Evolved Resistance to Placental Invasion Secondarily Confers Increased Survival in Melanoma Patients
Abstract
:1. Introduction
2. Results
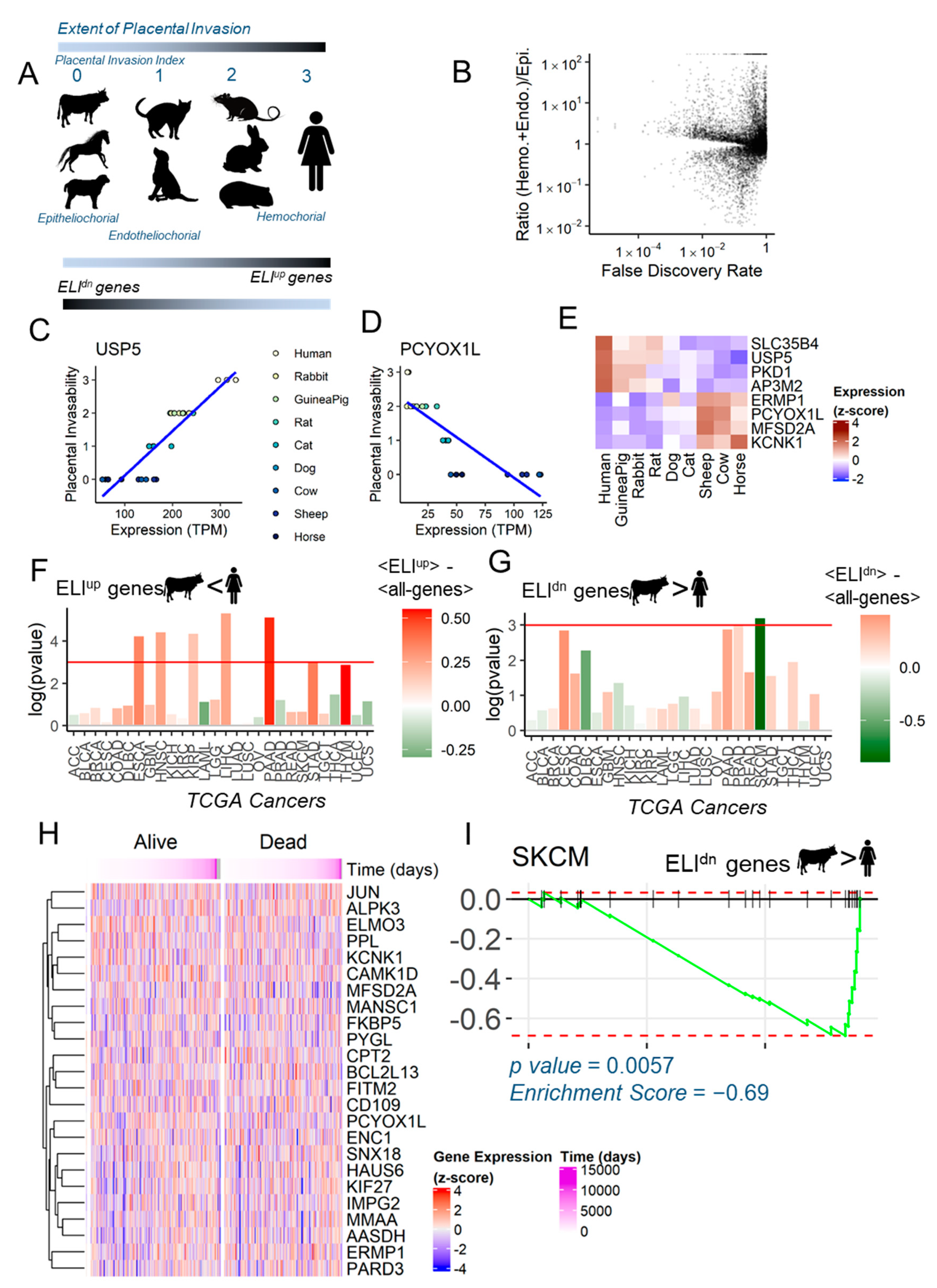
2.1. Endometrial Genes Correlated with Placental Invasion Among Mammals
2.2. Anti-Invasive ELIdn Genes Are Negatively Enriched in Human Skin Cancers
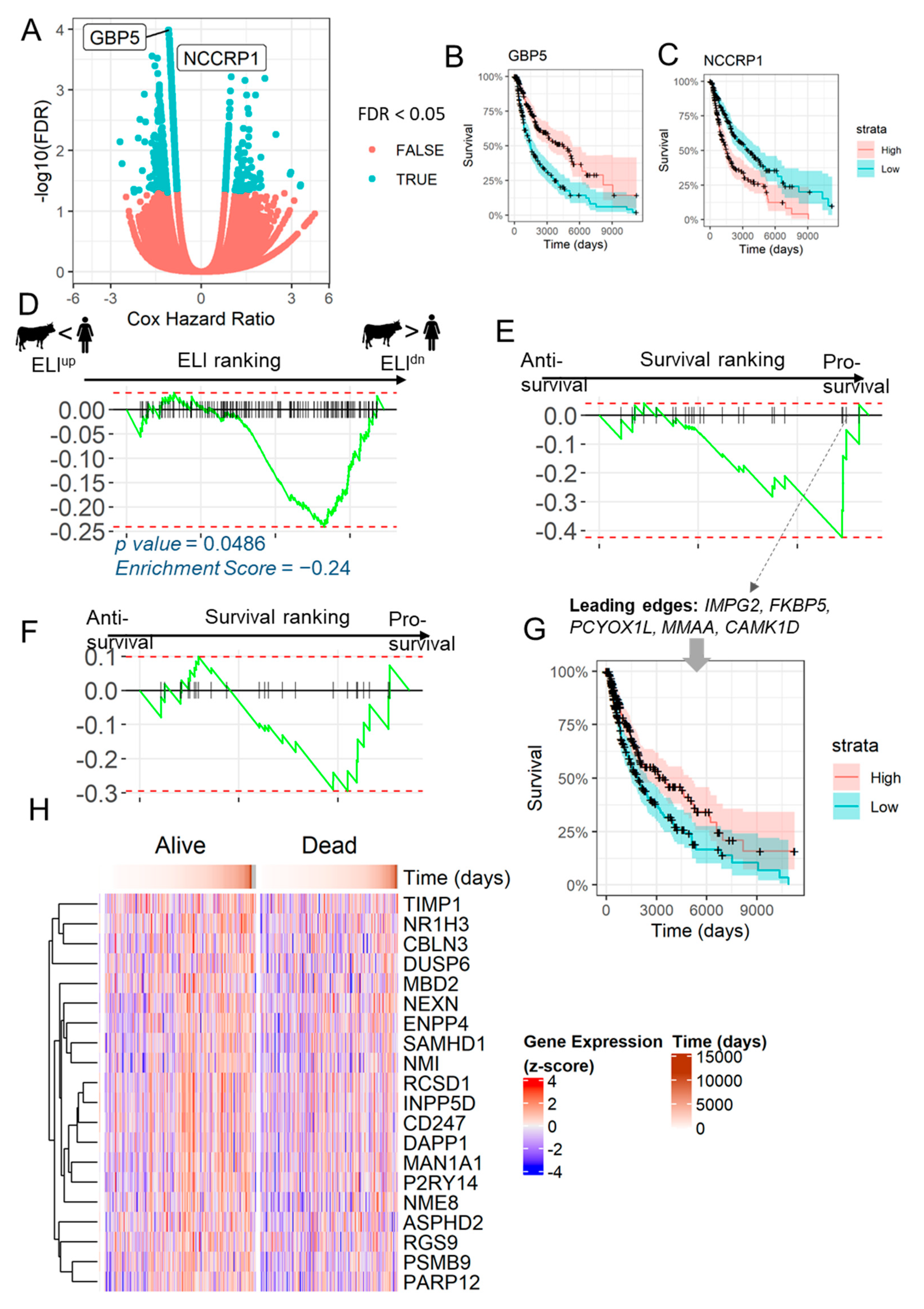
2.3. Anti-Invasive ELIdn Genes Are Associated with Increased Survival in Human Melanoma Patients
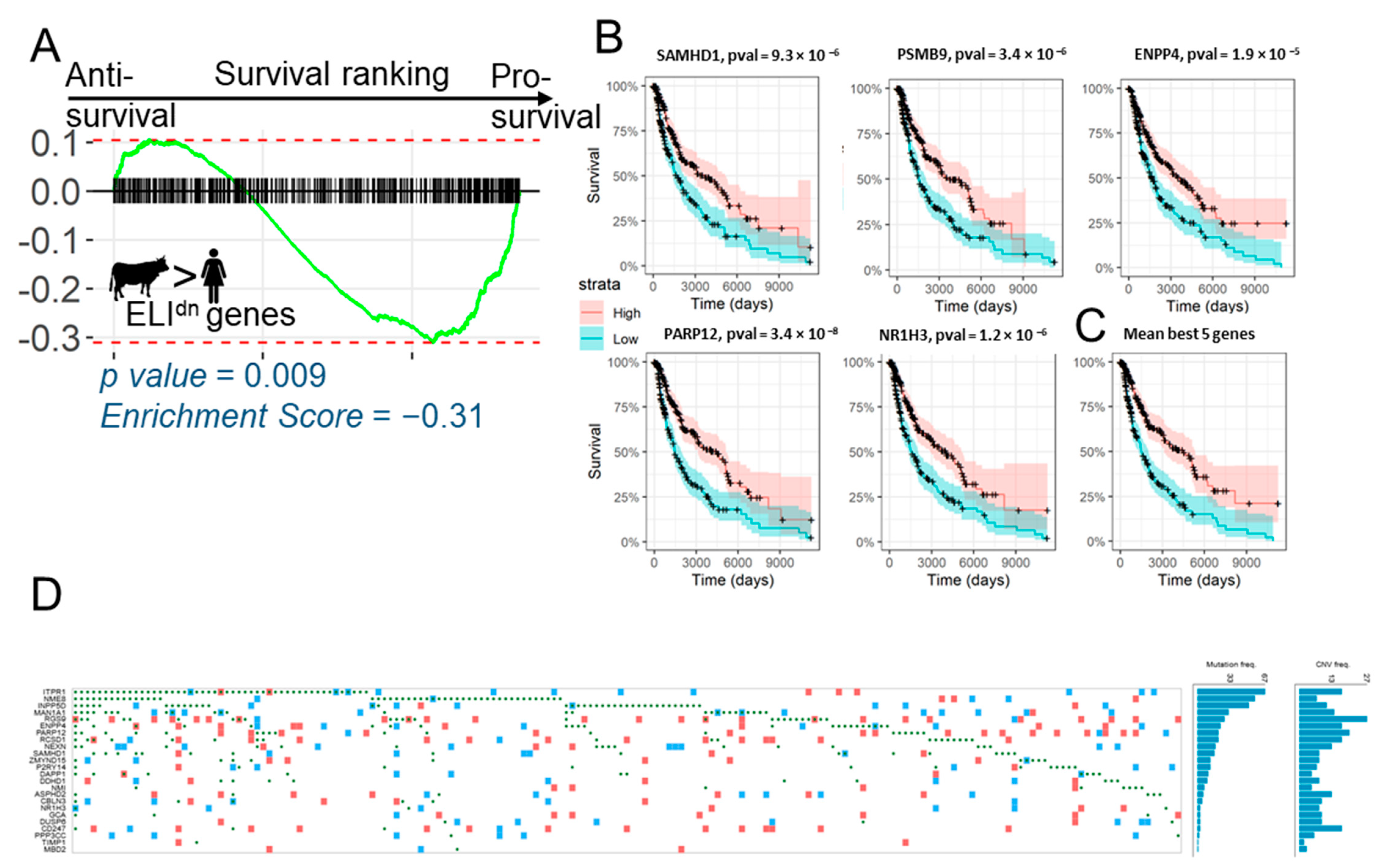
2.4. Loss of Anti-Invasive ELIdn Genes is Associated with Decreased Survival in Melanoma Patients
3. Methods
3.1. Data Sources
3.2. Statistical Analysis
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- D’Souza, A.W.; Wagner, G.P. Malignant cancer and invasive placentation: A case for positive pleiotropy between endometrial and malignancy phenotypes. Evol. Med. Public Health 2014, 2014, 136–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, G.P.; Kshitiz; Levchenko, A. Comments on Boddy et al., 2020: Available data suggests positive relationship between placental invasion and malignancy. Evol. Med. Public Health 2020, 2020, 211–214. [Google Scholar] [CrossRef]
- Boddy, A.M.; Abegglen, L.M.; Pessier, A.P.; Aktipis, A.; Schiffman, J.D.; Maley, C.C.; Witte, C. Lifetime cancer prevalence and life history traits in mammals. Evol. Med. Public Health 2020, 2020, 187–195. [Google Scholar] [CrossRef]
- Priester, W.A.; Mantel, N. Occurrence of tumors in domestic animals. Data from 12 United States and Canadian colleges of veterinary medicine. J. Natl. Cancer Inst. 1971, 47, 1333–1344. [Google Scholar]
- Seluanov, A.; Gladyshev, V.N.; Vijg, J.; Gorbunova, V. Mechanisms of cancer resistance in long-lived mammals. Nat. Rev. Cancer 2018, 18, 433–441. [Google Scholar] [CrossRef]
- Afzal, J.; Maziarz, J.D.; Hamidzadeh, A.; Liang, C.; Erkenbrack, E.M.; Kim, H.N.; Haeger, J.D.; Pfarrer, C.; Hoang, T.; Ott, T.; et al. Evolution of placental invasion and cancer metastasis are causally linked. Nat. Ecol. Evol. 2019, 3, 1743–1753. [Google Scholar] [CrossRef]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef] [Green Version]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Suhail, Y.; Cain, M.P.; Vanaja, K.; Kurywchak, P.A.; Levchenko, A.; Kalluri, R.; Kshitiz. Systems Biology of Cancer Metastasis. Cell Syst. 2019, 9, 109–127. [Google Scholar] [CrossRef] [Green Version]
- Hutchenreuther, J.; Leask, A. Why target the tumor stroma in melanoma? J. Cell Commun. Signal. 2018, 12, 113–118. [Google Scholar] [CrossRef] [Green Version]
- Shelley, A.J.; Doubova, M.; Langley, A.R.; Beecker, J. Timing of Clinical Detection and Biopsy in Invasive Melanoma and Breslow Depth: A Chart Review. J. Cutan. Med. Surg. 2019, 23, 229–230. [Google Scholar] [CrossRef] [PubMed]
- Laing, R.W.; Carter, N. Measurement of Breslow depth. J. Pathol. 1992, 166, 420. [Google Scholar] [PubMed]
- Li, X.; Ma, Q.; Xu, Q.; Duan, W.; Lei, J.; Wu, E. Targeting the cancer-stroma interaction: A potential approach for pancreatic cancer treatment. Curr. Pharm. Des. 2012, 18, 2404–2415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siletz, A.; Kniazeva, E.; Jeruss, J.S.; Shea, L.D. Transcription factor networks in invasion-promoting breast carcinoma-associated fibroblasts. Cancer Microenviron. 2013, 6, 91–107. [Google Scholar] [CrossRef] [Green Version]
- Houthuijzen, J.M.; Jonkers, J. Cancer-associated fibroblasts as key regulators of the breast cancer tumor microenvironment. Cancer Metastasis Rev. 2018, 37, 577–597. [Google Scholar] [CrossRef]
- Hu, G.; Xu, F.; Zhong, K.; Wang, S.; Huang, L.; Chen, W. Activated Tumor-infiltrating Fibroblasts Predict Worse Prognosis in Breast Cancer Patients. J. Cancer 2018, 9, 3736–3742. [Google Scholar] [CrossRef] [Green Version]
- Suhail, Y.; Maziarz, J.D.; Dighe, A.; Wagner, G.P.; Kshitiz. Cis-Regulatory Differences Explaining Evolved Levels of Endometrial Invasibility in Eutherian Mammals. bioRxiv 2020. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research; Weinstein, J.N.; Collisson, E.A.; Mills, G.B.; Shaw, K.R.; Ozenberger, B.A.; Ellrott, K.; Shmulevich, I.; Sander, C.; Stuart, J.M. The Cancer Genome Atlas Pan-Cancer analysis project. Nat. Genet. 2013, 45, 1113–1120. [Google Scholar] [CrossRef]
- Consortium, G.T. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef]
- Ma, X.; Dighe, A.; Maziarz, J.D.; Newmann, E.; Erkenbrack, E.M.; Hei, Y.Y.; Liu, Y.; Suhail, Y.; Kshitiz; Pak, I.; et al. Human vulnerability to cancer malignancy is enhanced by evolution of higher mesenchymal CD44 expression compared to other mammals. bioRxiv 2020. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Xu, H.; Wang, Q.; Fu, P.; Huang, T.; Anas, O.; Zhao, H.; Xiong, N. Pard3 suppresses glioma invasion by regulating RhoA through atypical protein kinase C/NF-kappaB signaling. Cancer Med. 2019, 8, 2288–2302. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wan, F.; Sheng, H.; Shi, G.; Shen, Y.; Lin, G.; Dai, B.; Zhu, Y.; Ye, D. NR1H3 Expression is a Prognostic Factor of Overall Survival for Patients with Muscle-Invasive Bladder Cancer. J. Cancer 2017, 8, 852–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, C.; Qiu, Y.; Liu, J.; Feng, H.; Shen, S.; Saiyin, H.; Yu, W.; Wei, Y.; Yu, L.; Su, W.; et al. PARP12 (ARTD12) suppresses hepatocellular carcinoma metastasis through interacting with FHL2 and regulating its stability. Cell Death Dis. 2018, 9, 856. [Google Scholar] [CrossRef] [Green Version]
- Kodigepalli, K.M.; Li, M.; Bonifati, S.; Panfil, A.R.; Green, P.L.; Liu, S.L.; Wu, L. SAMHD1 inhibits epithelial cell transformation in vitro and affects leukemia development in xenograft mice. Cell Cycle 2018, 17, 2564–2576. [Google Scholar] [CrossRef] [Green Version]
- Anderson, A.M.; Kalimutho, M.; Harten, S.; Nanayakkara, D.M.; Khanna, K.K.; Ragan, M.A. The metastasis suppressor RARRES3 as an endogenous inhibitor of the immunoproteasome expression in breast cancer cells. Sci. Rep. 2017, 7, 39873. [Google Scholar] [CrossRef] [Green Version]
- Tang, Z.; Li, C.; Kang, B.; Gao, G.; Li, C.; Zhang, Z. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017, 45, W98–W102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Andersen, P.K.; Gill, R.D. Cox’s regression model for counting processes: A large sample study. Ann. Stat. 1982, 10, 1100–1120. [Google Scholar] [CrossRef]
- Therneau, T.M.; Li, H. Computing the Cox model for case cohort designs. Lifetime Data Anal. 1999, 5, 99–112. [Google Scholar] [CrossRef]
- Therneau, T.M.; Grambsch, P.M. Modeling Survival Data: Extending the Cox Model; Springer: New York, NY, USA, 2000. [Google Scholar]
- Korotkevich, G.; Sukhov, V.; Sergushichev, A. Fast gene set enrichment analysis. bioRxiv 2019. [Google Scholar] [CrossRef] [Green Version]
- Pereira, B.A.; Vennin, C.; Papanicolaou, M.; Chambers, C.R.; Herrmann, D.; Morton, J.P.; Cox, T.R.; Timpson, P. CAF Subpopulations: A New Reservoir of Stromal Targets in Pancreatic Cancer. Trends Cancer 2019, 5, 724–741. [Google Scholar] [CrossRef] [Green Version]
- Suva, M.L.; Tirosh, I. Single-Cell RNA Sequencing in Cancer: Lessons Learned and Emerging Challenges. Mol. Cell 2019, 75, 7–12. [Google Scholar] [CrossRef]
- Ren, X.; Kang, B.; Zhang, Z. Understanding tumor ecosystems by single-cell sequencing: Promises and limitations. Genome Biol. 2018, 19, 211. [Google Scholar] [CrossRef] [PubMed]
- Krtolica, A.; Campisi, J. Integrating epithelial cancer, aging stroma and cellular senescence. Adv. Gerontol. 2003, 11, 109–116. [Google Scholar] [PubMed]
- Ruhland, M.K.; Loza, A.J.; Capietto, A.H.; Luo, X.; Knolhoff, B.L.; Flanagan, K.C.; Belt, B.A.; Alspach, E.; Leahy, K.; Luo, J.; et al. Stromal senescence establishes an immunosuppressive microenvironment that drives tumorigenesis. Nat. Commun. 2016, 7, 11762. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Kohli, J.; Demaria, M. Senescent Cells in Cancer Therapy: Friends or Foes? Trends Cancer 2020, 6, 838–857. [Google Scholar] [CrossRef]
- Pothuraju, R.; Rachagani, S.; Junker, W.M.; Chaudhary, S.; Saraswathi, V.; Kaur, S.; Batra, S.K. Pancreatic cancer associated with obesity and diabetes: An alternative approach for its targeting. J. Exp. Clin. Cancer Res. 2018, 37, 319. [Google Scholar] [CrossRef] [Green Version]
- Filer, A.; Raza, K.; Salmon, M.; Buckley, C.D. Targeting stromal cells in chronic inflammation. Discov. Med. 2007, 7, 20–26. [Google Scholar]
- Hillers, L.E.; D’Amato, J.V.; Chamberlin, T.; Paderta, G.; Arendt, L.M. Obesity-Activated Adipose-Derived Stromal Cells Promote Breast Cancer Growth and Invasion. Neoplasia 2018, 20, 1161–1174. [Google Scholar] [CrossRef]
- Quail, D.F.; Dannenberg, A.J. The obese adipose tissue microenvironment in cancer development and progression. Nat. Rev. Endocrinol. 2019, 15, 139–154. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suhail, Y.; Afzal, J.; Kshitiz. Evolved Resistance to Placental Invasion Secondarily Confers Increased Survival in Melanoma Patients. J. Clin. Med. 2021, 10, 595. https://doi.org/10.3390/jcm10040595
Suhail Y, Afzal J, Kshitiz. Evolved Resistance to Placental Invasion Secondarily Confers Increased Survival in Melanoma Patients. Journal of Clinical Medicine. 2021; 10(4):595. https://doi.org/10.3390/jcm10040595
Chicago/Turabian StyleSuhail, Yasir, Junaid Afzal, and Kshitiz. 2021. "Evolved Resistance to Placental Invasion Secondarily Confers Increased Survival in Melanoma Patients" Journal of Clinical Medicine 10, no. 4: 595. https://doi.org/10.3390/jcm10040595
APA StyleSuhail, Y., Afzal, J., & Kshitiz. (2021). Evolved Resistance to Placental Invasion Secondarily Confers Increased Survival in Melanoma Patients. Journal of Clinical Medicine, 10(4), 595. https://doi.org/10.3390/jcm10040595






