Abstract
Early insertion of a supraglottic airway (SGA) device could improve chest compression fraction by allowing providers to perform continuous chest compressions or by shortening the interruptions needed to deliver ventilations. SGA devices do not require the same expertise as endotracheal intubation. This study aimed to determine whether the immediate insertion of an i-gel® while providing continuous chest compressions with asynchronous ventilations could generate higher CCFs than the standard 30:2 approach using a face-mask in a simulation of out-of-hospital cardiac arrest. A multicentre, parallel, randomised, superiority, simulation study was carried out. The primary outcome was the difference in CCF during the first two minutes of resuscitation. Overall and per-cycle CCF quality of compressions and ventilations parameters were also compared. Among thirteen teams of two participants, the early insertion of an i-gel® resulted in higher CCFs during the first two minutes (89.0% vs. 83.6%, p = 0.001). Overall and per-cycle CCF were consistently higher in the i-gel® group, even after the 30:2 alternation had been resumed. In the i-gel® group, ventilation parameters were enhanced, but compressions were significantly shallower (4.6 cm vs. 5.2 cm, p = 0.007). This latter issue must be addressed before clinical trials can be considered.
1. Introduction
1.1. Background
High-quality chest compressions are mandatory to increase the probability of survival after out-of-hospital cardiac arrest (OHCA) [1,2]. One of the key determinants of survival and a favourable neurological outcome is the chest compression fraction (CCF), i.e., the proportion of time spent performing compressions during cardiopulmonary resuscitation (CPR) [3,4,5,6,7,8,9,10]. An increase of 10% in CCF can even yield an 11% increase in survival [6].
The optimal airway management strategy for OHCA is still debated [11,12,13,14,15,16,17], and airway management manoeuvres might lead to interruptions in chest compressions, thereby preventing rescuers from achieving high CCFs. Even though endotracheal intubation (ETI) is often considered as the gold standard, this technique requires great skill that must be maintained by regular practice to be both safe and efficient [18,19,20]. However, most prehospital providers are either insufficiently trained in ETI or lack the possibility of maintaining this skill. Supraglottic airway (SGA) devices might therefore represent an adequate solution in many settings, as their use does not require the same level of expertise. There are many different SGA devices, some of which are easier to use than others. Converse to traditional laryngeal mask airways, the i-gel® (Intersurgical Ltd., Wokingham, UK) embeds a thermoplastic cuff that does not need to be inflated, making its insertion quicker and allowing high success rates to be achieved [21,22,23,24,25,26,27,28,29,30,31]. In addition, SGA devices seem to confer a certain degree of airway protection, as the rates of regurgitation and aspiration do not significantly differ from those seen with ETI in case of OHCA [32] and are much lower than those reported when a bag-valve-mask (BVM) device is used [33,34]. Moreover, the use of the i-gel® is not associated with significant leaks during the delivery of continuous chest compressions [35]. This ensures a high reliability of end-tidal CO2 measurements, an important feature, as capnography is increasingly used to monitor and optimise CPR quality and to detect ROSC [1].
Our hypothesis was that, in case of OHCA, the early insertion of an i-gel® device without prior BVM ventilation could improve CCF while allowing a similar time than the standard approach to elapse before the first effective ventilation could be delivered.
1.2. Objectives
The primary aim of this study was to determine whether the immediate insertion of an i-gel® while providing continuous chest compressions, followed by asynchronous ventilations (experimental approach) during the two minutes following the first rhythm analysis allows for the generation of higher CCFs than the 30 compressions: 2 ventilations (standard) approach in a simulated model of OHCA.
The secondary objective was to compare the impact of this approach on CPR quality and ventilation parameters [1,36,37].
2. Materials and Methods
2.1. Study Design
This was a multicentre, parallel, randomised, superiority, simulation study designed in accordance with the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) statement [38], the detailed protocol of which was previously published [39]. Results are reported according to the Consolidated Standards of Reporting Trials (CONSORT) statement (Table S1) [40]. The trial was prospectively registered: NCT04736446 (3 February 2021).
2.2. Setting
In Switzerland, paramedics follow a 3-year training curriculum and are trained to treat most prehospital emergencies autonomously. They can administer several Advanced Life-Support (ALS) treatments according to emergency care protocols validated by prehospital medical directors. Emergency Medical Technicians (EMTs) graduate after a 1-year curriculum and are trained to assist paramedics. The ALS teams can either be composed of two paramedics or of a paramedic teaming with an EMT. Even though these aspects are standardised, the Swiss federal system generates considerable inter-cantonal heterogeneity between Emergency Medical Services (EMS). In some regions, EMS operating in the same areas have different treatment protocols, as each service has its own medical director who can almost singlehandedly decide upon the extent of delegations. This study was performed in four different EMS where paramedics applied the standard 30:2 approach and did not routinely use the i-gel® in case of OHCA.
2.3. Participants and Recruitment
All registered paramedics and EMTs actively working in either of these EMS were eligible for inclusion. Study team members and participants who did not attend the training path were excluded. Participants were told that the study was about OHCA management but were not informed of its specific objectives. The study was conducted on the premises of each EMS.
2.4. Study Sequence
The study sequence is summarised in Figure 1.
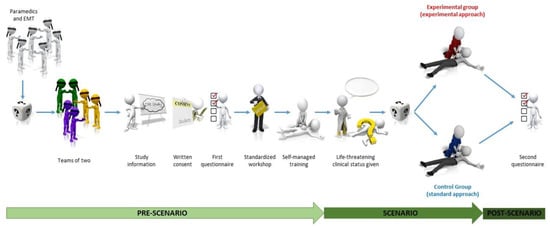
Figure 1.
Study sequence.
2.4.1. Randomisation
Randomisation was two-tiered. First, to ensure that there would be at least one paramedic per team, an online balanced team generator was used [41]. Intra-cluster randomisation with a 1:1 ratio was then performed using an online randomiser, with each trial center representing a cluster [42]. Opaque, sealed envelopes were used to ensure the concealment of allocation.
2.4.2. Standardised Workshop
The use of the i-gel® device was taught to each team separately by one of the investigators (Loric Stuby, LSt) in accordance with Peyton’s approach [43,44,45], based on a standard operating procedure (Files S1 and S2). Twenty minutes were dedicated to this workshop. This took into account the time required to gather the participants’ consent and to allow them to complete a short demographic questionnaire.
2.4.3. Training Session of Experimental Approach
Following the workshop, each team was allowed 20 min of self-training supported by a custom-made demonstration video (https://cpr2-intro.swiss-cpr-studies.ch/, accessed on 30 November 2021). During this training session, an experimental approach developed by David Thurre (DT) and LSt was practised. This approach consists of the immediate placement of an i-gel® device by one team member immediately after the first rhythm analysis, without prior BVM ventilation. Meanwhile, the second team member provides continuous chest compressions. Ventilations are given asynchronously at a rate of 10 per minute once the SGA device is in place [1,2]. After two minutes of continuous chest compressions, 30:2 CPR is provided “over-the-head” by one rescuer while the other performs ALS actions.
2.4.4. Study Scenario
Another custom-made video, which presented the manikin’s and defibrillator’s characteristics, was displayed to the participants upon entering the study room (https://cpr2-briefing.swiss-cpr-studies.ch/, accessed on 30 November 2021). The scenario was then presented in exactly the same way to all participants regardless of the study site: “This is Michael, a 50-year-old man who suddenly collapsed 10 min ago. He is now unconscious, pale, and does not seem to be breathing. Medical reinforcement is already underway and will arrive in about ten minutes. No first responder has been dispatched by the emergency medical call center, and there is no bystander nearby”. Only then could the leader open the opaque, sealed envelope containing the specific airway management strategy the team has to apply. There was no further interaction between participants and the study team until the scenario was stopped. The simulated patient was apneic and in refractory ventricular fibrillation (VF) regardless of the number of defibrillation attempts. The scenario was stopped at T0 (first compression) + 10 min.
At the end of the scenario, participants were asked to fill a last questionnaire designed to assess their satisfaction regarding the approach they had applied and the cognitive load they had perceived during the study scenario. Participants were asked to withhold information regarding the aim and course of the study until data collection had been completed in all four EMS.
2.5. Equipment
Apart from the multiparametric monitor/defibrillator, which was provided with the simulation manikin (Laerdal SimMan 3G, Laerdal Medical, Stavanger, Norway), all teams had access to their usual resuscitation equipment. A size 4 i-gel® device (Intersurgical Ltd., Wokingham, UK) and a lubricant recommended by the manikin’s manufacturer were added to this equipment.
2.6. Study Outcomes
2.6.1. Primary Outcome
The primary outcome was the CCF achieved during the first two minutes of CPR (starting from first compression).
2.6.2. Secondary Outcomes
Secondary outcomes were: overall (entire 10-min scenario) and per-cycle CCF (at 2–4, 4–6, 6–8 and 8–10 min); depth of compressions; proportion of compressions within (5 to 6 cm), below (<5 cm) and above (>6 cm) target value; compression rate; proportion of compressions within (100–120 compressions per minute—cpm), below (<100 cpm) and above target values (>120 cpm); proportion of compressions with complete chest recoil (<5 mm deviation from the reference value); time to first shock; time to first effective ventilation (defined as > 300 mL [46,47,48,49,50]); proportions of ventilations within, (300–700 mL), below (<300 mL) and above (>700 mL) target values; number of ventilations delivered; minute ventilation volume; provider satisfaction; and self-assessed cognitive load.
2.7. Data Collection, Extraction and Curation
Most data were automatically collected through the manikin’s sensors and extracted to a comma-separated values (CSV) file, thereby preventing assessment bias. Variables of interest were automatically generated using a custom-coded PHP script [51]. Data gathered on paper questionnaires were entered in duplicate using EpiData (The EpiData Association, att. Jens Lauritsen, Enghavevej 34, DK5230 Odense M, Denmark, Europe) [52], and any discrepancy was listed and resolved to minimise copying and typing errors. Missing data were treated as such. No imputation technique was used. The minute ventilation volume was calculated by dividing the total volume delivered by the ventilation time (corresponding to 600 s scenario time minus the time to first ventilation). All data that could have allowed the data analyst (DT) to identify group allocation were removed (e.g., number of insertion attempts). The groups were renamed “Teysachaux” and “Moléson”, and the curated databases were sent in a Stata DTA file format for blinded statistical analyses. All investigators were able to access the curated and coded data sets [53].
2.8. Sample Size Calculation
Few data were available to help with the sample size calculation. We estimated that the mean time needed to deliver two ventilations would amount to approximately 4 s, thereby adding up to 20 s per cycle. We also estimated that the initial rhythm analysis should take about 8 s, thus increasing the no-compression time to around 28 s out of the first 120 s cycle (23% of no-flow, worth 77% of CCF in the control group). In the experimental group, we estimated that the no-flow should be lower, as ventilations were to be provided without interrupting compressions (8 s only for rhythm analysis: 7% of no-flow, worth 93% of CCF). Based on observational data from pilot tests and case reviews, data variability was estimated with a standard deviation of 12. Twenty-six teams were therefore required to have a 90% chance of detecting, at the 5% significance level, a difference in CCF from 77% in the control group to 93% in the experimental group. In the original study protocol [39], the sample size calculation resulted in 24 teams because it was calculated with an online calculator [54] rather than with Stata.
2.9. Statistical Analysis
Variables were described using either mean (SD or 95% CI) or median [Q1; Q3] according to normality. A Student’s t-test or the Mann–Whitney U test were used accordingly. Proportions were reported with their 95% CI. Cognitive load was treated as a continuous variable. User satisfaction was assessed graphically, then tested using Fisher’s exact test. There was a minor protocol deviation, as we had initially planned to dichotomise this variable. To avoid losing valuable information, it was, however, decided not to proceed with dichotomisation. This decision was taken after protocol publication but before completion of data collection and statistical analyses. A two-sided p-value lower than 0.05 was considered significant. All statistical analyses were performed using Stata V15.1 (StataCorp. 2017. Stata Statistical Software: Release 15. College Station, TX, USA, StataCorp LLC).
3. Results
Fifty-two participants were recruited (44 paramedics and 8 EMTs) between March and May 2021. They were divided into 26 teams allocated equally to both groups (Figure 2). Their characteristics are detailed in Table 1. All were analysed by original assigned groups.
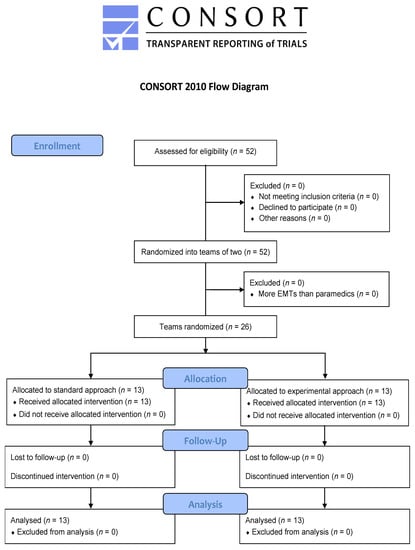
Figure 2.
Study flowchart.

Table 1.
Participants’ characteristics.
In the experimental group, the successful insertion rate was of 84.6% (11/13) at first try and of 100% at the second one. Among this group, 11/13 teams switched to 30:2 CPR, as expected (2 min after the first rhythm analysis). The CCF was consistently higher when the experimental approach was used (Table 2).

Table 2.
Chest compression fraction, overall and per cycle expressed as mean (95% CI).
Applying the experimental approach led to shallower compressions with a difference higher than 0.6 cm (median (Q1; Q3) 4.6 cm (4.3; 5.0) versus 5.2 cm (4.9; 5.3), p = 0.007). Accordingly, the mean proportion of compressions within the depth target (5 to 6 cm) was lower in the experimental group (41.7% (95% CI 28.2–55.3) versus 66.5% (95% CI 51.5–81.4), p = 0.01). All out-of-target compressions were too shallow, as there was no value above target (> 6 cm) in either group (Figure 3).
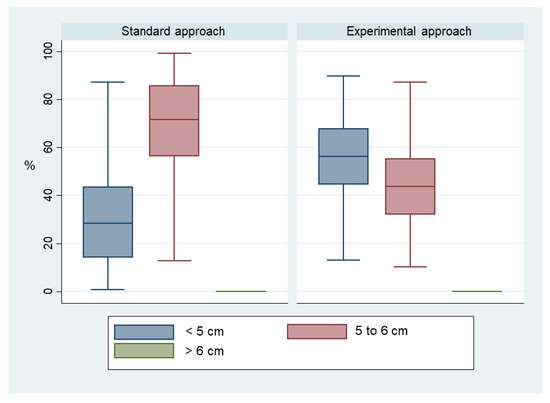
Figure 3.
Proportions of compressions below, within and above depth target value.
Mean compressions rates were similar in both groups (116 cpm (95% CI 112–120) in the experimental group versus 115 cpm (95% CI 110–119), p = 0.65), with a similar proportion of within-target compressions between groups (median (Q1; Q3) 78.2% (52.9; 92.6) versus 89.9% (62.3; 96.9), p = 0.40) (Figure S1). There was no difference regarding chest recoil (median (Q1; Q3) 97.9% (86.7; 99.6) in the experimental group versus 98.9% (92.4; 99.5), p = 0.90).
Time to first shock was also similar (mean 41.6 s (95% CI 36.1–47.2) in the experimental group versus 40.9 (95% CI 34.7–47.1), p = 0.85). There was no difference regarding time to first ventilation (median (Q1; Q3) 109.0 s (90.3; 126.5) in the experimental group versus 102.6 (93.1; 110.1), p = 0.74). The number of shocks delivered was slightly higher in the experimental group (median (Q1; Q3) 5 (5; 5) versus 4 (4; 5), p = 0.05).
The total number of ventilations over the 10 min scenario was more than twice as high in the experimental group (mean 39 (95% CI 33–46) versus 19 (95% CI 16–23), p < 0.001), corresponding to a higher minute ventilation (median (Q1; Q3) 2374 mL/min (2134; 2672) versus 794 mL/min (689; 1285). The proportion of ventilations within target volume was also significantly higher in this group (median (Q1; Q3) 94.6% (88.9; 98.0) versus 81.8% (65.0; 85.7), p = 0.003) (Figure 4).
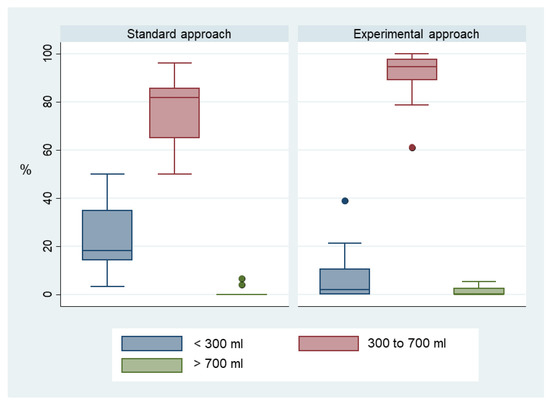
Figure 4.
Proportions of ventilations below, within and above volume target value.
Mean ventilation volume was higher in the experimental group (481 mL (95% CI 430–533) versus 385 mL (95% CI 347–423), p = 0.003); combined with the number of ventilations, this led to a higher minute ventilation (median (Q1; Q3) i-gel® 2044 mL (1709; 2185) versus 662 mL (540; 998), p < 0.001).
There was no difference in the self-assessed cognitive load (4.4 (95% CI 3.4–5.3) in the experimental group versus 4.7 (95% CI 3.9–5.5), p = 0.61). Satisfaction was higher in the experimental group (p = 0.01) (Figure S2).
4. Discussion
In this simulation study, the immediate insertion of an i-gel® device led to significantly higher CCFs. This difference can be explained by the ability to immediately provide continuous chest compressions and by the avoidance of interruptions otherwise necessary to provide ventilations when using a BVM device. CCF was consistently higher during the following cycles, even during one rescuer CPR (applying 30:2 scheme). This implies that using an i-gel® can help decrease no-flow time by overriding the time required to adequately place the ventilation mask. In contradiction with Vogt et al.’s study [55] but similarly to Cereceda-Sánchez et al.’s findings [56], ventilations were also of higher quality when the i-gel® device was used. Indeed, the number of ventilations provided was more than twice as high in the experimental group, while hyper-insufflations remained anecdotic. These endpoints should be investigated in a pediatric population, as the experimental approach we have described could offer a substantial advantage in pediatric OHCA where ventilations are paramount [57].
The number of shocks delivered was slightly higher in the experimental group. As time to first shock and to first ventilation were similar between groups, it seems that, contrarily to the King Laryngeal Tube [58], the i-gel® device does not delay the provision of these critical actions.
The present study also supports the use of a short and well-focused learning pathway, as paramedics were able to correctly insert the i-gel® in all cases and without delay to critical actions. Even though a face-to-face workshop is certainly mandatory, the theoretical parts could easily be provided as asynchronous, distance learning interventions [59,60,61].
In our scenario, OHCA had to be managed by two providers only, which represents a worst-case situation. Actual clinical outcomes might prove superior to those we report as CPR-trained bystanders might be assigned to perform high-quality chest compressions while professionals focus on other aspects of OHCA-management, such as team coordination, ventilations, rhythm analysis and ALS treatments.
Compressions were unexpectedly shallower in the experimental group. Two main hypotheses could explain this fact. First, most compressions were delivered in an over-the-head position [62,63,64], and the cumbersome airway management apparatus (i-gel® + antibacterial filter + BVM) coupled with a shallower i-gel® insertion depth in the manikin might have prevented the adequate provision of chest compressions. Second, participants might have been too focused on the i-gel® device, the use of which was new to them, thus downgrading the quality of compressions. Regardless of the actual reasons explaining these shallower compressions, this issue must be fixed before implementation of the i-gel® in the field can be considered. Further studies should now be carried out to assess the most effective way of maintaining an adequate compression depth. These studies could consider two different approaches: forcing participants to deliver compressions from the patient’s side [65], using a feedback device [66,67,68,69] or both together.
Strengths and Limitations
The main limitation is the simulated context, and clinical studies will be needed to validate this experimental approach once the issue regarding the shallow compressions has been addressed. Another limitation is that the recruitment strategy could have biased the sample through a self-selection phenomenon. Randomisation should, however, have mitigated this phenomenon.
The resuscitation sequence outlined in the demonstration video might have been different than the sequence some participants were used to. In this video, both providers initially performed CPR together and switched to single-rescuer CPR only two minutes after the first shock had been delivered to allow the other rescuer to gain intravenous access. This might have induced, in both groups, a different strategy than the one they would have chosen in their actual practice, thus affecting BLS quality. Nevertheless, randomisation should have smoothened this effect out. Moreover, it is necessary to highlight that the proposed approach may delay IV cannulation and the administration of medications. While this is hardly relevant regarding the chosen scenario of a VF, this should be assessed and, more specifically, in case of asystole or pulseless electrical activity.
Some outcomes, such as satisfaction, might be overestimated, as many participants were eager to increase their skills and knowledge and hoped to autonomously use the i-gel® device in the future. In addition, a Hawthorne effect cannot be ruled out, as participants knew that they were being observed and that the results would be submitted for publication [70]. This might have happened despite the fact that participants were unaware of the study outcomes, as it was hardly possible to blind them to the type of intervention.
The strengths of this trial reside in its multicentre design, which allowed the inclusion of paramedics and EMTs from different EMS. The design was also rather original, and we believe that our results address a gap regarding the optimal approach in the initial management of OHCA, while raising other questions.
5. Conclusions
After minimal training, paramedics and EMTs using our experimental approach achieved a higher CCF and better ventilation parameters than those applying their standard of care. Our results show that the early insertion of an i-gel® device enables these enhancements in an adult OHCA simulation without delaying time to first ventilation or to first shock. However, chest compressions were shallower when the i-gel® was used, and further studies are needed to understand and address this issue before clinical trials can be considered.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jcm11010217/s1, Table S1: CONSORT 2010 Checklist, File S1: Standard Operating Procedure (French version), File S2: Standard Operating Procedure (English version), Figure S1: Compression rate, Figure S2: Satisfaction.
Author Contributions
Conceptualisation, L.S. (Loric Stuby) and D.T.; methodology, L.S. (Loric Stuby), D.T. and L.S. (Laurent Suppan); software, L.J. and L.S. (Loric Stuby); validation, L.S. (Laurent Suppan), L.S. (Loric Stuby), D.T. and L.J.; formal analysis, D.T.; investigation, D.T., L.J. and L.S. (Loric Stuby); resources, J.S., L.S. (Loric Stuby), L.J. and D.T.; data curation, L.S. (Loric Stuby); writing—original draft preparation, L.S. (Loric Stuby); writing—review and editing, L.S. (Laurent Suppan)., T.S., E.P., J.S., M.B., D.T., L.J. and L.S. (Loric Stuby); visualisation, L.S. (Loric Stuby); supervision, L.S. (Laurent Suppan) and L.S. (Loric Stuby); project administration, L.S. (Loric Stuby) and D.T.; funding acquisition, J.S., L.S. (Loric Stuby) and D.T. All authors have read and agreed to the published version of the manuscript.
Funding
“Swissrescue.ch—Website for Prehospital Healthcare Providers” provided funding for the manikin rental. EMS “Genève TEAM Ambulances” lent an ambulance to carry the manikins and provided the study’s supplies (syringes, needles, factices medications, etc…). Protocol’s publication fees were paid by a grant from the EMS “Compagnie d’Ambulances de l’Hôpital du Valais”, Martigny, Valais, and by private funds. The present article’s publication fees were paid by a grant from the Funded by the Presidents’ Conference as part of the section projects of the Swiss Paramedics Association. Apart from that, this research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and review and approval were waived by the regional Ethics Committee (CER-Vaud—Commission Cantonale d’Ethique de la Recherche sur l’Être Humain, Lausanne, Switzerland)—REQ-2020-01491—as such, studies do not fall under the Swiss Federal Human Research Act.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the participants to publish this paper.
Data Availability Statement
The datasets generated and analysed during the current study are available in Mendeley Data repository, (https://data.mendeley.com/datasets/98rf9psgvb/1) (accessed on 30 November 2021).
Acknowledgments
Thanks to the EMS for providing the premises and their warm welcome and to all the paramedics and EMTs for their voluntary participation. Specifically, thanks to André Baumann, Karine Corbat, Gilles Lemasson, Olivier Cassard, Jean-François Pillet, Marc Solioz and Cédric Frioud for the support.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript or in the decision to publish the results.
Trial Registration: NCT04736446 (3 February 2021).
References
- Panchal, A.R.; Bartos, J.A.; Cabañas, J.G.; Donnino, M.W.; Drennan, I.R.; Hirsch, K.G.; Kudenchuk, P.J.; Kurz, M.C.; Lavonas, E.J.; Morley, P.T.; et al. Part 3: Adult Basic and Advanced Life Support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2020, 142, S366–S468. [Google Scholar] [CrossRef]
- Soar, J.; Böttiger, B.W.; Carli, P.; Couper, K.; Deakin, C.D.; Djärv, T.; Lott, C.; Olasveengen, T.; Paal, P.; Pellis, T.; et al. European Resuscitation Council Guidelines 2021: Adult advanced life support. Resuscitation 2021, 161, 115–151. [Google Scholar] [CrossRef] [PubMed]
- Rea, T.; Olsufka, M.; Yin, L.; Maynard, C.; Cobb, L. The relationship between chest compression fraction and outcome from ventricular fibrillation arrests in prolonged resuscitations. Resuscitation 2014, 85, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Vaillancourt, C.; Everson-Stewart, S.; Christenson, J.; Andrusiek, D.; Powell, J.; Nichol, G.; Cheskes, S.; Aufderheide, T.P.; Berg, R.; Stiell, I. The impact of increased chest compression fraction on return of spontaneous circulation for out-of-hospital cardiac arrest patients not in ventricular fibrillation. Resuscitation 2011, 82, 1501–1507. [Google Scholar] [CrossRef] [Green Version]
- Uppiretla, A.K.; Gangalal, G.M.; Rao, S.; Bosco, D.D.; Shareef, S.M.; Sampath, V. Effects of Chest Compression Fraction on Return of Spontaneous Circulation in Patients with Cardiac Arrest: A Brief Report. Adv. J. Emerg. Med. 2019, 4, e8. [Google Scholar] [CrossRef] [PubMed]
- Christenson, J.; Andrusiek, D.; Everson-Stewart, S.; Kudenchuk, P.; Hostler, D.; Powell, J.; Callaway, C.; Bishop, D.; Vaillancourt, C.; Davis, D.; et al. Chest Compression Fraction Determines Survival in Patients with out-of-Hospital Ventricular Fibrillation. Circulation 2009, 120, 1241–1247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaillancourt, C.; Petersen, A.; Meier, E.N.; Christenson, J.; Menegazzi, J.J.; Aufderheide, T.P.; Nichol, G.; Berg, R.; Callaway, C.W.; Idris, A.H.; et al. The impact of increased chest compression fraction on survival for out-of-hospital cardiac arrest patients with a non-shockable initial rhythm. Resuscitation 2020, 154, 93–100. [Google Scholar] [CrossRef]
- Wik, L.; Olsen, J.-A.; Persse, D.; Sterz, F.; Lozano, M.; Brouwer, M.A.; Westfall, M.; Souders, C.M.; Travis, D.T.; Herken, U.R.; et al. Why do some studies find that CPR fraction is not a predictor of survival? Resuscitation 2016, 104, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Bobrow, B.J.; Clark, L.L.; Ewy, G.A.; Chikani, V.; Sanders, A.B.; Berg, R.A.; Richman, P.B.; Kern, K.B. Minimally Interrupted Cardiac Resuscitation by Emergency Medical Services for out-of-Hospital Cardiac Arrest. JAMA 2008, 299, 1158–1165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shimizu, K.; Wakasugi, M.; Kawagishi, T.; Hatano, T.; Fuchigami, T.; Okudera, H. Effect of Advanced Airway Management by Paramedics during out-of-Hospital Cardiac Arrest on Chest Compression Fraction and Return of Spontaneous Circulation. Open Access Emerg. Med. 2021, 13, 305–310. [Google Scholar] [CrossRef]
- Newell, C.; Grier, S.; Soar, J. Airway and ventilation management during cardiopulmonary resuscitation and after successful resuscitation. Crit. Care 2018, 22, 190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granfeldt, A.; Avis, S.R.; Nicholson, T.C.; Holmberg, M.J.; Moskowitz, A.; Coker, A.; Berg, K.M.; Parr, M.; Donnino, M.W.; Soar, J.; et al. Advanced airway management during adult cardiac arrest: A systematic review. Resuscitation 2019, 139, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Olasveengen, T.; Mancini, M.; Berg, R.; Brooks, S.; Castren, M.; Chung, S.; Considine, J.; Escalante, R. CPR: Chest Compression to Ventilation Ratio—EMS Delivered (BLS): Systematic Review. Available online: https://costr.ilcor.org/document/cpr-chest-compression-to-ventilation-ratio-ems-delivered (accessed on 12 February 2021).
- Carney, N.; Totten, A.M.; Cheney, T.; Jungbauer, R.; Neth, M.R.; Weeks, C.; Davis-O’Reilly, C.; Fu, R.; Yu, Y.; Chou, R.; et al. Prehospital Airway Management: A Systematic Review. Prehospital Emerg. Care 2021, 1–12. [Google Scholar] [CrossRef]
- Malinverni, S.; Bartiaux, M.; Cavallotto, F.; De Longueville, D.; Mols, P.; Gorlicki, J.; Adnet, F. Does endotracheal intubation increases chest compression fraction in out of hospital cardiac arrest: A substudy of the CAAM trial. Resuscitation 2019, 137, 35–40. [Google Scholar] [CrossRef]
- Kurz, M.C.; Prince, D.K.; Christenson, J.; Carlson, J.; Stub, D.; Cheskes, S.; Lin, S.; Aziz, M.; Austin, M.; Vaillancourt, C.; et al. Association of advanced airway device with chest compression fraction during out-of-hospital cardiopulmonary arrest. Resuscitation 2016, 98, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Jensen, J.L.; Cheung, K.W.; Tallon, J.M.; Travers, A.H. Comparison of tracheal intubation and alternative airway techniques performed in the prehospital setting by paramedics: A systematic review. Can. J. Emerg. Med. 2010, 12, 135–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saracoglu, A.; Saracoglu, K. Advanced airway management in out-of-hospital cardiac arrest—to intubate or not to intubate: A narrative review of the existing literature. Anaesthesiol. Intensive Ther. 2020, 52, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Buis, M.L.; Maissan, I.M.; Hoeks, S.E.; Klimek, M.; Stolker, R.J. Defining the learning curve for endotracheal intubation using direct laryngoscopy: A systematic review. Resuscitation 2016, 99, 63–71. [Google Scholar] [CrossRef]
- Chan, M.; Fehlmann, C.A.; Pasquier, M.; Suppan, L.; Savoldelli, G.L. Endotracheal Intubation Success Rate in an Urban, Supervised, Resident-Staffed Emergency Mobile System: An 11-Year Retrospective Cohort Study. J. Clin. Med. 2020, 9, 238. [Google Scholar] [CrossRef] [Green Version]
- Duckett, J.; Fell, P.; Han, K.; Kimber, C.; Taylor, C. Introduction of the i-gel supraglottic airway device for prehospital airway management in a UK ambulance service. Emerg. Med. J. 2014, 31, 505–507. [Google Scholar] [CrossRef]
- Wharton, N.M.; Gibbison, B.; Gabbott, D.A.; Haslam, G.M.; Muchatuta, N.; Cook, T.M. I-gel insertion by novices in manikins and patients. Anaesthesia 2008, 63, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Leventis, C.; Chalkias, A.; Sampanis, M.A.; Foulidou, X.; Xanthos, T. Emergency airway management by paramedics: Comparison between standard endotracheal intubation, Laryngeal Mask Airway, and I-Gel. Eur. J. Emerg. Med. 2014, 21, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Castle, N.; Owen, R.; Hann, M.; Naidoo, R.; Reeves, D. Assessment of the speed and ease of insertion of three supraglottic airway devices by paramedics: A manikin study. Emerg. Med. J. 2010, 27, 860–863. [Google Scholar] [CrossRef]
- Goliasch, G.; Ruetzler, A.; Fischer, H.; Frass, M.; Sessler, D.I.; Ruetzler, K. Evaluation of advanced airway management in absolutely inexperienced hands: A randomized manikin trial. Eur. J. Emerg. Med. 2013, 20, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Ruetzler, K.; Roessler, B.; Potura, L.; Priemayr, A.; Robak, O.; Schuster, E.; Frass, M. Performance and skill retention of intubation by paramedics using seven different airway devices—A manikin study. Resuscitation 2011, 82, 593–597. [Google Scholar] [CrossRef]
- Chauhan, G.; Nayar, P.; Seth, A.; Gupta, K.; Panwar, M.; Agrawal, N. Comparison of clinical performance of the I-gel with LMA proseal. J. Anaesthesiol. Clin. Pharmacol. 2013, 29, 56–60. [Google Scholar] [CrossRef]
- Gabbott, D.A.; Beringer, R. The iGEL supraglottic airway: A potential role for resuscitation? Resuscitation 2007, 73, 161–162. [Google Scholar] [CrossRef]
- Middleton, P.M.; Simpson, P.; Thomas, R.; Bendall, J.C. Higher insertion success with the i-gel® supraglottic airway in out-of-hospital cardiac arrest: A randomised controlled trial. Resuscitation 2014, 85, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Häske, D.; Gaier, G.; Heinemann, N.; Schempf, B.; Renz, J.-U. Minimal training for first responders with the i-gel™ leads to successful use in prehospital cardiopulmonary resuscitation. Resuscitation 2019, 134, 167–168. [Google Scholar] [CrossRef] [Green Version]
- Theiler, L.; Gutzmann, M.; Kleine-Brueggeney, M.; Urwyler, N.; Kaempfen, B.; Greif, R. i-gel ™ supraglottic airway in clinical practice: A prospective observational multicentre study. Br. J. Anaesth. 2012, 109, 990–995. [Google Scholar] [CrossRef] [Green Version]
- Benger, J.R.; Kirby, K.; Black, S.; Brett, S.J.; Clout, M.; Lazaroo, M.J.; Nolan, J.; Reeves, B.C.; Robinson, M.; Scott, L.J.; et al. Effect of a Strategy of a Supraglottic Airway Device vs. Tracheal Intubation during out-of-Hospital Cardiac Arrest on Functional Outcome. JAMA 2018, 320, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Stone, B.; Chantler, P.; Baskett, P. The incidence of regurgitation during cardiopulmonary resuscitation: A comparison between the bag valve mask and laryngeal mask airway: The AIRWAYS-2 randomized clinical trial. Resuscitation 1998, 38, 3–6. [Google Scholar] [CrossRef]
- Piegeler, T.; Roessler, B.; Goliasch, G.; Fischer, H.; Schlaepfer, M.; Lang, S.; Ruetzler, K. Evaluation of six different airway devices regarding regurgitation and pulmonary aspiration during cardio-pulmonary resuscitation (CPR)—A human cadaver pilot study. Resuscitation 2016, 102, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Häske, D.; Schempf, B.; Gaier, G.; Niederberger, C. Performance of the i-gel™ during pre-hospital cardiopulmonary resuscitation. Resuscitation 2013, 84, 1229–1232. [Google Scholar] [CrossRef]
- Soar, J.; Berg, K.M.; Andersen, L.W.; Böttiger, B.W.; Cacciola, S.; Callaway, C.W.; Couper, K.; Cronberg, T.; D’Arrigo, S.; Deakin, C.D.; et al. Adult Advanced Life Support: 2020 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Resuscitation 2020, 156, A80–A119. [Google Scholar] [CrossRef]
- Nolan, J.P.; Maconochie, I.; Soar, J.; Olasveengen, T.M.; Greif, R.; Wyckoff, M.H.; Singletary, E.M.; Aickin, R.; Berg, K.M.; Mancini, M.E.; et al. Executive Summary: 2020 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Circulation 2020, 142, S2–S27. [Google Scholar] [CrossRef]
- Chan, A.-W.; Tetzlaff, J.M.; Altman, D.G.; Laupacis, A.; Gøtzsche, P.C.; Krleža-Jerić, K.; Hróbjartsson, A.; Mann, H.; Dickersin, K.; Berlin, J.A.; et al. SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials. Ann. Intern. Med. 2013, 158, 200–207. [Google Scholar] [CrossRef] [Green Version]
- Stuby, L.; Jampen, L.; Sierro, J.; Paus, E.; Spichiger, T.; Suppan, L.; Thurre, D. Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial. Healthcare 2021, 9, 354. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Keamk-Create Random and Balanced Teams. Available online: https://www.keamk.com/ (accessed on 5 January 2021).
- Create a Blocked Randomisation List. Sealed Envelope. Available online: https://www.sealedenvelope.com/simple-randomiser/v1/lists (accessed on 6 December 2020).
- Peyton, J.W.R. Teaching & Learning in Medical Practice; Manticore Europe Ltd.: Rickmansworth, Herts, UK, 1998; ISBN 978-1-900887-00-7. [Google Scholar]
- Stuby, L.; Currat, L.; Gartner, B.; Mayoraz, M.; Harbarth, S.; Suppan, L.; Suppan, M. Impact of Face-to-Face Teaching in Addition to Electronic Learning on Personal Protective Equipment Doffing Proficiency in Student Paramedics: Protocol for a Randomized Controlled Trial. JMIR Res. Protoc. 2021, 10, e26927. [Google Scholar] [CrossRef]
- Giacomino, K.; Caliesch, R.; Sattelmayer, K.M. The effectiveness of the Peyton’s 4-step teaching approach on skill acquisition of procedures in health professions education: A systematic review and meta-analysis with integrated meta-regression. PeerJ 2020, 8, e10129. [Google Scholar] [CrossRef]
- Dörges, V.; Ocker, H.; Hagelberg, S.; Wenzel, V.; Schmucker, P. Optimisation of tidal volumes given with self-inflatable bags without additional oxygen. Resuscitation 2000, 43, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Dörges, V.; Ocker, H.; Hagelberg, S.; Wenzel, V.; Idris, A.H.; Schmucker, P. Smaller tidal volumes with room-air are not sufficient to ensure adequate oxygenation during bag–valve–mask ventilation. Resuscitation 2000, 44, 37–41. [Google Scholar] [CrossRef]
- Baskett, P.; Nolan, J.; Parr, M. Tidal volumes which are perceived to be adequate for resuscitation. Resuscitation 1996, 31, 231–234. [Google Scholar] [CrossRef]
- Aramendi, E.; Lu, Y.; Chang, M.P.; Elola, A.; Irusta, U.; Owens, P.; Idris, A.H. A novel technique to assess the quality of ventilation during pre-hospital cardiopulmonary resuscitation. Resuscitation 2018, 132, 41–46. [Google Scholar] [CrossRef]
- Neto, A.S.; Cardoso, S.O.; Manetta, J.A.; Pereira, V.G.M.; Espósito, D.C.; Pasqualucci, M.D.O.P.; Damasceno, M.C.T.; Schultz, M.J. Association between Use of Lung-Protective Ventilation with Lower Tidal Volumes and Clinical Outcomes among Patients without Acute Respiratory Distress Syndrome: A Meta-Analysis. JAMA 2012, 308, 1651–1659. [Google Scholar] [CrossRef] [Green Version]
- Stuby, L. CPR—Early Insertion Effect of a Supraglottic Airway Device on CCF in Simulated OHCA—PHP Code for Data Extraction from SimMan® 3G. Mendeley Data, V1. 2021. Available online: https://data.mendeley.com/datasets/s8d2gpfhyw/1 (accessed on 30 November 2021).
- Christiansen, T.; Lauritsen, J. EpiData Software. Available online: https://www.epidata.dk/ (accessed on 18 December 2020).
- Stuby, L.; Thurre, D.; Jampen, L.; Spichiger, T.; Sierro, J.; Bergeron, M.; Paus, E. CPR-Early Insertion Effect of a Supraglottic Airway Device on CCF in simulated OHCA-Dataset. Mendeley Data, V1. 2021. Available online: https://data.mendeley.com/datasets/98rf9psgvb/1 (accessed on 30 November 2021).
- Sealed Envelope. Power Calculator for Continuous Outcome Superiority Trial. Available online: https://www.sealedenvelope.com/power/continuous-superiority/ (accessed on 8 June 2021).
- Vogt, L.; Sellmann, T.; Wetzchewald, D.; Schwager, H.; Russo, S.; Marsch, S. Effects of Bag Mask Ventilation and Advanced Airway Management on Adherence to Ventilation Recommendations and Chest Compression Fraction: A Prospective Randomized Simulator-Based Trial. J. Clin. Med. 2020, 9, 2045. [Google Scholar] [CrossRef]
- Cereceda-Sánchez, F.; Molina-Mula, J.; Clar-Terradas, J.; Mascaró-Galmes, A.; Montero-París, P.; Martinez-Cuellar, N. 7 Bag-valve-mask vs. laryngeal mask (I-Gel®) during basic instrumental CPR with capnography monitoring: Preliminary results of a randomized cluster trial. BMJ Open 2019, 9, A3. [Google Scholar] [CrossRef]
- Lavonas, E.J.; Ohshimo, S.; Nation, K.; Van de Voorde, P.; Nuthall, G.; Maconochie, I.; Torabi, N.; Morrison, L.J.; DeCaen, A.; Atkins, D.; et al. International Liaison Committee on Resuscitation (ILCOR) Pediatric Life Support Task Force Advanced Airway Interventions for Paediatric Cardiac Arrest: A Systematic Review and Meta-Analysis. Resuscitation 2019, 138, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.L.; Walker, M.; Leroux, Y.; Carter, A. Chest Compression Fraction in Simulated Cardiac Arrest Management by Primary Care Paramedics: King Laryngeal Tube Airway Versus Basic Airway Management. Prehospital Emerg. Care 2013, 17, 285–290. [Google Scholar] [CrossRef]
- Koka, A.; Suppan, L.; Cottet, P.; Carrera, E.; Stuby, L.; Suppan, M. Teaching the National Institutes of Health Stroke Scale to Paramedics (E-Learning vs. Video): Randomized Controlled Trial. J. Med Internet Res. 2020, 22, e18358. [Google Scholar] [CrossRef]
- Suppan, L.; Abbas, M.; Stuby, L.; Cottet, P.; Larribau, R.; Golay, E.; Iten, A.; Harbarth, S.; Gartner, B.; Suppan, M. Effect of an E-Learning Module on Personal Protective Equipment Proficiency among Prehospital Personnel: Web-Based Randomized Controlled Trial. J. Med. Internet Res. 2020, 22, e21265. [Google Scholar] [CrossRef]
- Suppan, L.; Stuby, L.; Gartner, B.; Larribau, R.; Iten, A.; Abbas, M.; Harbarth, S.; Suppan, M. Impact of an e-learning module on personal protective equipment knowledge in student paramedics: A randomized controlled trial. Antimicrob. Resist. Infect. Control. 2020, 9, 1–9. [Google Scholar] [CrossRef]
- Perkins, G.D.; Stephenson, B.T.; Smith, C.M.; Gao, F. A comparison between over-the-head and standard cardiopulmonary resuscitation. Resuscitation 2004, 61, 155–161. [Google Scholar] [CrossRef]
- Chi, C.-H.; Tsou, J.-Y.; Su, F.-C. Comparison of chest compression kinematics associated with over-the-head and standard cardiopulmonary resuscitation. Am. J. Emerg. Med. 2009, 27, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Ćwiertnia, M.; Kawecki, M.; Ilczak, T.; Mikulska, M.; Dutka, M.; Bobiński, R. Comparison of standard and over-the-head method of chest compressions during cardiopulmonary resuscitation—A simulation study. BMC Emerg. Med. 2019, 19, 73. [Google Scholar] [CrossRef] [Green Version]
- Maisch, S.; Issleib, M.; Kuhls, B.; Mueller, J.; Horlacher, T.; Goetz, A.E.; Schmidt, G.N. A Comparison between over-The-Head and Standard Cardiopulmonary Resuscitation Performed by Two Rescuers: A Simulation Study. J. Emerg. Med. 2010, 39, 369–376. [Google Scholar] [CrossRef]
- Davis, D.P.; Graham, P.G.; Husa, R.D.; Lawrence, B.; Minokadeh, A.; Altieri, K.; Sell, R.E. A performance improvement-based resuscitation programme reduces arrest incidence and increases survival from in-hospital cardiac arrest. Resuscitation 2015, 92, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Bobrow, B.J.; Vadeboncoeur, T.F.; Stolz, U.; Silver, A.E.; Tobin, J.M.; Crawford, S.A.; Mason, T.K.; Schirmer, J.; Smith, G.A.; Spaite, D. The Influence of Scenario-Based Training and Real-Time Audiovisual Feedback on out-of-Hospital Cardiopulmonary Resuscitation Quality and Survival from out-of-Hospital Cardiac Arrest. Ann. Emerg. Med. 2013, 62, 47–56.e1. [Google Scholar] [CrossRef]
- Smith, K.; Dyson, K.; Stub, D.; Magnuson, N.; Anastasopoulos, K.; Bernard, S. 29 Feasibility of using a defibrillator to provide real-time and post-event feedback to paramedics on the quality of their CPR. BMJ Open 2019, 9, A11. [Google Scholar] [CrossRef]
- Lakomek, F.; Lukas, R.-P.; Brinkrolf, P.; Mennewisch, A.; Steinsiek, N.; Gutendorf, P.; Sudowe, H.; Heller, M.; Kwiecien, R.; Zarbock, A.; et al. Real-time feedback improves chest compression quality in out-of-hospital cardiac arrest: A prospective cohort study. PLoS ONE 2020, 15, e0229431. [Google Scholar] [CrossRef] [Green Version]
- Campbell, J.P.; Maxey, V.A.; Watson, W.A. Hawthorne Effect: Implications for Prehospital Research. Ann. Emerg. Med. 1995, 26, 590–594. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).