Osteoporosis Detection by Physical Function Tests in Resident Health Exams: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry
Abstract
1. Introduction
2. Materials and Methods
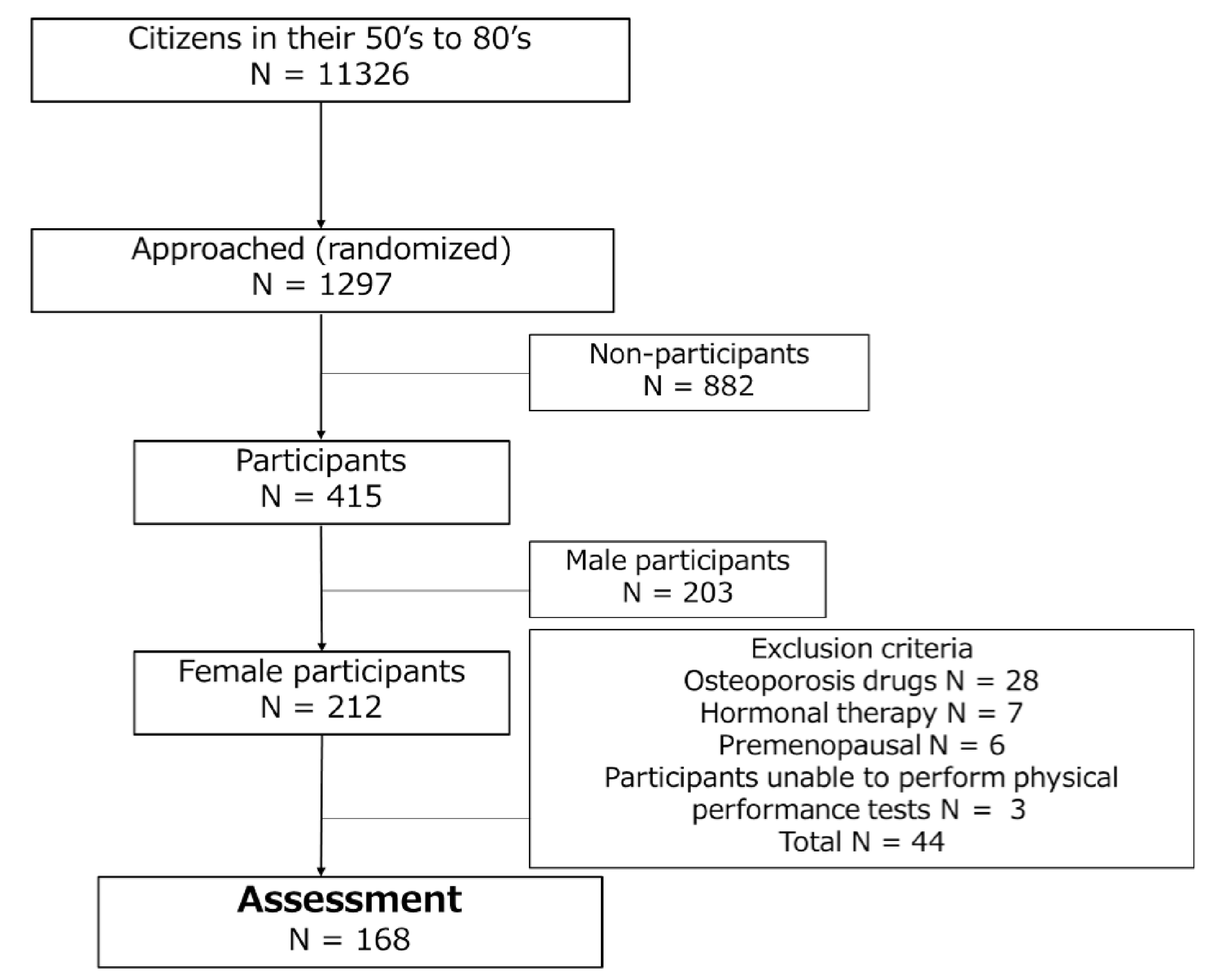
2.1. Participants
2.2. Osteoporosis Diagnosis
2.3. Physical Performance Tests
2.4. Statistical Analysis
3. Results
3.1. Baseline Data and Osteoporosis Prevalence
3.2. Physical Performance Test Results and Correlations with BMD
3.3. Physical Performance Tests Associated with Osteoporosis
3.4. Osteoporosis Screening by Physical Performance Tests
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech. Rep. Ser. 1994, 843, 1–129.
- Nguyen, N.D.; Center, J.R.; Eisman, J.A.; Nguyen, T.V. Bone loss, weight loss, and weight fluctuation predict mortality risk in elderly men and women. J. Bone Miner. Res. 2007, 22, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Yoshida, H. Low bone mineral density at femoral neck is a predictor of increased mortality in elderly Japanese women. Osteoporos. Int. 2010, 21, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Portal Site of Official Statistics of Japan Website. Statistics Dashboard. Available online: https://dashboard.e-stat.go.jp/dataSearch (accessed on 25 November 2020).
- Goeree, R.; Blackhouse, G.; Adachi, J. Cost-effectiveness of alternative treatments for women with osteoporosis in Canada. Curr. Med. Res. Opin. 2006, 22, 1425–1436. [Google Scholar] [CrossRef]
- Lo, S.S. Bone health status of postmenopausal Chinese women. Hong Kong Med. J. 2015, 21, 536–541. [Google Scholar] [CrossRef][Green Version]
- Tong, H.; Zong, H.; Xu, S.Q.; Wang, X.R.; Gong, X.; Xu, J.H.; Cheng, M. Osteoporosis Self-Assessment Tool as a Screening Tool for Predicting Osteoporosis in Elderly Chinese Patients With Established Rheumatoid Arthritis. J. Clin. Densitom. 2019, 22, 321–328. [Google Scholar] [CrossRef]
- Kanis, J.A.; Oden, A.; Johansson, H.; Borgström, F.; Ström, O.; McCloskey, E. FRAX and its applications to clinical practice. Bone 2009, 44, 734–743. [Google Scholar] [CrossRef]
- Miyakoshi, N.; Hongo, M.; Mizutani, Y.; Shimada, Y. Prevalence of sarcopenia in Japanese women with osteopenia and osteoporosis. J. Bone Miner. Metab. 2013, 31, 556–561. [Google Scholar] [CrossRef]
- Chan, D.C.; Lee, W.T.; Lo, D.H.; Leung, J.C.; Kwok, A.W.; Leung, P.C. Relationship between grip strength and bone mineral density in healthy Hong Kong adolescents. Osteoporos. Int. 2008, 19, 1485–1495. [Google Scholar] [CrossRef]
- Bettis, T.; Kim, B.J.; Hamrick, M.W. Impact of muscle atrophy on bone metabolism and bone strength: Implications for muscle-bone crosstalk with aging and disuse. Osteoporos. Int. 2018, 29, 1713–1720. [Google Scholar] [CrossRef]
- Brotto, M.; Bonewald, L. Bone and muscle: Interactions beyond mechanical. Bone 2015, 80, 109–114. [Google Scholar] [CrossRef]
- Brotto, M.; Johnson, M.L. Endocrine crosstalk between muscle and bone. Curr. Osteoporos. Rep. 2014, 12, 135–141. [Google Scholar] [CrossRef]
- Coupland, C.A.; Cliffe, S.J.; Bassey, E.J.; Grainge, M.J.; Hosking, D.J.; Chilvers, C.E. Habitual physical activity and bone mineral density in postmenopausal women in England. Int. J. Epidemiol. 1999, 28, 241–246. [Google Scholar] [CrossRef]
- Cousins, J.M.; Petit, M.A.; Paudel, M.L.; Taylor, B.C.; Hughes, J.M.; Cauley, J.A.; Zmuda, J.M.; Cawthon, P.M.; Ensrud, K.E.; Osteoporotic Fractures in Men (MrOS) Study Group. Muscle power and physical activity are associated with bone strength in older men: The osteoporotic fractures in men study. Bone 2010, 47, 205–211. [Google Scholar] [CrossRef]
- Kitamura, K.; Nakamura, K.; Kobayashi, R.; Oshiki, R.; Saito, T.; Oyama, M.; Takahashi, S.; Nishiwaki, T.; Iwasaki, M.; Yoshihara, A. Physical activity and 5-year changes in physical performance tests and bone mineral density in postmenopausal women: The Yokogoshi Study. Maturitas 2011, 70, 80–84. [Google Scholar] [CrossRef]
- Locquet, M.; Beaudart, C.; Bruyere, O.; Kanis, J.A.; Delandsheere, L.; Reginster, J.Y. Bone health assessment in older people with or without muscle health impairment. Osteoporos. Int. 2018, 29, 1057–1067. [Google Scholar] [CrossRef]
- Ikegami, S.; Uchiyama, S.; Nakamura, Y.; Mukaiyama, K.; Hirabayashi, H.; Kamimura, M.; Nonaka, K.; Kato, H. Factors that characterize bone health with aging in healthy postmenopausal women. J. Bone Miner. Metab. 2015, 33, 440–447. [Google Scholar] [CrossRef]
- Tokida, R.; Uehara, M.; Ikegami, S.; Takahashi, J.; Nishimura, H.; Sakai, N.; Kato, H. Association Between Sagittal Spinal Alignment and Physical Function in the Japanese General Elderly Population: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry. J. Bone Joint Surg. Am. 2019, 101, 1698–1706. [Google Scholar] [CrossRef]
- Orimo, H.; Hayashi, Y.; Fukunaga, M.; Sone, T.; Fujiwara, S.; Shiraki, M.; Hagino, H.; Hosoi, T.; Ohta, H.; Yoneda, T.; et al. Diagnostic criteria for primary osteoporosis: Year 2000 revision. J. Bone Miner. Metab. 2001, 19, 331–337. [Google Scholar] [CrossRef]
- Nakamura, K.; Ogata, T. Locomotive Syndrome: Definition and Management. Clin. Rev. Bone Miner. Metab. 2016, 14, 56–67. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Tachiki, T.; Kouda, K.; Dongmei, N.; Tamaki, J.; Iki, M.; Kitagawa, J.; Takahira, N.; Sato, Y.; Kajita, E.; Fujita, Y.; et al. Muscle strength is associated with bone health independently of muscle mass in postmenopausal women: The Japanese population-based osteoporosis study. J. Bone Miner. Metab. 2019, 37, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Elhakeem, A.; Hartley, A.; Luo, Y.; Goertzen, A.L.; Hannam, K.; Clark, E.M.; Leslie, W.D.; Tobias, J.H. Lean mass and lower limb muscle function in relation to hip strength, geometry and fracture risk indices in community-dwelling older women. Osteoporos. Int. 2019, 30, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.; Nordstrom, A.; Nordstrom, P. Objectively measured physical activity is associated with parameters of bone in 70-year-old men and women. Bone 2015, 81, 72–79. [Google Scholar] [CrossRef]
- Reid, K.F.; Naumova, E.N.; Carabello, R.J.; Phillips, E.M.; Fielding, R.A. Lower extremity muscle mass predicts functional performance in mobility-limited elders. J. Nutr. Health Aging 2008, 12, 493–498. [Google Scholar] [CrossRef]
- Muranaga, S.; Hirano, K. Development of a convenient way to predict ability to walk, using a two-step test. J. Showa Med. Assoc. 2003, 63, 301–308. [Google Scholar]
- Demura, S.; Yamada, T. The maximal double step length test can evaluate more adequately the decrease of physical function with age, than the maximal single step length test. Arch. Gerontol. Geriatr. 2010, 53, 21–24. [Google Scholar] [CrossRef]
- Ashe, M.; Liu-Ambrose, T.; Cooper, D.; Khan, K.; McKay, H. Muscle power is related to tibial bone strength in older women. Osteoporos. Int. 2008, 19, 1725–1732. [Google Scholar] [CrossRef]

| Age Stratum (Years) | N | Height (cm) | Weight (kg) | BMI (kg/m2) | L2-4 T-Score | Femoral Neck T-Score | Total Hip T-Score |
|---|---|---|---|---|---|---|---|
| 50–59 | 42 | 158.1 ± 5.1 | 55.0 ± 8.8 | 22.0 ± 3.9 | −0.2 ± 1.5 | −0.9 ± 0.9 | −0.6 ± 1.1 |
| 60–69 | 58 | 153.0 ± 5.1 | 52.4 ± 7.6 | 22.4 ± 2.8 | −0.8 ± 1.5 | −1.3 ± 0.9 | −1.0 ± 0.9 |
| 70–79 | 39 | 150.3 ± 5.5 | 51.6 ± 7.6 | 22.8 ± 3.1 | −0.9 ± 1.6 | −1.6 ± 0.9 | −1.1 ± 1.0 |
| 80–89 | 29 | 145.4 ± 5.8 | 50.4 ± 7.6 | 23.8 ± 3.1 | −0.7 ± 1.8 | −2.0 ± 0.7 | −1.9 ± 0.8 |
| Total | 168 | 152.3 ± 6.8 | 52.3 ± 8.0 | 22.6 ± 3.2 | −0.6 ± 1.6 | −1.4 ± 0.9 | −1.1 ± 1.0 |
| Age Stratum (Years) | Normal | Osteopenia | Osteoporosis | FRAX Major Osteoporotic † | FRAX Hip Fracture † |
|---|---|---|---|---|---|
| 50–59 | 22 (52.3%) | 16 (38.0%) | 4 (9.5%) | 4.3 ± 1.4 | 0.5 ± 0.7 |
| 60–69 | 13 (22.4%) | 34 (58.6%) | 11 (19.0%) | 6.9 ± 1.7 | 0.9 ± 0.7 |
| 70–79 | 9 (23.0%) | 19 (48.7%) | 11 (28.2%) | 9.4 ± 3.7 | 2.2 ± 2.1 |
| 80–89 | 2 (6.9%) | 17 (58.6%) | 10 (34.5%) | 14.7 ± 4.0 | 5.8 ± 2.9 |
| Total | 46 (27.4%) | 86 (51.2%) | 36 (21.4%) | 8.2 ± 4.4 | 1.9 ± 2.5 |
| Age Stratum (Years) | Grip Strength (kg) | Knee Extension (%Weight) | One-Leg Standing (Sec) | Two-Step Test (No Unit) | Stand-Up Test (Points) | Locomo25 (Points) |
|---|---|---|---|---|---|---|
| 50–59 | 25.1 ± 4.7 | 1.37 ± 0.36 | 47.6 ± 15.8 | 1.54 ± 0.13 | 4.4 ± 0.9 | 5.0 ± 4.6 |
| 60–69 | 21.8 ± 3.7 | 1.22 ± 0.36 | 44.2 ± 16.7 | 1.45 ± 0.14 | 4.4 ± 0.8 | 4.4 ± 4.6 |
| 70–79 | 20.7 ± 4.2 | 0.94 ± 0.34 | 24.6 ± 15.6 | 1.36 ± 0.19 | 3.5 ± 1.0 | 9.9 ± 10.2 |
| 80–89 | 16.9 ± 3.9 | 0.73 ± 0.36 | 9.1 ± 9.1 | 1.05 ± 0.25 | 3.0 ± 1.0 | 20.4 ± 15.3 |
| Total | 21.5 ± 4.9 | 1.10 ± 0.42 | 34.4 ± 20.9 | 1.38 ± 0.24 | 4.0 ± 1.1 | 8.6 ± 10.4 |
| L2-4 T-Score | Femoral Neck T-Score | Total Hip T-Score | ||||
|---|---|---|---|---|---|---|
| rho | p-Value | rho | p-Value | rho | p-Value | |
| Grip strength | 0.24 | <0.01 * | 0.38 | <0.01 * | 0.37 | <0.01 * |
| Knee extension | 0.03 | 0.73 | 0.25 | <0.01 * | 0.23 | <0.01 * |
| One-leg standing | 0.02 | 0.81 | 0.31 | <0.01 * | 0.25 | <0.01 * |
| Two-step test | 0.01 | 0.92 | 0.39 | <0.01 * | 0.43 | <0.01 * |
| Stand-up test | −0.11 | 0.17 | 0.12 | 0.10 | 0.17 | 0.02 * |
| Locomo25 | 0.10 | 0.18 | −0.19 | 0.01 * | −0.22 | <0.01 * |
| Age | −0.10 | 0.17 | −0.40 | <0.01 * | −0.41 | <0.01 * |
| BMI | 0.19 | 0.01 * | 0.11 | 0.17 | 0.17 | 0.02 * |
| Candidate | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Odds Ratio | p-Value | Odds Ratio | p-Value | |
| Grip strength (−1 kg) | 1.09 (1.01–1.19) | 0.03 * | ||
| Knee extension (−1%weight) | 1.31 (0.53–3.18) | 0.55 | ||
| One-leg standing (−1 sec) | 1.02 (1.01–1.04) | 0.01 * | ||
| Two-step test (−1) | 10.4 (2.30–46.7) | <0.01 * | 31.5 (5.29–188.0) | <0.01 * |
| Stand-up test (−1 point) | 1.12 (0.79–1.59) | 0.50 | ||
| Locomo25 (+1 point) | 1.02 (0.98–1.05) | 0.21 | ||
| Age (+1 year) | 1.06 (1.02–1.10) | <0.01 * | ||
| BMI (−1 kg/m2) | 1.18 (1.02–1.34) | 0.01 * | 1.3 (1.11–1.53) | <0.01 * |
| BMI/TST Score | 1.24 | 1.26 | 1.28 | 1.3 | 1.32 | 1.34 | 1.36 | 1.38 |
|---|---|---|---|---|---|---|---|---|
| 21.0 | 22.0 | 7.3 | 8.6 | 7.3 | 7.3 | 6.1 | 5.8 | 4.5 |
| 21.6 | 12.8 | 6.4 | 7.3 | 6.6 | 6.7 | 5.8 | 5.5 | 4.4 |
| 22.2 | 9.8 | 5.9 | 5.5 | 5.2 | 5.5 | 4.4 | 4.0 | 3.7 |
| 22.4 | 11.0 | 6.6 | 6.1 | 5.8 | 5.3 | 4.3 | 3.7 | 3.4 |
| 23.0 | 9.2 | 6.1 | 5.8 | 5.5 | 4.3 | 3.4 | 3.1 | 2.9 |
| 23.4 | 10.1 | 6.7 | 6.3 | 6.0 | 4.6 | 3.7 | 3.1 | 3.0 |
| 23.8 | 5.0 | 4.0 | 4.0 | 4.0 | 3.4 | 2.8 | 2.4 | 2.4 |
| 24.0 | 5.0 | 4.0 | 4.0 | 4.0 | 3.4 | 2.8 | 2.4 | 2.4 |
| BMI/TST Score | 1.24 | 1.26 | 1.28 | 1.3 | 1.32 | 1.34 | 1.36 | 1.38 |
|---|---|---|---|---|---|---|---|---|
| 21.0 | 0.8 | 0.7 | 0.7 | 0.7 | 0.8 | 0.8 | 0.7 | 0.7 |
| 21.6 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.8 | 0.7 | 0.6 |
| 22.2 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.6 |
| 22.4 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.6 | 0.5 |
| 23.0 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.6 | 0.6 | 0.5 |
| 23.4 | 0.7 | 0.6 | 0.6 | 0.6 | 0.7 | 0.6 | 0.6 | 0.5 |
| 23.8 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.6 | 0.6 | 0.5 |
| 24.0 | 0.7 | 0.7 | 0.7 | 0.7 | 0.7 | 0.6 | 0.6 | 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osawa, R.; Ikegami, S.; Horiuchi, H.; Tokida, R.; Kato, H.; Takahashi, J. Osteoporosis Detection by Physical Function Tests in Resident Health Exams: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry. J. Clin. Med. 2021, 10, 1896. https://doi.org/10.3390/jcm10091896
Osawa R, Ikegami S, Horiuchi H, Tokida R, Kato H, Takahashi J. Osteoporosis Detection by Physical Function Tests in Resident Health Exams: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry. Journal of Clinical Medicine. 2021; 10(9):1896. https://doi.org/10.3390/jcm10091896
Chicago/Turabian StyleOsawa, Ryuji, Shota Ikegami, Hiroshi Horiuchi, Ryosuke Tokida, Hiroyuki Kato, and Jun Takahashi. 2021. "Osteoporosis Detection by Physical Function Tests in Resident Health Exams: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry" Journal of Clinical Medicine 10, no. 9: 1896. https://doi.org/10.3390/jcm10091896
APA StyleOsawa, R., Ikegami, S., Horiuchi, H., Tokida, R., Kato, H., & Takahashi, J. (2021). Osteoporosis Detection by Physical Function Tests in Resident Health Exams: A Japanese Cohort Survey Randomly Sampled from a Basic Resident Registry. Journal of Clinical Medicine, 10(9), 1896. https://doi.org/10.3390/jcm10091896






