Estimating the Prevalence of Knee Pain and the Association between Illness Perception Profiles and Self-Management Strategies in the Frederiksberg Cohort of Elderly Individuals with Knee Pain: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Period
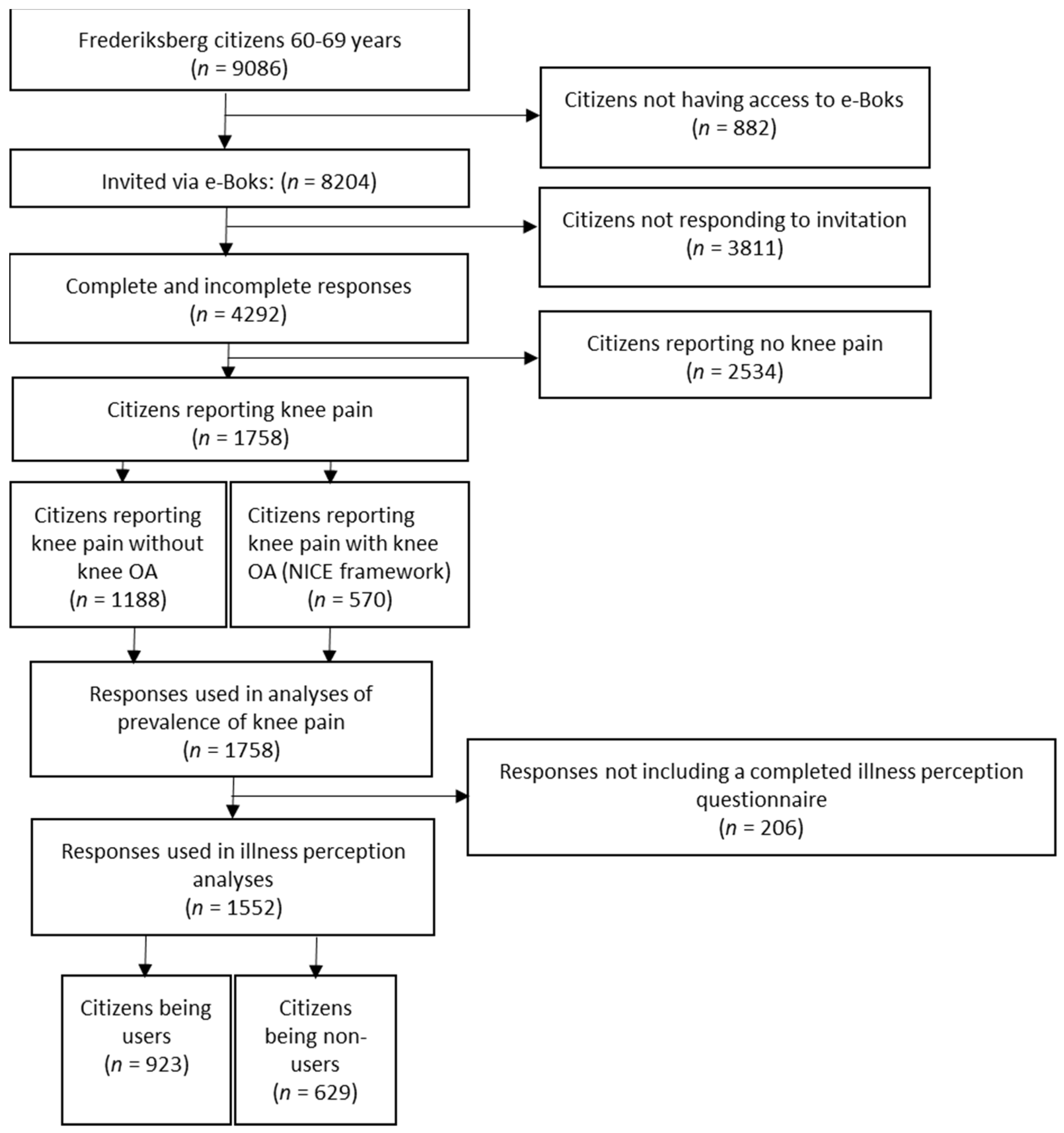
2.2. Participants
2.3. Variables and Outcome Measures
2.3.1. The Brief Illness Perception Questionnaire
2.3.2. Knee Injury and Osteoarthritis Outcome Score
2.3.3. Current Knee Pain and Self-Reported Knee Osteoarthritis
- Age over 45
- Activity related knee pain and
- Morning joint-related stiffness that lasts no longer than 30 min
2.3.4. Health Related Quality of Life
2.4. Self-Management Strategies
2.5. Use of CAMs
- Vitamins and minerals were defined as any supplement containing only vitamins or minerals, taken by the respondent in order to relieve knee pain or promote health. Seventeen predefined choices for the use of vitamins or minerals were given (Supplementary file 1).
- Dietary supplements/herbal medicines (in this manuscript referred to as “dietary supplements”) were defined as any supplement not being a vitamin/mineral. Twenty-five predefined choices for the use of dietary supplements were given including fish oil, rosehip, ginger, glucosamine, probiotics, and medical cannabinoids (Supplementary file 1).
- Non-medical treatments were defined as an (active) treatment (normally) not being delivered by a medical doctor or another authorized health professional. Sixteen predefined choices for alternative treatments were provided, including: acupuncture, acupressure, cranio-sacral therapy, hypnosis, and kinesiology (Supplementary file 1).
2.6. Use of Conventional Products and Treatments
3. Statistical Analyses
4. Results
4.1. Self-Management Strategies for Knee Pain
4.2. Brief Illness Perception Scores in Users and Non-Users
4.3. Health-Related Outcome Measures
4.4. Cluster Analysis of Brief-IPQ Scores
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Apold, H.; Meyer, H.E.; Nordsletten, L.; Furnes, O.; Baste, V.; Flugsrud, G.B. Risk factors for knee replacement due to primary osteoarthritis, a population based, prospective cohort study of 315,495 individuals. BMC Musculoskelet. Disord. 2014, 15, 217. [Google Scholar] [CrossRef] [PubMed]
- Järvholm, B.; Lewold, S.; Malchau, H.; Vingård, E. Age, bodyweight, smoking habits and the risk of severe osteoarthritis in the hip and knee in men. Eur. J. Epidemiol. 2005, 20, 537–542. [Google Scholar] [CrossRef]
- Sharma, L.; Kapoor, D.; Issa, S. Epidemiology of osteoarthritis: An update. Curr. Opin. Rheumatol. 2006, 18, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Neogi, T.; Felson, D.; Niu, J.; Nevitt, M.; Lewis, C.E.; Aliabadi, P.; Sack, B.; Torner, J.; Bradley, L.; Zhang, Y. Association between radiographic features of knee osteoarthritis and pain: Results from two cohort studies. BMJ 2009, 339. [Google Scholar] [CrossRef] [PubMed]
- Peat, G.; Thomas, E.; Duncan, R.; Wood, L. Is a “false-positive” clinical diagnosis of knee osteoarthritis just the early diagnosis of pre–radiographic disease? Arthritis Care Res. 2010, 62, 1502–1506. [Google Scholar] [CrossRef] [PubMed]
- Thorstensson, C.; Andersson, M.; Jönsson, H.; Saxne, T.; Petersson, I. Natural course of knee osteoarthritis in middle-aged subjects with knee pain: 12-year follow-up using clinical and radiographic criteria. Ann. Rheum. Dis. 2009, 68, 1890–1893. [Google Scholar] [CrossRef]
- Peat, G.; McCarney, R.; Croft, P. Knee pain and osteoarthritis in older adults: A review of community burden and current use of primary health care. Ann. Rheum. Dis. 2001, 60, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Brazier, J.E.; Kearns, B.; Relton, C.; Smith, C.; Cooper, C.L. Examining the impact of 11 long-standing health conditions on health-related quality of life using the EQ-5D in a general population sample. Eur. J. Health Econ. 2015, 16, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Lau-Walker, M. Predicting self-efficacy using illness perception components: A patient survey. Br. J. Health Psychol. 2006, 11, 643–661. [Google Scholar] [CrossRef] [PubMed]
- Kaptein, A.A.; Bijsterbosch, J.; Scharloo, M.; Hampson, S.E.; Kroon, H.M.; Kloppenburg, M. Using the common sense model of illness perceptions to examine osteoarthritis change: A 6-year longitudinal study. Health Psychol. 2010, 29, 56. [Google Scholar] [CrossRef]
- Goodwin, J.S.; Black, S.A.; Satish, S. Aging versus disease: The opinions of older black, Hispanic, and non-Hispanic white Americans about the causes and treatment of common medical conditions. J. Am. Geriatr. Soc. 1999, 47, 973–979. [Google Scholar] [CrossRef]
- Frostholm, L.; Fink, P.; Christensen, K.S.; Toft, T.; Oernboel, E.; Olesen, F.; Weinman, J. The patients’ illness perceptions and the use of primary health care. Psychosom. Med. 2005, 67, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Arden, N.K.; Perry, T.A.; Bannuru, R.R.; Bruyère, O.; Cooper, C.; Haugen, I.K.; Hochberg, M.C.; McAlindon, T.E.; Mobasheri, A.; Reginster, J.-Y. Non-surgical management of knee osteoarthritis: Comparison of ESCEO and OARSI 2019 guidelines. Nat. Rev. Rheumatol. 2020, 1–8. [Google Scholar] [CrossRef]
- Zhang, W.; Moskowitz, R.; Nuki, G.; Abramson, S.; Altman, R.; Arden, N.; Bierma-Zeinstra, S.; Brandt, K.; Croft, P.; Doherty, M. OARSI recommendations for the management of hip and knee osteoarthritis, part I: Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthr. Cartil. 2007, 15, 981–1000. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Moskowitz, R.; Nuki, G.; Abramson, S.; Altman, R.; Arden, N.; Bierma-Zeinstra, S.; Brandt, K.; Croft, P.; Doherty, M. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthr. Cartil. 2008, 16, 137–162. [Google Scholar] [CrossRef]
- Zhang, W.; Nuki, G.; Moskowitz, R.; Abramson, S.; Altman, R.; Arden, N.; Bierma-Zeinstra, S.; Brandt, K.; Croft, P.; Doherty, M. OARSI recommendations for the management of hip and knee osteoarthritis: Part III: Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthr. Cartil. 2010, 18, 476–499. [Google Scholar] [CrossRef]
- Farha, N.; Spencer, A.; McGervey, M. Outpatient Evaluation of Knee Pain. Med Clin. N. Am. 2020, 105, 117–136. [Google Scholar] [CrossRef] [PubMed]
- Dantas, L.O.; De Fátima Salvini, T.; McAlindon, T.E. Knee osteoarthritis: Key treatments and implications for physical therapy. Braz. J. Phys. Ther. 2020. [Google Scholar] [CrossRef] [PubMed]
- Inam, S.H.A.; Riaz, B.; Jamil, H.; Rafique, D.; Siddiqi, U.A.; Iqbal, M.; Sherwani, N.Z.F.; Khan, W. Do Patients with Osteoarthritis get Weight Loss Counseling? Cureus 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Mbizo, J.; Okafor, A.; Sutton, M.A.; Burkhart, E.N.; Stone, L.M. Complementary and Alternative Medicine Use by Normal Weight, Overweight, and Obese Patients with Arthritis or Other Musculoskeletal Diseases. J. Altern. Complement. Med. 2016, 22, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Al-Windi, A. Predictors of herbal medicine use in a Swedish health practice. Pharmacoepidemiol. Drug Saf. 2004, 13, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Biltoft-Jensen, A.A.; Hindborg, H.; Christensen, T.; Saxholt, E.; Bredsdorff, L.; Knudsen, V.K.; Ravn-Haren, G.; Fødevareinstituttet, D. De fleste får nok vitaminer og mineraler fra kosten alene. E-artikel DTU Fødevareinstitutet 2016, 2016, 1–13. [Google Scholar]
- Clarke, T.C.; Black, L.I.; Stussman, B.J.; Barnes, P.M.; Nahin, R.L. Trends in the use of complementary health approaches among adults: United States, 2002–2012. Natl. Health Stat. Rep. 2015, 79, 1. [Google Scholar]
- Djuv, A.; Nilsen, O.G.; Steinsbekk, A. The co-use of conventional drugs and herbs among patients in Norwegian general practice: A cross-sectional study. BMC Complement. and Altern. Med. 2013, 13, 295. [Google Scholar] [CrossRef]
- Phang, J.K.; Kwan, Y.H.; Hendra, G.; Tan, V.I.C.; Thumboo, J.; Østbye, T.; Fong, W. Complementary and alternative medicine for rheumatic diseases: A systematic review of randomized controlled trials. Complement. Ther. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Schnabel, K.; Binting, S.; Witt, C.M.; Teut, M. Use of complementary and alternative medicine by older adults—A cross-sectional survey. BMC Geriatr. 2014, 14, 38. [Google Scholar] [CrossRef]
- Ginnerup-Nielsen, E.M.; Henriksen, M.; Christensen, R.; Heitmann, B.L.; Altman, R.; March, L.; Woolf, A.; Karlsen, H.; Bliddal, H. Prevalence of self-reported knee symptoms and management strategies among elderly individuals from Frederiksberg municipality: Protocol for a prospective and pragmatic Danish cohort study. BMJ Open 2019, 9, e028087. [Google Scholar] [CrossRef]
- Vandenbroucke, J.P.; Von Elm, E.; Altman, D.G.; Gotzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M.; Initiative, S. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Ann. Intern. Med. 2007, 147, W163–W194. [Google Scholar] [CrossRef]
- e-Boks. Available online: https://www.e-boks.com/danmark/en (accessed on 15 December 2020).
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Kiadaliri, A.A.; Lohmander, L.S.; Moradi-Lakeh, M.; Petersson, I.F.; Englund, M. High and rising burden of hip and knee osteoarthritis in the Nordic region, 1990–2015: Findings from the Global Burden of Disease Study 2015. Acta Orthop. 2018, 89, 177–183. [Google Scholar] [CrossRef]
- Moss-Morris, R.; Weinman, J.; Petrie, K.; Horne, R.; Cameron, L.; Buick, D. The revised illness perception questionnaire (IPQ-R). Psychol. Health 2002, 17, 1–16. [Google Scholar] [CrossRef]
- Broadbent, E.; Petrie, K.J.; Main, J.; Weinman, J. The Brief Illness Perception Questionnaire. Available online: http://ipq.h.uib.no//pdf/B-IPQ-Danish.pdf (accessed on 25 May 2020).
- Broadbent, E.; Petrie, K.J.; Main, J.; Weinman, J. The brief illness perception questionnaire. J Psychosom. Res. 2006, 60, 631–637. [Google Scholar] [CrossRef]
- Broadbent, E.; Wilkes, C.; Koschwanez, H.; Weinman, J.; Norton, S.; Petrie, K.J. A systematic review and meta-analysis of the Brief Illness Perception Questionnaire. Psychol. Health 2015, 30, 1361–1385. [Google Scholar] [CrossRef]
- Costa, E.C.; Vale, S.; Sobral, M.; Graca Pereira, M. Illness perceptions are the main predictors of depression and anxiety symptoms in patients with chronic pain. Psychol. Health Med. 2016, 21, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.; Dziedzic, K.; Thomas, E.; Baker, S.; Croft, P. The illness perceptions associated with health and behavioural outcomes in people with musculoskeletal hand problems: Findings from the North Staffordshire Osteoarthritis Project (NorStOP). Rheumatology 2007. [Google Scholar] [CrossRef] [PubMed]
- Wilski, M.; Tasiemski, T. Illness perception, treatment beliefs, self-esteem, and self-efficacy as correlates of self-management in multiple sclerosis. Acta Neurol. Scand. 2016, 133, 338–345. [Google Scholar] [CrossRef]
- Weinman, J.; Petrie, K.J.; Moss-Morris, R.; Horne, R. The illness perception questionnaire: A new method for assessing the cognitive representation of illness. Psychol. Health 1996, 11, 431–445. [Google Scholar] [CrossRef]
- Roos, E.M.; Roos, H.P.; Lohmander, L.S.; Ekdahl, C.; Beynnon, B.D. Knee Injury and Osteoarthritis Outcome Score (KOOS)—Development of a self-administered outcome measure. J. Orthop. Sports Phys. Ther. 1998, 28, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Roos, E.M.; Toksvig-Larsen, S. Knee injury and Osteoarthritis Outcome Score (KOOS)–Validation and comparison to the WOMAC in total knee replacement. Health Qual. Life Outcomes 2003, 1, 17. [Google Scholar] [CrossRef] [PubMed]
- Collins, N.; Prinsen, C.; Christensen, R.; Bartels, E.; Terwee, C.; Roos, E. Knee Injury and Osteoarthritis Outcome Score (KOOS): Systematic review and meta-analysis of measurement properties. Osteoarthr. Cartil. 2016, 24, 1317–1329. [Google Scholar] [CrossRef] [PubMed]
- Monticone, M.; Ferrante, S.; Salvaderi, S.; Motta, L.; Cerri, C. Responsiveness and minimal important changes for the Knee Injury and Osteoarthritis Outcome Score in subjects undergoing rehabilitation after total knee arthroplasty. Am. J. Phys. Med. Rehabil. 2013, 92, 864–870. [Google Scholar] [CrossRef]
- National Institute for Health and Clinical Excellence. Osteoarthritis: National Clinical Guideline for Care and Management in Adults; National Institute for Health and Clinical Excellence: London, UK, 2014. [Google Scholar]
- Rabin, R.; Charro, F.D. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Lauritsen, J. Danske Normtal for Euroqol-5d. Available online: http://uag.dk/simpelfunktion/pdf/eq5dknorm.pdf (accessed on 21 September 2020).
- Wittrup-Jensen, K.U.; Lauridsen, J.; Gudex, C.; Pedersen, K.M. Generation of a Danish TTO value set for EQ-5D health states. Scand. J. Public Health 2009, 37, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Rubin, D.B. Multiple Imputation for Nonresponse in Surveys; John Wiley & Sons: Hoboken, NJ, USA, 2004; Chapter 1; Volume 81. [Google Scholar]
- Bland, J.M.; Altman, D.G. Statistics notes: Bootstrap resampling methods. BMJ 2015, 350. [Google Scholar] [CrossRef] [PubMed]
- Frostholm, L.; Hornemann, C.; Ørnbøl, E.; Fink, P.; Mehlsen, M. Using Illness Perceptions to Cluster Chronic Pain Patients. Clin. J. Pain 2018, 34, 991–999. [Google Scholar] [CrossRef]
- Clatworthy, J.; Hankins, M.; Buick, D.; Weinman, J.; Horne, R. Cluster analysis in illness perception research: A Monte Carlo study to identify the most appropriate method. Psychol. Health 2007, 22, 123–142. [Google Scholar] [CrossRef]
- Postler, A.; Ramos, A.L.; Goronzy, J.; Günther, K.-P.; Lange, T.; Schmitt, J.; Zink, A.; Hoffmann, F. Prevalence and treatment of hip and knee osteoarthritis in people aged 60 years or older in Germany: An analysis based on health insurance claims data. Clin. Interv. Aging 2018, 13, 2339. [Google Scholar] [CrossRef]
- Henriksen, M.; Christensen, R.; Klokker, L.; Bartholdy, C.; Bandak, E.; Ellegaard, K.; Boesen, M.P.; Riis, R.G.C.; Bartels, E.M.; Bliddal, H. Evaluation of the benefit of corticosteroid injection before exercise therapy in patients with osteoarthritis of the knee: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 923–930. [Google Scholar] [CrossRef]
- Hayati, M.; Yazdi, Z.; Abbasi, M. Comparison of non-steroidal anti-inflammatory drugs and knee kinesio taping in early osteoarthritis pain: A randomized controlled trial. J. Bodyw. Mov. Ther. 2019, 23, 666–670. [Google Scholar] [CrossRef]
- Marot, V.; Murgier, J.; Carrozzo, A.; Reina, N.; Monaco, E.; Chiron, P.; Berard, E.; Cavaignac, E. Determination of normal KOOS and WOMAC values in a healthy population. Knee Surg. Sports Traumatol. Arthrosc. 2019, 27, 541–548. [Google Scholar] [CrossRef]
- Sørensen, J.; Davidsen, M.; Gudex, C.; Pedersen, K.M.; Brønnum-Hansen, H. Danish EQ-5D population norms. Scand. J. Public Health 2009, 37, 467–474. [Google Scholar] [CrossRef]
- Sawyer, A.T.; Harris, S.L.; Koenig, H.G. Illness perception and high readmission health outcomes. Health Psychol. Open 2019, 6, 2055102919844504. [Google Scholar] [CrossRef]
- Raftery, M.N.; Sarma, K.; Murphy, A.W.; De la Harpe, D.; Normand, C.; McGuire, B.E. Chronic pain in the Republic of Ireland—community prevalence, psychosocial profile and predictors of pain-related disability: Results from the Prevalence, Impact and Cost of Chronic Pain (PRIME) study, part 1. Pain 2011, 152, 1096–1103. [Google Scholar] [CrossRef]
- Bedson, J.; Mottram, S.; Thomas, E.; Peat, G. Knee pain and osteoarthritis in the general population: What influences patients to consult? Fam. Pract. 2007, 24, 443–453. [Google Scholar] [CrossRef]
- Capewell, S.; Dowrick, C. Healthful diet and physical activity for cardiovascular disease prevention in adults without known risk factors: Is behavioral counselling necessary? JAMA Intern. Med. 2017, 177, 1254–1255. [Google Scholar] [CrossRef]
- Rose, G. Sick individuals and sick populations. Int. J. Epidemiol. 2001, 30, 427–432. [Google Scholar] [CrossRef]
- Sacks, G.; Swinburn, B.; Lawrence, M. Obesity Policy Action framework and analysis grids for a comprehensive policy approach to reducing obesity. Obes. Rev. 2009, 10, 76–86. [Google Scholar] [CrossRef]
- Lowe, R.; Porter, A.; Snooks, H.; Button, L.; Evans, B.A. The association between illness representation profiles and use of unscheduled urgent and emergency health care services. Br. J. Health Psychol. 2011, 16, 862–879. [Google Scholar] [CrossRef] [PubMed]
- Rivera, E.; Corte, C.; Steffen, A.; DeVon, H.A.; Collins, E.G.; McCabe, P.J. Illness representation and self-care ability in older adults with chronic disease. Geriatrics 2018, 3, 45. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Dean, S.; Hay-Smith, E.J.C.; Taylor, W.; Baxter, G.D. Musculoskeletal pain and treatment choice: An exploration of illness perceptions and choices of conventional or complementary therapies. Disabil. Rehabil. 2010, 32, 1645–1657. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.W.; Hansen, E.H.; Rasmussen, N.K. Use of natural medicines in the Danish population: A national cross-sectional survey. Ann. Pharmacother. 2005, 39, 1534–1538. [Google Scholar] [CrossRef]

| Users | Non-Users | Difference | p * | |
|---|---|---|---|---|
| Median (95% CI) | ||||
| N (%) | 923 (59.5) | 629 (40.5) | N/A | |
| Demographics | ||||
| Women, N (%) | 612 (66.3) | 381 (60.6) | N/A | 0.021 |
| age years, median (IQR) | 64 (62–67) | 64 (62–67) | N/A | 0.35 |
| BMI (kg/m2), median (IQR) | 26.6 (23.6–30.5) | 24.8 (22.6–27.9) | −1.8 (−2.27 to −1.33) | <0.0001 |
| Knee OA ¶ | 374 (40.5) | 125 (19.9) | N/A | <0.0001 |
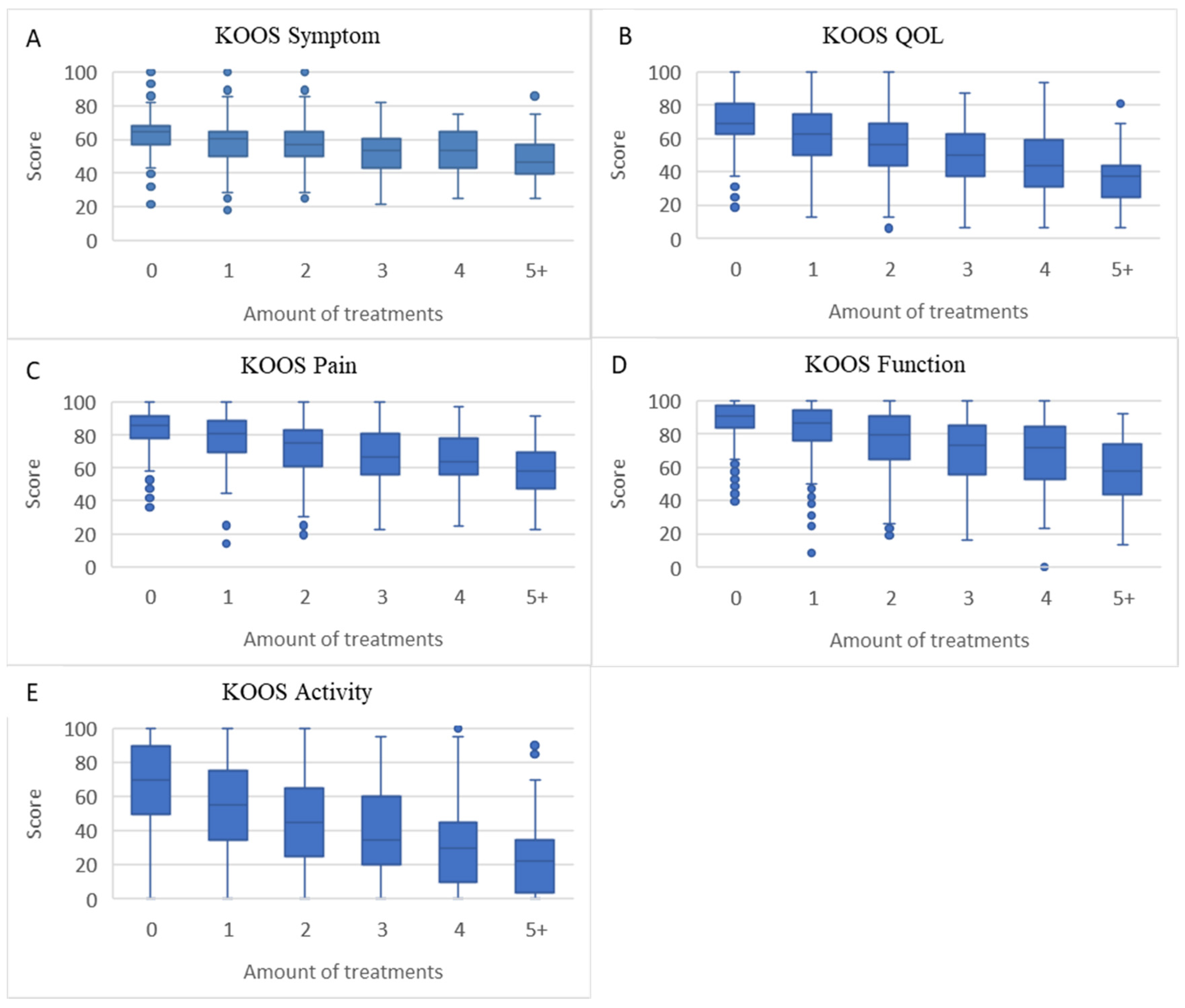
| KOOS. 0–100 score (median, IQR) | ||||
| KOOS symptoms | 57.1 (50.0–64.3) | 64.3 (57.1–67.9) | 7.2 (5.9 to 8.5) | <0.0001 |
| KOOS QOL | 56.2 (43.8–68.8) | 68.8 (62.5–81.2) | 12.6 (11 to 14.2) | <0.0001 |
| KOOS pain | 75.0 (61.1–86.1) | 86.1(77.8–91.7) | 11.1 (9.7 to 12.5) | <0.0001 |
| KOOS function | 80.9 (66.2–91.2) | 91.2 (83.8–97.1) | 10.3 (8.8 to 11.8) | <0.0001 |
| KOOS Sports and recreation | 45 (25–70) | 70.0 (50–90) | 25.0 (21.7 to 28.3) | <0.0001 |
| EQ-5D Index (median, IQR) | 0.776 (0.723–0.824) | 0.824 (0.776–1.000) | 0.048 (0.039 to 0.057) | <0.0001 |
| Current VAS pain, 0–100 mm (median, IQR) | 25 (10.75–50) | 12 (3–25) | −13 (−15 to −11) | <0.0001 |
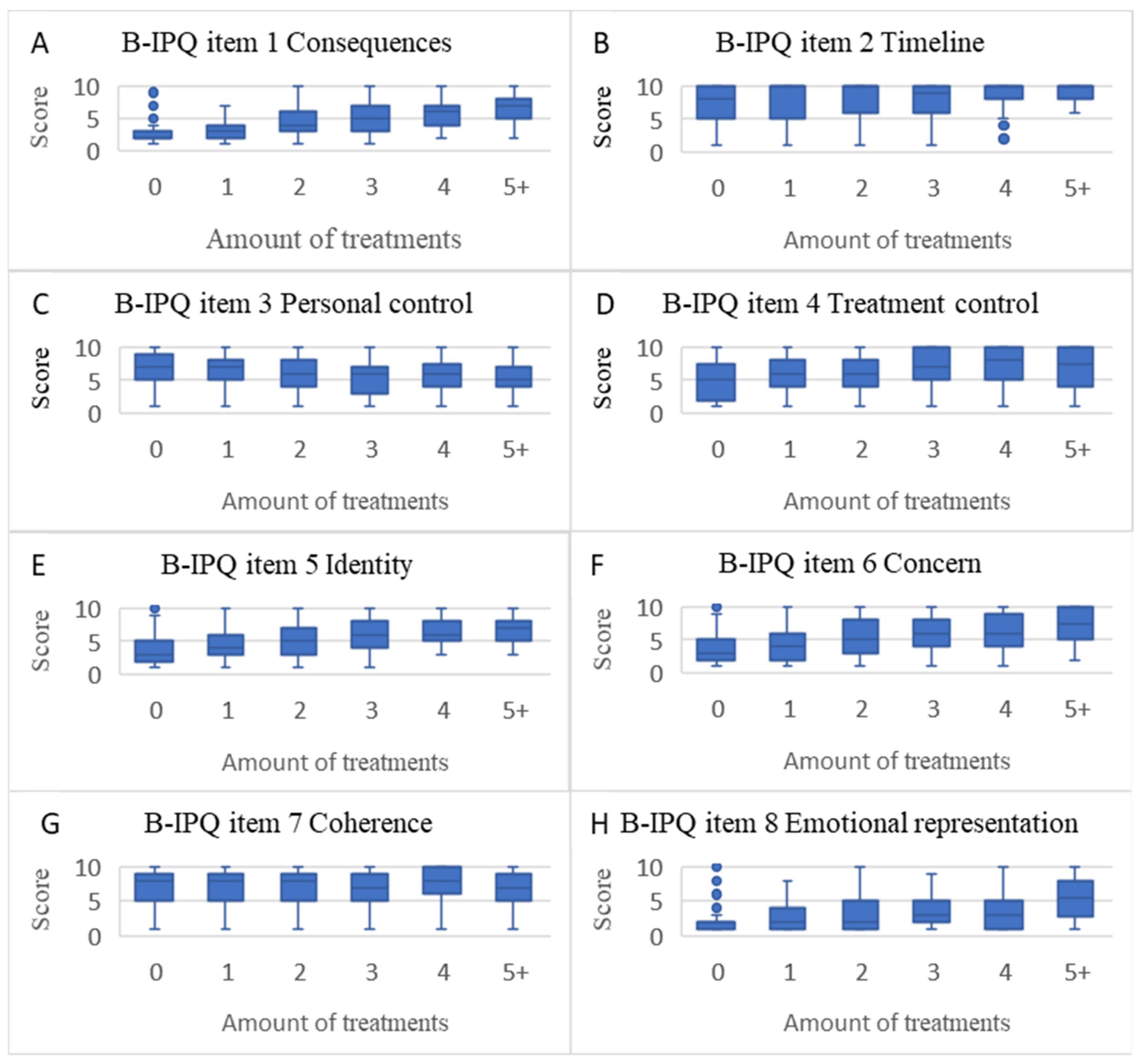
| Brief-IPQ, 1–10 score (median, IQR) | ||||
| consequences B-IPQ 1 | 4.0 (2–6) | 2.0 (2–3) | −2.0 (−2.3 to −1.7) | <0.0001 |
| timeline B-IPQ 2 | 10.0 (6–10) | 8.0 (5–10) | −2.0 (−3.1 to −0.9) | <0.0001 |
| personal control B-IPQ 3 | 6.0 (4–8) | 7.0 (5–9) | 1.0 (0.6 to 1.5) | <0.0001 |
| treatment control B-IPQ 4 | 7.0 (5–9) | 5.0 (2–7.5) | −2.0 (−2.4 to −1.6) | <0.0001 |
| identity B-IPQ 5 | 5.0 (3–7) | 3.0 (2–5) | −2.0 (−2.3 to −1.7) | <0.0001 |
| concern B-IPQ 6 | 5.0 (3–7) | 3.0 (2–5) | −2.0 (−2.4 to −1.6) | <0.0001 |
| coherence B-IPQ 7 | 8.0 (5–9) | 8.0 (5–9) | 0 | 0.275 |
| emotional representation B-IPQ 8 | 2.0 (1–5) | 2.0 (1–2) | 0 | <0.0001 |
| Cluster 1 “Concerned Optimists” (n = 642) | Cluster 2 “Unconcerned Confident” (n = 910) | Difference | ||
|---|---|---|---|---|
| Median (IQR) | Median (IQR) | Median (95% CI) | p * | |
| Demographics | ||||
| Women (N, %) | 416 (64.8) | 577 (63.4) | N/A | 0.57 |
| Age (median, IQR) | 64 (62–67) | 64 (62–67) | N/A | 0.476 |
| BMI (median, IQR) | 26.8 (23.8–30.7) | 25.2 (22.8–28.36) | 1.6 (1.1 to 2.1) | <0.0001 |
| Knee OA ¶ | 321 (50) | 178 (19.6) | N/A | <0.0001 |
| KOOS. 0–100 score (median, IQR) | ||||
| KOOS Symptoms | 53.6 (46.4–60.7) | 64.3 (57.1–67.9) | −10.7 (−12.1 to −9.3) | <0.0001 |
| KOOS Qol | 50 (37.5–56.2) | 75 (62.5–81.2) | −25 (−25.7 to −24.3) | <0.0001 |
| KOOS Pain | 66.7 (55.6–77.8) | 86.1 (80.6–91.7) | −19.4 (−20.0 to −18.8) | <0.0001 |
| KOOS Function | 72.1 (57.4–83,8) | 92.6 (85.30–97.1) | −20.5 (−21.3 to −19.8) | <0.0001 |
| KOOS Sports and recreation | 35 (20–51.25) | 70 (55–85) | −35 (−38 to −32) | <0.0001 |
| EQ5D Index, median (IQR) | 0.756 (0.723–0.824) | 0.824 (0.818–1.000) | −0.068 (−0.074 to −0.062) | <0.0001 |
| Current VAS Pain, median (IQR) | 40 (20–62) | 11 (3–24) | 29 (26.4 to 31.6) | <0.0001 |
| User types median (IQR) | ||||
| Non-users (N, %) | 161 (25.1) | 468 (51.4) | N/A | <0.0001 |
| (only) CAM users | 106 (10.6) | 167 (18.4) | N/A | <0.0001 |
| (only) Pharmacological treatment users # | 28 (4.4) | 10 (1.1) | N/A | |
| (only)Non-pharmacological treatment users ¤ | 90 (14) | 131 (14.4) | N/A | |
| Two or more treatment types § | 257 (40) | 134 (14.7) | N/A | |
| Brief-IPQ, 1–10 score (median, IQR) | ||||
| consequences B-IPQ 1 | 5 (4–7) | 2 (2–3) | 3 (2.9 to 3.1) | <0.0001 |
| timeline B-IPQ 2 | 10 (8–10) | 8 (4–10) | 2 (1.7 to 2.3) | <0.0001 |
| personal control B-IPQ 3 | 5 (3–7) | 8 (5–9) | −3 (−3.4 to −2.6) | <0.0001 |
| treatment control B-IPQ 4 | 8 (6–10) | 5 (2–7) | 3 (2.7 to 3.3) | <0.0001 |
| identity B-IPQ 5 | 6 (5–7) | 3 (2–4) | 3 (2.9 to 3.1) | <0.0001 |
| concern B-IPQ 6 | 7 (5–8) | 2 (2–3) | 5 (4.7 to 5.3) | <0.0001 |
| coherence B-IPQ 7 | 7 (5–9) | 8 (5–9) | −1 (−2.8 to 0.8) | 0.275 |
| emotional representation B-IPQ 8 | 4 (2–6) | 1 (1–2) | 3 (2.9 to 3.1) | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ginnerup-Nielsen, E.; Christensen, R.; Heitmann, B.L.; Altman, R.D.; March, L.; Woolf, A.; Bliddal, H.; Henriksen, M. Estimating the Prevalence of Knee Pain and the Association between Illness Perception Profiles and Self-Management Strategies in the Frederiksberg Cohort of Elderly Individuals with Knee Pain: A Cross-Sectional Study. J. Clin. Med. 2021, 10, 668. https://doi.org/10.3390/jcm10040668
Ginnerup-Nielsen E, Christensen R, Heitmann BL, Altman RD, March L, Woolf A, Bliddal H, Henriksen M. Estimating the Prevalence of Knee Pain and the Association between Illness Perception Profiles and Self-Management Strategies in the Frederiksberg Cohort of Elderly Individuals with Knee Pain: A Cross-Sectional Study. Journal of Clinical Medicine. 2021; 10(4):668. https://doi.org/10.3390/jcm10040668
Chicago/Turabian StyleGinnerup-Nielsen, Elisabeth, Robin Christensen, Berit L Heitmann, Roy D. Altman, Lyn March, Anthony Woolf, Henning Bliddal, and Marius Henriksen. 2021. "Estimating the Prevalence of Knee Pain and the Association between Illness Perception Profiles and Self-Management Strategies in the Frederiksberg Cohort of Elderly Individuals with Knee Pain: A Cross-Sectional Study" Journal of Clinical Medicine 10, no. 4: 668. https://doi.org/10.3390/jcm10040668
APA StyleGinnerup-Nielsen, E., Christensen, R., Heitmann, B. L., Altman, R. D., March, L., Woolf, A., Bliddal, H., & Henriksen, M. (2021). Estimating the Prevalence of Knee Pain and the Association between Illness Perception Profiles and Self-Management Strategies in the Frederiksberg Cohort of Elderly Individuals with Knee Pain: A Cross-Sectional Study. Journal of Clinical Medicine, 10(4), 668. https://doi.org/10.3390/jcm10040668






