczematous dermatitis (11) | TgAbs, TPOAbs, ANCA, ASCA neg; IAA, IA2Abs, GADA, ANA, dsDNAAbs pos | het. IVS9+6 G>A |
| 6 | M | 3.57 | Atopic dermatitis (3), combined immunodeficiency (4), CMC (4) | TgAbs, TPOAbs, IAA, IA2Abs, GADA, TRGAbs, dsDNAAbs neg; ANA, ENA, RoAbs, SCL70Abs, LaAbs, SMAbs, RNPAbs, JO1Abs pos | het. S278R het. IVS9+6 G>A |
| 7 | F | 1.83 | CMC (1), pseudohypoparathyroidism (4), psychomotor retardation (4), coordination disorder (5), congenital onychopathy (12), pityriasis capitis (12), subclinical HT (12) | TgAbs, TPOAbs, ANCA, dsDNAAbs, PM/Scl 100 Abs neg; ANA pos | WT |
| 8 | M | 7.83 | Vitiligo (10), HT (10), inhalants allergy (10), selective IgA deficiency (12) | TRGAbs, anti-gliadin Abs, EMA, APCA, AMA, ANA, ANCA, ASCA, ENA, dsDNAAbs neg; TgAbs, TPOAbs pos | WT |
| 9 | M | 4.29 | Candidiasis, alopecia areata (4), subclinical HT (4) (familiarity for HT and T1D) | TgAbs, TPOAbs, IAA, IA2Abs, GADA, TRGAbs, APCA, ANA, ANCA, ENA neg | het. S278R |
| 10 | F | 2.16 | Childhood obesity (6), CMC (9), recurrent infections (9), T1D (9), preclinical hypoparathyroidism, het. MTHFR C677T homocysteine 9 mutation (9), leukopenia (10) | TgAbs, TPOAbs, anti-TSH-receptor Abs, TRGAbs, anti-gliadin Abs, ANA, anti-cardiolipin Abs, anti-beta2 glycoprotein Abs neg; IAA, GADA pos | WT |
| 11 | F | 8.3 | Tubulointerstitial nephritis (4), polyallergy (urticaria and food allergy) (9), chronic renal failure (10), frequent asthma episodes (9), bronchopneumopathy with pulmonary bronchiectasis, hypereosinophilia (9), chronic pancreatitis (10) | ANA, aPLAbs neg; ANCA, ASCA, MPOAbs, anti-cardiolipin Abs pos | WT |
| 12 | F | 10.99 | Alopecia (few months), HT (9), CMC, bronchitis, asthma, urinary tract infections, food allergies, failure to thrive, hypogammaglobulinemia, eczema, vitiligo, chronic gastritis, morphea | TgAbs, TPOAbs neg | WT |
| 13 | M | 11.18 | Addison’s disease, frequent infections, HT | Diabetes-related Abs, anti-gliadin Abs neg; TgAbs, TPOAbs, 21OHAbs pos | het. S278R |
| 14 | M | 11.18 | CMC, HT, autoimmune pancytopenia | TgAbs, TPOAbs pos | WT |
| 15 | F | 7.03 | Alopecia (7), recurrent infections | TgAbs, TPOAbs, IAA, IA2Abs, GADA, anti-adrenal Abs, ENA, ASMA, ARA, APCA, ANCA, dsDNAAbs, LKMAbs, LC1Abs, anti-ribosome Abs neg; ANA pos | WT |
| 16 | F | 10.51 | HT (11), T1D (11), CMC | TPOAbs, IA2Abs, TRGAbs neg; TgAbs, IAA, GADA pos | WT |
| 17 | F | 5.71 | Turner syndrome, alopecia (3), HT, nail mycosis, cheilitis | TgAbs, TPOAbs pos | het. IVS9+6 G>A |
| 18 | M | 2.8 | Alopecia (3), recurrent respiratory infections, overweight (5) | TgAbs, TPOAbs, IAA, GADA, TRGAbs, APCA, anti-adrenal Abs, AMA, ANA, ARA, LC1Abs, LKMAbs, anti-ribosome Abs neg; IA2Abs, ASMA, ENA pos | WT |
| 19 | F | CMC, HT, autoimmune hypophysitis | WT | ||
| 20 | M | 9.06 | Alopecia (8), CVID (10), allergic rhinitis (10), onychodystrophy (12), palmar-plantar psoriasis (12), lactose intolerance (15) [54] | TgAbs, TPOAbs, IAA, IA2Abs, GADA, APCA, ANA, ASCA, ASMA, LKMAbs, dsDNAAbs, ENA, AMA, LC1Abs, anti-ribosome Abs, SCL70Abs neg | het. S250C |
| 21 | M | 5.57 | Hypoparathyroidism (12) | TgAbs, TPOAbs, IAA, IA2Abs, GADA, 21OHAbs neg | het. S278R het. IVS9+6 G>A |
| 22 | F | 4.66 | T1D (7) | TgAbs, IA2Abs, TRGAbs neg; GADA pos | het. S278R het. IVS9+6 G>A |
| 23 | M | 2.31 | T1D (2), linguistic retardation (2), celiac disease (21) | TgAbs, TPOAbs neg; TRGAbs pos | het. S278R het. IVS9+6 G>A |
| 24 | F | 2.34 | Congenital cataract, growth retardation (5), HT (5), autoimmune haemolytic anemia (7), juvenile idiopathic arthritis (9), A20 haploinsufficiency (16) | IAA, IA2Abs, APCA, anti-adrenal Abs, AMA, ANCA, ASMA, ARA, dsDNAAbs, LKMAbs, LC1Abs, anti-ribosome Abs neg; TgAbs, TPOAbs, GADA, ANA, ENA pos | het. S278R het. IVS9+6 G>A |
| 25 | F | 3.72 | T1D, HT (21), vitiligo (21) | Anti-TSH-receptor Abs, IA2Abs, GADA, TRGAbs, anti-adrenal Abs neg; TgAbs, TPOAbs, IAA pos | het. S278R het. IVS9+6 G>A |
| 26 | F | 4.81 | T1D, Down’s syndrome, Basedow’s disease (32) | TgAbs, TPOAbs, TRGAbs, APCA, anti-adrenal Abs, AMA, ARA, LKMAbs, LC1Abs, anti-ribosome Abs neg; ASMA pos | het. S278R |
| 27 | F | 11.25 | HT, celiac disease | IAA, IA2Abs, GADA, TRGAbs neg; TgAbs, TPOAbs pos | het. S278R |
| 28 | F | 12.43 | Alopecia areata (5), parapsoriasis (12), HT (12), mycosis fungoides (13) | het. S278R | |
| 29 | M | 8.41 | Isolated hypoparathyroidism (8) | TgAbs, TPOAbs, TRGAbs, anti-adrenal Abs neg | het. S278R |
| 30 | F | 5.78 | Alopecia (6), onychodystrophy (13) | TgAbs, TPOAbs, IAA, IA2Abs, TRGAbs, APCA, anti-adrenal Abs, AMA, ASMA, ARA, LKMAbs, LC1Abs, anti-ribosome Abs neg; GADA, ANA pos | het. S278R |
| 31 | F | 10.91 | Primary hypoparathyroidism | het. S278R | |
| 32 | M | 10.08 | Hypoparathyroidism | het. S278R | |
| 33 | F | Addison’s disease, vitiligo | het. S278R het. IVS9+6 G>A | ||
| 34 | F | 14.35 | Central hypoadrenalism (15), HT (15), bronchiectasis (15), CIDP (21) | IAA, GADA, ANA, ANCA neg; TgAbs, TPOAbs pos | WT |
| 35 | F | 1 | Early menarche from ovarian cyst (9), alopecia (16), HT (17), subclinical hypoadrenalism (17) | TgAbs, TRGAbs, APCA, anti-adrenal Abs, AMA, ASMA, ARA, LKMAbs, LC1Abs, SLA/LPAbs, Sp100Abs, gp210Abs, anti-cardiolipin Abs neg; TPOAbs, ANA pos | het. S278R |
| 36 | F | Hypoparathyroidism, Addison’s disease, secondary ovarian failure | WT | ||
| 37 | F | 1 | Alopecia (3), HT (7) | TgAbs, IAA, IA2Abs, TRGAbs, APCA, anti-adrenal Abs, AMA, ASMA, ARA, LKMAbs, LC1Abs, anti-ribosome Abs, anti-cardiolipin Abs neg; TPOAbs, GADA, ANA pos | het. S278R |
| 38 | F | 6.15 | Alopecia (1), nail dystrophy, HT (5), allergic rhinitis (11), arthralgia (11), recurrent infections in pediatric age | IAA, GADA, TRGAbs, APCA, ANCA, ASMA, ARA, dsDNAAbs, LKMAbs, anti-ribosome Abs neg; TgAbs, TPOAbs, IA2Abs, ANA pos | het. IVS9+6 G>A |
| 39 | M | 3.84 | T1D, GH deficit, HT, autoimmune leukopenia | TRGAbs, anti-adrenal Abs neg; TgAbs, TPOAbs pos | het. IVS9+6 G>A |
| 40 | F | 15.42 | Addison’s disease, HT, celiac disease (4) | TgAbs, TPOAbs, IAA, IA2Abs, GADA, APCA, ANA, AMA, ASMA, LKMAbs, ARA, LC1Abs, anti-ribosome Abs neg; TRGAbs, anti-adrenal Abs pos | WT |
| 41 | M | alopecia, HT, celiac disease | IAA, IA2Abs, GADA, anti-adrenal Abs, dsDNAAbs, aPLAbs, anti-cardiolipin Abs neg; TgAbs, TPOAbs, TRGAbs, ANA, ANCA pos | het. S278R | |
| 42 | F | Addison’s disease, HT, psoriasis | WT | ||
| 43 | F | 7.97 | Hypoadrenalism (8), hypothyroidism (10) | IAA, IA2Abs, GADA, TRGAbs, AMA, ASMA, ARA, LKMAbs, LC1Abs, anti-ribosome Abs neg; TgAbs, TPOAbs, APCA, anti-adrenal Abs pos | WT |
| 44 | F | 5.64 | Celiac disease (5), T1D (7), HT (7) | TgAbs, TRGAbs neg; TPOAbs, IAA, IA2Abs, GADA pos | WT |
| 45 | F | 5.19 | T1D (4), autoimmune hepatitis (5), HT (5) | TPOAbs, TRGAbs, APCA, ANA, AMA, ASMA, ARA, ANCA, LKMAbs, LC1Abs, anti-ribosome Abs neg; TgAbs pos | WT |
| 46 | F | 1.8 | T1D (2), celiac disease (3), autoimmune hepatitis (13) | TgAbs, TPOAbs, TRGAbs, APCA, ANCA, ANA, AMA, ASMA, ARA, LKMAbs, LC1Abs, anti-ribosome Abs neg; IAA, IA2Abs, GADA pos | WT |
| 47 | F | 11.71 | Arthralgia (4), psoriasis (6), HT (9), T1D (9), gastritis (12) | TgAbs, TRGAbs, APCA, anti-adrenal Abs, dsDNAAbs, AMA, ASMA, ARA, ASCA, LKMAbs, LC1Abs, anti-ribosome Abs neg; TPOAbs, ANA, ANCA pos | WT |
| 48 | F | 14.77 | Addison’s disease, MERS | IAA, IA2Abs, GADA, APCA, ANA, ENA, AMA, ASMA, ARA, LKMAbs, LC1Abs, aPLAbs, anti-cardiolipin Abs, anti-ribosome Abs, anti-beta2 microglobulin Abs neg; TRGAbs, anti-adrenal Abs pos | het. S278R |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fierabracci, A.; Belcastro, E.; Carbone, E.; Pagliarosi, O.; Palma, A.; Pacillo, L.; Giancotta, C.; Zangari, P.; Finocchi, A.; Cancrini, C.; et al. In Search for the Missing Link in APECED-like Conditions: Analysis of the AIRE Gene in a Series of 48 Patients. J. Clin. Med. 2022, 11, 3242. https://doi.org/10.3390/jcm11113242
Fierabracci A, Belcastro E, Carbone E, Pagliarosi O, Palma A, Pacillo L, Giancotta C, Zangari P, Finocchi A, Cancrini C, et al. In Search for the Missing Link in APECED-like Conditions: Analysis of the AIRE Gene in a Series of 48 Patients. Journal of Clinical Medicine. 2022; 11(11):3242. https://doi.org/10.3390/jcm11113242
Chicago/Turabian StyleFierabracci, Alessandra, Eugenia Belcastro, Elena Carbone, Olivia Pagliarosi, Alessia Palma, Lucia Pacillo, Carmela Giancotta, Paola Zangari, Andrea Finocchi, Caterina Cancrini, and et al. 2022. "In Search for the Missing Link in APECED-like Conditions: Analysis of the AIRE Gene in a Series of 48 Patients" Journal of Clinical Medicine 11, no. 11: 3242. https://doi.org/10.3390/jcm11113242
APA StyleFierabracci, A., Belcastro, E., Carbone, E., Pagliarosi, O., Palma, A., Pacillo, L., Giancotta, C., Zangari, P., Finocchi, A., Cancrini, C., Delfino, D. V., Cappa, M., & Betterle, C. (2022). In Search for the Missing Link in APECED-like Conditions: Analysis of the AIRE Gene in a Series of 48 Patients. Journal of Clinical Medicine, 11(11), 3242. https://doi.org/10.3390/jcm11113242








