Vaginal Probiotics for Reproductive Health and Related Dysbiosis: Systematic Review and Meta-Analysis
Abstract
1. Introduction
1.1. Microbiota Colonization Sites in Women’s Reproductive System
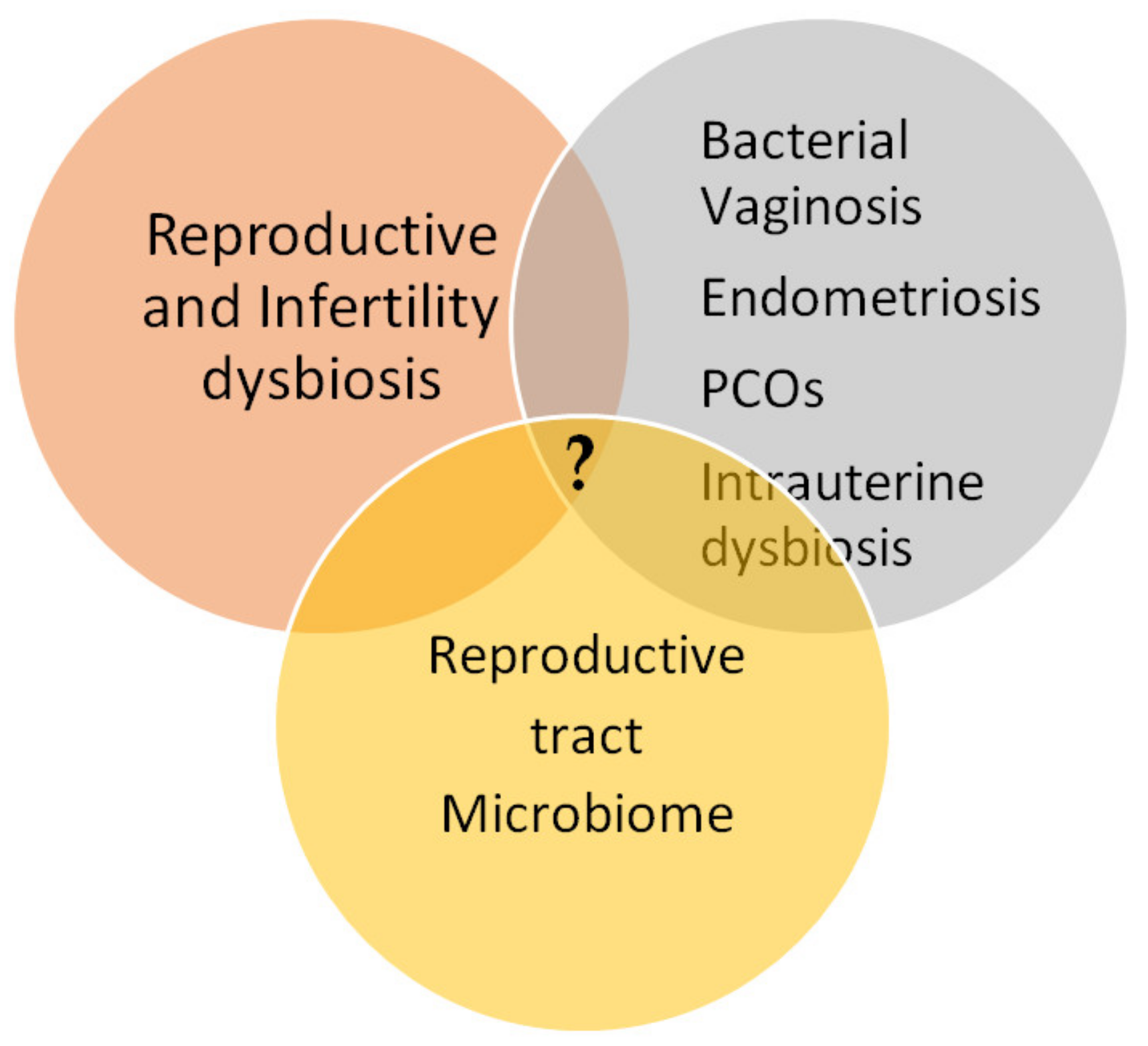
1.2. Microbial Dysbiosis Associated with Reproductive System Diseases
1.3. Probiotics for Reproductive Health Interventions
1.4. Administration Routes of Probiotics in Reproductive Dysbiosis
2. Materials and Methods
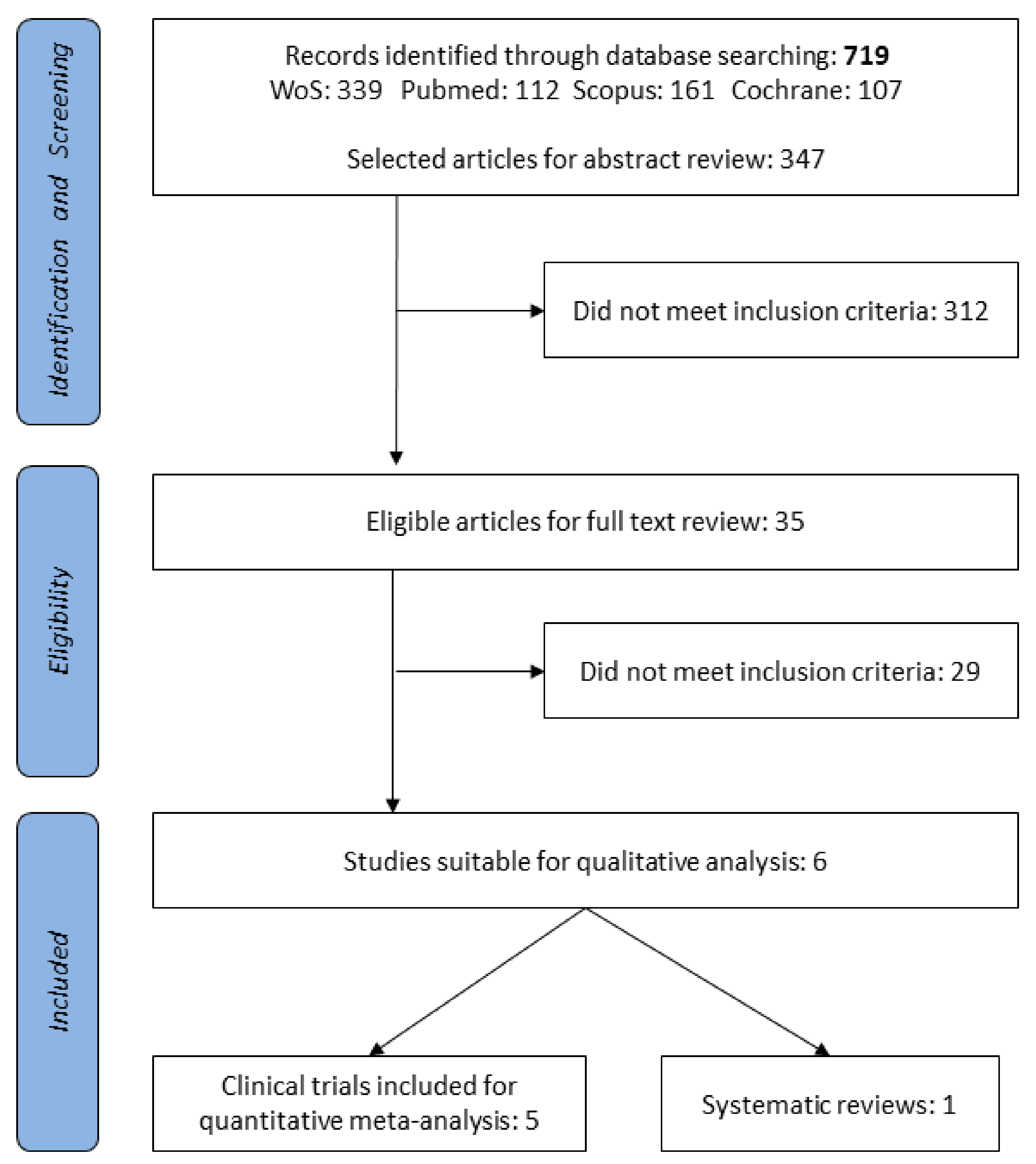
2.1. Eligibility Criteria and Search Strategy
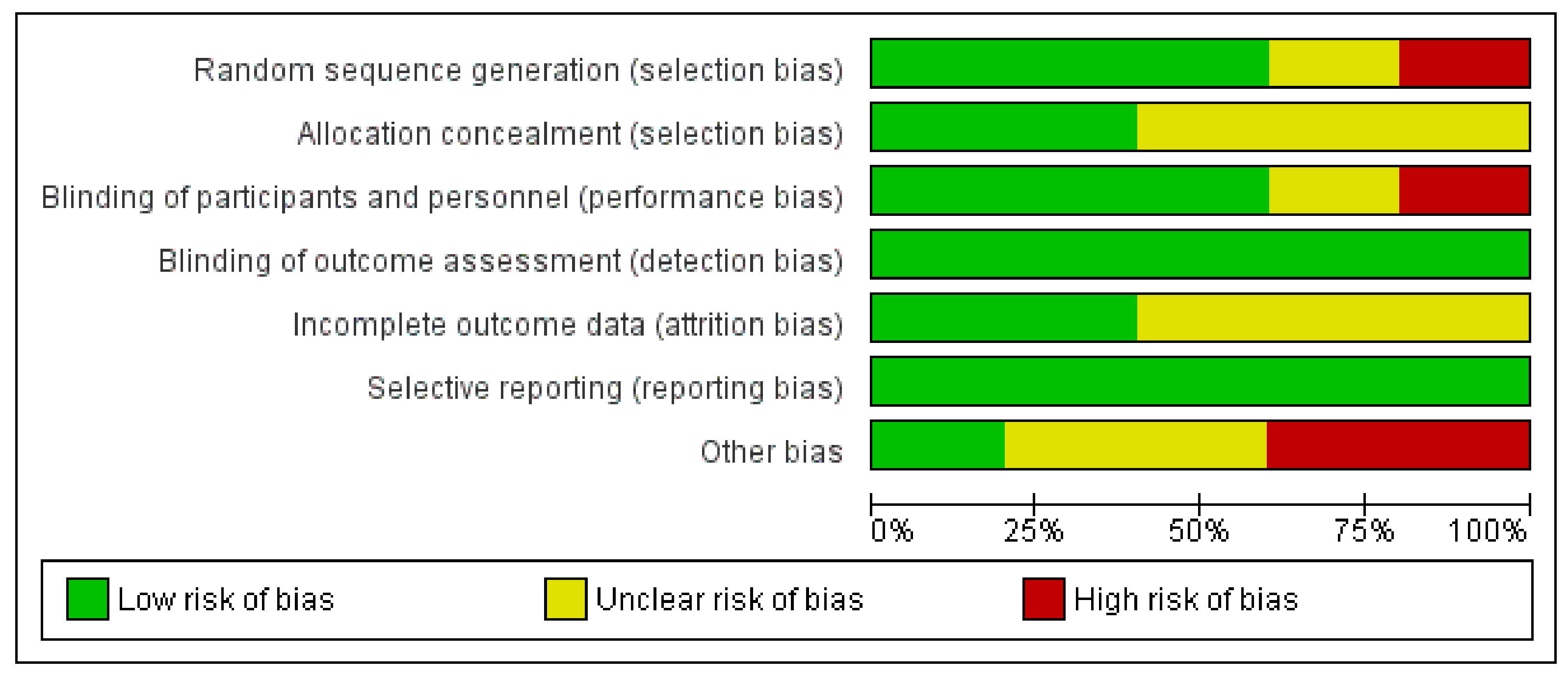
2.2. Data Extraction, Analysis, and Risk of Bias (Quality) Assessment
2.3. Statistical Analysis
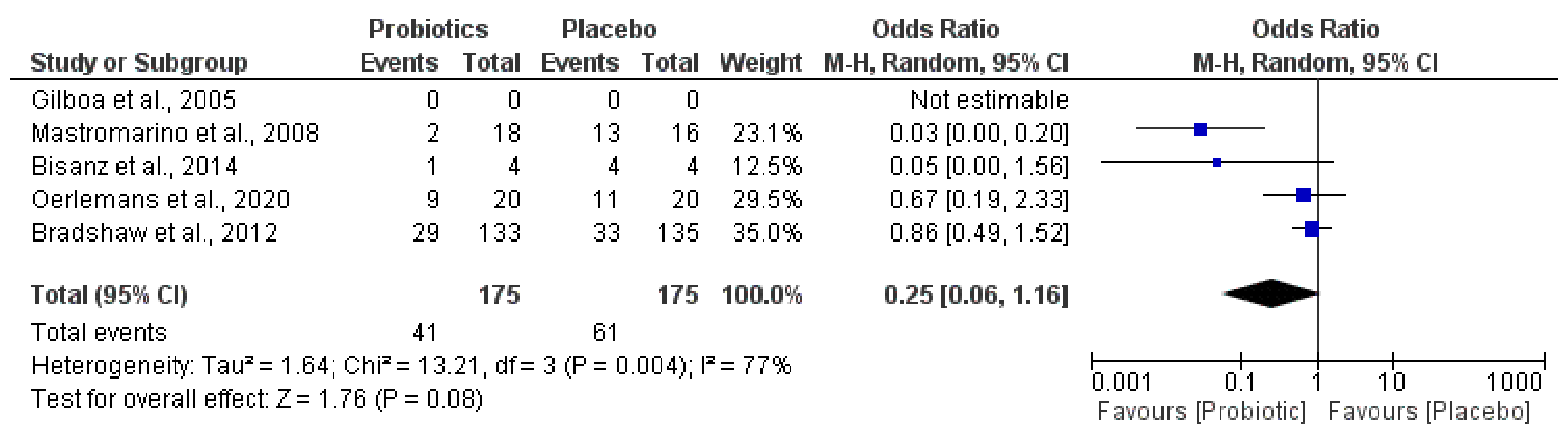
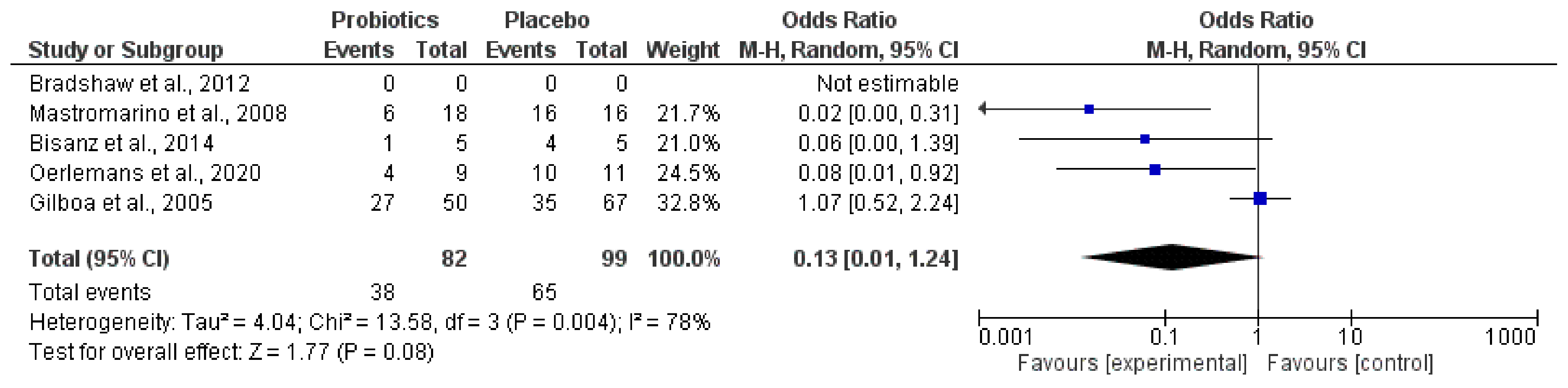
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- García-Velasco, J.A.; Menabrito, M.; Catalán, I.B. What Fertility Specialists Should Know about the Vaginal Microbiome: A Review. Reprod. Biomed. Online 2017, 35, 103–112. [Google Scholar] [CrossRef]
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen-Gut Microbiome Axis: Physiological and Clinical Implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef]
- Vazquez, F.; Fernández-Blázquez, A.; García, B. Vaginosis. Vaginal Microbiota. Enf. Infec. Microbiol. Clin. 2019, 37, 592–601. [Google Scholar] [CrossRef]
- Torres, P.J.; Siakowska, M.; Banaszewska, B.; Pawelczyk, L.; Duleba, A.J.; Kelley, S.T.; Thackray, V.G. Gut Microbial Diversity in Women With Polycystic Ovary Syndrome Correlates With Hyperandrogenism. J. Clin. Endocrinol. Metab. 2018, 103, 1502–1511. [Google Scholar] [CrossRef]
- Fontané, L.; Benaiges, D.; Goday, A.; Llauradó, G.; Pedro-Botet, J. Influence of the Microbiota and Probiotics in Obesity. Clin. Investig. Arterioscler. 2018, 30, 271–279. [Google Scholar] [CrossRef]
- Rondanelli, M.; Faliva, M.A.; Perna, S.; Giacosa, A.; Peroni, G.; Castellazzi, A.M. Using Probiotics in Clinical Practice: Where Are We Now? A Review of Existing Meta-Analyses. Gut Microbes 2017, 8, 521–543. [Google Scholar] [CrossRef]
- Aguilera, M.; Gálvez-Ontiveros, Y.; Rivas, A. Endobolome, a New Concept for Determining the Influence of Microbiota Disrupting Chemicals (MDC) in Relation to Specific Endocrine Pathogenesis. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Ata, B.; Yildiz, S.; Turkgeldi, E.; Brocal, V.P.; Dinleyici, E.C.; Moya, A.; Urman, B. The Endobiota Study: Comparison of Vaginal, Cervical and Gut Microbiota Between Women with Stage 3/4 Endometriosis and Healthy Controls. Sci. Rep. 2019, 9, 2204. [Google Scholar] [CrossRef] [PubMed]
- Pramanick, R.; Mayadeo, N.; Warke, H.; Begum, S.; Aich, P.; Aranha, C. Vaginal Microbiota of Asymptomatic Bacterial Vaginosis and Vulvovaginal Candidiasis: Are They Different from Normal Microbiota? Microb. Pathog. 2019, 134, 103599. [Google Scholar] [CrossRef]
- Garcia-Grau, I.; Perez-Villaroya, D.; Bau, D.; Gonzalez-Monfort, M.; Vilella, F.; Moreno, I.; Simon, C. Taxonomical and Functional Assessment of the Endometrial Microbiota in A Context of Recurrent Reproductive Failure: A Case Report. Pathogens 2019, 8, 205. [Google Scholar] [CrossRef]
- Moreno, I.; Garcia-Grau, I.; Bau, D.; Perez-Villaroya, D.; Gonzalez-Monfort, M.; Vilella, F.; Romero, R.; Simón, C. The First Glimpse of the Endometrial Microbiota in Early Pregnancy. Am. J. Obstet. Gynecol. 2020, 222, 296–305. [Google Scholar] [CrossRef]
- Perez-Muñoz, M.E.; Arrieta, M.-C.; Ramer-Tait, A.E.; Walter, J. A Critical Assessment of the “Sterile Womb” and “in Utero Colonization” Hypotheses: Implications for Research on the Pioneer Infant Microbiome. Microbiome 2017, 5, 48. [Google Scholar] [CrossRef] [PubMed]
- De Goffau, M.C.; Lager, S.; Sovio, U.; Gaccioli, F.; Cook, E.; Peacock, S.J.; Parkhill, J.; Charnock-Jones, D.S.; Smith, G.C.S. Human Placenta Has No Microbiome but Can Contain Potential Pathogens. Nature 2019, 572, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Sood, R.; Zehnder, J.L.; Druzin, M.L.; Brown, P.O. Gene Expression Patterns in Human Placenta. Proc. Natl. Acad. Sci. USA 2006, 103, 5478–5483. [Google Scholar] [CrossRef]
- Bohbot, J.M.; Cardot, J.M. Vaginal Impact of the Oral Administration of Total Freeze-Dried Culture of LCR 35 in Healthy Women. Infect. Dis. Obstet. Gynecol. 2012, 2012, 503648. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, P.; Prabha, V. Evaluation of Profertility Effect of Probiotic Lactobacillus plantarum 2621 in a Murine Model. Indian J. Med. Res. 2015, 142, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Salah, R.M.; Allam, A.M.; Magdy, A.M.; Mohamed, A.S. Bacterial Vaginosis and Infertility: Cause or Association? Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 167, 59–63. [Google Scholar] [CrossRef]
- Van Oostrum, N.; De Sutter, P.; Meys, J.; Verstraelen, H. Risks Associated with Bacterial Vaginosis in Infertility Patients: A Systematic Review and Meta-Analysis. Hum. Reprod. 2013, 28, 1809–1815. [Google Scholar] [CrossRef]
- Halis, G.; Arici, A. Endometriosis and Inflammation in Infertility. Ann. N. Y. Acad. Sci. 2004, 1034, 300–315. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, Z.; Jiang, S.; Bai, X.; Ma, C.; Peng, Q.; Chen, K.; Chang, H.; Fang, T.; Zhang, H. Probiotic Bifidobacterium Lactis V9 Regulates the Secretion of Sex Hormones in Polycystic Ovary Syndrome Patients through the Gut-Brain Axis. mSystems 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Espinoza, J.; Chaiworapongsa, T.; Kalache, K. Infection and Prematurity and the Role of Preventive Strategies. Semin. Neonatol. 2002, 7, 259–274. [Google Scholar] [CrossRef]
- Basavaprabhu, H.N.; Sonu, K.S.; Prabha, R. Mechanistic Insights into the Action of Probiotics against Bacterial Vaginosis and Its Mediated Preterm Birth: An Overview. Microb. Pathog. 2020, 141, 104029. [Google Scholar] [CrossRef]
- Moreno, I.; Simon, C. Relevance of Assessing the Uterine Microbiota in Infertility. Fertil. Steril. 2018, 110, 337–343. [Google Scholar] [CrossRef]
- Krauss-Silva, L.; Moreira, M.E.L.; Alves, M.B.; Braga, A.; Camacho, K.G.; Batista, M.R.R.; Almada-Horta, A.; Rebello, M.R.; Guerra, F. A Randomised Controlled Trial of Probiotics for the Prevention of Spontaneous Preterm Delivery Associated with Bacterial Vaginosis: Preliminary Results. Trials 2011, 12, 239. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, G.E.; Heinemann, C.; Baroja, M.L.; Bruce, A.W.; Beuerman, D.; Madrenas, J.; Reid, G. Oral Administration of the Probiotic Combination Lactobacillus Rhamnosus GR-1 and L. Fermentum RC-14 for Human Intestinal Applications. Int. Dairy J. 2002, 12, 191–196. [Google Scholar] [CrossRef]
- Verna, E.C.; Lucak, S. Use of Probiotics in Gastrointestinal Disorders: What to Recommend? Ther. Adv. Gastroenterol. 2010, 3, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.B.-M.; Lee, B.H. New Perspectives on Probiotics in Health and Disease. Food Sci. Hum. Wellness 2015, 4, 56–65. [Google Scholar] [CrossRef]
- Doron, S.; Snydman, D.R. Risk and Safety of Probiotics. Clin. Infect. Dis. 2015, 60 (Suppl. 2), S129–S134. [Google Scholar] [CrossRef]
- De Goffau, M.C.; Lager, S.; Salter, S.J.; Wagner, J.; Kronbichler, A.; Charnock-Jones, D.S.; Peacock, S.J.; Smith, G.C.S.; Parkhill, J. Recognizing the Reagent Microbiome. Nat. Microbiol. 2018, 3, 851–853. [Google Scholar] [CrossRef]
- Mombelli, B.; Gismondo, M.R. The Use of Probiotics in Medical Practice. Int. J. Antimicrob. Agents 2000, 16, 531–536. [Google Scholar] [CrossRef]
- McDonald, B.D.; Jabri, B.; Bendelac, A. Diverse Developmental Pathways of Intestinal Intraepithelial Lymphocytes. Nat. Rev. Immunol. 2018, 18, 514–525. [Google Scholar] [CrossRef]
- Ngugi, B.M.; Hemmerling, A.; Bukusi, E.A.; Kikuvi, G.; Gikunju, J.; Shiboski, S.; Fredricks, D.N.; Cohen, C.R. Effects of Bacterial Vaginosis-Associated Bacteria and Sexual Intercourse on Vaginal Colonization with the Probiotic Lactobacillus Crispatus CTV-05. Sex. Transm. Dis. 2011, 38, 1020–1027. [Google Scholar] [CrossRef]
- Fernández, L.; Cárdenas, N.; Arroyo, R.; Manzano, S.; Jiménez, E.; Martín, V.; Rodríguez, J.M. Prevention of Infectious Mastitis by Oral Administration of Lactobacillus salivarius PS2 During Late Pregnancy. Clin. Infect. Dis. 2016, 62, 568–573. [Google Scholar] [CrossRef]
- Tomusiak, A.; Strus, M.; Heczko, P.B.; Adamski, P.; Stefański, G.; Mikołajczyk-Cichońska, A.; Suda-Szczurek, M. Efficacy and Safety of a Vaginal Medicinal Product Containing Three Strains of Probiotic Bacteria: A Multicenter, Randomized, Double-Blind, and Placebo-Controlled Trial. Drug Des. Dev. Ther. 2015, 9, 5345–5354. [Google Scholar] [CrossRef] [PubMed]
- Kyono, K.; Hashimoto, T.; Kikuchi, S.; Nagai, Y.; Sakuraba, Y. A Pilot Study and Case Reports on Endometrial Microbiota and Pregnancy Outcome: An Analysis Using 16S RRNA Gene Sequencing among IVF Patients, and Trial Therapeutic Intervention for Dysbiotic Endometrium. Reprod. Med. Biol. 2019, 18, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Moreno, I.; Codoñer, F.M.; Vilella, F.; Valbuena, D.; Martinez-Blanch, J.F.; Jimenez-Almazán, J.; Alonso, R.; Alamá, P.; Remohí, J.; Pellicer, A.; et al. Evidence That the Endometrial Microbiota Has an Effect on Implantation Success or Failure. Am. J. Obstet. Gynecol. 2016, 215, 684–703. [Google Scholar] [CrossRef] [PubMed]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.K.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal Microbiome of Reproductive-Age Women. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4680–4687. [Google Scholar] [CrossRef]
- Romero, R.; Hassan, S.S.; Gajer, P.; Tarca, A.L.; Fadrosh, D.W.; Bieda, J.; Chaemsaithong, P.; Miranda, J.; Chaiworapongsa, T.; Ravel, J. The Vaginal Microbiota of Pregnant Women Who Subsequently Have Spontaneous Preterm Labor and Delivery and Those with a Normal Delivery at Term. Microbiome 2014, 2, 18. [Google Scholar] [CrossRef]
- Romero, R.; Chaiworapongsa, T.; Kuivaniemi, H.; Tromp, G. Bacterial Vaginosis, the Inflammatory Response and the Risk of Preterm Birth: A Role for Genetic Epidemiology in the Prevention of Preterm Birth. Am. J. Obstet. Gynecol. 2004, 190, 1509–1519. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Watson, P.F.; Petrie, A. Method Agreement Analysis: A Review of Correct Methodology. Theriogenology 2010, 73, 1167–1179. [Google Scholar] [CrossRef]
- Canese, K. An Updated PubMed Is on Its Way. NLM Technical Bulletin. March–April. 2019. Available online: https://www.nlm.nih.gov/pubs/techbull/ma19/ma19_pubmed_update.html (accessed on 20 January 2021).
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0 (updated March 2011); The Cochrane Collaboration; John Wiley & Sons Ltd.: Chichester, UK, 2011; Available online: https://handbook-5-1.cochrane.org/ (accessed on 20 January 2021).
- López-Moreno, A.; Aguilera, M. Probiotics Dietary Supplementation for Modulating Endocrine and Fertility Microbiota Dysbiosis. Nutrients 2020, 12, 757. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Betsi, G.I.; Athanasiou, S. Probiotics for the Treatment of Women with Bacterial Vaginosis. Clin. Microbiol. Infect. 2007, 13, 657–664. [Google Scholar] [CrossRef]
- Ozkinay, E.; Terek, M.C.; Yayci, M.; Kaiser, R.; Grob, P.; Tuncay, G. The Effectiveness of Live Lactobacilli in Combination with Low Dose Oestriol (Gynoflor) to Restore the Vaginal Flora after Treatment of Vaginal Infections. Br. J. Obstet. Gynaecol. 2005, 112, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Oerlemans, E.F.M.; Bellen, G.; Claes, I.; Henkens, T.; Allonsius, C.N.; Wittouck, S.; van den Broek, M.F.L.; Wuyts, S.; Kiekens, F.; Donders, G.G.G.; et al. Impact of a Lactobacilli-Containing Gel on Vulvovaginal Candidosis and the Vaginal Microbiome. Sci. Rep. 2020, 10, 7976. [Google Scholar] [CrossRef] [PubMed]
- Mastromarino, P.; Macchia, S.; Meggiorini, L.; Trinchieri, V.; Mosca, L.; Perluigi, M.; Midulla, C. Effectiveness of Lactobacillus-Containing Vaginal Tablets in the Treatment of Symptomatic Bacterial Vaginosis. Clin. Microbiol. Infect. 2009, 15, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Bisanz, J.E.; Seney, S.; McMillan, A.; Vongsa, R.; Koenig, D.; Wong, L.; Dvoracek, B.; Gloor, G.B.; Sumarah, M.; Ford, B.; et al. A Systems Biology Approach Investigating the Effect of Probiotics on the Vaginal Microbiome and Host Responses in a Double Blind, Placebo-Controlled Clinical Trial of Post-Menopausal Women. PLoS ONE 2014, 9, e104511. [Google Scholar] [CrossRef]
- Bradshaw, C.S.; Pirotta, M.; De Guingand, D.; Hocking, J.S.; Morton, A.N.; Garland, S.M.; Fehler, G.; Morrow, A.; Walker, S.; Vodstrcil, L.A.; et al. Efficacy of Oral Metronidazole with Vaginal Clindamycin or Vaginal Probiotic for Bacterial Vaginosis: Randomised Placebo-Controlled Double-Blind Trial. PLoS ONE 2012, 7, e34540. [Google Scholar] [CrossRef]
- Gilboa, Y.; Bar-Hava, I.; Fisch, B.; Ashkenazi, J.; Voliovitch, I.; Borkowski, T.; Orvieto, R. Does Intravaginal Probiotic Supplementation Increase the Pregnancy Rate in IVF-Embryo Transfer Cycles? Reprod. Biomed. Online 2005, 11, 71–75. [Google Scholar] [CrossRef]
- The Human Microbiome Project Consortium. Structure, Function and Diversity of the Healthy Human Microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [PubMed]
- The Human Microbiome Project Consortium. A Framework for Human Microbiome Research. Nature 2012, 486, 215–221. [Google Scholar] [CrossRef] [PubMed]
- González, A.; Vázquez-Baeza, Y.; Knight, R. SnapShot: The Human Microbiome. Cell 2014, 158, 690. [Google Scholar] [CrossRef]
- Shao, X.; Cheng, H.; Zhou, J.; Zhang, J.; Zhu, Y.; Yang, C.; Di Narzo, A.; Yu, J.; Shen, Y.; Li, Y.; et al. Prenatal Exposure to Ambient Air Multi-Pollutants Significantly Impairs Intrauterine Fetal Development Trajectory. Ecotoxicol. Environ. Saf. 2020, 201, 110726. [Google Scholar] [CrossRef] [PubMed]
- D’Argenio, V. The Prenatal Microbiome: A New Player for Human Health. High-Throughput 2018, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Onderdonk, A.B.; Delaney, M.L.; Fichorova, R.N. The Human Microbiome during Bacterial Vaginosis. Clin. Microbiol. Rev. 2016, 29, 223–238. [Google Scholar] [CrossRef]
- Gao, C.-J.; Kannan, K. Phthalates, Bisphenols, Parabens, and Triclocarban in Feminine Hygiene Products from the United States and Their Implications for Human Exposure. Environ. Int. 2020, 136, 105465. [Google Scholar] [CrossRef]
- Cherpes, T.L.; Hillier, S.L.; Meyn, L.A.; Busch, J.L.; Krohn, M.A. A Delicate Balance: Risk Factors for Acquisition of Bacterial Vaginosis Include Sexual Activity, Absence of Hydrogen Peroxide-Producing Lactobacilli, Black Race, and Positive Herpes Simplex Virus Type 2 Serology. Sex. Transm. Dis. 2008, 35, 78–83. [Google Scholar] [CrossRef]
- Witkin, S.S.; Linhares, I.M. Why Do Lactobacilli Dominate the Human Vaginal Microbiota? Br. J. Obstet. Gynaecol. 2017, 124, 606–611. [Google Scholar] [CrossRef]
- Verstraelen, H.; Senok, A.C. Vaginal Lactobacilli, Probiotics, and IVF. Reprod. Biomed. Online 2005, 11, 674–675. [Google Scholar] [CrossRef]
- Bagga, R.; Arora, P. Genital Micro-Organisms in Pregnancy. Front. Public Health 2020, 8, 225. [Google Scholar] [CrossRef]
- Melgaço, A.C.C.; Blohem Pessoa, W.F.; Freire, H.P.; Evangelista de Almeida, M.; Santos Barbosa, M.; Passos Rezende, R.; Timenetsky, J.; Miranda Marques, L.; Romano, C.C. Potential of Maintaining a Healthy Vaginal Environment by Two Lactobacillus Strains Isolated from Cocoa Fermentation. BioMed Res. Int. 2018, 2018, 7571954. [Google Scholar] [CrossRef]
- Reid, G.; Beuerman, D.; Heinemann, C.; Bruce, A.W. Probiotic Lactobacillus Dose Required to Restore and Maintain a Normal Vaginal Flora. FEMS Immunol. Med. Microbiol. 2001, 32, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Reid, G.; Charbonneau, D.; Erb, J.; Kochanowski, B.; Beuerman, D.; Poehner, R.; Bruce, A.W. Oral Use of Lactobacillus Rhamnosus GR-1 and L. Fermentum RC-14 Significantly Alters Vaginal Flora: Randomized, Placebo-Controlled Trial in 64 Healthy Women. FEMS Immunol. Med. Microbiol. 2003, 35, 131–134. [Google Scholar] [CrossRef]
- Reid, G.; Burton, J.; Hammond, J.-A.; Bruce, A.W. Nucleic Acid-Based Diagnosis of Bacterial Vaginosis and Improved Management Using Probiotic Lactobacilli. J. Med. Food 2004, 7, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, C.; Donders, G.G.; Palmeira-de-Oliveira, R.; Queiroz, J.A.; Tomaz, C.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, A. Bacteriocin Production of the Probiotic Lactobacillus Acidophilus KS400. AMB Express 2018, 8, 153. [Google Scholar] [CrossRef]
- McMillan, A.; Rulisa, S.; Gloor, G.B.; Macklaim, J.M.; Sumarah, M.; Reid, G. Pilot Assessment of Probiotics for Pregnant Women in Rwanda. PLoS ONE 2018, 13, e0195081. [Google Scholar] [CrossRef]
- Anukam, K.C.; Duru, M.U.; Eze, C.C.; Egharevba, J.; Aiyebelehin, A.; Bruce, A.; Reid, G. Oral Use of Probiotics as an Adjunctive Therapy to Fluconazole in the Treatment of Yeast Vaginitis: A Study of Nigerian Women in an Outdoor Clinic. Microbial Ecol. Health Dis. 2009, 21, 72–77. [Google Scholar] [CrossRef]
- Sniffen, J.C.; McFarland, L.V.; Evans, C.T.; Goldstein, E.J.C. Choosing an Appropriate Probiotic Product for Your Patient: An Evidence-Based Practical Guide. PLoS ONE 2018, 13, e0209205. [Google Scholar] [CrossRef] [PubMed]
- Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. Effect of Probiotics Supplementations on Health Status of Athletes. Int. J. Environ. Res. Public Health 2019, 16, 4469. [Google Scholar] [CrossRef]
- Hyman, R.W.; Herndon, C.N.; Jiang, H.; Palm, C.; Fukushima, M.; Bernstein, D.; Vo, K.C.; Zelenko, Z.; Davis, R.W.; Giudice, L.C. The Dynamics of the Vaginal Microbiome during Infertility Therapy with in Vitro Fertilization-Embryo Transfer. J. Assist Reprod. Genet. 2012, 29, 105–115. [Google Scholar] [CrossRef]
- Chang, C.-J.; Lin, T.-L.; Tsai, Y.-L.; Wu, T.-R.; Lai, W.-F.; Lu, C.-C.; Lai, H.-C. Next Generation Probiotics in Disease Amelioration. J. Food Drug Anal. 2019, 27, 615–622. [Google Scholar] [CrossRef]
- Lai, K.-P.; Chung, Y.-T.; Li, R.; Wan, H.-T.; Wong, C.K.-C. Bisphenol A Alters Gut Microbiome: Comparative Metagenomics Analysis. Environ. Pollut. 2016, 218, 923–930. [Google Scholar] [CrossRef]
- O’Toole, P.W.; Marchesi, J.R.; Hill, C. Next-Generation Probiotics: The Spectrum from Probiotics to Live Biotherapeutics. Nat. Microbiol. 2017, 2, 17057. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Chen, Y.; Chen, T. Vaginal Microbiota Transplantation for the Treatment of Bacterial Vaginosis: A Conceptual Analysis. FEMS Microbiol. Lett. 2019, 366, fnz025. [Google Scholar] [CrossRef] [PubMed]
- DeLong, K.; Zulfiqar, F.; Hoffmann, D.E.; Tarzian, A.J.; Ensign, L.M. Vaginal Microbiota Transplantation: The Next Frontier. J. Law Med. Ethics 2019, 47, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Lev-Sagie, A.; Goldman-Wohl, D.; Cohen, Y.; Dori-Bachash, M.; Leshem, A.; Mor, U.; Strahilevitz, J.; Moses, A.E.; Shapiro, H.; Yagel, S.; et al. Vaginal Microbiome Transplantation in Women with Intractable Bacterial Vaginosis. Nat. Med. 2019, 25, 1500–1504. [Google Scholar] [CrossRef]

| Parameters | Inclusion Criteria |
|---|---|
| Population | Human |
| Intervention | Probiotics strains and doses |
| Comparison | Vaginal probiotics versus placebo |
| Outcome | Fertility parameters |
| Setting | Clinical trials (CTs) |
| Reference | Population Sample (n) | Probiotic Strains | Probiotic Doses | Probiotic Administration Time (Weeks) | Disorders/Diseases | Clinical Effects and Health Parameter Modifications |
|---|---|---|---|---|---|---|
| Oerlemans et al. [48] | 20 women with vulvovaginal candidiasis (VVC) | Lactobacillus pentosus KCA1, Lactobacillus plantarum WCFS1, and Lactobacillus rhamnosus GG | 2.5 × 109–2.5 × 1010 CFU/day | 1.5 | Vulvovaginal candidiasis | Probiotic formulation restores the vaginal microbiota in 45% of women. The other 55% of women needed rescue medication (fluconazole), but, at the end of the study, these women presented a larger reduction in the amount of Lactobacillus sp. compared to the other group. |
| Mastromarino et al. [49] | 39 women | Florisia®: Lactobacillus brevis (CD2), Lactobacillus salivarius subsp. salicinius (FV2), and L. plantarum (FV9) | ≥109 CFU/day | 1 | Bacterial vaginosis | This probiotic product of exogenous strains of Lactobacillus spp. administered intravaginally restored the healthy vaginal microbiota and it can be administered to treat bacterial vaginosis (BV) disorders. |
| Bisanz et al. [50] | 14 postmenopausal women | L. rhamnosus GR-1 and Lactobacillus reuteri RC-14 | 2.5 × 109 CFU/day | 3 (day) | Bacterial vaginosis | Total Lactobacillus increased and the proportion of Atopobium decreased. In addition, there was a trend for Gardnerella and Prevotella reduction. No changes in Nugent score and host metabolome. |
| Bradshaw et al. [51] | 450 healthy women | Lactobacillus acidophilus KS400 | >107 CFU/day | 12 | Recurrent bacterial vaginosis | Lactobacillus acidophilus KS400 administered vaginally in combination of oral metronidazole during an extended course did not cure recurrent bacterial vaginosis. |
| Gilboa et al. [52] | 117 women | Probiotic Femina®: Lactobacillus acidophilus, Bifidobacterium bifidum, and Bifidobacterium longum | 6 × 109 CFU/treatment | 1 (day) | In vitro fertilization (IVF)–embryo transfer cycle | Probiotic Femina® did not affect the vaginal colonization of Lactobacillus during oocyte retrieval or embryo transfer and did not improve the pregnancy rate. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Moreno, A.; Aguilera, M. Vaginal Probiotics for Reproductive Health and Related Dysbiosis: Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 1461. https://doi.org/10.3390/jcm10071461
López-Moreno A, Aguilera M. Vaginal Probiotics for Reproductive Health and Related Dysbiosis: Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2021; 10(7):1461. https://doi.org/10.3390/jcm10071461
Chicago/Turabian StyleLópez-Moreno, Ana, and Margarita Aguilera. 2021. "Vaginal Probiotics for Reproductive Health and Related Dysbiosis: Systematic Review and Meta-Analysis" Journal of Clinical Medicine 10, no. 7: 1461. https://doi.org/10.3390/jcm10071461
APA StyleLópez-Moreno, A., & Aguilera, M. (2021). Vaginal Probiotics for Reproductive Health and Related Dysbiosis: Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 10(7), 1461. https://doi.org/10.3390/jcm10071461







