Altered Cerebral Curvature in Preterm Infants Is Associated with the Common Genetic Variation Related to Autism Spectrum Disorder and Lipid Metabolism
Abstract
:1. Introduction
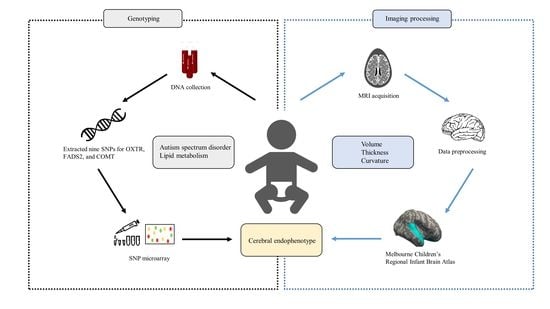
2. Materials and Methods
2.1. Study Design and Participants
2.2. Genotyping
2.3. MRI Scanning Procedure
2.4. MRI Acquisitions
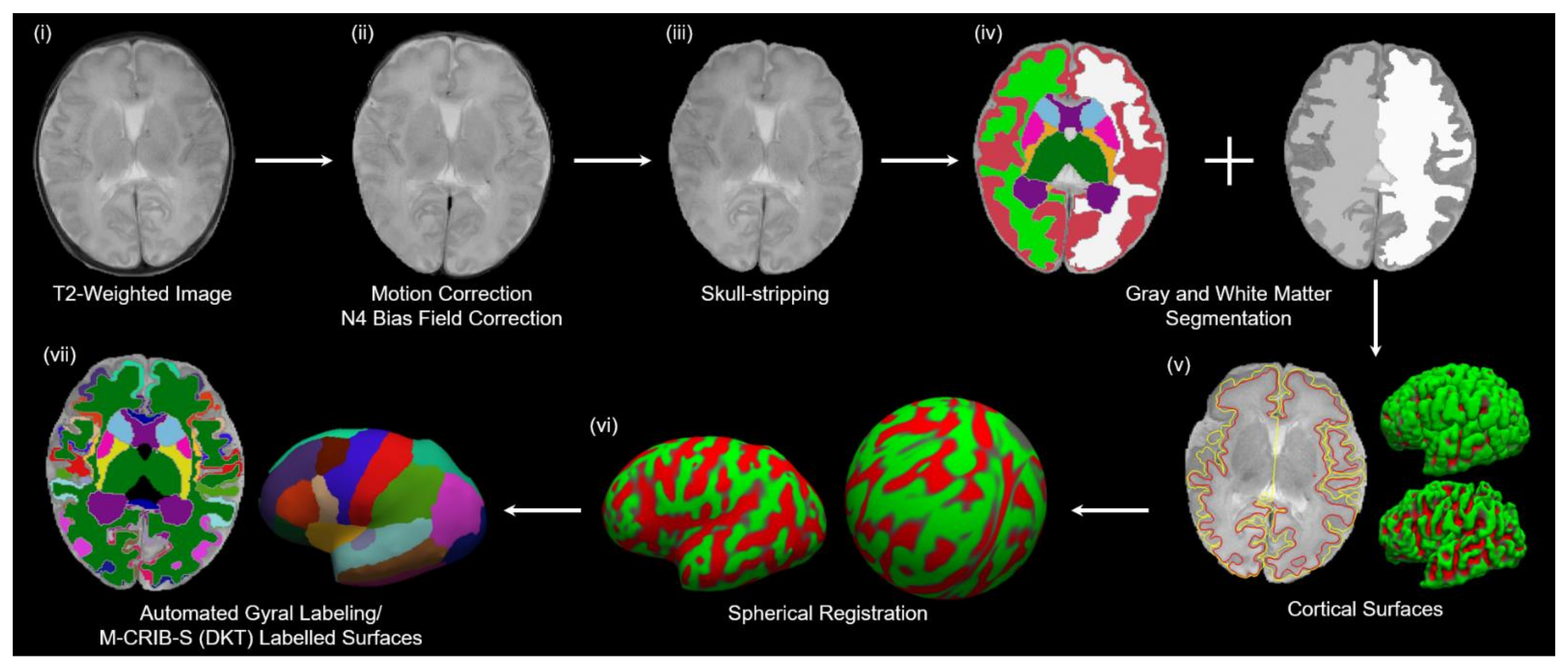
2.5. Image Processing
2.6. Neurodevelopmental Assessment
2.7. Statistical Analysis
3. Results
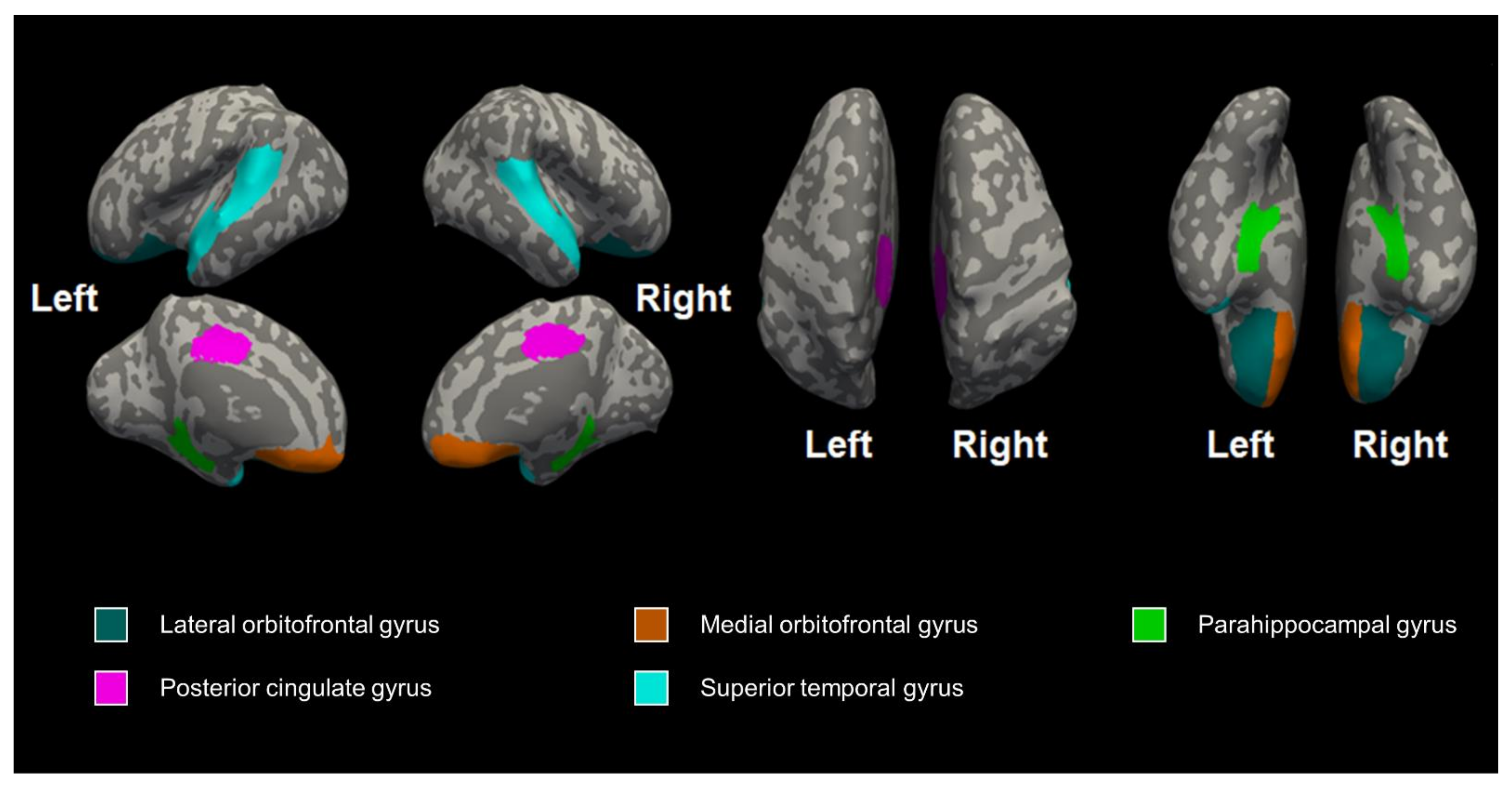
3.1. The Differences in Cerebral Volume, Thickness, and Curvature between the Preterm and Control Groups
3.2. The Effect of Genotype on Cerebral Volume, Thickness, and Curvature
3.3. The Neurodevelopmental Follow-Up
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Walani, S.R. Global burden of preterm birth. Int. J. Gynaecol. Obstet. 2020, 150, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Hee Chung, E.; Chou, J.; Brown, K.A. Neurodevelopmental outcomes of preterm infants: A recent literature review. Transl. Pediatr. 2020, 9, S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Ball, G.; Boardman, J.P.; Rueckert, D.; Aljabar, P.; Arichi, T.; Merchant, N.; Gousias, I.S.; Edwards, A.D.; Counsell, S.J. The effect of preterm birth on thalamic and cortical development. Cereb Cortex 2012, 22, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Kiss, J.Z.; Vasung, L.; Petrenko, V. Process of cortical network formation and impact of early brain damage. Curr. Opin. Neurol. 2014, 27, 133–141. [Google Scholar] [CrossRef]
- Demirci, N.; Holland, M.A. Cortical thickness systematically varies with curvature and depth in healthy human brains. Hum. Brain Mapp. 2022, 43, 2064–2084. [Google Scholar] [CrossRef]
- Grantham-McGregor, S.; Cheung, Y.B.; Cueto, S.; Glewwe, P.; Richter, L.; Strupp, B. Developmental potential in the first 5 years for children in developing countries. Lancet 2007, 369, 60–70. [Google Scholar] [CrossRef] [Green Version]
- Alexander, B.; Kelly, C.E.; Adamson, C.; Beare, R.; Zannino, D.; Chen, J.; Murray, A.L.; Loh, W.Y.; Matthews, L.G.; Warfield, S.K.; et al. Changes in neonatal regional brain volume associated with preterm birth and perinatal factors. Neuroimage 2019, 185, 654–663. [Google Scholar] [CrossRef]
- Nam, K.W.; Castellanos, N.; Simmons, A.; Froudist-Walsh, S.; Allin, M.P.; Walshe, M.; Murray, R.M.; Evans, A.; Muehlboeck, J.S.; Nosarti, C. Alterations in cortical thickness development in preterm-born individuals: Implications for high-order cognitive functions. Neuroimage 2015, 115, 64–75. [Google Scholar] [CrossRef] [Green Version]
- Garcia, K.E.; Robinson, E.C.; Alexopoulos, D.; Dierker, D.L.; Glasser, M.F.; Coalson, T.S.; Ortinau, C.M.; Rueckert, D.; Taber, L.A.; Van Essen, D.C.; et al. Dynamic patterns of cortical expansion during folding of the preterm human brain. Proc. Natl. Acad. Sci. USA 2018, 115, 3156–3161. [Google Scholar] [CrossRef] [Green Version]
- Padilla, N.; Alexandrou, G.; Blennow, M.; Lagercrantz, H.; Aden, U. Brain Growth Gains and Losses in Extremely Preterm Infants at Term. Cereb Cortex 2015, 25, 1897–1905. [Google Scholar] [CrossRef] [Green Version]
- Kline, J.E.; Sita Priyanka Illapani, V.; He, L.; Parikh, N.A. Automated brain morphometric biomarkers from MRI at term predict motor development in very preterm infants. Neuroimage Clin. 2020, 28, 102475. [Google Scholar] [CrossRef] [PubMed]
- Ajayi-Obe, M.; Saeed, N.; Cowan, F.M.; Rutherford, M.A.; Edwards, A.D. Reduced development of cerebral cortex in extremely preterm infants. Lancet 2000, 356, 1162–1163. [Google Scholar] [CrossRef]
- Lee, J.Y.; Choi, Y.H.; Cha, J.H.; Lee, J.Y.; Lee, Y.J.; Kim, B.H.; Kim, I.K.; Lee, J.M.; Lee, H.J. Altered asymmetries of the structural networks comprising the fronto-limbic brain circuitry of preterm infants. Sci. Rep. 2021, 11, 1318. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhong, M.; Yao, S.; Jin, X.; Liu, Y.; Tan, C.; Zhu, X.; Yi, J. Hemispheric asymmetry of the frontolimbic cortex in young adults with borderline personality disorder. Acta Psychiatr. Scand. 2017, 136, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.S.; Jernigan, T.L.; Vestergaard, M.; Mortensen, E.L.; Baare, W.F.C. Neuroticism is linked to microstructural left-right asymmetry of fronto-limbic fibre tracts in adolescents with opposite effects in boys and girls. Neuropsychologia 2018, 114, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Toller, G.; Adhimoolam, B.; Rankin, K.P.; Huppertz, H.J.; Kurthen, M.; Jokeit, H. Right fronto-limbic atrophy is associated with reduced empathy in refractory unilateral mesial temporal lobe epilepsy. Neuropsychologia 2015, 78, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Counsell, S.J.; Rutherford, M.A.; Cowan, F.M.; Edwards, A.D. Magnetic resonance imaging of preterm brain injury. Arch. Dis. Child. Fetal Neonatal Ed. 2003, 88, F269–F274. [Google Scholar] [CrossRef]
- Kline, J.E.; Illapani, V.S.P.; He, L.; Altaye, M.; Logan, J.W.; Parikh, N.A. Early cortical maturation predicts neurodevelopment in very preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2020, 105, 460–465. [Google Scholar] [CrossRef]
- Dubois, J.; Alison, M.; Counsell, S.J.; Hertz-Pannier, L.; Huppi, P.S.; Benders, M. MRI of the Neonatal Brain: A Review of Methodological Challenges and Neuroscientific Advances. J. Magn. Reson. Imaging 2021, 53, 1318–1343. [Google Scholar] [CrossRef]
- Thompson, P.M.; Cannon, T.D.; Narr, K.L.; van Erp, T.; Poutanen, V.P.; Huttunen, M.; Lonnqvist, J.; Standertskjold-Nordenstam, C.G.; Kaprio, J.; Khaledy, M.; et al. Genetic influences on brain structure. Nat. NeuroSci. 2001, 4, 1253–1258. [Google Scholar] [CrossRef]
- Dempfle, A.; Scherag, A.; Hein, R.; Beckmann, L.; Chang-Claude, J.; Schafer, H. Gene-environment interactions for complex traits: Definitions, methodological requirements and challenges. Eur. J. Hum. Genet. 2008, 16, 1164–1172. [Google Scholar] [CrossRef] [PubMed]
- Leviton, A.; Gressens, P.; Wolkenhauer, O.; Dammann, O. Systems approach to the study of brain damage in the very preterm newborn. Front. Syst. NeuroSci. 2015, 9, 58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boardman, J.P.; Walley, A.; Ball, G.; Takousis, P.; Krishnan, M.L.; Hughes-Carre, L.; Aljabar, P.; Serag, A.; King, C.; Merchant, N.; et al. Common genetic variants and risk of brain injury after preterm birth. Pediatrics 2014, 133, e1655–e1663. [Google Scholar] [CrossRef] [PubMed]
- Bearden, C.E.; Freimer, N.B. Endophenotypes for psychiatric disorders: Ready for primetime? Trends Genet. 2006, 22, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Preston, G.A.; Weinberger, D.R. Intermediate phenotypes in schizophrenia: A selective review. Dialogues Clin. NeuroSci. 2005, 7, 165–179. [Google Scholar] [CrossRef]
- Kendler, K.S.; Neale, M.C. Endophenotype: A conceptual analysis. Mol. Psychiatry 2010, 15, 789–797. [Google Scholar] [CrossRef] [Green Version]
- Prasad, K.M.; Keshavan, M.S. Structural cerebral variations as useful endophenotypes in schizophrenia: Do they help construct “extended endophenotypes”? Schizophr. Bull. 2008, 34, 774–790. [Google Scholar] [CrossRef]
- Cha, J.H.; Ahn, J.H.; Kim, Y.J.; Lee, B.G.; Kim, J.I.; Park, H.K.; Kim, B.N.; Lee, H.J. Impact of Preterm Birth on Neurodevelopmental Disorders in South Korea: A Nationwide Population-Based Study. J. Clin. Med. 2022, 11, 2476. [Google Scholar] [CrossRef]
- Afif, I.Y.; Manik, A.R.; Munthe, K.; Maula, M.I.; Ammarullah, M.I.; Jamari, J.; Winarni, T.I. Physiological Effect of Deep Pressure in Reducing Anxiety of Children with ASD during Traveling: A Public Transportation Setting. Bioengineering 2022, 9, 157. [Google Scholar] [CrossRef]
- Behnia, F.; Parets, S.E.; Kechichian, T.; Yin, H.; Dutta, E.H.; Saade, G.R.; Smith, A.K.; Menon, R. Fetal DNA methylation of autism spectrum disorders candidate genes: Association with spontaneous preterm birth. Am. J. Obstet. Gynecol. 2015, 212, 533.e1–533.e9. [Google Scholar] [CrossRef]
- Koletzko, B.; Reischl, E.; Tanjung, C.; Gonzalez-Casanova, I.; Ramakrishnan, U.; Meldrum, S.; Simmer, K.; Heinrich, J.; Demmelmair, H. FADS1 and FADS2 Polymorphisms Modulate Fatty Acid Metabolism and Dietary Impact on Health. Annu. Rev. Nutr. 2019, 39, 21–44. [Google Scholar] [CrossRef] [PubMed]
- Dutt, A.; Shaikh, M.; Ganguly, T.; Nosarti, C.; Walshe, M.; Arranz, M.; Rifkin, L.; McDonald, C.; Chaddock, C.A.; McGuire, P.; et al. COMT gene polymorphism and corpus callosum morphometry in preterm born adults. Neuroimage 2011, 54, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.M.; Kim, S.J.; Kistner-Griffin, E.; Guter, S.; Cook, E.H.; Jacob, S. ASD and Genetic Associations with Receptors for Oxytocin and Vasopressin-AVPR1A, AVPR1B, and OXTR. Front. NeuroSci. 2016, 10, 516. [Google Scholar] [CrossRef] [PubMed]
- Esmaiel, N.N.; Ashaat, E.A.; Mosaad, R.; Fayez, A.; Ibrahim, M.; Abdallah, Z.Y.; Issa, M.Y.; Salem, S.; Ramadan, A.; El Wakeel, M.A.; et al. The potential impact of COMT gene variants on dopamine regulation and phenotypic traits of ASD patients. Behav. Brain Res. 2020, 378, 112272. [Google Scholar] [CrossRef]
- Sun, C.; Zou, M.; Wang, X.; Xia, W.; Ma, Y.; Liang, S.; Hao, Y.; Wu, L.; Fu, S. FADS1-FADS2 and ELOVL2 gene polymorphisms in susceptibility to autism spectrum disorders in Chinese children. BMC Psychiatry 2018, 18, 283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, Y.; Kaneko, N.; Nunokawa, A.; Shibuya, M.; Egawa, J.; Someya, T. Oxytocin receptor (OXTR) gene and risk of schizophrenia: Case-control and family-based analyses and meta-analysis in a Japanese population. Psychiatry Clin. NeuroSci. 2012, 66, 622. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jandacek, R.; Rider, T.; Tso, P.; McNamara, R.K. Elevated delta-6 desaturase (FADS2) expression in the postmortem prefrontal cortex of schizophrenic patients: Relationship with fatty acid composition. Schizophr. Res. 2009, 109, 113–120. [Google Scholar] [CrossRef]
- Fan, J.B.; Zhang, C.S.; Gu, N.F.; Li, X.W.; Sun, W.W.; Wang, H.Y.; Feng, G.Y.; St Clair, D.; He, L. Catechol-O-methyltransferase gene Val/Met functional polymorphism and risk of schizophrenia: A large-scale association study plus meta-analysis. Biol. Psychiatry 2005, 57, 139–144. [Google Scholar] [CrossRef]
- WHO: Recommended definitions, terminology and format for statistical tables related to the perinatal period and use of a new certificate for cause of perinatal deaths. Modifications recommended by FIGO as amended October 14, 1976. Acta Obstet. Gynecol. Scand. 1977, 56, 247–253. [Google Scholar]
- Blencowe, H.; Cousens, S.; Oestergaard, M.Z.; Chou, D.; Moller, A.B.; Narwal, R.; Adler, A.; Vera Garcia, C.; Rohde, S.; Say, L.; et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet 2012, 379, 2162–2172. [Google Scholar] [CrossRef] [Green Version]
- Committee on Drugs, American Academy of Pediatrics. Guidelines for the ethical conduct of studies to evaluate drugs in pediatric populations. Pediatrics 1995, 95, 286–294. [Google Scholar] [CrossRef]
- Copeland, A.; Silver, E.; Korja, R.; Lehtola, S.J.; Merisaari, H.; Saukko, E.; Sinisalo, S.; Saunavaara, J.; Lähdesmäki, T.; Parkkola, R.; et al. Infant and Child MRI: A Review of Scanning Procedures. Front. Neurosci. 2021, 15, 632. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Tourville, J. 101 labeled brain images and a consistent human cortical labeling protocol. Front. NeuroSci. 2012, 6, 171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adamson, C.L.; Alexander, B.; Ball, G.; Beare, R.; Cheong, J.L.Y.; Spittle, A.J.; Doyle, L.W.; Anderson, P.J.; Seal, M.L.; Thompson, D.K. Parcellation of the neonatal cortex using Surface-based Melbourne Children’s Regional Infant Brain atlases (M-CRIB-S). Sci. Rep. 2020, 10, 4359. [Google Scholar] [CrossRef] [Green Version]
- Tustison, N.J.; Avants, B.B.; Cook, P.A.; Zheng, Y.; Egan, A.; Yushkevich, P.A.; Gee, J.C. N4ITK: Improved N3 bias correction. IEEE Trans. Med. Imaging 2010, 29, 1310–1320. [Google Scholar] [CrossRef] [Green Version]
- Makropoulos, A.; Gousias, I.S.; Ledig, C.; Aljabar, P.; Serag, A.; Hajnal, J.V.; Edwards, A.D.; Counsell, S.J.; Rueckert, D. Automatic whole brain MRI segmentation of the developing neonatal brain. IEEE Trans. Med. Imaging 2014, 33, 1818–1831. [Google Scholar] [CrossRef]
- Fischl, B. FreeSurfer. Neuroimage 2012, 62, 774–781. [Google Scholar] [CrossRef] [Green Version]
- Yim, C.H.; Kim, G.H.; Eun, B.L. Usefulness of the Korean Developmental Screening Test for infants and children for the evaluation of developmental delay in Korean infants and children: A single-center study. Korean J. Pediatr. 2017, 60, 312–319. [Google Scholar] [CrossRef]
- Chung, H.J.; Yang, D.; Kim, G.H.; Kim, S.K.; Kim, S.W.; Kim, Y.K.; Kim, Y.A.; Kim, J.S.; Kim, J.K.; Kim, C.; et al. Development of the Korean Developmental Screening Test for Infants and Children (K-DST). Clin. Exp. Pediatr. 2020, 63, 438–446. [Google Scholar] [CrossRef]
- Ball, G.; Seidlitz, J.; O'Muircheartaigh, J.; Dimitrova, R.; Fenchel, D.; Makropoulos, A.; Christiaens, D.; Schuh, A.; Passerat-Palmbach, J.; Hutter, J.; et al. Cortical morphology at birth reflects spatiotemporal patterns of gene expression in the fetal human brain. PLoS Biol. 2020, 18, e3000976. [Google Scholar] [CrossRef]
- Alexander-Bloch, A.F.; Raznahan, A.; Vandekar, S.N.; Seidlitz, J.; Lu, Z.; Mathias, S.R.; Knowles, E.; Mollon, J.; Rodrigue, A.; Curran, J.E.; et al. Imaging local genetic influences on cortical folding. Proc. Natl. Acad. Sci. USA 2020, 117, 7430–7436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hedderich, D.M.; Bauml, J.G.; Berndt, M.T.; Menegaux, A.; Scheef, L.; Daamen, M.; Zimmer, C.; Bartmann, P.; Boecker, H.; Wolke, D.; et al. Aberrant gyrification contributes to the link between gestational age and adult IQ after premature birth. Brain 2019, 142, 1255–1269. [Google Scholar] [CrossRef] [PubMed]
- Arpino, C.; D'Argenzio, L.; Ticconi, C.; Di Paolo, A.; Stellin, V.; Lopez, L.; Curatolo, P. Brain damage in preterm infants: Etiological pathways. Ann. Ist. Super. Sanita 2005, 41, 229–237. [Google Scholar] [PubMed]
- Krishnan, M.L.; Wang, Z.; Aljabar, P.; Ball, G.; Mirza, G.; Saxena, A.; Counsell, S.J.; Hajnal, J.V.; Montana, G.; Edwards, A.D. Machine learning shows association between genetic variability in PPARG and cerebral connectivity in preterm infants. Proc. Natl. Acad. Sci. USA 2017, 114, 13744–13749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lohmann, G.; von Cramon, D.Y.; Steinmetz, H. Sulcal variability of twins. Cereb Cortex 1999, 9, 754–763. [Google Scholar] [CrossRef] [Green Version]
- Atkinson, E.G.; Rogers, J.; Cheverud, J.M. Evolutionary and developmental implications of asymmetric brain folding in a large primate pedigree. Evolution 2016, 70, 707–715. [Google Scholar] [CrossRef]
- Israel, S.; Lerer, E.; Shalev, I.; Uzefovsky, F.; Riebold, M.; Laiba, E.; Bachner-Melman, R.; Maril, A.; Bornstein, G.; Knafo, A.; et al. The oxytocin receptor (OXTR) contributes to prosocial fund allocations in the dictator game and the social value orientations task. PLoS ONE 2009, 4, e5535. [Google Scholar] [CrossRef]
- Tost, H.; Kolachana, B.; Hakimi, S.; Lemaitre, H.; Verchinski, B.A.; Mattay, V.S.; Weinberger, D.R.; Meyer-Lindenberg, A. A common allele in the oxytocin receptor gene (OXTR) impacts prosocial temperament and human hypothalamic-limbic structure and function. Proc. Natl. Acad. Sci. USA 2010, 107, 13936–13941. [Google Scholar] [CrossRef] [Green Version]
- Boardman, J.P.; Craven, C.; Valappil, S.; Counsell, S.J.; Dyet, L.E.; Rueckert, D.; Aljabar, P.; Rutherford, M.A.; Chew, A.T.; Allsop, J.M.; et al. A common neonatal image phenotype predicts adverse neurodevelopmental outcome in children born preterm. Neuroimage 2010, 52, 409–414. [Google Scholar] [CrossRef]
- Ligam, P.; Haynes, R.L.; Folkerth, R.D.; Liu, L.; Yang, M.; Volpe, J.J.; Kinney, H.C. Thalamic damage in periventricular leukomalacia: Novel pathologic observations relevant to cognitive deficits in survivors of prematurity. Pediatr. Res. 2009, 65, 524–529. [Google Scholar] [CrossRef] [Green Version]
- Meyer, M.; Jurek, B.; Alfonso-Prieto, M.; Ribeiro, R.; Milenkovic, V.M.; Winter, J.; Hoffmann, P.; Wetzel, C.H.; Giorgetti, A.; Carloni, P.; et al. Structure-function relationships of the disease-linked A218T oxytocin receptor variant. Mol. Psychiatry 2022, 27, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Li, Z.; Su, Y. The association between oxytocin receptor gene polymorphism (OXTR) and trait empathy. J. Affect. Disord 2012, 138, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E.T. The cingulate cortex and limbic systems for emotion, action, and memory. Brain Struct. Funct. 2019, 224, 3001–3018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leech, R.; Sharp, D.J. The role of the posterior cingulate cortex in cognition and disease. Brain 2014, 137, 12–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maddock, R.J.; Garrett, A.S.; Buonocore, M.H. Posterior cingulate cortex activation by emotional words: fMRI evidence from a valence decision task. Hum. Brain Mapp. 2003, 18, 30–41. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhang, R.; Jiang, F.; Zhang, H.; Zhao, A.; Xu, B.; Jin, L.; Wang, T.; Jia, W.; Jia, W.; et al. FADS1-FADS2 genetic polymorphisms are associated with fatty acid metabolism through changes in DNA methylation and gene expression. Clin. Epigenetics 2018, 10, 113. [Google Scholar] [CrossRef] [PubMed]
- Caspi, A.; Williams, B.; Kim-Cohen, J.; Craig, I.W.; Milne, B.J.; Poulton, R.; Schalkwyk, L.C.; Taylor, A.; Werts, H.; Moffitt, T.E. Moderation of breastfeeding effects on the IQ by genetic variation in fatty acid metabolism. Proc. Natl. Acad. Sci. USA 2007, 104, 18860–18865. [Google Scholar] [CrossRef] [Green Version]
- Brookes, K.J.; Chen, W.; Xu, X.; Taylor, E.; Asherson, P. Association of fatty acid desaturase genes with attention-deficit/hyperactivity disorder. Biol. Psychiatry 2006, 60, 1053–1061. [Google Scholar] [CrossRef]
- Lotta, T.; Vidgren, J.; Tilgmann, C.; Ulmanen, I.; Melén, K.; Julkunen, I.; Taskinen, J. Kinetics of human soluble and membrane-bound catechol O-methyltransferase: A revised mechanism and description of the thermolabile variant of the enzyme. Biochemistry 1995, 34, 4202–4210. [Google Scholar] [CrossRef]
- Huber, J.C.; Schneeberger, C.; Tempfer, C.B. Genetic modeling of estrogen metabolism as a risk factor of hormone-dependent disorders. Maturitas 2002, 41 (Suppl. S1), S55–S64. [Google Scholar] [CrossRef]
- Dumontheil, I.; Roggeman, C.; Ziermans, T.; Peyrard-Janvid, M.; Matsson, H.; Kere, J.; Klingberg, T. Influence of the COMT genotype on working memory and brain activity changes during development. Biol. Psychiatry 2011, 70, 222–229. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Gene | SNP Name | Chromosome | Coordinate (Position) | Source | Variant | East Asian MAF | Hardy–Weinberg Equilibrium Unaffected, P | Gene Location |
|---|---|---|---|---|---|---|---|---|
| OXTR | rs1042778 | 3 | 8794545 | dbSNP 153 | G/T | 0.093 | 1.000 | 3′UTR |
| OXTR | rs2268493 | 3 | 8800840 | dbSNP 153 | T/C | 0.210 | 1.000 | Intron |
| OXTR | rs53576 | 3 | 8804371 | dbSNP 153 | A/G | 0.419 | 1.000 | Intron |
| FADS2 | rs174570 | 11 | 61597212 | dbSNP 153 | C/T | 0.387 | 1.000 | Intron |
| FADS2 | rs174576 | 11 | 61603510 | dbSNP 153 | C/A | 0.556 | 0.857 | Intron |
| FADS2 | rs498793 | 11 | 61624705 | dbSNP 153 | C/T | 0.107 | 1.000 | Intron |
| COMT | rs740603 | 22 | 19945177 | dbSNP 153 | A/G | 0.426 | 1.000 | Intron |
| COMT | rs165774 | 22 | 19952561 | dbSNP 153 | G/A | 0.149 | 0.797 | Intron |
| COMT | rs174696 | 22 | 19953176 | dbSNP 153 | C/T | 0.422 | 1.000 | Intron |
| Characteristics | Preterm (n = 20) | Control (n = 6) | p-Value |
|---|---|---|---|
| Sex | 0.182 | ||
| Male | 13 (65.0%) | 2 (33.3%) | |
| Female | 7 (35.0%) | 4 (66.7%) | |
| Ethnicity | |||
| Asian | 20 (100%) | 6 (100%) | NA |
| Scan age, mean ± SD | 37.60 ± 2.04 | 40.00 ± 1.79 | 0.016 |
| Birth weight, mean ± SD | 1624.00 ± 710.40 | 3373.33 ± 27.53 | <0.001 |
| Gestational age, mean ± SD | 31.00 ± 3.88 | 38.83 ± 0.98 | <0.001 |
| Moderate-to-severe BPD | 3 (15%) | 0 (0%) | 0.412 |
| Moderate-to-severe ROP | 2 (10%) | 0 (0%) | 0.420 |
| K-DST | |||
| <-2 SD in any domain | 2 (10%) | 0 (0%) | 0.509 |
| <-2 SD in gross motor domain | 1 (5%) | 0 (0%) | 0.648 |
| <-2 SD in fine motor domain | 0 (0%) | 0 (0%) | NA |
| <-2 SD in cognition domain | 0 (0%) | 0 (0%) | NA |
| <-2 SD in language domain | 1 (5%) | 0 (0%) | 0.648 |
| <-2 SD in sociality domain | 0 (0%) | 0 (0%) | NA |
| Characteristics | Very Preterm (n = 9) | Late Preterm (n = 11) | Control (n = 6) | p-Value | FDR |
|---|---|---|---|---|---|
| Volume | |||||
| Total gray-matter volume | 98,268.70 ± 11,511.49 | 109,480.18 ± 15,158.94 | 116,044.91 ± 10,982.82 | 0.043 | 0.072 |
| Left hemisphere cortical gray matter | 34,697.14 ± 4374.11 | 36,820.41 ± 5824.50 | 38,987.16 ± 5452.27 | 0.317 | 0.317 |
| Right hemisphere cortical gray matter | 32,924.48 ± 3688.58 | 36,505.99 ± 5754.01 | 38,354.64 ± 3806.95 | 0.091 | 0.114 |
| Left-hemisphere cerebral white matter | 63,135.95 ± 7821.90 | 73,906.42 ± 10,090.29 | 72,867.42 ± 9780.54 | 0.040 | 0.072 |
| Right-hemisphere cerebral white matter | 60,970.21 ± 8171.73 | 70,194.39 ± 7638.09 | 69,560.52 ± 6089.01 | 0.027 | 0.072 |
| Thickness | |||||
| Left lateral orbitofrontal gyrus | 1.08 ± 0.15 | 1.15 ± 0.20 | 1.15 ± 0.23 | 0.661 | 0.826 |
| Right lateral orbitofrontal gyrus | 1.03 ± 0.12 | 1.11 ± 0.21 | 1.09 ± 0.12 | 0.522 | 0.826 |
| Left medial orbitofrontal gyrus | 1.14 ± 0.17 | 1.16 ± 0.15 | 1.18 ± 0.16 | 0.877 | 0.937 |
| Right medial orbitofrontal gyrus | 1.19 ± 0.23 | 1.22 ± 0.14 | 1.12 ± 0.14 | 0.574 | 0.826 |
| Left parahippocampal gyrus | 1.06 ± 0.22 | 1.01 ± 0.14 | 1.10 ± 0.07 | 0.584 | 0.826 |
| Right parahippocampal gyrus | 0.98 ± 0.25 | 1.07 ± 0.19 | 1.03 ± 0.14 | 0.588 | 0.826 |
| Left posterior cingulate gyrus | 1.34 ± 0.19 | 1.33 ± 0.12 | 1.31 ± 0.07 | 0.937 | 0.937 |
| Right posterior cingulate gyrus | 1.30 ± 0.15 | 1.36 ± 0.08 | 1.36 ± 0.13 | 0.551 | 0.826 |
| Left superior temporal gyrus | 1.41 ± 0.17 | 1.38 ± 0.13 | 1.45 ± 0.16 | 0.645 | 0.826 |
| Right superior temporal gyrus | 1.34 ± 0.14 | 1.46 ± 0.17 | 1.42 ± 0.14 | 0.256 | 0.826 |
| Curvature | |||||
| Left lateral orbitofrontal gyrus | 1.76 ± 0.74 | 1.96 ± 0.51 | 2.47 ± 0.30 | 0.075 | 0.107 |
| Right lateral orbitofrontal gyrus | 1.73 ± 0.46 | 2.23 ± 0.58 | 2.43 ± 0.70 | 0.064 | 0.107 |
| Left medial orbitofrontal gyrus | 1.35 ± 0.63 | 1.63 ± 0.31 | 2.00 ± 0.38 | 0.045 | 0.090 |
| Right medial orbitofrontal gyrus | 1.20 ± 0.40 | 1.79 ± 0.40 | 1.86 ± 0.59 | 0.011 | 0.028 |
| Left parahippocampal gyrus | 0.55 ± 0.16 | 0.52 ± 0.31 | 0.62 ± 0.31 | 0.735 | 0.735 |
| Right parahippocampal gyrus | 0.35 ± 0.16 | 0.49 ± 0.52 | 0.55 ± 0.27 | 0.571 | 0.634 |
| Left posterior cingulate gyrus | 0.54 ± 0.27 | 1.01 ± 0.30 | 1.00 ± 0.40 | 0.006 | 0.028 |
| Right posterior cingulate gyrus | 0.76 ± 0.41 | 0.98 ± 0.29 | 0.89 ± 0.23 | 0.324 | 0.405 |
| Left superior temporal gyrus | 4.41 ± 0.81 | 5.35 ± 0.62 | 5.69 ± 0.99 | 0.009 | 0.028 |
| Right superior temporal gyrus | 3.70 ± 0.62 | 4.64 ± 0.83 | 5.32 ± 0.57 | <0.001 | 0.010 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Ahn, J.-H.; Lee, J.Y.; Jang, Y.H.; Kim, Y.-E.; Kim, J.I.; Kim, B.-N.; Lee, H.J. Altered Cerebral Curvature in Preterm Infants Is Associated with the Common Genetic Variation Related to Autism Spectrum Disorder and Lipid Metabolism. J. Clin. Med. 2022, 11, 3135. https://doi.org/10.3390/jcm11113135
Kim H, Ahn J-H, Lee JY, Jang YH, Kim Y-E, Kim JI, Kim B-N, Lee HJ. Altered Cerebral Curvature in Preterm Infants Is Associated with the Common Genetic Variation Related to Autism Spectrum Disorder and Lipid Metabolism. Journal of Clinical Medicine. 2022; 11(11):3135. https://doi.org/10.3390/jcm11113135
Chicago/Turabian StyleKim, Hyuna, Ja-Hye Ahn, Joo Young Lee, Yong Hun Jang, Young-Eun Kim, Johanna Inhyang Kim, Bung-Nyun Kim, and Hyun Ju Lee. 2022. "Altered Cerebral Curvature in Preterm Infants Is Associated with the Common Genetic Variation Related to Autism Spectrum Disorder and Lipid Metabolism" Journal of Clinical Medicine 11, no. 11: 3135. https://doi.org/10.3390/jcm11113135