Patient-Reported Outcomes, Health-Related Quality of Life, and Clinical Outcomes for Urothelial Cancer Patients Receiving Chemo- or Immunotherapy: A Real-Life Experience
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
- Age ≥ 18 years.
- Initiating chemotherapy as a neoadjuvant treatment, or chemo- or immunotherapy as a standard therapy (outside clinical trials) for metastatic disease.
- Able to read Danish.
- No serious cognitive impairment.
2.2. Study Design
2.3. Questionnaires
2.3.1. EORTC
2.3.2. HADS
2.3.3. PRO-CTCAE
2.4. Outcomes
2.5. Statistics
3. Results
3.1. Inclusion
3.2. Clinical Data
3.3. Questionnaire Completion Rate
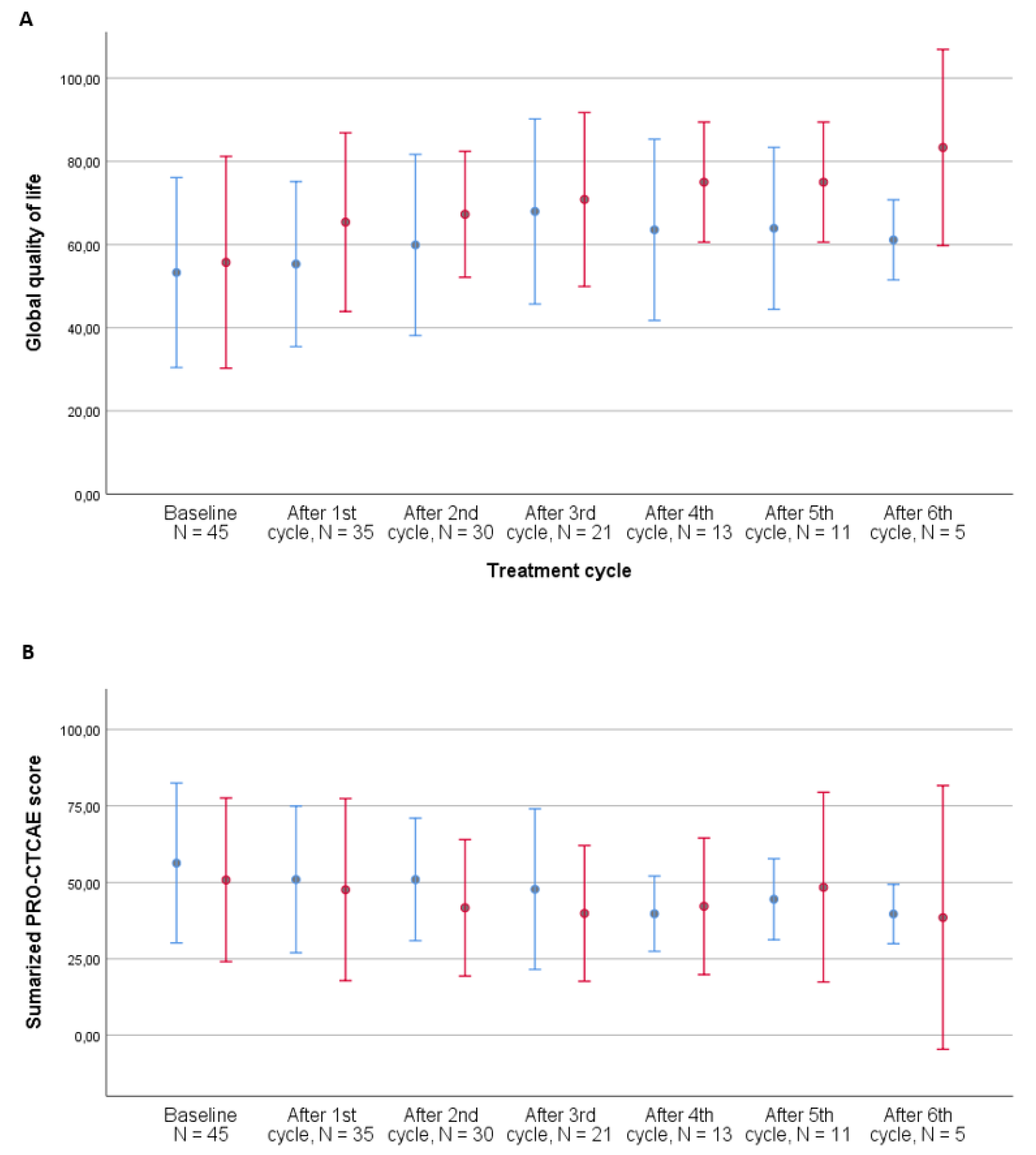
3.4. Health-Related Quality of Life
3.5. PRO-CTCAE Symptoms
3.6. Anxiety and Depression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guancial, E.A.; Roussel, B.; Bergsma, D.P.; Bylund, K.C.; Sahasrabudhe, D.; Messing, E.; Mohile, S.G.; Fung, C. Bladder cancer in the elderly patient: Challenges and solutions. Clin. Interv. Aging 2015, 10, 939–949. [Google Scholar] [CrossRef]
- Scosyrev, E.; Wu, G.; Golijanin, D.; Messing, E. Non-bladder cancer mortality in patients with urothelial cancer of the bladder. Urol. Oncol. 2013, 31, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, T.L.; Teiblum, S.; Paludan, M.; Poulsen, L.O.; Jorgensen, A.Y.; Bruun, K.H.; Hallas, J.; Herrstedt, J. Significance of age and comorbidity on treatment modality, treatment adherence, and prognosis in elderly ovarian cancer patients. Gynecol. Oncol. 2012, 127, 367–374. [Google Scholar] [CrossRef]
- Chagpar, R.; Xing, Y.; Chiang, Y.J.; Feig, B.W.; Chang, G.J.; You, Y.N.; Cormier, J.N. Adherence to stage-specific treatment guidelines for patients with colon cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012, 30, 972–979. [Google Scholar] [CrossRef]
- Sternberg, C.N.; Bellmunt, J.; Sonpavde, G.; Siefker-Radtke, A.O.; Stadler, W.M.; Bajorin, D.F.; Dreicer, R.; George, D.J.; Milowsky, M.I.; Theodorescu, D.; et al. ICUD-EAU International Consultation on Bladder Cancer 2012: Chemotherapy for urothelial carcinoma-neoadjuvant and adjuvant settings. Eur. Urol. 2013, 63, 58–66. [Google Scholar] [CrossRef]
- Meeks, J.J.; Bellmunt, J.; Bochner, B.H.; Clarke, N.W.; Daneshmand, S.; Galsky, M.D.; Hahn, N.M.; Lerner, S.P.; Mason, M.; Powles, T.; et al. A systematic review of neoadjuvant and adjuvant chemotherapy for muscle-invasive bladder cancer. Eur. Urol. 2012, 62, 523–533. [Google Scholar] [CrossRef]
- Vale, C.L. Neoadjuvant chemotherapy in invasive bladder cancer: Update of a systematic review and meta-analysis of individual patient data advanced bladder cancer (ABC) meta-analysis collaboration. Eur. Urol. 2005, 48, 202–205. [Google Scholar] [CrossRef]
- Taarnhoj, G.A.; Johansen, C.; Pappot, H. Quality of life in bladder cancer patients receiving medical oncological treatment; a systematic review of the literature. Health Qual. Life Outcomes 2019, 17, 20. [Google Scholar] [CrossRef]
- Donnelly, C.B.; Wotherspoon, A.C.; Morris, M.; Wilson, R.H.; Chen, J.J.; Cairnduff, V.; Morgan, E.; Devlin, A.; Gavin, A.T. A population-level investigation of cancer clinical trials participation in a UK region. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. (ECP) 2017, 26, S229–S235. [Google Scholar] [CrossRef]
- Unger, J.M.; Cook, E.; Tai, E.; Bleyer, A. The Role of Clinical Trial Participation in Cancer Research: Barriers, Evidence, and Strategies. Am. Soc. Clin. Oncol. Educ. Book Am. Soc. Clin. Oncol. Annu. Meet. 2016, 35, 185–198. [Google Scholar] [CrossRef]
- Zauderer, M.G.; Sima, C.S.; Korc-Grodzicki, B.; Kris, M.G.; Krug, L.M. Toxicity of initial chemotherapy in older patients with lung cancers. J. Geriatr. Oncol. 2013, 4, 64–70. [Google Scholar] [CrossRef][Green Version]
- Basch, E.; Deal, A.M.; Kris, M.G.; Scher, H.I.; Hudis, C.A.; Sabbatini, P.; Rogak, L.; Bennett, A.V.; Dueck, A.C.; Atkinson, T.M.; et al. Symptom Monitoring With Patient-Reported Outcomes During Routine Cancer Treatment: A Randomized Controlled Trial. J. Clin. Oncol. 2016, 34, 557–565. [Google Scholar] [CrossRef]
- Maggiore, R.J.; Dale, W.; Gross, C.P.; Feng, T.; Tew, W.P.; Mohile, S.G.; Owusu, C.; Klepin, H.D.; Lichtman, S.M.; Gajra, A.; et al. Polypharmacy and potentially inappropriate medication use in older adults with cancer undergoing chemotherapy: Effect on chemotherapy-related toxicity and hospitalization during treatment. J. Am. Geriatr. Soc. 2014, 62, 1505–1512. [Google Scholar] [CrossRef]
- Basch, E.; Deal, A.M.; Dueck, A.C.; Scher, H.I.; Kris, M.G.; Hudis, C.; Schrag, D. Overall Survival Results of a Trial Assessing Patient-Reported Outcomes for Symptom Monitoring During Routine Cancer Treatment. JAMA 2017. [Google Scholar] [CrossRef] [PubMed]
- Denis, F.; Lethrosne, C.; Pourel, N.; Molinier, O.; Pointreau, Y.; Domont, J.; Bourgeois, H.; Senellart, H.; Tremolieres, P.; Lizee, T.; et al. Randomized Trial Comparing a Web-Mediated Follow-up With Routine Surveillance in Lung Cancer Patients. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef]
- Lancaster, G.A.; Dodd, S.; Williamson, P.R. Design and analysis of pilot studies: Recommendations for good practice. J. Eval. Clin. Pract. 2004, 10, 307–312. [Google Scholar] [CrossRef]
- Viechtbauer, W.; Smits, L.; Kotz, D.; Bude, L.; Spigt, M.; Serroyen, J.; Crutzen, R. A simple formula for the calculation of sample size in pilot studies. J. Clin. Epidemiol. 2015, 68, 1375–1379. [Google Scholar] [CrossRef]
- Schougaard, L.M.; Larsen, L.P.; Jessen, A.; Sidenius, P.; Dorflinger, L.; de Thurah, A.; Hjollund, N.H. AmbuFlex: Tele-patient-reported outcomes (telePRO) as the basis for follow-up in chronic and malignant diseases. Qual. Life Res. Int. J. Qual. Life Asp. Treat. Care Rehabil. 2016, 25, 525–534. [Google Scholar] [CrossRef]
- Taarnhøj, G.A.; Lindberg, H.; Dohn, L.H.; Omland, L.H.; Hjøllund, N.H.; Johansen, C.; Pappot, H. Electronic reporting of patient-reported outcomes in a fragile and comorbid population during cancer therapy-a feasibility study. Health Qual. Life Outcomes 2020, 18, 225. [Google Scholar] [CrossRef]
- Aaronson, N.K.; Ahmedzai, S.; Bergman, B.; Bullinger, M.; Cull, A.; Duez, N.J.; Filiberti, A.; Flechtner, H.; Fleishman, S.B.; de Haes, J.C.; et al. The European Organization for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. J. Natl. Cancer Inst. 1993, 85, 365–376. [Google Scholar] [CrossRef]
- Gutierrez-Ibarluzea, I.; Simpson, S.; Benguria-Arrate, G. Early awareness and alert systems: An overview of EuroScan methods. Int. J. Technol. Assess. Health Care 2012, 28, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Porter, M.P.; Penson, D.F. Health related quality of life after radical cystectomy and urinary diversion for bladder cancer: A systematic review and critical analysis of the literature. J. Urol. 2005, 173, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Cosco, T.D.; Doyle, F.; Ward, M.; McGee, H. Latent structure of the Hospital Anxiety And Depression Scale: A 10-year systematic review. J. Psychosom. Res. 2012, 72, 180–184. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- Stern, A.F. The hospital anxiety and depression scale. Occup. Med. Lond. 2014, 64, 393–394. [Google Scholar] [CrossRef]
- Basch, E.; Reeve, B.B.; Mitchell, S.A.; Clauser, S.B.; Minasian, L.M.; Dueck, A.C.; Mendoza, T.R.; Hay, J.; Atkinson, T.M.; Abernethy, A.P.; et al. Development of the National Cancer Institute′s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef]
- Baeksted, C.; Nissen, A.; Pappot, H.; Bidstrup, P.E.; Mitchell, S.A.; Basch, E.; Dalton, S.O.; Johansen, C. Danish Translation and Linguistic Validation of the U.S. National Cancer Institute′s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). J. Pain Symptom Manag. 2016, 52, 292–297. [Google Scholar] [CrossRef]
- Taarnhoj, G.A.; Lindberg, H.; Johansen, C.; Pappot, H. Patient-reported outcomes item selection for bladder cancer patients in chemo- or immunotherapy. J. Patient Rep. Outcomes 2019, 3, 56. [Google Scholar] [CrossRef]
- Kaufman, D.S.; Shipley, W.U.; Feldman, A.S. Bladder cancer. Lancet 2009, 374, 239–249. [Google Scholar] [CrossRef]
- Osoba, D.; Rodrigues, G.; Myles, J.; Zee, B.; Pater, J. Interpreting the significance of changes in health-related quality-of-life scores. J. Clin. Oncol. 1998, 16, 139–144. [Google Scholar] [CrossRef]
- Roychowdhury, D.; Hayden, A.; Liepa, A.M. Health-related quality-of-life parameters as independent prognostic factors in advanced or metastatic bladder cancer. J. Clin. Oncol. 2003, 21, 673–678. [Google Scholar] [CrossRef]
- Joly, F.; Houede, N.; Noal, S.; Chevreau, C.; Priou, F.; Chinet-Charrot, P.; Rolland, F.; Flechon, A.; Henry-Amar, M.; Culine, S. Do patients with advanced urothelial carcinoma benefit from weekly paclitaxel chemotherapy? A GETUG phase II study. Clin. Genitourin. Cancer 2009, 7, E28–E33. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, D.J.; Bellmunt, J.; Fradet, Y.; Lee, J.L.; Fong, L.; Vogelzang, N.J.; Climent, M.A.; Petrylak, D.P.; Choueiri, T.K.; Necchi, A.; et al. Health-Related Quality-of-Life Analysis From KEYNOTE-045: A Phase III Study of Pembrolizumab Versus Chemotherapy for Previously Treated Advanced Urothelial Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 1579–1587. [Google Scholar] [CrossRef]
- Quinten, C.; Coens, C.; Ghislain, I.; Zikos, E.; Sprangers, M.A.; Ringash, J.; Martinelli, F.; Ediebah, D.E.; Maringwa, J.; Reeve, B.B.; et al. The effects of age on health-related quality of life in cancer populations: A pooled analysis of randomized controlled trials using the European Organisation for Research and Treatment of Cancer (EORTC) QLQ-C30 involving 6024 cancer patients. Eur. J. Cancer 2015, 51, 2808–2819. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.M.; Smith, D.C.; Montie, J.; Hayman, J.A.; Sullivan, M.A.; Kent, E.; Griffith, K.A.; Esper, P.; Sandler, H.M. Prospective quality-of-life assessment in patients receiving concurrent gemcitabine and radiotherapy as a bladder preservation strategy. Urology 2004, 64, 69–73. [Google Scholar] [CrossRef]
- Juul, T.; Petersen, M.A.; Holzner, B.; Laurberg, S.; Christensen, P.; Gronvold, M. Danish population-based reference data for the EORTC QLQ-C30: Associations with gender, age and morbidity. Qual. Life Res. 2014, 23, 2183–2193. [Google Scholar] [CrossRef]
- Nolte, S.; Liegl, G.; Petersen, M.A.; Aaronson, N.K.; Costantini, A.; Fayers, P.M.; Groenvold, M.; Holzner, B.; Johnson, C.D.; Kemmler, G.; et al. General population normative data for the EORTC QLQ-C30 health-related quality of life questionnaire based on 15,386 persons across 13 European countries, Canada and the Unites States. Eur. J. Cancer 2019, 107, 153–163. [Google Scholar] [CrossRef]
- Grossman, H.B.; Natale, R.B.; Tangen, C.M.; Speights, V.O.; Vogelzang, N.J.; Trump, D.L.; White, R.W.d.; Sarosdy, M.F.; Wood, D.P.; Raghavan, D.; et al. Neoadjuvant Chemotherapy plus Cystectomy Compared with Cystectomy Alone for Locally Advanced Bladder Cancer. N. Engl. J. Med. 2003, 349, 859–866. [Google Scholar] [CrossRef]
- Holmsten, K.; Jensen, N.V.; Mouritsen, L.S.; Jonsson, E.; Mellnert, C.; Agerbaek, M.; Nilsson, C.; Moe, M.; Carus, A.; Ofverholm, E.; et al. Vinflunine/gemcitabine versus carboplatin/gemcitabine as first-line treatment in cisplatin-ineligible patients with advanced urothelial carcinoma: A randomised phase II trial (VINGEM). Eur. J. Cancer 2019. [Google Scholar] [CrossRef]
- Boeri, L.; Soligo, M.; Frank, I.; Boorjian, S.A.; Thompson, R.H.; Tollefson, M.; Tarrel, R.; Quevedo, F.J.; Cheville, J.C.; Karnes, R.J. Clinical predictors and survival outcome of patients receiving suboptimal neoadjuvant chemotherapy and radical cystectomy for muscle-invasive bladder cancer: A single-center experience. World J. Urol. 2019, 37, 2409–2418. [Google Scholar] [CrossRef] [PubMed]
- Niegisch, G.; Lorch, A.; Droller, M.J.; Lavery, H.J.; Stensland, K.D.; Albers, P. Neoadjuvant chemotherapy in patients with muscle-invasive bladder cancer: Which patients benefit? Eur. Urol. 2013, 64, 355–357. [Google Scholar] [CrossRef] [PubMed]
- International Collaboration of Trialists. on behalf of the Medical Research Council Advanced Bladder Cancer Working Party (now the National Cancer Research Institute Bladder Cancer Clinical Studies Group) the European Organisation for Research and Treatment of Cancer Genito-Urinary Tract Cancer Group; The Australian Bladder Cancer Study Group; The National Cancer Institute of Canada Clinical Trials Group; Finnbladder, Norwegian Bladder Cancer Study Group; Club Urologico Espanol de Tratamiento Oncologico Group. International Phase III Trial Assessing Neoadjuvant Cisplatin, Methotrexate, and Vinblastine Chemotherapy for Muscle-Invasive Bladder Cancer: Long-Term Results of the BA06 30894 Trial. J. Clin. Oncol. 2011, 29, 2171–2177. [Google Scholar] [CrossRef] [PubMed]
- Wagland, R.; Richardson, A.; Ewings, S.; Armes, J.; Lennan, E.; Hankins, M.; Griffiths, P. Prevalence of cancer chemotherapy-related problems, their relation to health-related quality of life and associated supportive care: A cross-sectional survey. Supportive Care Cancer Off. J. Multinatl. Assoc. Supportive Care Cancer 2016, 24, 4901–4911. [Google Scholar] [CrossRef]
- Dueck, A.C.; Mendoza, T.R.; Mitchell, S.A.; Reeve, B.B.; Castro, K.M.; Rogak, L.J.; Atkinson, T.M.; Bennett, A.V.; Denicoff, A.M.; O’Mara, A.M.; et al. Validity and Reliability of the US National Cancer Institute′s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). JAMA Oncol. 2015, 1, 1051–1059. [Google Scholar] [CrossRef]
- Lim, G.Y.; Tam, W.W.; Lu, Y.; Ho, C.S.; Zhang, M.W.; Ho, R.C. Prevalence of Depression in the Community from 30 Countries between 1994 and 2014. Sci. Rep. 2018, 8, 2861. [Google Scholar] [CrossRef] [PubMed]
- Krajewski, W.; Koscielska-Kasprzak, K.; Rymaszewska, J.; Zdrojowy, R. How different cystoscopy methods influence patient sexual satisfaction, anxiety, and depression levels: A randomized prospective trial. Qual. Life Res. 2017, 26, 625–634. [Google Scholar] [CrossRef]
- Velikova, G.; Booth, L.; Smith, A.B.; Brown, P.M.; Lynch, P.; Brown, J.M.; Selby, P.J. Measuring quality of life in routine oncology practice improves communication and patient well-being: A randomized controlled trial. J. Clin. Oncol. 2004, 22, 714–724. [Google Scholar] [CrossRef]
- Ruland, C.M.; Andersen, T.; Jeneson, A.; Moore, S.; Grimsbo, G.H.; Borosund, E.; Ellison, M.C. Effects of an internet support system to assist cancer patients in reducing symptom distress: A randomized controlled trial. Cancer Nurs. 2013, 36, 6–17. [Google Scholar] [CrossRef]


| Clinical Data | Total N = 79 (%) | Neoadjuvant Group N = 26 (%) | Metastatic Group N = 53 (%) |
|---|---|---|---|
| Gender | |||
| Men | 64 (81) | 21 (81) | 43 (81) |
| Women | 15 (19) | 5 (19) | 10 (19) |
| Median age, years (range) | 68 (35–82) | 66 (52–77) | 68 (35–82) |
| Disease | |||
| Neoadjuvant | 26 (33) | - | - |
| Metastatic | 53 (67) | - | - |
| Treatment * | |||
| Cisplatin-gemcitabine | 46 (59) | 25 (96) | 21 (40) |
| Carboplatin-gemcitabine | 9 (12) | 1 (4) | 8 (15) |
| Vinflunine | 2 (3) | 0 (0) | 2 (4) |
| Vinflunine-Gemcitabine | 1 (1) | 0 (0) | 1 (2) |
| Pembrolizumab | 20 (26) | 0 (0) | 20 (39) |
| Treatment completion | |||
| No | 43 (54) | 11 (42) | 32 (60) |
| Yes | 36 (46) | 15 (58) | 21 (40) |
| Reason for discontinuation | |||
| Progression | 18 (42) | 3 (27) | 15 (47) |
| Nephrotoxicity | 8 (19) | 4 (36) | 4 (13) |
| Death | 4 (9) | 0 (0) | 1 (3) |
| Hematological toxicity | 2 (5) | 0 (0) | 2 (6) |
| Declining performance status | 2 (5) | 1 (9) | 1 (3) |
| Infection | 2 (5) | 0 (0) | 2 (6) |
| Dyspnea | 1 (2) | 1 (9) | 0 (0) |
| Constipation | 1 (2) | 1 (9) | 0 (0) |
| Colitis | 1 (2) | 0 (0) | 1 (3) |
| Neuropathy | 1 (2) | 0 (0) | 1 (3) |
| Otologic toxicity | 1 (2) | 1 (9) | 0 (0) |
| Intolerable decrease in QoL | 1 (2) | 0 (0) | 1 (3) |
| Other | 1 (2) | 0 (0) | 1 (3) |
| Admission to hospital | |||
| No | 32 (41) | 15 (58) | 17 (32) |
| Yes | 47 (59) | 11 (42) | 36 (68) |
| Reason for hospital admission | |||
| Hematological toxicity | 12 (26) | 3 (27) | 9 (25) |
| Infection | 11 (23) | 3 (27) | 8 (22) |
| Thrombo-embolic events | 6 (13) | 1 (9) | 5 (14) |
| Pain | 4 (9) | 0 (0) | 4 (11) |
| Nephrological toxicity | 3 (6) | 1 (9) | 2 (6) |
| Dyspnea | 2 (4) | 0 (0) | 2 (6) |
| Nausea/vomiting | 2 (4) | 1 (9) | 1 (3) |
| Constipation | 1 (2) | 1 (9) | 0 (0) |
| Pneumonitis | 1 (2) | 0 (0) | 1 (3) |
| Nephritis | 1 (2) | 0 (0) | 1 (3) |
| Dehydration | 1 (2) | 1 (9) | 0 (0) |
| Declining performance status | 1 (2) | 0 (0) | 1 (3) |
| Ileus | 1 (2) | 0 (0) | 1 (3) |
| Other | 1 (2) | 0 (0) | 1 (3) |
| Neoadjuvant Group | ||||||||
| Baseline | After 4 Cycles | |||||||
| Mean | Min | Max | SD * | Mean | Min | Max | SD * | |
| Global Quality of Life | 73 | 25 | 100 | 19 | 72 | 25 | 100 | 23 |
| Pain ** | 19 | 0 | 100 | 23 | 7 | 0 | 33 | 11 |
| Fatigue ** | 25 | 0 | 67 | 19 | 31 | 0 | 78 | 25 |
| Urinary symptoms ** | 38 | 0 | 76 | 23 | 17 | 0 | 38 | 14 |
| Metastatic Group | ||||||||
| Baseline | After 3 cycles *** | |||||||
| Mean | Min | Max | SD * | Mean | Min | Max | SD * | |
| Global Quality of Life | 54 | 0 | 100 | 24 | 69 | 33 | 100 | 21 |
| Pain ** | 38 | 0 | 100 | 37 | 15 | 0 | 50 | 18 |
| Fatigue ** | 48 | 0 | 100 | 26 | 38 | 0 | 89 | 28 |
| Urinary symptoms ** | 25 | 0 | 76 | 21 | 18 | 5 | 43 | 12 |
| Symptom | Total | Baseline | After 1st Cycle | After 2nd Cycle | After 3rd Cycle | After 4th Cycle | After 5th Cycle | After 6th Cycle |
|---|---|---|---|---|---|---|---|---|
| % | % | % | % | % | % | % | % | |
| Reports, N | 449 | 71 | 59 | 50 | 38 | 26 | 17 | 8 |
| Frequent urination | 37 | 45 | 35 | 38 | 35 | 36 | 31 | 13 |
| Fatigue | 35 | 34 | 37 | 38 | 26 | 27 | 59 | 28 |
| General pain | 31 | 45 | 42 | 27 | 26 | 15 | 35 | 13 |
| Dry mouth | 23 | 23 | 28 | 20 | 26 | 23 | 18 | 13 |
| Swelling of arms or legs | 23 | 21 | 18 | 25 | 24 | 31 | 41 | 0 |
| Abdominal pain | 21 | 27 | 19 | 22 | 21 | 19 | 18 | 0 |
| Decreased appetite | 21 | 23 | 19 | 20 | 19 | 24 | 24 | 0 |
| Insomnia | 20 | 34 | 19 | 17 | 13 | 19 | 6 | 0 |
| Shortness of breath | 20 | 13 | 24 | 29 | 18 | 19 | 18 | 0 |
| Nausea | 19 | 14 | 16 | 22 | 24 | 16 | 24 | 25 |
| Urinary urgency | 18 | 40 | 27 | 22 | 19 | 20 | 13 | 13 |
| Muscle pain | 18 | 19 | 21 | 16 | 16 | 12 | 18 | 13 |
| Constipation | 15 | 14 | 21 | 12 | 16 | 15 | 12 | 0 |
| Sad | 15 | 21 | 16 | 16 | 11 | 8 | 6 | 13 |
| Decreased libido | 14 | 27 | 8 | 9 | 8 | 16 | 6 | 13 |
| Joint pain | 13 | 15 | 14 | 12 | 8 | 12 | 18 | 25 |
| Taste changes | 13 | 8 | 10 | 16 | 16 | 19 | 18 | 13 |
| Heart palpitations | 12 | 14 | 12 | 16 | 8 | 4 | 6 | 13 |
| Dry skin | 12 | 13 | 10 | 10 | 13 | 12 | 12 | 13 |
| Numbness & tingling | 11 | 13 | 3 | 8 | 11 | 12 | 30 | 25 |
| Painful urination | 11 | 22 | 7 | 8 | 11 | 8 | 0 | 0 |
| Diarrhea | 9 | 14 | 7 | 8 | 5 | 12 | 0 | 13 |
| Urinary incontinence | 9 | 16 | 9 | 7 | 5 | 8 | 6 | 0 |
| Anxiety | 9 | 19 | 9 | 2 | 5 | 8 | 0 | 0 |
| Discouraged | 9 | 8 | 9 | 13 | 8 | 4 | 12 | 0 |
| Headache | 8 | 11 | 8 | 6 | 3 | 8 | 12 | 13 |
| Ringing in ears | 8 | 3 | 7 | 6 | 11 | 15 | 12 | 13 |
| Cough | 7 | 5 | 11 | 6 | 5 | 12 | 6 | 0 |
| Dizziness | 7 | 5 | 5 | 8 | 11 | 4 | 6 | 13 |
| Increased sweating | 6 | 13 | 5 | 4 | 8 | 0 | 0 | 0 |
| Heartburn | 6 | 6 | 12 | 6 | 3 | 4 | 0 | 0 |
| Hair loss | 6 | 0 | 9 | 6 | 8 | 4 | 12 | 13 |
| Itching | 6 | 8 | 5 | 4 | 3 | 8 | 6 | 0 |
| Chills | 6 | 6 | 9 | 2 | 3 | 12 | 0 | 0 |
| Difficulty swallowing | 5 | 6 | 5 | 2 | 8 | 0 | 12 | 0 |
| Hot flashes/flushes | 4 | 10 | 5 | 0 | 3 | 0 | 0 | 0 |
| Concentration | 3 | 6 | 0 | 4 | 3 | 4 | 0 | 0 |
| Mouth/throat sores | 3 | 3 | 2 | 4 | 3 | 0 | 6 | 0 |
| Memory | 2 | 3 | 0 | 4 | 3 | 4 | 0 | 0 |
| Blurred vision | 2 | 2 | 2 | 0 | 3 | 4 | 0 | 0 |
| Vomiting | 2 | 2 | 0 | 2 | 3 | 0 | 0 | 13 |
| Rash§ (yes/no) | 65 | 65 | 64 | 63 | 68 | 62 | 77 | 63 |
| Change in usual urine color § (yes/no) | 24 | 27 | 19 | 22 | 26 | 27 | 24 | 38 |
| Pain/swelling at injection site § (yes/no) | 24 | 11 | 23 | 20 | 40 | 39 | 35 | 13 |
| Hives § (yes/no) | 8 | 5 | 7 | 12 | 11 | 12 | 0 | 13 |
| Confidence Interval | p-Value | |||
|---|---|---|---|---|
| Estimate | Lower | Upper | ||
| Global quality of life | ||||
| Chemotherapy | 1.07 | −0.07 | 2.21 | 0.066 |
| Immunotherapy | 1.62 | 0.26 | 2.97 | 0.02 |
| Summarized PRO-CTCAE score | ||||
| Chemotherapy | −0.82 | −0.23 | 0.69 | 0.286 |
| Immunotherapy | −1.89 | −3.42 | −0.16 | 0.032 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taarnhøj, G.A.; Lindberg, H.; Johansen, C.; Pappot, H. Patient-Reported Outcomes, Health-Related Quality of Life, and Clinical Outcomes for Urothelial Cancer Patients Receiving Chemo- or Immunotherapy: A Real-Life Experience. J. Clin. Med. 2021, 10, 1852. https://doi.org/10.3390/jcm10091852
Taarnhøj GA, Lindberg H, Johansen C, Pappot H. Patient-Reported Outcomes, Health-Related Quality of Life, and Clinical Outcomes for Urothelial Cancer Patients Receiving Chemo- or Immunotherapy: A Real-Life Experience. Journal of Clinical Medicine. 2021; 10(9):1852. https://doi.org/10.3390/jcm10091852
Chicago/Turabian StyleTaarnhøj, Gry Assam, Henriette Lindberg, Christoffer Johansen, and Helle Pappot. 2021. "Patient-Reported Outcomes, Health-Related Quality of Life, and Clinical Outcomes for Urothelial Cancer Patients Receiving Chemo- or Immunotherapy: A Real-Life Experience" Journal of Clinical Medicine 10, no. 9: 1852. https://doi.org/10.3390/jcm10091852
APA StyleTaarnhøj, G. A., Lindberg, H., Johansen, C., & Pappot, H. (2021). Patient-Reported Outcomes, Health-Related Quality of Life, and Clinical Outcomes for Urothelial Cancer Patients Receiving Chemo- or Immunotherapy: A Real-Life Experience. Journal of Clinical Medicine, 10(9), 1852. https://doi.org/10.3390/jcm10091852






