Early Prognostic Indicators of Subsequent Hospitalization in Patients with Mild COVID-19
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Statistical Analyses
3. Results
3.1. Demographic, Clinical and Laboratory Characteristics and Outcome
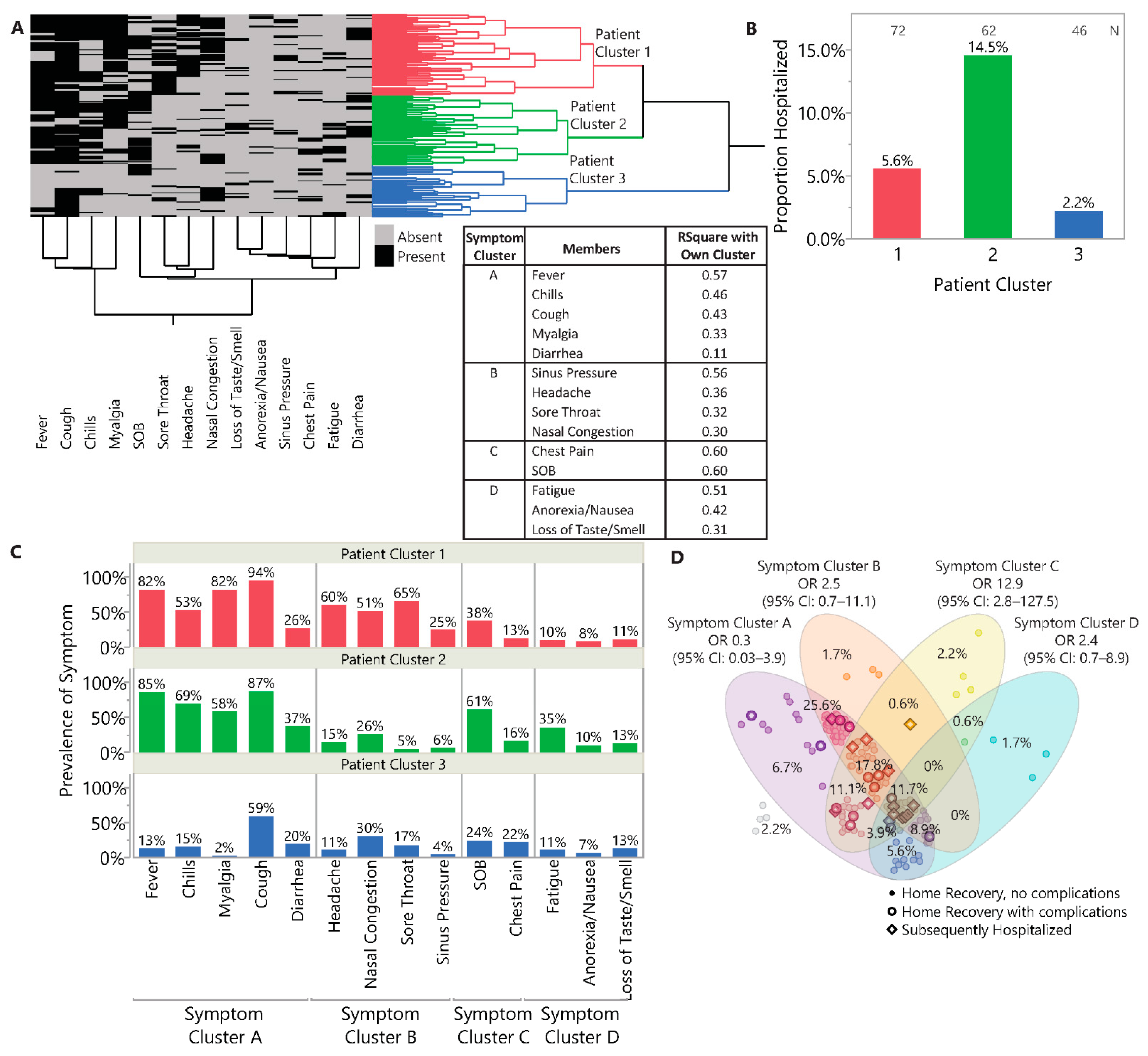
3.2. Symptomatology and Symptom Clusters
3.3. Comorbidities and Risk Factors
3.4. Duration of Illness and Viral Load: Association with Symptoms
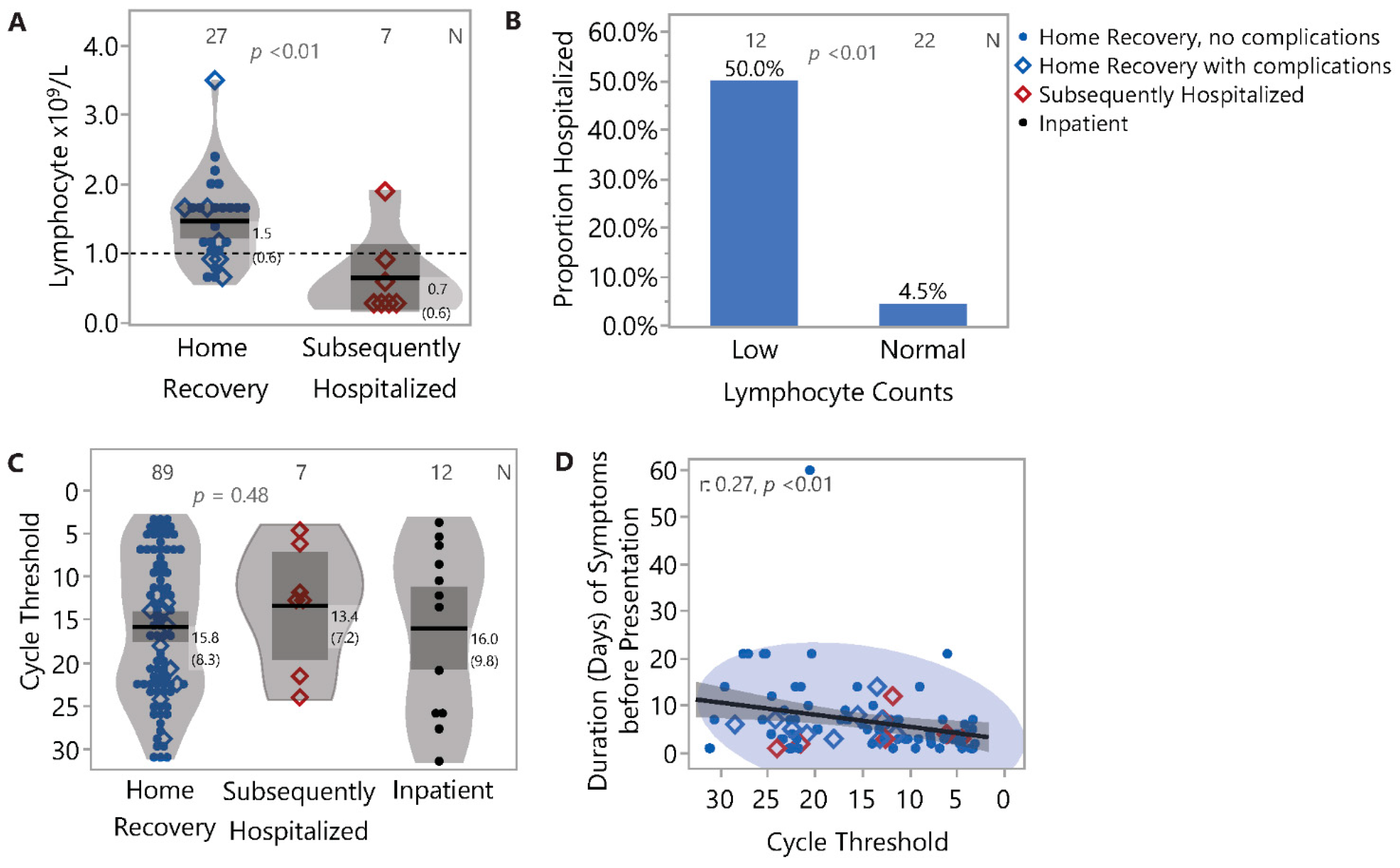
3.5. Outcome Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petrilli, C.M.; Jones, S.A.; Yang, J.; Rajagopalan, H.; O’Donnell, L.; Chernyak, Y.; Tobin, K.A.; Cerfolio, R.J.; Francois, F.; Horwitz, L.I. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: Prospective cohort study. BMJ 2020, 369, m1966. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; Cohen, S.L.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Suleyman, G.; Fadel, R.A.; Malette, K.M.; Hammond, C.; Abdulla, H.; Entz, A.; Demertzis, Z.; Hanna, Z.; Failla, A.; Dagher, C.; et al. Clinical Characteristics and Morbidity Associated with Coronavirus Disease 2019 in a Series of Patients in Metropolitan Detroit. JAMA Netw. Open 2020, 3, e2012270. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Hong, C.M.; Kim, D.H.; Lee, T.H.; Lee, J. Clinical Course of Asymptomatic and Mildly Symptomatic Patients with Coronavirus Disease Admitted to Community Treatment Centers, South Korea. Emerg. Infect. Dis. 2020, 26, 2346–2352. [Google Scholar] [CrossRef]
- Struyf, T.; Deeks, J.J.; Dinnes, J.; Takwoingi, Y.; Davenport, C.; Leeflang, M.M.; Spijker, R.; Hooft, L.; Emperador, D.; Dittrich, S.; et al. Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19 disease. Cochrane Database Syst. Rev. 2020, 2020, CD013665. [Google Scholar] [CrossRef]
- Lapostolle, F.; Schneider, E.; Vianu, I.; Dollet, G.; Roche, B.; Berdah, J.; Michel, J.; Goix, L.; Chanzy, E.; Petrovic, T.; et al. Clinical features of 1487 COVID-19 patients with outpatient management in the Greater Paris: The COVID-call study. Intern. Emerg. Med. 2020, 15, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Attanasi, M.; Pasini, S.; Caronni, A.; Pellegrino, G.M.; Faverio, P.; Di Pillo, S.; Cimino, M.M.; Cipolla, G.; Chiarelli, F.; Centanni, S.; et al. Inpatient Care during the COVID-19 Pandemic: A Survey of Italian Physicians. Respiration 2020, 99, 667–677. [Google Scholar] [CrossRef] [PubMed]
- Jehi, L.; Ji, X.; Milinovich, A.; Erzurum, S.; Merlino, A.; Gordon, S.; Young, J.B.; Kattan, M.W. Development and validation of a model for individualized prediction of hospitalization risk in 4,536 patients with COVID-19. PLoS ONE 2020, 15, e0237419. [Google Scholar] [CrossRef] [PubMed]
- Novelli, L.; Raimondi, F.; Ghirardi, A.; Pellegrini, D.; Capodanno, D.; Sotgiu, G.; Guagliumi, G.; Senni, M.; Russo, F.M.; Lorini, F.L.; et al. At the peak of Covid-19 age and disease severity but not comorbidities are predictors of mortality. Covid-19 burden in Bergamo, Italy. Panminerva Med. 2020, 63, 51–61. [Google Scholar]
- Liu, J.; Li, H.; Luo, M.; Liu, J.; Wu, L.; Lin, X.; Li, R.; Wang, Z.; Zhong, H.; Zheng, W.; et al. Lymphopenia predicted illness severity and recovery in patients with COVID-19: A single-center, retrospective study. PLoS ONE 2020, 15, e0241659. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Meng, M.; Kumar, R.; Wu, Y.; Huang, J.; Deng, Y.; Weng, Z.; Yang, L. Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A systemic review and meta-analysis. Int. J. Infect. Dis. 2020, 96, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Bolouri, H.; Speake, C.; Skibinski, D.; Long, S.A.; Hocking, A.M.; Campbell, D.J.; Hamerman, J.A.; Malhotra, U.; Buckner, J.H. The COVID-19 immune landscape is dynamically and reversibly correlated with disease severity. J. Clin. Investig. 2021, 131. [Google Scholar] [CrossRef] [PubMed]
- Kelleni, M.T. SARS CoV-2 viral load might not be the right predictor of COVID-19 mortality. J. Infect. 2021, 82, e35. [Google Scholar] [CrossRef] [PubMed]
- Fajnzylber, J.; Regan, J.; Coxen, K.; Corry, H.; Wong, C.; Rosenthal, A.; Worrall, D.; Giguel, F.; Piechocka Trocha, A.; Atyeo, C.; et al. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat. Commun. 2020, 11, 5493. [Google Scholar] [CrossRef]
- Westblade, L.F.; Brar, G.; Pinheiro, L.C.; Paidoussis, D.; Rajan, M.; Martin, P.; Goyal, P.; Sepulveda, J.L.; Zhang, L.; George, G.; et al. SARS-CoV-2 Viral Load Predicts Mortality in Patients with and without Cancer Who Are Hospitalized with COVID-19. Cancer Cell 2020, 38, 661–671. [Google Scholar] [CrossRef]
- Aggarwal, S.; Aggarwal, S.; Aggarwal, A.; Jain, K.; Minhas, S. High Viral Load and Poor Ventilation: Cause of High Mortality From COVID-19. Asia Pac. J. Public Health 2020, 32, 377–378. [Google Scholar] [CrossRef]
- Lee, S.; Lee, E.; Lee, C.; Kim, H.; Rhee, H.; Park, S.Y.; Son, H.-J.; Yu, S.; Park, J.W.; Choo, E.J.; et al. Clinical Course and MolecularViral Shedding Among Asymptomatic and Symptomatic Patients With SARS-CoV-2 Infection in a Community Treatment Centerin the Republic of Korea. JAMA Intern. Med. 2020, 180, 1447–1452. [Google Scholar] [CrossRef]
- Tobin, M.J.; Laghi, F.; Jubran, A. Why COVID-19 Silent Hypoxemia Is Baffling to Physicians. Am. J. Respir. Crit. Care Med. 2020, 202, 356–360. [Google Scholar] [CrossRef]
- Herrmann, J.; Mori, V.; Bates, J.H.T.; Suki, B. Modeling lung perfusion abnormalities to explain early COVID-19 hypoxemia. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Burke, R.M.; Killerby, M.E.; Newton, S.; Ashworth, C.E.; Berns, A.L.; Brennan, S.; Bressler, J.M.; Bye, E.; Crawford, R.; Morano, L.H.; et al. Symptom Profiles of a Convenience Sample of Patients with COVID-19—United States, January–April 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Trimaille, A.; Thachil, J.; Marchandot, B.; Curtiaud, A.; Leonard-Lorant, I.; Carmona, A.; Matsushita, K.; Sato, C.; Sattler, L.; Grunebaum, L.; et al. D-Dimers Level as a Possible Marker of Extravascular Fibrinolysis in COVID-19 Patients. J. Clin. Med. 2020, 10, 39. [Google Scholar] [CrossRef] [PubMed]
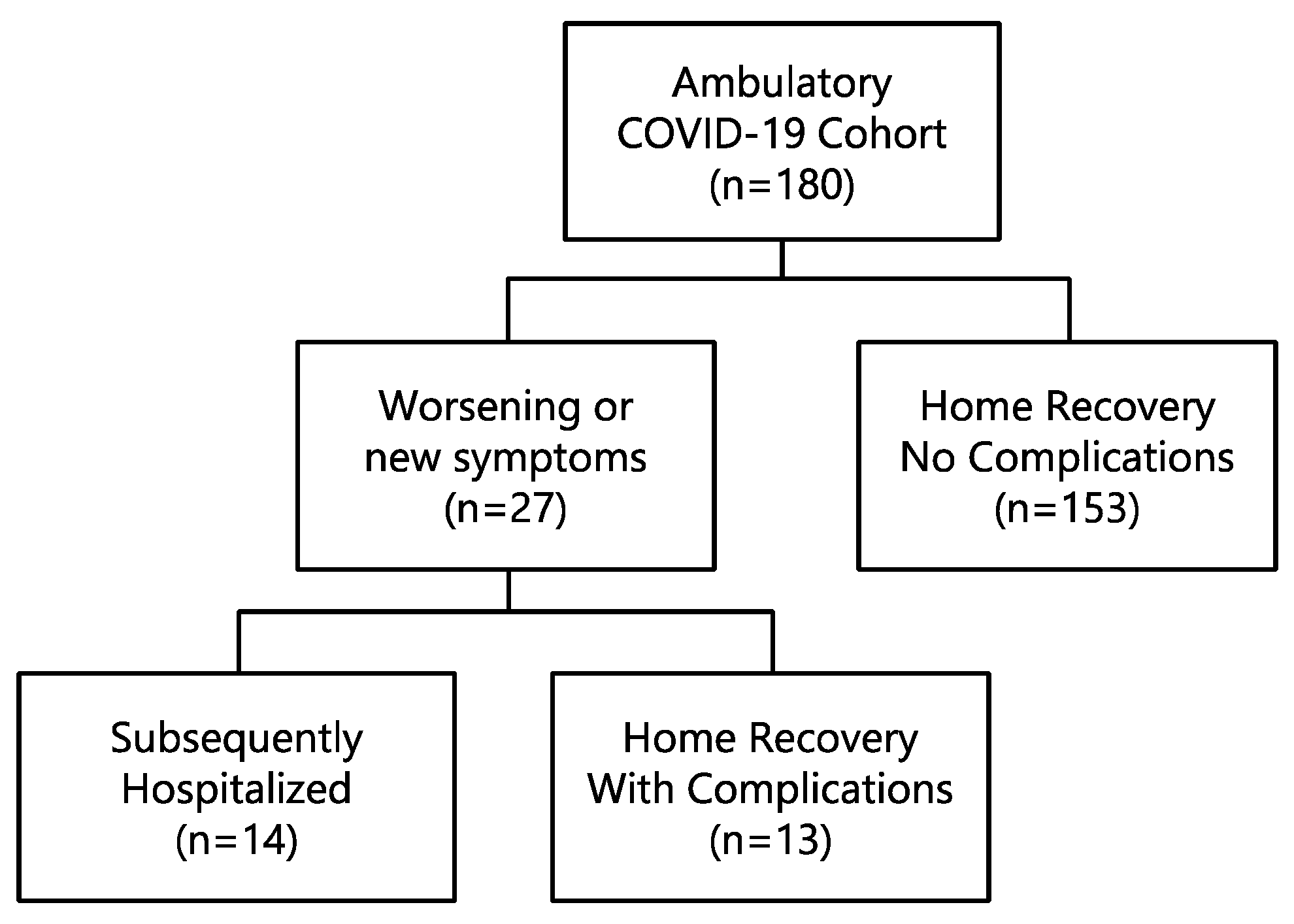


| Characteristics | Home Recovery No Complications (N = 153) | Home Recovery with Complications (N = 13) | Subsequently Hospitalized (N = 14) | p Value |
|---|---|---|---|---|
| Age | 0.05 1 | |||
| N | 153 | 13 | 14 | |
| Median | 52.6 | 56.5 | 56 | |
| Range | (18.2–81.8) | (43.5–76.6) | (41.0–83.5) | |
| Female, N (%) | 77 (50.3%) | 4 (30.8%) | 3 (21.4%) | 0.06 2 |
| Duration from Symptom Onset (days) | 0.54 1 | |||
| N | 147 | 13 | 14 | |
| Median | 5 | 4 | 4 | |
| Range | (1.0–60.0) | (1.0–14.0) | (1.0–12.0) | |
| Comorbidity, N (%) | 61 (39.9%) | 10 (76.9%) | 12 (85.7%) | 0.0003 2 |
| Race, N (%) | 0.62 2 | |||
| Am. Indian, AK Nat., Nat. Hawaiian | 5 (3.3%) | 0 (0.0%) | 0 (0.0%) | |
| Asian | 20 (13.1%) | 3 (23.1%) | 2 (14.3%) | |
| Black, African American | 9 (5.9%) | 0 (0.0%) | 0 (0.0%) | |
| Hispanic | 19 (12.4%) | 1 (7.7%) | 3 (21.4%) | |
| Other | 25 (16.3%) | 1 (7.7%) | 0 (0.0%) | |
| White | 75 (49.0%) | 8 (61.5%) | 9 (64.3%) | |
| BMI Category, N (%) | 0.63 3 | |||
| Obese | 48 (39.0%) | 6 (50.0%) | 7 (50.0%) | |
| Overweight | 40 (32.5%) | 5 (41.7%) | 5 (35.7%) | |
| Normal | 34 (27.6%) | 1 (8.3%) | 2 (14.3%) | |
| Underweight | 1 (0.8%) | 0 (0.0%) | 0 (0.0%) | |
| Lymphocyte × 109/L | 0.01 1 | |||
| N | 20 | 7 | 7 | |
| Median | 1.6 | 1.1 | 0.3 | |
| Range | (0.6–2.4) | (0.7–3.5) | (0.3–1.9) | |
| Neutrophil × 109/L | 0.87 1 | |||
| N | 20 | 7 | 7 | |
| Median | 3.5 | 3.9 | 6.2 | |
| Range | (1.5–8.9) | (1.8–5.1) | (0.8–11.5) | |
| Cycle Threshold | 0.46 1 | |||
| N | 79 | 10 | 7 | |
| Median | 15.1 | 16.8 | 12.6 | |
| Range | (3.2–31.2) | (11.5–28.5) | (4.6–24.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ylescupidez, A.; Rips, A.; Bahnson, H.T.; Speake, C.; Verma, P.; Hocking, A.M.; Buckner, J.H.; Malhotra, U. Early Prognostic Indicators of Subsequent Hospitalization in Patients with Mild COVID-19. J. Clin. Med. 2021, 10, 1562. https://doi.org/10.3390/jcm10081562
Ylescupidez A, Rips A, Bahnson HT, Speake C, Verma P, Hocking AM, Buckner JH, Malhotra U. Early Prognostic Indicators of Subsequent Hospitalization in Patients with Mild COVID-19. Journal of Clinical Medicine. 2021; 10(8):1562. https://doi.org/10.3390/jcm10081562
Chicago/Turabian StyleYlescupidez, Alyssa, Aaron Rips, Henry T. Bahnson, Cate Speake, Punam Verma, Anne M. Hocking, Jane H. Buckner, and Uma Malhotra. 2021. "Early Prognostic Indicators of Subsequent Hospitalization in Patients with Mild COVID-19" Journal of Clinical Medicine 10, no. 8: 1562. https://doi.org/10.3390/jcm10081562
APA StyleYlescupidez, A., Rips, A., Bahnson, H. T., Speake, C., Verma, P., Hocking, A. M., Buckner, J. H., & Malhotra, U. (2021). Early Prognostic Indicators of Subsequent Hospitalization in Patients with Mild COVID-19. Journal of Clinical Medicine, 10(8), 1562. https://doi.org/10.3390/jcm10081562






