Polymorphism of Interleukin-1 Gene Cluster in Polish Patients with Acute Coronary Syndrome
Abstract
1. Introduction
2. Materials and Methods
- -
- >18 years of age,
- -
- Having signed an informed consent to participate in the study including genetic tests.
- -
- An isolated critical change in the main left coronary artery,
- -
- Coronary artery spasm and coronary embolism,
- -
- Endocrine disorders or other conditions contributing to the imbalance between oxygen supply and demand in the myocardium (type 2) or necrosis of the myocardium associated with treatment procedures (type 4a and 4b).
2.1. Group Characteristics
2.2. Study Protocol
2.3. Genotyping
2.4. Statistical Methods
3. Results
- -
- For locus -31 of the IL-1B gene: T—66%, C—34%,
- -
- For locus -511: C—65.8%, T—34.2%,
- -
- For the IL-1RN gene—allele 1: 77.4%; 2: 18.6%; 3: 3.8%, and 4: 0.2%.
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kukreti, S.; Konstantopoulos, K.; Smith, C.W.; McIntire, L.V. Molecular mechanisms of monocyte adhesion to interleukin-1beta-stimulated endothelial cells under physiologic flow conditions. Blood 1997, 89, 4104–4111. [Google Scholar] [CrossRef]
- Ferreira, A.M.; McNeil, C.J.; Stallaert, K.M.; Rogers, K.A.; Sanding, M. Interleukin-1beta reduces transcellular monocyte diapedesis and compromises endothelial adherence junction integrity. Microcirculation 2005, 12, 563–579. [Google Scholar] [CrossRef]
- Siwik, D.A.; Chang, D.L.; Colucci, W.S. Interleukin-1beta and tumor necrosis factor-alpha decreases collagen synthesis and increase matrix metalloproteinase activity in cardiac fibroblasts in vitro. Circ. Res. 2000, 86, 1259–1265. [Google Scholar] [CrossRef]
- Zheng, Y.; Gardner, S.E.; Clarke, M.C. Cell death, damage-associated molecular patterns, and sterile inflammation in cardiovascular disease. Arter. Thromb. Vasc. Biol. 2011, 31, 2781–2786. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, S.; Tolley, N.D.; Dixon, D.A.; McIntyre, T.M.; Pescott, S.M.; Zimmermann, G.A.; Weyrich, A.S. Activated platelets mediate inflammatory signaling by regulated interleukin 1beta synthesis. J. Cell Biol. 2001, 154, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.T.; McIntyre, T.M. Lipopolysaccharide signaling without a nucleus: Kinase cascades stimulate platelet shedding of proinflammatory IL-1b-rich microparticles. J. Immunol. 2011, 186, 5489–5496. [Google Scholar] [CrossRef] [PubMed]
- Arendt, W.P. The balance between IL-1 and IL-Ra in disease. Cytokine Growth Factor. Rev. 2002, 13, 323–340. [Google Scholar] [CrossRef]
- Martinon, F.; Petrilli, V.; Mayor, A.; Tardivel, A.; Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 2006, 440, 237–241. [Google Scholar] [CrossRef]
- Casini-Raggi, V.; Kam, L.; Chong, Y.J.; Fiocchi, C.; Pizarro, T.T.; Cominelli, F. Mucosal imbalance of IL-1 and IL-1 receptor antagonist in inflammatory bowel disease. A novel mechanism of chronic intestinal inflammation. J. Immunol. 1995, 154, 2434–2440. [Google Scholar] [PubMed]
- Dewberry, R.; Holden, H.; Crossman, D.; Francis, S. Interleukin-1 receptor antagonist expression in human endothelial cells and atherosclerosis. Arter. Thromb. Vasc. Biol. 2000, 20, 2394–2400. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Thuren, T.; Zalewski, A.; Libby, P. Interleukin-1b inhibition and the prevention of recurrent cardiovascular events: Rationale and design of the canakinumab anti-inflammatory thrombosis outcomes study (CANTOS). Am. Heart J. 2011, 162, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerosis Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.-C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N. Engl. J. Med. 2019, 381, 2497–2505. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; White, H.D. Joint ESC/ACCF/AHA/WHF Task Force for the Redefinition of Myocardial Infarction. Universal definition of myocardial infarction. Circulation 2007, 116, 2634–2653. [Google Scholar] [CrossRef] [PubMed]
- Van de Werf, F.; Bax, J.; Betriu, A.; Blomstrom-Lundquist, C.; Crea, F.; Falk, V.; Filippatos, G.; Fox, K.; Huber, K.; Kastrati, A.; et al. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: The Task Force on the Management of ST-Segment Elevation Acute Myocardial Infarction of the European Society of Cardiology. Eur. Heart J. 2008, 29, 2909–2945. [Google Scholar] [PubMed]
- Bassand, J.P.; Hamm, C.W. New European guidelines for the management of patients with unstable angina/non-ST-segment elevation myocardial infarction—What are the new and key messages. Pol. Arch Med. Wewn 2007, 117, 391–393. [Google Scholar]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Simoons, M.L.; Chaitman, B.R.; White, H.D.; Katus, H.A.; Apple, F.S.; Lindahl, B.; Morrow, D.A.; et al. Third universal definition of myocardial infarction. Eur. Heart J. 2012, 33, 2551–2567. [Google Scholar] [CrossRef]
- Gensini, G.G. A more meaningful scoring system for determining the severity of coronary heart disease. Am. J. Cardiol. 1983, 51, 606. [Google Scholar] [CrossRef]
- Rentrop, K.P.; Cohen, M.; Blanke, H.; Philips, R.A. Changes in collateral channel filling immediately after controlled coronary artery occlusion by an agioplasty balloon in human subjects. JACC 1985, 5, 587–592. [Google Scholar] [CrossRef]
- Luotola, K.; Pietilä, A.; Alanne, M.; Lanki, T.; Loo, B.M.; Jula, A.; Perola, M.; Peters, A.; Zeller, T.; Blankenberg, S.; et al. Genetic variation of the interleukin-1 family and nongenetic factors determining the interleukin-1 receptor antagonist phenotypes. Metabol. Clin. Exp. 2010, 59, 1520–1527. [Google Scholar] [CrossRef] [PubMed]
- Gorący, J.; Gorący, I.; Safranow, K.; Taryma, O.; Adler, G.; Ciechanowicz, A.A. Lack of association of interleukin-1 gene cluster polymorphisms with angiographically documented coronary artery disease: Demonstration of association with hypertension in the Polish population. Arch. Med. Res. 2011, 42, 426–432. [Google Scholar] [CrossRef]
- Rad, R.; Prinz, C.; Neu, B.; Neuhofer, M.; Zeitner, M.; Voland, P.; Becker, I.; Schepp, W.; Gerhard, M. Synergistic effect of Helicobacter pylori virulence factors and interleukin-1 polymorphisms for the development of severe histological changes in the gastric mucosa. J. Infect Dis. 2003, 188, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Eklund, C.; Jahan, F.; Pessi, T.; Lehtimäki, T.; Hurme, M. Interleukin 1B gene polymorphism is associated with baseline C-reactive protein levels in healthy individuals. Eur. Cytokine Netw. 2003, 14, 168–171. [Google Scholar] [PubMed]
- Brown, B.D.; Nsengimana, J.; Barret, J.H.; Lawrence, M.A.; Steiter, L.; Cheng, S. An evaluation of inflammatory gene polymorphisms in sibships discordant for premature coronary artery disease: The GRACE-IMMUNE study. BMC Med. 2010, 8, 5. [Google Scholar] [CrossRef]
- Iacoviello, L.; Di Castelnuovo, A.; Gattone, M.; Pezzini, A.; Assanelli, D.; Lorenzet, L. Polymorphisms of the interleukin-1b gene affect the risk of myocardial infarction and ischemic stroke at young age and the response of the mononuclear cells to stimulation in vitro. Arter. Thromb. Vasc. Biol. 2004, 25, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Latella, M.; de Gaetano, M.; Di Castelnuovo, A.; Napoleone, E.; Lorenzet, R.; Gattone, M. Interleukin 1 gene cluster, myocardial infarction at young age and inflammatory response of human mononuclear cells. Immunol. Invest. 2009, 38, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, P.S.; Sheikine, Y.; Jatta, K.; Ghaderi, M.; Samnegård, A.; Eriksson, P.; Sirsjö, A. A functional interleukin-1 receptor antagonist influences atherosclerosis development—The interleukin 1ß: Interleukin-1 receptor agonist balance in atherosclerosis. Circ J. 2009, 73, 1531–1536. [Google Scholar] [CrossRef]
- Oda, K.; Tanaka, N.; Arai, T.; Araki, J.; Song, Y.; Zhang, L.; Kuchiba, A.; Hosoi, T.; Shirasawa, T.; Muramatsu, M.; et al. Polymorphisms in pro- and anti-inflammatory cytokine genes and susceptibility to atherosclerosis: A pathological study of 1503 consecutive autopsy cases. Hum. Mol. Gen. 2007, 16, 592–599. [Google Scholar] [CrossRef]
- Gorący, I.; Kaczmarczyk, M.; Ciechanowicz, A.; Lewandowska, K.; Jakubiszyn, P.; Bodnar, O. Polymorphism of Interleukin 1B May Modulate the Risk of Ischemic Stroke in Polish Patients. Medicina 2019, 55, 558. [Google Scholar] [CrossRef]
- Jagielska, J.; Kapopara, P.R.; Salguero, G.; Scherr, M.; Schűtt, H.; Grote, K.; Schieffer, B.; Bavendiek, U. Interleukin-1b assembles a proangiogenic signaling module consisting of caveolin-1, tumor necrosis factor receptor-associated factor 6, p38-mitogen-activated protein kinase (MAPK), and MAPK-activated protein kinase 2 in endothelial cells. Arter. Thromb. Vasc. Biol. 2012, 32, 1280–1288. [Google Scholar] [CrossRef]
- Yang, L.; Guo, X.G.; Du, C.Q.; Jang, J.X.; Jiang, D.M.; Li, B.; Zhou, W.-J.; Zhang, F.-R. Interleukin-1 beta increases activity of human endothelial progenitor cells. Inflammation 2012, 35, 1242–1250. [Google Scholar] [CrossRef] [PubMed]
- Ørn, S.; Ueland, T.; Manhenke, C.; Sandanger, Ø.; Godang, K.; Yndestad, A.; Mollnes, T.E.; Dickstein, K.; Aukrust, P. Increased interleukin-1β levels are associated with left ventricular hypertrophy and remodelling following acute ST segment elevation myocardial infarction treated by primary percutaneous coronary intervention. J. Intern. Med. 2012, 272, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Rechciński, T.; Wierzbowska-Drabik, K.; Wejner-Mik, P.; Szymańska, B.; Jerczyńska, H.; Lipiec, P.; Kasprzak, J.D. An attempt to find a relationship between pro-inflammatory interleukin-1 beta or its natural receptor antagonist concentration and hypertrophy of heart muscle in myocardial infarction survivors. Eur. Heart J. Cardiovasc. Imaging 2014. [Google Scholar] [CrossRef]
- Samiani, N.J.; Erdmann, J.; Hall, A.S.; Henstenberg, C.; Mangina, M.; Mayer, B. Genomwide association analysis of coronary artery disease. N. Engl. J. Med. 2007, 357, 443–453. [Google Scholar] [CrossRef]
- Poloński, L.; Gąsior, M.; Gierlotka, M.; Kalarus, Z.; Ciesliński, A.; Dubiel, J.S.; Gil, R.J.; Ruzyłło, W.; Trusz-Gluza, M.; Zembala, M.; et al. Polish Registry of Acute Coronary Syndromes (PL-ACS). Characteristics, treatments and outcomes of patients with acute coronary syndromes in Poland. Kardiol. Pol. 2007, 65, 861–872. [Google Scholar] [PubMed]
- Poloński, L.; Gąsior, M.; Gierlotka, M.; Wilczek, K.; Kalarus, Z.; Dubiel, J.S.; Rużyłło, W.; Banasiak, W.; Opolski, G.; Zembala, M.; et al. What has changed in the treatment of ST-segment elevation myocardial infarction in Poland in 2003–2009? data from the Polish Registry of Acute Coronary Syndromes (PL-ACS). Kardiol. Pol. 2011, 69, 1109–1118. [Google Scholar] [PubMed]
- Niccoli, G.; Giubilato, S.; Di Vito, L.; Leo, A.; Cosentino, N.; Pitocco, D.; Marco, V.; Ghirlanda, G.; Prati, F.; Crea, F. Severity of coronary atherosclerosis in patients with a first acute coronary event: A diabetes paradox. Eur. Heart J. 2013, 34, 729–741. [Google Scholar] [CrossRef]
- Rechciński, T.; Grębowska, A.; Kurpesa, M.; Peruga, Z.; Dziuba, M.; Krzemińska-Pakuła, M.; Rudnicka, W.; Chmiela, M. CD14 gene polymorphism 159C/T in a group of patients with coronary artery disease from a population with high morbidity of cardiovascular diseases. Kardiol. Pol. 2007, 65, 237–244. [Google Scholar] [PubMed]
- Wen, A.Q.; Weng, J.; Feng, K.; Zhu, P.F.; Wang, Z.G.; Jiang, J.X. Effects of haplotypes in the interleukin 1b promoter on lipopolisaccharide-induced interleukin 1b expression. Shock 2006, 26, 25–30. [Google Scholar] [CrossRef]
- Rogus, J.; Beck, J.D.; Offenbacher, S.; Huttner, K.; Iacoviello, L.; Latella, M.C.; De Gaetano, M.; Wang, H.-Y.; Kornman, K.S.; Duff, G.W. IL-1B gene promoter haplotype pairs predict clinical levels of interleukin-1b and C-reactive protein. Hum. Genet. 2008, 123, 387–398. [Google Scholar] [CrossRef]
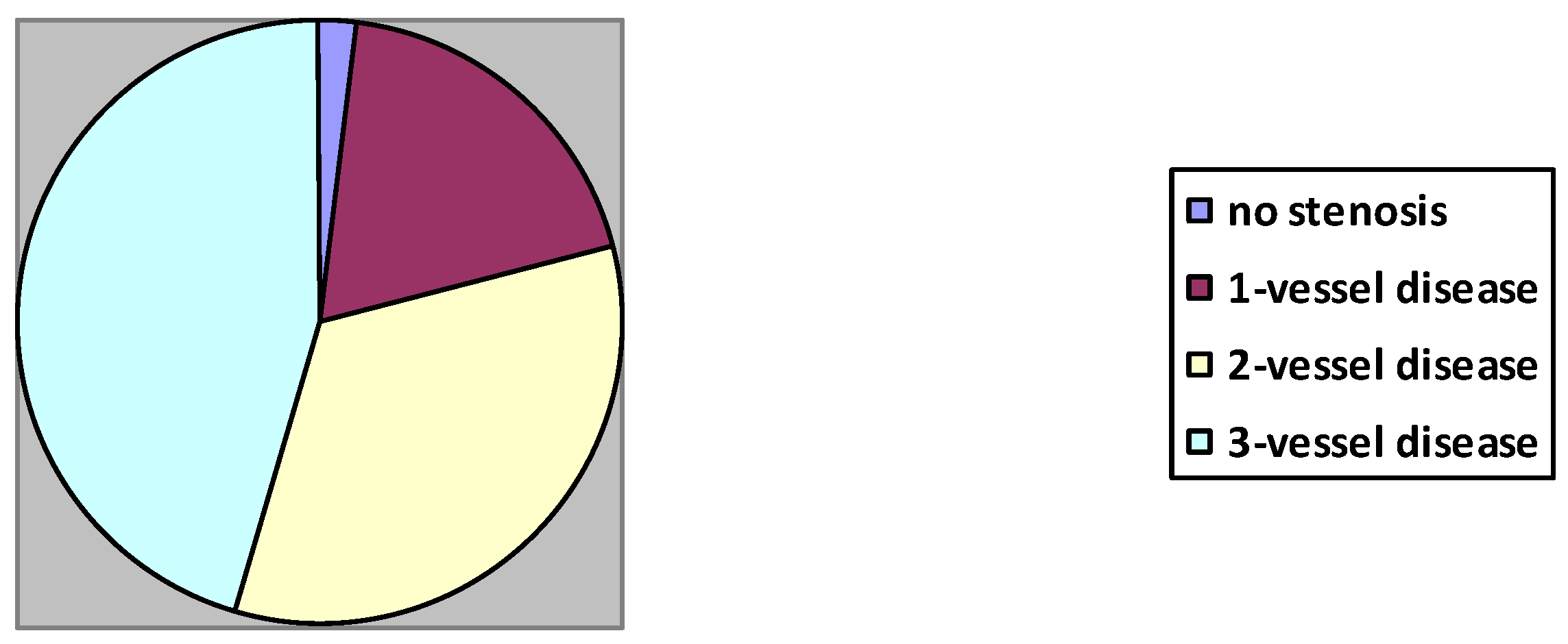
| Numeric Symbol | Number of 86 Base Pair Repeats |
|---|---|
| 1 | 4 |
| 2 | 2 |
| 3 | 5 |
| 4 | 3 |
| 5 | 6 |
| Parameter/Variable | The Value/Frequency of Occurrence n (%) |
|---|---|
| Age (years) during the index ACS—range | 39–83 |
| The median age [interquartile range] (years) | 59 [53–65] |
| Mean age ± SD (years) | 59.2 ± 9 |
| Males | 187 (74.8%) |
| Risk Factor/Disease Entity | Value/Frequency of Occurrence (%) |
|---|---|
| Arterial hypertension | 180 (72.0%) |
| Type 2 diabetes mellitus | 70 (28.0%) |
| Smoking | 111 (44.4%) |
| Obesity | 91 (36.4%) |
| BMI: minimum–maximum | 18.4–41.1 |
| BMI: median [interquartile range] | 28.1 [25.5–31.0] |
| Hyperlipidemia | 188 (75.2%) |
| Cerebral stroke | 20 (8.0%) |
| Atherosclerosis of arteries of lower limbs | 6 (2.4%) |
| Abdominal aortic aneurysm | 6 (2.4%) |
| Intracranial aneurysm | 9 (3.6%) |
| Chronic inflammatory diseases * | 5 (2.0%) |
| Locus | Allele/Genotype | The Real Number/Frequency of Alleles/Genotypes n (%) | The Theoretical Number/Frequency in Accordance with the Hardy-Weinberg Equation n (%) | Significance of the Differences in the Distribution of the Frequencies of Genotypes (chi2 Test) |
|---|---|---|---|---|
| -31 | C/ T/ /CC /CT /TT | 170 (34.0) 330 (66.0) 24 (9.6) 122 (48.8) 104 (41.6) | × × 28.9 (11.56) 112.2 (44.88) 108.9 (43.56) | p = 0.38 |
| -511 | T/ C/ /TT /CT /CC | 171 (34.2) 329 (65.8) 24 (9.6) 123 (49.2) 103 (41.2) | × × 29.2 (11.7) 112.6 (45.0) 108.2 (43.3) | p = 0.34 |
| -RN | 1/ 2/ 3/ 4/ /11 /22 /33 /44 /12 /13 /14 /23 /24 /34 | 387 (77.4) 93 (18.6) 19 (3.8) 1 (0.2) 159 (63.6) 17 (6.8) 5 (2.0) 0 (0) 59 (23.6) 9 (3.6) 1 (0.4) 0 (0) 0 (0) 0 (0) | × × × × 149.7 (59.9) 8.6 (3.5) 0.4 (0.1) <0.1 (<0.1) 71.9 (28.8) 14.7 (5.8) 0.8 (0.3) 3.5 (1.4) 0.2 (<0.1) < 0.1 (<0.1) | p = 0.0001 |
| No. | The Variant with Three Polymorphisms -31 -511 -RN | The Real Number and Frequency n (%) Letter Symbol | The Theoretical Number and Frequency in Accordance with the Hardy-Weinberg Equation n (%) | The Significance of Distribution Differences of the Genetic Variants—Real and Theoretical (chi2 Test) |
|---|---|---|---|---|
| 69 Variants not Represented in the Study Group | 0 (0) | 72.62 (29.06) | ||
| i ii iii iv v vi vii viii ix x xi xii xiii xiv xv xvi xvi xviii xix xx xxi | CT CT 11 TT CT 11 CT CC 11 TT CC 11 CT CT 12 TT CC 12 TT CC 13 CT TT 11 CC CT 11 CC CT 12 CT CT 13 CC TT 11 CT CT 22 TT CC 22 CC TT 12 CT TT 22 TT CC 14 CC TT 22 CT CT 33 CT TT 33 TT CC 33 | 67 (26.8) L 3 (1.2) 2 (0.8) 78 (31.2) XL 39 (15.6) M 10 (4.0) S 6 (2.4) 1 (0.4) 2 (0.8) 1 (0.4) 3 (1.2) 6 (2.4) 5 (2.0) 5 (2.0) 9 (3.6) XS 1 (0.4) 1 (0.4) 6 (2.4) 3 (1.2) 1 (0.4) 1 (0.4) | 30.25 (12.1) 29.36 (11.7) 29.1 (11.6) 28.24 (11.3) 14.53 (5.8) 13.57 (5.4) 2.77 (1.1) 7.86 (3.1) 7.79 (3.1) 3.74 (1.5) 2.97 (1.2) 2.02 (0.8) 1.74 (0.7) 1.63 (0.6) 0.97 (0.4) 0.45 (0.2) 0.14 (<0.1) 0.12 (<0.1) 0.07 (<0.1) 0.01 (<0.1) 0.01 (<0.1) | <0.001 |
| The Parameter | -31 TT n = 104 | -31 CT n = 122 | -31 CC n = 24 | Significance (chi2 Test) | |
|---|---|---|---|---|---|
| ASC <60 years of age | n | 57 | 60 | 12 | 0.69 |
| % | 54.8 | 49.2 | 50 | ||
| Collateral circulation | n | 35 | 45 | 10 | 0.67 |
| % | 34.0 | 37.2 | 43.5 | ||
| Gensini Score | mean ± SD | 48 ± 35 | 45 ± 29 | 51 ± 41 | 0.94 |
| Minimum–maximum | 1–188 | 4–148 | 6–140 | ||
| The median [25–75%] | 40 [24–65] | 39 [24–63] | 34 [19–83] | ||
| The number of arteries with stenosis ≥70%: 0/1/2/3 | The number of patients | 4/39/38/23 | 1/61/40/20 | 0/14/5/5 | 0.76 |
| % | 4/37.5/36.5/22 | 0.8/50/32.8/16.4 | 0/58/21/21 | ||
| Left ventricular hypertrophy: absent/present | n | 15/110 | 12/87 | 6/20 | 0.81 1 |
| % | 6/44 | 4.8/34.8 | 2.4/8 | ||
| The Parameter | -511 CC n = 103 | -511 CT n = 123 | -511 TT n = 24 | Significance (chi2 Test) | |
|---|---|---|---|---|---|
| ASC <60 years of age | n | 56 | 62 | 11 | 0.70 |
| % | 54.4 | 50.4 | 45.8 | ||
| Collateral circulation | n | 34 | 46 | 10 | 0.66 |
| % | 33.3 | 38.0 | 41.6 | ||
| Gensini Score | mean ± SD | 47 ± 34 | 47 ± 31 | 51 ± 41 | 0.99 |
| Minimum–maximum | 1–188 | 4–154 | 6–140 | ||
| The median [25–75%] | 40 [23–62] | 39 [24–66] | 37 [20–80] | ||
| The number of arteries with stenosis ≥70%: 0/1/2/3 | The number of patients | 4/40/36/23 | 1/61/41/20 | 0/13/6/5 | 0.88 |
| % | 3.9/38.8/35/22.3 | 0.8/49.6/33.3/16.3 | 0/54/25/21 | ||
| Left ventricular hypertrophy: absent/present | n | 15/110 | 12/87 | 6/20 | 0.81 1 |
| % | 6/44 | 4.8/34.8 | 2.4/8 | ||
| The Parameter | RN 11 n = 159 | RN 12 n = 59 | RN 22 n = 17 | RN 13 n = 9 | RN 33 n = 5 | RN 14 n = 1 | Significance (chi2 Test) | |
|---|---|---|---|---|---|---|---|---|
| ASC < 60 years of age | N | 77 | 40 | 5 | 5 | 2 | 0 | 0.04 |
| % | 48.4 | 67.8 | 29.4 | 55.6 | 40.0 | 0 | ||
| Collateral circulation | N | 56 | 21 | 7 | 4 | 2 | 0 | 0.94 |
| % | 35.7 | 35.6 | 43.7 | 44.4 | 40 | 0 | ||
| Gensini Score | mean ± SD | 48.4 ± 34.0 | 48.2 ± 34.3 | 39.4 ± 22.3 | 42.1 ± 33.8 | 48.2 ± 19.3 | 14 | 0.78 |
| Minimum–maximum | 3.5–188 | 1–148 | 6–80 | 2–88 | 24–77 | 14–14 | ||
| The median [25–75%] | 39.8 [24–74] | 40.0 [24–80] | 38.5 [23–54] | 36.5 [4–80] | 49 [40–51] | 14 | ||
| The number of arteries with stenosis ≥70%: 0/1/2/3 | The number of patients | 2/68/56/33 | 1/32/15/11 | 0/9/6/2 | 2/2/4/1 | 0/2/2/1 | 0/0/1/0 | 0.99 |
| % | 1.2/42.8/ /35.3/ /20.7 | 1.6/54.3/ /25.5/ /18.6 | 0/53/35/ /12 | 22.2/22.2/ /44.5/11.1 | 0/40/40/20 | 0/0/100//0 | ||
| Left ventricular hypertrophy: absent/present | N | 29/130 | 12/47 | 6/11 | 2/7 | 1/4 | 0/1 | 0.87 1 |
| % | 11.6/52 | 4.8/18.8 | 2.4/4.4 | 0.8/2.8 | 0.4/1.6 | 0/0.4 | ||
| The Parameter | The Genetic Variant at the 3 Loci Numbers in the Subgroups Variant Symbol | Significance (Fisher’s Exact Test) | |||||
|---|---|---|---|---|---|---|---|
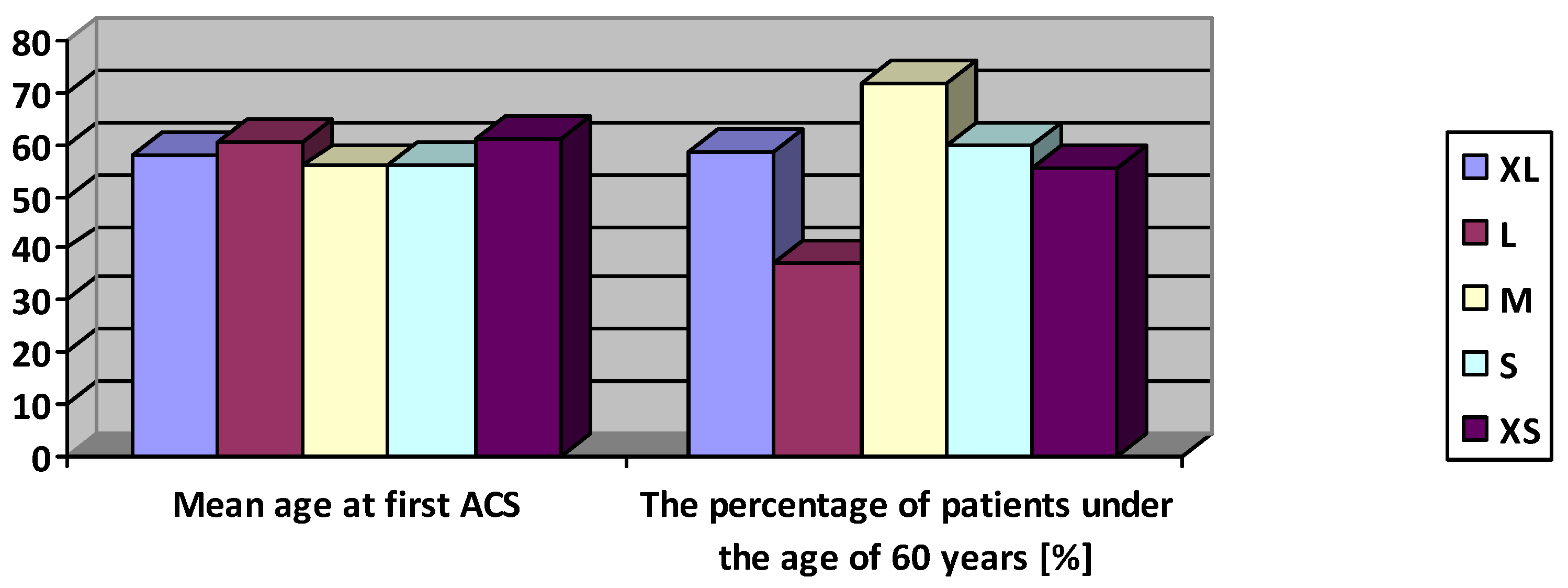
| -31 TT -511 CC -RN 11 n = 78 XL | -31 CT -511 CT -RN 11 n = 67 L | -31 CT -511 CT -RN 12 n = 39 M | -31 TT -511 CC -RN 12 n = 10 S | -31 CC -511 TT -RN 12 n = 9 XS | |||
| ACS <60 years of age | n | 46 | 25 | 28 | 6 | 5 | 0.07 |
| % | 58.9 | 37.3 | 71.8 | 60 | 55.6 | ||
| Collateral circulation | N | 26 | 24 | 14 | 3 | 4 | 0.63 |
| % | 33.3 | 36.4 | 35.9 | 30.0 | 44.4 | ||
| Gensini Score | mean ± SD | 50.2 ± 35.8 | 43.8 ± 28.1 | 46.9 ± 34.1 | 36.9 ± 18.4 | 69.6 ± 43.2 | 0.62 |
| Minimum–maximum | 3.5–188 | 4–112 | 4–148 | 1–59 | 16–140 | ||
| The median [25–75%] | 40 [24–80] | 38 [24–64] | 39.5 [24–80] | 41.25 [21–52] | 80 [32–86] | 0.58 | |
| % of distribution acc. to the tertiles 1:2:3 | 33:31:36 | 34:32:34 | 36:32:32 | 34:30:36 | 22:22:56 | ||
| The number of arteries with stenosis >70%: 0/1/2/3 | Number of patients | 1/31/27/ /19 | 1/29/26/ /11 | 0/25/9/5 | 1/5/2/2 | 0/2/3/4 | 0.97 |
| % | 1.4/39.7/ /34.6/24.3 | 1.6/43.2// /38.8/16.4 | 0/64.1/ /23.1/12.8 | 10/50/ /20/20 | 0/22.2/ /33.3/44.5 | ||
| Parameter/Result of Examination/Treatment | The Value/Frequency of Occurrence n (%) |
|---|---|
| ACS 1 with ST segment elevation | 177 (70.8%) |
| ACS without ST segment elevation | 73 (29.2%) |
| Primary PCI 2 with stent implantation | 193 (77.2%) |
| Primary balloon PCI | 4 (1.6%) |
| Pharmacological treatment of ACS alone | 19 (7.6%) |
| Surgical revascularization after ACS (in three patients after pPCI 2) | 37 (14.8%) |
| Gensini Score Minimum–maximum median [interquartile range] mean ± SD | 1–188 40.0 [24.0–66.0] 47.45 ± 33.1 |
| Left ventricular ejection fraction (%) Minimum–maximum median [interquartile range] mean ± SD | 22–60 47 [40–54] 46.5 ± 9.5 |
| Left ventricular hypertrophy | 217 (86.8%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rechciński, T.; Szymańska, B.; Wierzbowska-Drabik, K.; Chmiela, M.; Matusiak, A.; Kurpesa, M.; Wróblewski, J.; Kasprzak, J.D. Polymorphism of Interleukin-1 Gene Cluster in Polish Patients with Acute Coronary Syndrome. J. Clin. Med. 2021, 10, 990. https://doi.org/10.3390/jcm10050990
Rechciński T, Szymańska B, Wierzbowska-Drabik K, Chmiela M, Matusiak A, Kurpesa M, Wróblewski J, Kasprzak JD. Polymorphism of Interleukin-1 Gene Cluster in Polish Patients with Acute Coronary Syndrome. Journal of Clinical Medicine. 2021; 10(5):990. https://doi.org/10.3390/jcm10050990
Chicago/Turabian StyleRechciński, Tomasz, Bożena Szymańska, Karina Wierzbowska-Drabik, Magdalena Chmiela, Agnieszka Matusiak, Małgorzata Kurpesa, Janusz Wróblewski, and Jarosław D. Kasprzak. 2021. "Polymorphism of Interleukin-1 Gene Cluster in Polish Patients with Acute Coronary Syndrome" Journal of Clinical Medicine 10, no. 5: 990. https://doi.org/10.3390/jcm10050990
APA StyleRechciński, T., Szymańska, B., Wierzbowska-Drabik, K., Chmiela, M., Matusiak, A., Kurpesa, M., Wróblewski, J., & Kasprzak, J. D. (2021). Polymorphism of Interleukin-1 Gene Cluster in Polish Patients with Acute Coronary Syndrome. Journal of Clinical Medicine, 10(5), 990. https://doi.org/10.3390/jcm10050990








