Systematic Review of Incidence Studies of Pneumonia in Persons with Spinal Cord Injury
Abstract
1. Introduction
2. Materials and Methods
2.1. Types of Studies
2.2. Literature Searches
Eligibility Criteria
2.3. Study Records
2.4. Data Extraction
2.5. Data Processing
2.6. Risk-of-Bias Assessment
2.7. Data Synthesis
3. Results
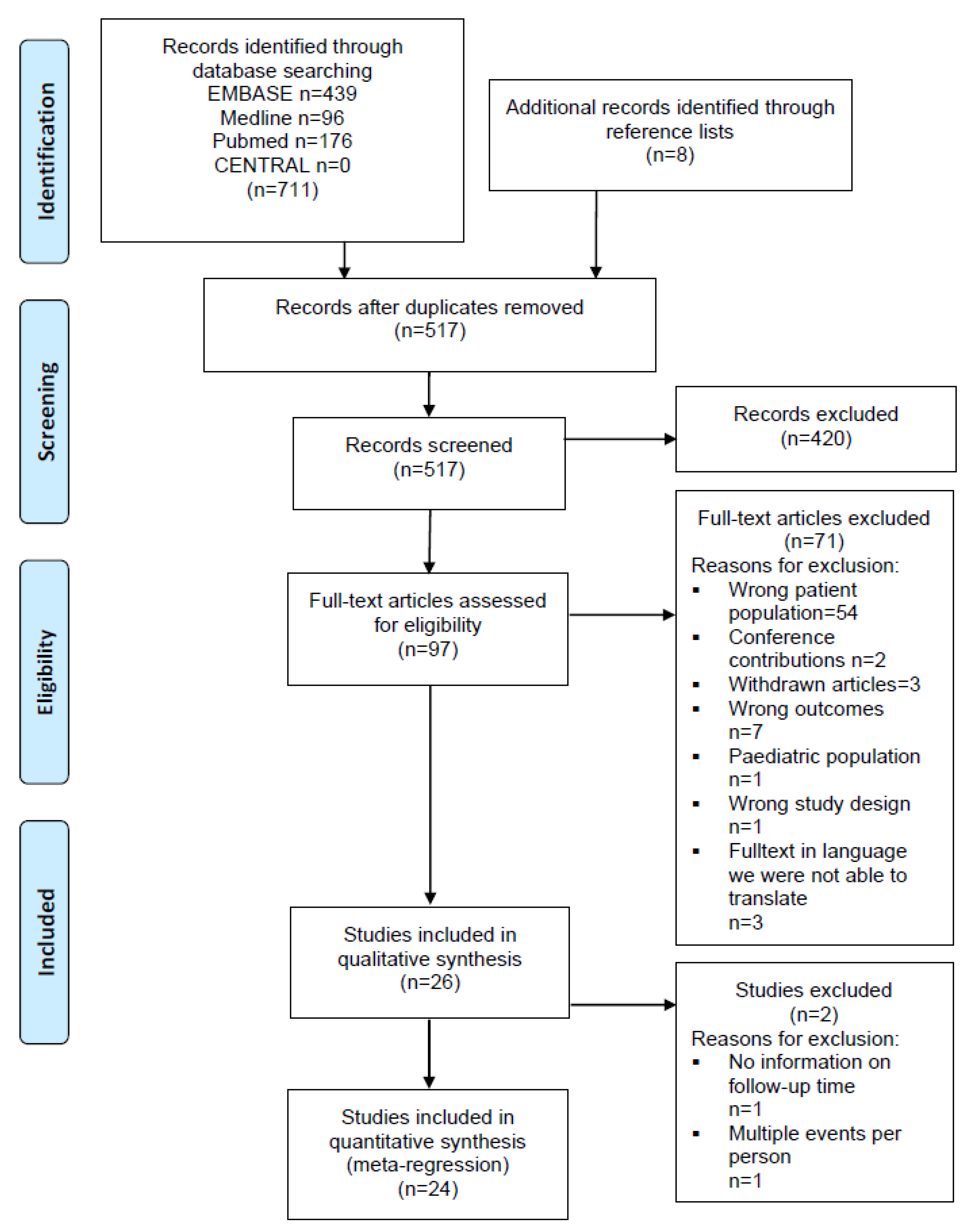
3.1. Studies Identified
3.2. Risk of Bias
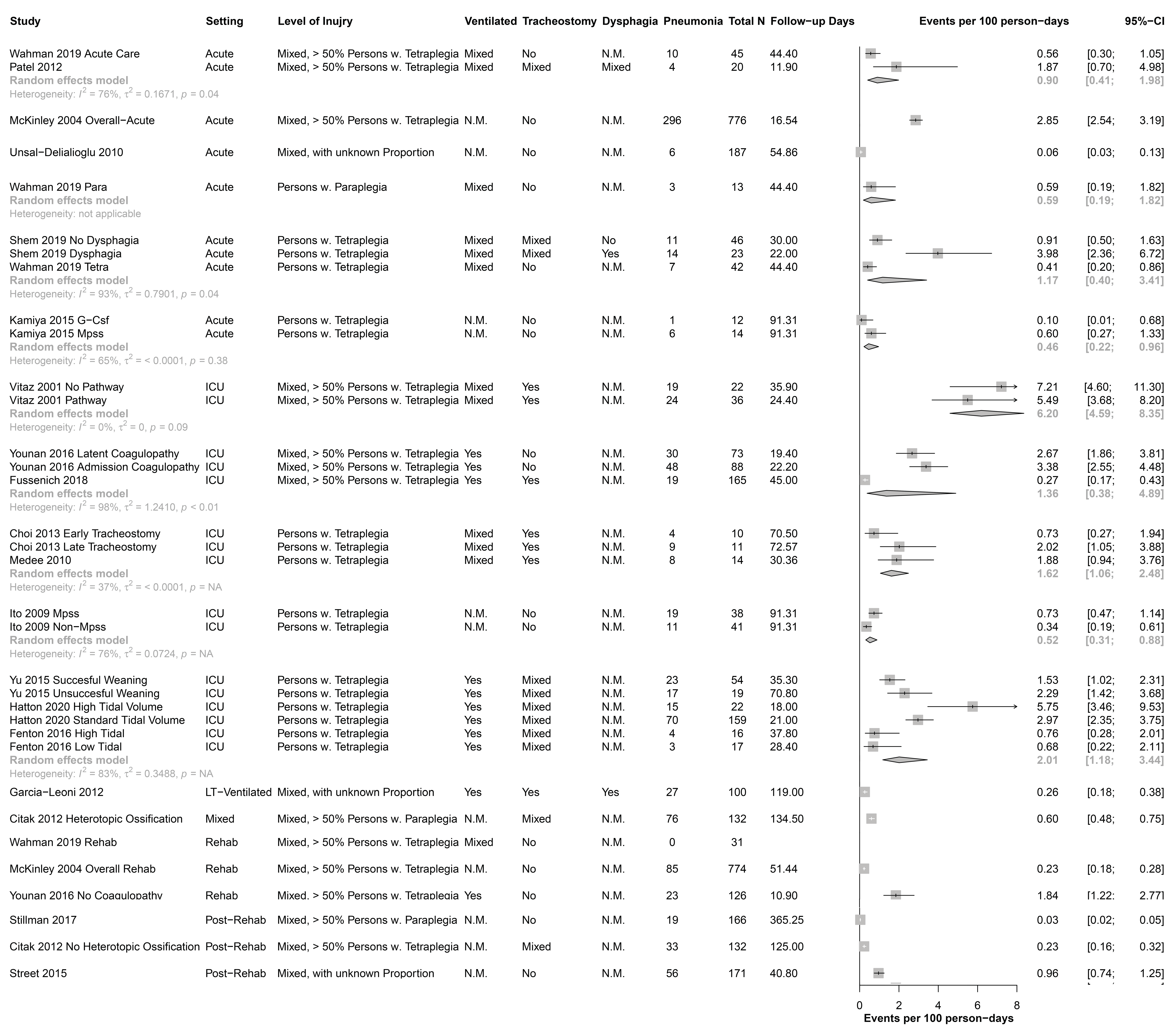
3.3. Incidence Rates per 100 Person-Days and Incidence-Rate Ratios
3.4. Sensitivity Analyses
4. Discussion
4.1. Overall Completeness of Evidence
4.2. Covartiates
4.2.1. Setting
4.2.2. Level of Injury
4.2.3. Mechanical Ventilation
4.2.4. Tracheostomy
4.2.5. Dysphagia
4.2.6. General Aspects
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marsolais, E.B.; Boninger, M.L.; McCormick, P.C.; Love, L.; Mackelprang, R.W.; Dalsey, W.C. Respiratory management following spinal cord injury: A clinical practice guideline for health-care professionals. J. Spinal Cord Med. 2005, 28, 259–293. [Google Scholar]
- Wang, A.Y.; Jaeger, R.J.; Yarkony, G.M.; Turba, R.M. Cough in spinal cord injured patients: The relationship between motor level and peak expiratory flow. Spinal Cord 1997, 35, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Lanig, I.S.; Peterson, W.P. The respiratory system in spinal cord injury. Phys. Med. Rehabil. Clin. N. Am. 2000, 11, 29–43. [Google Scholar] [CrossRef]
- Savic, G.; DeVivo, M.J.; Frankel, H.L.; Jamous, M.A.; Soni, B.M.; Charlifue, S. Causes of death after traumatic spinal cord injury-a 70-year British study. Spinal Cord 2017, 55, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.B.; Groomes, T.E. Incidence of respiratory complications following spinal cord injury. Arch. Phys. Med. Rehabil. 1994, 75, 270–275. [Google Scholar] [CrossRef]
- Berlly, M.; Shem, K. Respiratory management during the first five days after spinal cord injury. J. Spinal Cord Med. 2007, 30, 309–318. [Google Scholar] [CrossRef] [PubMed]
- CDC. Causes of Pneumonia. Available online: https://www.cdc.gov/pneumonia/causes.html (accessed on 18 May 2021).
- Torres, A.; Blasi, F.; Peetermans, W.E.; Viegi, G.; Welte, T. The aetiology and antibiotic management of community-acquired pneumonia in adults in Europe: A literature review. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1065–1079. [Google Scholar] [CrossRef] [PubMed]
- Welte, T.; Torres, A.; Nathwani, D. Clinical and economic burden of community-acquired pneumonia among adults in Europe. Thorax 2012, 67, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Agostinello, J.; Battistuzzo, C.R.; Batchelor, P.E. Early clinical predictors of pneumonia in critically ill spinal cord injured individuals: A retrospective cohort study. Spinal Cord 2019, 57, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Osterthun, R.; Post, M.W.; van Asbeck, F.W.; van Leeuwen, C.M.; van Koppenhagen, C.F. Causes of death following spinal cord injury during inpatient rehabilitation and the first five years after discharge. A Dutch cohort study. Spinal Cord 2014, 52, 483–488. [Google Scholar] [CrossRef]
- Ahoniemi, E.; Pohjolainen, T.; Kautiainen, H. Survival after spinal cord injury in Finland. J. Rehabil. Med. 2011, 43, 481–485. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lidal, I.B.; Snekkevik, H.; Aamodt, G.; Hjeltnes, N.; Biering-Sorensen, F.; Stanghelle, J.K. Mortality after spinal cord injury in Norway. J. Rehabil. Med. 2007, 39, 145–151. [Google Scholar] [CrossRef]
- Weaver, F.M.; Smith, B.; Evans, C.T.; Kurichi, J.E.; Patel, N.; Kapur, V.K.; Burns, S.P. Outcomes of outpatient visits for acute respiratory illness in veterans with spinal cord injuries and disorders. Am. J. Phys. Med. Rehabil. 2006, 85, 718–726. [Google Scholar] [CrossRef]
- Chen, C.F.; Lien, I.N.; Wu, M.C. Respiratory function in patients with spinal cord injuries: Effects of posture. Paraplegia 1990, 28, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.; DiMarco, A.F.; Hoit, J.D.; Garshick, E. Respiratory dysfunction and management in spinal cord injury. Respir. Care 2006, 51, 853–868. [Google Scholar]
- Emmi, V. [Guidelines for treatment of pneumonia in intensive care units]. Infez. Med. 2005, 7–17. Available online: https://pubmed.ncbi.nlm.nih.gov/16801748/ (accessed on 18 May 2021).
- Hayashi, T.; Fujiwara, Y.; Sakai, H.; Kubota, K.; Kawano, O.; Mori, E.; Takao, T.; Masuda, M.; Morishita, Y.; Maeda, T. The time course of dysphagia following traumatic cervical spinal cord injury: A prospective cohort study. Spinal Cord 2020, 58, 53–57. [Google Scholar] [CrossRef]
- Shem, K.; Castillo, K.; Wong, S.; Chang, J. Dysphagia in individuals with tetraplegia: Incidence and risk factors. J. Spinal Cord Med. 2011, 34, 85–92. [Google Scholar] [CrossRef]
- Shem, K.; Castillo, K.; Wong, S.L.; Chang, J.; Kolakowsky-Hayner, S. Dysphagia and respiratory care in individuals with tetraplegia: Incidence, associated factors, and preventable complications. Top. Spinal Cord Inj. Rehabil. 2012, 18, 15–22. [Google Scholar] [CrossRef]
- Martin, N.D.; Marks, J.A.; Donohue, J.; Giordano, C.; Cohen, M.J.; Weinstein, M.S. The mortality inflection point for age and acute cervical spinal cord injury. J. Trauma 2011, 71, 380–385; discussion 385–386. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Moola, S.; Riitano, D.; Lisy, K. The development of a critical appraisal tool for use in systematic reviews addressing questions of prevalence. Int. J. Health Policy Manag. 2014, 3, 123–128. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Biering-Sørensen, F.; DeVivo, M.; Charlifue, S.; Chen, Y.; New, P.; Noonan, V.; Post, M.; Vogel, L. International spinal cord injury core data set (version 2.0)—including standardization of reporting. Spinal Cord 2017, 55, 759–764. [Google Scholar] [CrossRef] [PubMed]
- CDC. Lesson 3: Measures of Risk; Sectiion 2: Morbidity Frequency Measures. Available online: https://www.cdc.gov/csels/dsepd/ss1978/lesson3/section2.html#:~:text=The%20denominator%20of%20an%20incidence,be%20included%20in%20the%20numerator. (accessed on 18 May 2021).
- Hoy, D.; Brooks, P.; Woolf, A.; Blyth, F.; March, L.; Bain, C.; Baker, P.; Smith, E.; Buchbinder, R. Assessing risk of bias in prevalence studies: Modification of an existing tool and evidence of interrater agreement. J. Clin. Epidemiol. 2012, 65, 934–939. [Google Scholar] [CrossRef] [PubMed]
- Stijnen, T.; Hamza, T.H.; Ozdemir, P. Random effects meta-analysis of event outcome in the framework of the generalized linear mixed model with applications in sparse data. Stat. Med. 2010, 29, 3046–3067. [Google Scholar] [CrossRef]
- Oliveira, J.; Zagalo, C.; Cavaco-Silva, P. Prevention of ventilator-associated pneumonia. Rev. Port. Pneumol. 2014, 20, 152–161. [Google Scholar] [CrossRef]
- Shem, K.; Wong, J.; Dirlikov, B.; Castillo, K. Pharyngeal Dysphagia in Individuals With Cervical Spinal Cord Injury: A Prospective Observational Cohort Study. Top. Spinal Cord Inj. Rehabil. 2019, 25, 322–330. [Google Scholar] [CrossRef]
- Spittal, M.J.; Pirkis, J.; Gurrin, L.C. Meta-analysis of incidence rate data in the presence of zero events. BMC Med. Res. Methodol. 2015, 15, 42. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, B.R.; Sterne, J.A. Essential Medical Statistics; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. Evid. Based Ment. Health 2019, 22, 153–160. [Google Scholar] [CrossRef]
- Viechtbauer, W. Conducting Meta-Analyses in R with the metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Garcia-Leoni, M.E.; Moreno, S.; Garcia-Garrote, F.; Cercenado, E. Ventilator-associated pneumonia in long-term ventilator-assisted individuals. Spinal Cord 2010, 48, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Medee, B.; Girard, R.; Loukili, A.; Loiseau, K.; Tell, L.; Rode, G. Lower respiratory events in seated tracheotomized tetraplegic patients. Eur. J. Phys. Rehabil. Med. 2010, 46, 37–42. [Google Scholar] [PubMed]
- Choi, H.J.; Paeng, S.H.; Kim, S.T.; Lee, K.S.; Kim, M.S.; Jung, Y.T. The Effectiveness of Early Tracheostomy (within at least 10 Days) in Cervical Spinal Cord Injury Patients. J. Korean Neurosurg. Soc. 2013, 54, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Citak, M.; Suero, E.M.; Backhaus, M.; Aach, M.; Godry, H.; Meindl, R.; Schildhauer, T.A. Risk factors for heterotopic ossification in patients with spinal cord injury: A case-control study of 264 patients. Spine 2012, 37, 1953–1957. [Google Scholar] [CrossRef] [PubMed]
- Fenton, J.J.; Warner, M.L.; Lammertse, D.; Charlifue, S.; Martinez, L.; Dannels-McClure, A.; Kreider, S.; Pretz, C. A comparison of high vs. standard tidal volumes in ventilator weaning for individuals with sub-acute spinal cord injuries: A site-specific randomized clinical trial. Spinal Cord 2016, 54, 234–238. [Google Scholar] [CrossRef]
- Fussenich, W.; Hirschfeld Araujo, S.; Kowald, B.; Hosman, A.; Auerswald, M.; Thietje, R. Discontinuous ventilator weaning of patients with acute SCI. Spinal Cord 2018, 56, 461–468. [Google Scholar] [CrossRef]
- Hatton, G.E.; Mollett, P.J.; Du, R.E.; Wei, S.; Korupolu, R.; Wade, C.E.; Adams, S.D.; Kao, L.S. High tidal volume ventilation is associated with ventilator-associated pneumonia in acute cervical spinal cord injury. J. Spinal Cord Med. 2020, 44, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Sugimoto, Y.; Tomioka, M.; Kai, N.; Tanaka, M. Does high dose methylprednisolone sodium succinate really improve neurological status in patient with acute cervical cord injury?: A prospective study about neurological recovery and early complications. Spine 2009, 34, 2121–2124. [Google Scholar] [CrossRef]
- Kamiya, K.; Koda, M.; Furuya, T.; Kato, K.; Takahashi, H.; Sakuma, T.; Inada, T.; Ota, M.; Maki, S.; Okawa, A.; et al. Neuroprotective therapy with granulocyte colony-stimulating factor in acute spinal cord injury: A comparison with high-dose methylprednisolone as a historical control. Eur. Spine J. 2015, 24, 963–967. [Google Scholar] [CrossRef]
- McKinley, W.; Meade, M.A.; Kirshblum, S.; Barnard, B. Outcomes of early surgical management versus late or no surgical intervention after acute spinal cord injury. Arch. Phys. Med. Rehabil. 2004, 85, 1818–1825. [Google Scholar] [CrossRef] [PubMed]
- McKinley, W.O.; Tewksbury, M.A.; Godbout, C.J. Comparison of medical complications following nontraumatic and traumatic spinal cord injury. J. Spinal Cord Med. 2002, 25, 88–93. [Google Scholar] [CrossRef]
- Patel, A.; Smith, H.E.; Radcliff, K.; Yadlapalli, N.; Vaccaro, A.R. Odontoid fractures with neurologic deficit have higher mortality and morbidity. Clin. Orthop. Relat. Res. 2012, 470, 1614–1620. [Google Scholar] [CrossRef]
- Raab, A.M.; Krebs, J.; Perret, C.; Michel, F.; Hopman, M.T.; Mueller, G. Maximum Inspiratory Pressure is a Discriminator of Pneumonia in Individuals With Spinal-Cord Injury. Respir. Care 2016, 61, 1636–1643. [Google Scholar] [CrossRef]
- Smith, B.M.; Evans, C.T.; Kurichi, J.E.; Weaver, F.M.; Patel, N.; Burns, S.P. Acute respiratory tract infection visits of veterans with spinal cord injuries and disorders: Rates, trends, and risk factors. J. Spinal Cord Med. 2007, 30, 355–361. [Google Scholar] [CrossRef][Green Version]
- Stillman, M.D.; Barber, J.; Burns, S.; Williams, S.; Hoffman, J.M. Complications of Spinal Cord Injury Over the First Year after Discharge from Inpatient Rehabilitation. Arch. Phys. Med. Rehabil. 2017, 98, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Street, J.T.; Noonan, V.K.; Cheung, A.; Fisher, C.G.; Dvorak, M.F. Incidence of acute care adverse events and long-term health-related quality of life in patients with TSCI. Spine J. 2015, 15, 923–932. [Google Scholar] [CrossRef]
- Unsal-Delialioglu, S.; Kaya, K.; Sahin-Onat, S.; Kulakli, F.; Culha, C.; Ozel, S. Fever during rehabilitation in patients with traumatic spinal cord injury: Analysis of 392 cases from a national rehabilitation hospital in Turkey. J. Spinal Cord Med. 2010, 33, 243–248. [Google Scholar] [CrossRef][Green Version]
- Wahman, K.; Nilsson Wikmar, L.; Chlaidze, G.; Joseph, C. Secondary medical complications after traumatic spinal cord injury in Stockholm, Sweden: Towards developing prevention strategies. J. Rehabil. Med. 2019, 51, 513–517. [Google Scholar] [CrossRef]
- Younan, D.; Lin, E.; Griffin, R.; Vanlandingham, S.; Waters, A.; Harrigan, M.; Pittet, J.F.; Kerby, J.D. Early Trauma-Induced Coagulopathy is Associated with Increased Ventilator-Associated Pneumonia in Spinal Cord Injury Patients. Shock 2016, 45, 502–505. [Google Scholar] [CrossRef]
- Yu, W.K.; Ko, H.K.; Ho, L.I.; Wang, J.H.; Kou, Y.R. Synergistic impact of acute kidney injury and high level of cervical spinal cord injury on the weaning outcome of patients with acute traumatic cervical spinal cord injury. Injury 2015, 46, 1317–1323. [Google Scholar] [CrossRef]
- Vitaz, T.W.; McIlvoy, L.; Raque, G.H.; Spain, D.A.; Shields, C.B. Development and implementation of a clinical pathway for spinal cord injuries. J. Spinal Disord. 2001, 14, 271–276. [Google Scholar] [CrossRef]
- Chastre, J.; Fagon, J.Y. Ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 2002, 165, 867–903. [Google Scholar] [CrossRef] [PubMed]
- Berlowitz, D.J.; Tamplin, J. Respiratory muscle training for cervical spinal cord injury. Cochrane Database Syst. Rev. 2013, 7, CD008507. [Google Scholar] [CrossRef]
- NNIS System. National Nosocomial Infections Surveillance (NNIS) System Report, Data Summary from January 1990–May 1999, issued June 1999. A report from the NNIS System. Am. J. Infect. Control 1999, 27, 520–532. [Google Scholar]
- Bassetti, M.; Taramasso, L.; Giacobbe, D.R.; Pelosi, P. Management of ventilator-associated pneumonia: Epidemiology, diagnosis and antimicrobial therapy. Expert Rev. Anti-Infect. Ther. 2012, 10, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Kalil, A.C.; Metersky, M.L.; Klompas, M.; Muscedere, J.; Sweeney, D.A.; Palmer, L.B.; Napolitano, L.M.; O’Grady, N.P.; Bartlett, J.G.; Carratalà, J.; et al. Management of Adults with Hospital-acquired and Ventilator-associated Pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin. Infect. Dis. 2016, 63, e61–e111. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.T.; Lavela, S.L.; Weaver, F.M.; Priebe, M.; Sandford, P.; Niemiec, P.; Miskevics, S.; Parada, J.P. Epidemiology of hospital-acquired infections in veterans with spinal cord injury and disorder. Infect. Control Hosp. Epidemiol. 2008, 29, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef]
- Giuliano, K.K.; Baker, D.; Quinn, B. The epidemiology of nonventilator hospital-acquired pneumonia in the United States. Am. J. Infect. Control 2018, 46, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Galeiras Vazquez, R.; Rascado Sedes, P.; Mourelo Farina, M.; Montoto Marques, A.; Ferreiro Velasco, M.E. Respiratory management in the patient with spinal cord injury. BioMed Res. Int. 2013, 2013, 168757. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, C.D.; Childs, B.R.; Moore, T.A.; Vallier, H.A. Early Tracheostomy in Patients with Traumatic Cervical Spinal Cord Injury Appears Safe and May Improve Outcomes. Spine 2018, 43, 1110–1116. [Google Scholar] [CrossRef]
- Ganuza, J.R.; Forcada, A.G.; Gambarrutta, C.; Buigues, E.D.D.L.L.; Gonzalez, V.E.M.; Fuentes, F.P.; Luciani, A.A. Effect of technique and timing of tracheostomy in patients with acute traumatic spinal cord injury undergoing mechanical ventilation. J. Spinal Cord Med. 2011, 34, 76–84. [Google Scholar] [CrossRef]
- Arabi, Y.; Haddad, S.; Shirawi, N.; Al Shimemeri, A. Early tracheostomy in intensive care trauma patients improves resource utilization: A cohort study and literature review. Crit. Care 2004, 8, R347–R352. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Yang, Z.; Wang, J.; Liu, X.; Cao, Y.; Pan, Y.; Han, L.; Zhan, S. Prevalence of Pseudomonas aeruginosa and antimicrobial-resistant Pseudomonas aeruginosa in patients with pneumonia in mainland China: A systematic review and meta-analysis. Int. J. Infect. Dis. 2016, 49, 119–128. [Google Scholar] [CrossRef]
- Bath, P.M.; Lee, H.S.; Everton, L.F. Swallowing therapy for dysphagia in acute and subacute stroke. Cochrane Database Syst. Rev. 2018, 10, Cd000323. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.E. Aspiration pneumonitis and aspiration pneumonia. N. Engl. J. Med. 2001, 344, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.E.; Restrepo, M.I. New perspectives in aspiration community acquired Pneumonia. Expert Rev. Clin. Pharmacol. 2019, 12, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- The Cochrane Collaboration. Cochrane Handbook for Systematic Reviews of Interventions, Updated October 2019, 2nd ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2019. [Google Scholar]
| Citation | Year of Publication | Country | Type of Study | Sample Size (n) | Setting | Mean Age (±SD) | Sex Male(%) | SCI Classification Using AIS (%) | SCI Level of Injury (%) | Mechanical Ventilator Depen-Dency (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Choi et al. [39] Early Tracheostomy | 2013 | Korea | retro-spective | 10 | ICU | 54 ± 14 | 90 | A 20 B 20 C 60 D 0 | Tetra 100 | 70 |
| Choi et al. [39] Late Tracheostomy | 2013 | Korea | retrospective | 11 | ICU | 46 ± 17 | 91 | A 55 B 9 C 27 D 9 | Tetra 100 | 100 |
| Citak et al. [40] Heterotopic Ossification | 2012 | Germany | pro-spective | 132 | Mixed | 43 ± N.M. | 84 | A 83 B,C,D 17 | Tetra 55 Para 45 | N.M. |
| Citak et al. [40] No Heterotopic Ossification | 2012 | Germany | pro-spective | 132 | Mixed | 49 ± N.M. | 77 | A 46 B,C,D 54 | Tetra 49 Para 51 | N.M. |
| Fenton et al. [41] High Tidal | 2016 | USA | pro-spective | 16 | ICU | 39 ± 13 | 88 | A,B,C 100 | Tetra 100 | 100 |
| Fenton et al. [41] Low Tidal | 2016 | USA | pro-spective | 17 | ICU | 27 ± 7 | 65 | A,B,C 100 | Tetra 100 | 100 |
| Fussenich et al. [42] | 2018 | Germany | retro-spective | 165 | ICU | 57 ± 17 | 79 | A 46 B 12 C 32 D 11 | Tetra 82 Para 19 | 100 |
| Garcia-Leoni et al. [37] | 2010 | Spain | pro-spective | 100 | LT-Ventilated | 49 ± 17 | 75 | N.M. | Tetra 58 (missing data N.M.) | 100 |
| Hatton et al. [43] HighTidal Volume | 2020 | USA | retro-spective | 22 | ICU | 40 (27–51) | 77 | A 73 B,C,D 27 | Tetra 100 | 100 |
| Hatton et al. [43] Standard Tidal Voluma | 2020 | USA | retro-spective | 159 | ICU | 53 (35–70) | 79 | A 38 B,C,D 62 | Tetra 100 | 100 |
| Ito et al. [44] Mpss | 2009 | Japan | pro-spective | 38 | ICU | 55 ± N.M. | 79 | A 26 B 11 C 29 D 34 | Tetra 100 | N.M. |
| Ito et al. [44] Non-Mpss | 2009 | Japan | pro-spective | 41 | ICU | 80 | A 27 B 27 C 29 D 17 | Tetra 100 | N.M. | |
| Kamiya et al. [45] G-Csf | 2015 | Japan | retro-spective | 28 | Acute | 58 (38–72) | 75 | A 7 B 14 C 29 D 50 | Tetra 100 | N.M. |
| Kamiya et al. [45] Mpss | 2015 | Japan | retro-spective | 34 | Acute | 61 (18–85) | 79 | A 26 B 9 C 32 D 32 | Tetra 97 Unclear 3 | N.M. |
| * McKinley et al. [46] | 2004 | USA | retro-spective | 654 | Acute | 38 ± 16 | 79 | A 49 B,C,D 51 | Tetra 55 Para 45 | N.M. |
| McKinley et al. [47] Non-Traumatic | 2002 | USA | pro-spective | 38 | Acute | 55 ± 14 | 50 | A 5 B 29 C 34 D 31 | Tetra 34 Para 66 | N.M. |
| McKinley et al. [47] Traumatic | 2002 | USA | pro-spective | 79 | Acute | 39 ± 16 | 87 | A 46 B 22 C 18 D 15 | Tetra 42 Para 58 | N.M. |
| Medee et al. [38] | 2010 | France | retro-spective | 14 | ICU | 41 ± 18 | 57 | A,B 100 | Tetra 100 | 64 |
| Patel et al. [48] | 2012 | USA | retro-spective | 20 | Acute | 76 ± n.m | 65 | A 55 B 10 C 15 D 20 | Tetra 50 Para 5 Central Cord 45 | N.M. |
| Raab et al. [49] | 2016 | Switzer-land | retro-spective | 307 | Mixed | 53 ± 15 53 ± 18 | 81 | A 58 B 20 C 13 D 9 | N.M. | N.M. |
| Shem et al. [19] No Dysphagia | 2011 | USA | pro-spective | 17 | Acute | 35 ± 12 | 71 | N.M. | Tetra 100 | 41 |
| Shem et al. [19] Dysphagia | 2011 | USA | pro-spective | 12 | Acute | 49 ± 21 | 83 | N.M. | Tetra 100 | 67 |
| Shem et al. [20] No Dysphagia | 2012 | USA | pro-spective | 24 | Acute | 36 ± 13 | 71 | A 54 B,C,D 46 | Tetra 100 | 46 |
| Shem et al. [20] Dysphagia | 2012 | USA | pro-spective | 16 | Acute | 51 ± 18 | 88 | A 25 B,C,D 75 | Tetra 100 | 88 |
| Shem et al. [30] No Dysphagia | 2019 | USA | pro-spective | 53 | Acute | 39 ± 17 | 79 | A 47 B,C,D 53 | Tetra 100 | 40 |
| Shem et al. [30] Dysphagia | 2019 | USA | pro-spective | 23 | Acute | 48 ± 19 | 91 | A 35 B,C,D 65 | Tetra 100 | 65 |
| Smith et al. [50] | 2007 | USA | retro-spective | 18.693 | Mixed | 56 ± 14 | 98 | A 24 B,C,D 27 Unknown 32 | Tetra 33 Para 21 Missing 36 | N.M. |
| Stillman et al. [51] | 2017 | USA | pro-spective | 169 | Post-Rehab | 41 ± 16 | 79 | A,B,C 48 D 50 Unknown 2 | Tetra 23 Para 65 Unknown 2 | N.M. |
| Street et al. [52] | 2015 | Canada | pro-spective | 171 | Post-Rehab | 47 ± 20 | 81 | N.M. | N.M. | N.M. |
| Unsal-Delialioglu et al. [53] | 2010 | Turkey | retro-spective | 392 | Acute | 37 ± 14 | 76 | A 52 B 11 C 19 D 18 | N.M. | N.M. |
| # Wahman et al. [54] Para | 2019 | Sweden | pro-spective | 45 | Acute | 55 ± 17 | 60 | A 29 B,C,D 71 | Tetra 71 Para 29 | Yes (number N.M.) |
| Younan et al. [55] Latent Coagulopathy | 2016 | USA | retro-spective | 73 | ICU | 39 ± 17 | 82 | N.M. | ° Tetra N.M. Para N.M. | 100 |
| Younan et al. [55] Admission Coagulopathy | 2016 | USA | retro-spective | 88 | ICU | 39 ± 20 | 81 | N.M. | ° Tetra N.M. Para N.M. | 100 |
| Younan et al. [55] No Coagulopathy | 2016 | USA | retro-spective | 126 | Rehab | 44 ± 16 | 82 | N.M. | ° Tetra N.M.Para N.M. | 100 |
| Yu et al. [56] Successful Weaning | 2015 | Taiwan | retro-spective | 54 | ICU | 49 ± 19 | 83 | ∞ A 34B 10 C 10 D 4 Unknown 43 | Tetra 100 | 100 |
| Yu et al. [56] Unsuccessful Weaning | 2015 | Taiwan | retro-spective | 19 | ICU | 64 ± 17 | 84 | ∞ A 34 B 10 C 10 D 4 Unknown 43 | Tetra 100 | 100 |
| Vitaz et al. [57] No Pathway | 2001 | USA | retro-spective | 22 | ICU | 34 ± 10 | N.M. | N.M. | Tetra 86 Para 14 | Yes (number N.M.) |
| Vitaz et al. [57] Pathway | 2001 | USA | pro-spective | 36 | ICU | 33 ± 15 | N.M. | N.M. | Tetra 89 Para 11 | Yes (number N.M.) |
| Univariable Meta-Regression | Multivariable Meta-Regression | |||||
|---|---|---|---|---|---|---|
| Variable | Incidence Rate Ratio | 95% CI | p-Value | Incidence Rate Ratio | 95% CI | p-Value |
| Setting | ||||||
| Post-Rehab (Reference) | 1.00 | 1.00 | ||||
| Acute | 3.25 | 0.81 to 12.97 | 0.095 | 0.65 | 0.13 to 3.35 | 0.605 |
| ICU | 8.20 | 2.21 to 30.39 | 0.002 | 2.27 | 0.32 to 15.93 | 0.410 |
| Long-Term Ventilation | 1.33 | 0.12 to 14.6 | 0.817 | 0.97 | 0.05 to 17.64 | 0.983 |
| Mixed | 3.08 | 0.28 to 33.3 | 0.355 | 15.76 | 1.31 to 189.45 | 0.030 |
| Rehab | 3.23 | 0.49 to 21.41 | 0.224 | 0.75 | 0.1 to 5.9 | 0.785 |
| Level of Injury | ||||||
| Persons w. Paraplegia (Reference) | 1.00 | |||||
| Mixed, >50% Persons w. Paraplegia | 0.28 | 0.02 to 4.62 | 0.371 | 0.20 | 0.01 to 0.01 | 0.296 |
| Mixed, >50% Persons w. Tetraplegia | 2.54 | 0.22 to 30.11 | 0.459 | 3.64 | 0.41 to 0.41 | 0.244 |
| Persons w. Tetraplegia | 2.03 | 0.18 to 23.46 | 0.571 | 1.93 | 0.21 to 0.21 | 0.562 |
| Mixed, with unknown Proportion | 0.49 | 0.03 to 7.17 | 0.601 | 1.99 | 0.16 to 0.16 | 0.596 |
| Ventilation | ||||||
| Not mentioned (Reference) | 1.00 | |||||
| Mixed | 4.70 | 1.89 to 11.72 | 0.001 | 5.07 | 1.58 to 16.25 | 0.006 |
| Ventilated | 4.34 | 1.76 to 10.71 | 0.001 | 2.03 | 0.61 to 6.73 | 0.247 |
| Tracheostomy | ||||||
| No (Reference) | 1.00 | |||||
| Mixed | 2.55 | 0.99 to 6.57 | 0.053 | 1.17 | 0.42 to 3.21 | 0.763 |
| Yes | 2.52 | 0.85 to 7.48 | 0.096 | 0.41 | 0.1 to 1.69 | 0.217 |
| Dysphagia | ||||||
| No (Reference) | 1.00 | * | ||||
| Not mentioned | 0.95 | 0.07 to 12.18 | 0.968 | |||
| Mixed | 1.93 | 0.05 to 72.32 | 0.722 | |||
| Yes | 1.13 | 0.05 to 24.19 | 0.938 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raab, A.M.; Mueller, G.; Elsig, S.; Gandevia, S.C.; Zwahlen, M.; Hopman, M.T.E.; Hilfiker, R. Systematic Review of Incidence Studies of Pneumonia in Persons with Spinal Cord Injury. J. Clin. Med. 2022, 11, 211. https://doi.org/10.3390/jcm11010211
Raab AM, Mueller G, Elsig S, Gandevia SC, Zwahlen M, Hopman MTE, Hilfiker R. Systematic Review of Incidence Studies of Pneumonia in Persons with Spinal Cord Injury. Journal of Clinical Medicine. 2022; 11(1):211. https://doi.org/10.3390/jcm11010211
Chicago/Turabian StyleRaab, Anja Maria, Gabi Mueller, Simone Elsig, Simon C. Gandevia, Marcel Zwahlen, Maria T. E. Hopman, and Roger Hilfiker. 2022. "Systematic Review of Incidence Studies of Pneumonia in Persons with Spinal Cord Injury" Journal of Clinical Medicine 11, no. 1: 211. https://doi.org/10.3390/jcm11010211
APA StyleRaab, A. M., Mueller, G., Elsig, S., Gandevia, S. C., Zwahlen, M., Hopman, M. T. E., & Hilfiker, R. (2022). Systematic Review of Incidence Studies of Pneumonia in Persons with Spinal Cord Injury. Journal of Clinical Medicine, 11(1), 211. https://doi.org/10.3390/jcm11010211






