Mid-Regional Proadrenomedullin as a New Biomarker of Kidney and Cardiovascular Diseases—Is It the Future?
Abstract
1. Introduction
2. Adrenomedullin vs. Mid-Regional Proadrenomedullin
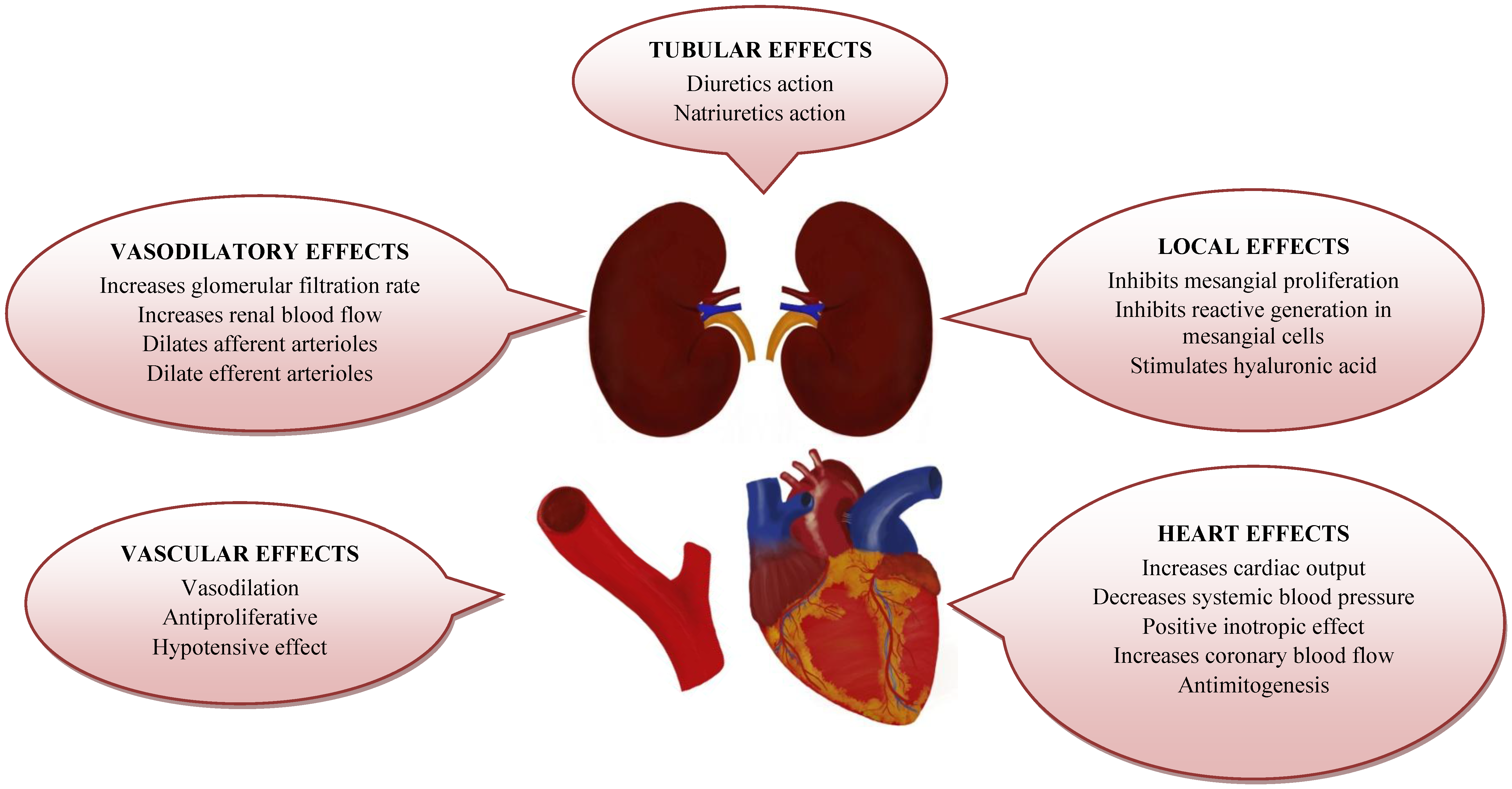
3. Impact of ADM on the Cardiovascular System
3.1. ADM and MR-ProADM in Heart Failure
Cardiorenal Syndrome
3.2. ADM and MR-ProADM in Myocardial Infarction
3.3. Significance of ADM and MR-ProADM in Hypertension
3.4. Possible Value of ADM or MR-ProADM in Other CVD
4. Significance of ADM in Kidney Diseases
4.1. ADM and MR-ProADM in Chronic Kidney Disease
4.2. Significance of MR-ProADM and ADM in Acute Kidney Injury (AKI)
4.3. ADM and MR-ProADM in Glomerulonephritis
4.4. MR-ProADM in End-Stage Kidney Disease (ESKD)
4.5. MR-ProADM after Renal Transplantation
5. NT-ProBNP vs. ADM and MR-ProADM
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global prevalence of chronic kidney disease–A Systematic Review And Meta-Analysis. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- KDOQI. Chronic Kidney Disease: Evaluation, Classification, and Stratification. 2012. Available online: https://www.kidney.org/professionals/guidelines/guidelines_commentaries/chronic-kidney-disease-classification (accessed on 17 June 2019).
- Aggarwal, R.; Petrie, B.; Bala, W.; Chiu, N. Mortality outcomes with intensive blood pressure targets in chronic kidney disease patients. Hypertension 2019, 73, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.; Kangawa, K.; Kawamoto, M.; Ichiki, Y.; Nakamura, S.; Matsuo, H.; Eto, T. Adrenomedullin: A novel hypotensive peptide isolated from human pheochromocytoma. Biochem. Biophys. Res. Commun. 1993, 192, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Hinson, J.P.; Kapas, S.; Smith, D.M. Adrenomedullin, a multifunctional regulatory peptide. Endocr. Rev. 2000, 21, 138–167. [Google Scholar] [CrossRef] [PubMed]
- Meeran, K.; O’Shea, D.; Upton, P.D.; Small, C.J.; Ghatei, M.A.; Byfield, P.H.; Bloom, S.R. Circulating adrenomedullin does not regulate systemic blood pressure but increases plasma prolactin after intravenous infusion in humans: A pharmacokinetic study. J. Clin. Endocrinol. Metab. 1997, 82, 95–100. [Google Scholar] [CrossRef]
- Elke, G.; Bloos, F.; Wilson, D.C.; Brunkhorst, F.M.; Briegel, J.; Reinhart, K.; Loeffler, M.; Kluge, S.; Nierhaus, A.; Jaschinski, U.; et al. The use of mid-regional proadrenomedullin to identify disease severity and treatment response to sepsis–A secondary analysis of a large randomised controlled trial. Crit. Care 2018, 22. [Google Scholar] [CrossRef]
- Kutz, A.; Hausfater, P.; Amin, D.; Amin, A.; Canavaggio, P.; Sauvin, G.; Bernard, M.; Conca, A.; Haubitz, S.; Struja, T.; et al. The triage-proadm score for an early risk stratification of medical patients in the emergency department–Development based on a multi-national, prospective, observational study. PLoS ONE 2016, 11, e0168076. [Google Scholar] [CrossRef]
- Lainchbury, J.G.; Troughton, R.W.; Lewis, L.K.; Yandle, T.G.; Richards, A.M.; Nicholls, M.G. Hemodynamic, hormonal, and renal effects of short-term adrenomedullin infusion in healthy volunteers. J. Clin. Endocrinol. Metab. 2000, 85, 5. [Google Scholar] [CrossRef]
- Hirata, Y.; Hayakawa, H.; Suzuki, Y.; Suzuki, E.; Ikenouchi, H.; Kohmoto, O.; Kimura, K.; Kitamura, K.; Eto, T.; Kangawa, K.; et al. Mechanisms of Adrenomedullin-Induced Vasodilation in the Rat Kidney. Hypertension 1995, 25, 790–795. [Google Scholar] [CrossRef]
- Nishimatsu, H.; Suzuki, E.; Nagata, D.; Moriyama, N.; Satonaka, H.; Walsh, K.; Sata, M.; Kangawa, K.; Matsuo, H.; Goto, A.; et al. Adrenomedullin induces endothelium-dependent vasorelaxation via the phosphatidylinositol 3-kinase/akt-dependent pathway in Rat Aorta. Circ. Res. 2001, 89, 63–70. [Google Scholar] [CrossRef]
- Tsuruda, T.; Kato, J.; Kitamura, K.; Kuwasako, K.; Imamura, T.; Koiwaya, Y.; Tsuji, T.; Kangawa, K.; Eto, T. Adrenomedullin: A possible autocrine or paracrine inhibitor of hypertrophy of cardiomyocytes. Hypertension 1998, 31, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Horio, T.; Nishikimi, T.; Yoshihara, F.; Nagaya, N.; Matsuo, H.; Takishita, S.; Kangawa, K. Production and secretion of adrenomedullin in cultured rat cardiac myocytes and nonmyocytes: Stimulation by interleukin-1β and tumor necrosis factor-α**this work was supported in part by special coordination funds for promoting science and technology (encouragement system of coe) from the science and technology agency of Japan, the ministry of health and welfare, and the human science foundation of Japan. Endocrinology 1998, 139, 4576–4580. [Google Scholar] [CrossRef] [PubMed]
- Lippton, H.; Chang, J.K.; Hao, Q.; Summer, W.; Hyman, A.L. Adrenomedullin dilates the pulmonary vascular bed in vivo. J. Appl. Physiol. 1994, 76, 2154–2156. [Google Scholar] [CrossRef] [PubMed]
- Lainchbury, J.G.; Cooper, G.J.S.; Coy, D.H.; Jiang, N.-Y.; Lewis, L.K.; Yandle, T.G.; Richards, A.M.; Nicholls, M.G. Adrenomedullin: A hypotensive hormone in man. Clin. Sci. 1997, 92, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Parkes, D.G.; May, C.N. Direct cardiac and vascular actions of adrenomedullin in conscious sheep. Br. J. Pharmcol. 1997, 120, 1179–1185. [Google Scholar] [CrossRef]
- Ikenouchi, H.; Kangawa, K.; Matsuo, H.; Hirata, Y. Negative inotropic effect of adrenomedullin in isolated adult rabbit cardiac ventricular myocytes. Circulation 1997, 95, 2318–2324. [Google Scholar] [CrossRef]
- Yoshimoto, R.; Mitsui-Saito, M.; Ozaki, H.; Karaki, H. Effects of adrenomedullin and calcitonin gene-related peptide on contractions of the rat aorta and porcine coronary artery. Br. J. Pharm. 1998, 123, 1645–1654. [Google Scholar] [CrossRef]
- Saita, M.; Shimokawa, A.; Kunitake, T.; Kato, K.; Hanamori, T.; Kitamura, K.; Eto, T.; Kannan, H. Central actions of adrenomedullin on cardiovascular parameters and sympathetic outflow in conscious rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1998, 274, R979–R984. [Google Scholar] [CrossRef]
- Szokodi, I.; Kinnunen, P.; Tavi, P.; Weckström, M.; Tóth, M.; Ruskoaho, H. Evidence for CAMP-Independent mechanisms mediating the effects of adrenomedullin, a new inotropic peptide. Circulation 1998, 97, 1062–1070. [Google Scholar] [CrossRef]
- Ihara, T.; Ikeda, U.; Tate, Y.; Ishibashi, S.; Shimada, K. Positive inotropic effects of adrenomedullin on rat papillary muscle. Eur. J. Pharmacol. 2000, 390, 167–172. [Google Scholar] [CrossRef]
- Saetrum Opgaard, O.; Hasbak, P.; de Vries, R.; Saxena, P.R.; Edvinsson, L. Positive inotropy mediated via cgrp receptors in isolated human myocardial trabeculae. Eur. J. Pharmacol. 2000, 397, 373–382. [Google Scholar] [CrossRef]
- Hirayama, N.; Kitamura, K.; Imamura, T.; Kato, J.; Koiwaya, Y.; Tsuji, T.; Kangawa, K.; Eto, T. Molecular forms of circulating adrenomedullin in patients with congestive heart failure. J. Endocrinol. 1999, 160, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Nishikimi, T.; Saito, Y.; Kitamura, K.; Ishimitsu, T.; Eto, T.; Kangawa, K.; Matsuo, H.; Omae, T.; Matsuoka, H. Increased plasma levels of adrenomedullin in patients with heart failure. J. Am. Coll. Cardiol. 1995, 26, 1424–1431. [Google Scholar] [CrossRef]
- von Haehling, S.; Filippatos, G.S.; Papassotiriou, J.; Cicoira, M.; Jankowska, E.A.; Doehner, W.; Rozentryt, P.; Vassanelli, C.; Struck, J.; Banasiak, W.; et al. Mid-Regional pro-Adrenomedullin as a novel predictor of mortality in patients with chronic heart failure. Eur. J. Heart Fail. 2010, 12, 484–491. [Google Scholar] [CrossRef]
- Nishikimi, T.; Nakagawa, Y. Adrenomedullin as a biomarker of heart failure. Heart Fail. Clin. 2018, 14, 49–55. [Google Scholar] [CrossRef]
- Molvin, J.; Jujic, A.; Bachus, E.; Gallo, W.; Tasevska-Dinevska, G.; Holm, H.; Melander, O.; Fedorowski, A.; Magnusson, M. Cardiovascular biomarkers predict post-discharge re-hospitalization risk and mortality among swedish heart failure patients. ESC Heart Fail. 2019, 6, 992–999. [Google Scholar] [CrossRef]
- Maisel, A.; Mueller, C.; Nowak, R.; Peacock, W.F.; Landsberg, J.W.; Ponikowski, P.; Mockel, M.; Hogan, C.; Wu, A.H.B.; Richards, M.; et al. Mid-Region pro-hormone markers for diagnosis and prognosis in acute dyspnea: Results from the bach (biomarkers in acute heart failure) trial. J. Am. Coll. Cardiol. 2010, 55, 2062–2076. [Google Scholar] [CrossRef]
- Xue, Y.; Taub, P.; Iqbal, N.; Fard, A.; Clopton, P.; Maisel, A. Mid-Region pro-Adrenomedullin adds predictive value to clinical predictors and framingham risk score for long-term mortality in stable outpatients with heart failure. Eur. J. Heart Fail. 2013, 15, 1343–1349. [Google Scholar] [CrossRef]
- Billebeau, G.; Vodovar, N.; Sadoune, M.; Launay, J.-M.; Beauvais, F.; Cohen-Solal, A. Effects of a cardiac rehabilitation programme on plasma cardiac biomarkers in patients with chronic heart failure. Eur J. Prev Cardiol. 2017, 24, 1127–1135. [Google Scholar] [CrossRef]
- Bhandari, S.S.; Davies, J.E.; Struck, J.; Ng, L.L. The midregional portion of proadrenomedullin is an independent predictor of left ventricular mass index in hypertension. Metab. Clin. Exp. 2010, 59, 7–13. [Google Scholar] [CrossRef]
- Pecková, M.; Charvat, J.; Schuck, O.; Zamrazil, V.; Bilek, R.; Hill, M.; Svab, P.; Horackova, M. Plasma adrenomedullin and subclinical cardiorenal syndrome in patients with Type 2 diabetes mellitus. J. Int Med. Res. 2012, 40, 1552–1559. [Google Scholar] [CrossRef] [PubMed]
- Yoshitomi, Y.; Nishikimi, T.; Kojima, S.; Kuramochi, M.; Takishita, S.; Matsuoka, H.; Miyata, A.; Matsuo, H.; Kangawa, K. Plasma levels of adrenomedullin in patients with acute myocardial infarction. Clin. Sci. 1998, 94, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.Q.; O’Brien, R.J.; Struck, J.; Quinn, P.; Morgenthaler, N.; Squire, I.; Davies, J.; Bergmann, A.; Ng, L.L. Prognostic value of midregional pro-adrenomedullin in patients with acute myocardial infarction: The lamp (leicester acute myocardial infarction peptide) study. J. Am. Coll. Cardiol. 2007, 49, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Arrigo, M.; Parenica, J.; Ganovska, E.; Pavlusova, M.; Mebazaa, A. Plasma bio-adrenomedullin is a marker of acute heart failure severity in patients with acute coronary syndrome. Int. J. Cardiol. Heart Vasc. 2019, 22, 174–176. [Google Scholar] [CrossRef] [PubMed]
- Walter, T.; Brueckmann, M.; Lang, S.; Sauer, T.; Fiedler, E.; Papassotiriou, J.; Behnes, M.; Elmas, E.; Borggrefe, M.; Bertsch, T. Comparison of long-term prognostic value of n-terminal-probnp and midregional-pro-adrenomedullin in patients with acute myocardial infarction. Clin. Lab. 2010, 56, 303–309. [Google Scholar] [PubMed]
- Nagata, S.; Yamasaki, M.; Kawano, A.; Kitamura, K. Developments of human adrenomedullin-IgG1 Fc fusion proteins. J. Biochem. 2019, 166, 157–162. [Google Scholar] [CrossRef]
- Ishimitsu, T.; Nishikimi, T.; Saito, Y.; Kitamura, K.; Eto, T.; Kangawa, K.; Matsuo, H.; Omae, T.; Matsuoka, H. Plasma levels of adrenomedullin, a newly identified hypotensive peptide, in patients with hypertension and renal failure. J. Clin. Investig. 1994, 94, 2158–2161. [Google Scholar] [CrossRef]
- Kato, J.; Kitamura, K.; Matsui, E.; Tanaka, M.; Ishizaka, Y.; Kita, T.; Kangawa, K.; Eto, T. Plasma adrenomedullin and natriuretic peptides in patients with essential or malignant hypertension. Hypertens. Res. 1999, 22, 61–65. [Google Scholar] [CrossRef]
- Kato, J.; Kitamura, K.; Eto, T. Plasma adrenomedullin level and development of hypertension. J. Hum. Hypertens. 2006, 20, 566–570. [Google Scholar] [CrossRef]
- Hu, W.; Zhou, P.-H.; Zhang, X.-B.; Xu, C.-G.; Wang, W. Plasma concentrations of adrenomedullin and natriuretic peptides in patients with essential hypertension. Exp. Med. 2015, 9, 1901–1908. [Google Scholar] [CrossRef]
- Charles, C.J.; Lainchbury, J.G.; Nicholls, M.G.; Rademaker, M.T.; Richards, A.M.; Troughton, R.W. Adrenomedullin and the renin-angiotensin-aldosterone system. Regul. Pept. 2003, 112, 41–49. [Google Scholar] [CrossRef]
- Letizia, C.; Subioli, S.; Cerci, S.; Caliumi, C.; Verrelli, C.; Delfini, E.; Celi, M.; Scuro, L.; D’Erasmo, E. High plasma adrenomedullin concentrations in patients with high-renin essential hypertension. J. Renin Angiotensin Aldosterone Syst. 2002, 3, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Caliumi, C.; Cianci, R.; Celi, M.; Cerci, S.; Cotesta, D.; Petramala, L.; Fontana, S.; Letizia, C. Plasma adrenomedullin concentrations in patients with renovascular or malignant hypertension. Minerva Cardioangiol. 2004, 52, 313–322. [Google Scholar] [PubMed]
- Then, C.; Rottenkolber, M.; Lechner, A.; Meisinger, C.; Heier, M.; Koenig, W.; Peters, A.; Rathmann, W.; Bidlingmaier, M.; Reincke, M.; et al. Altered relation of the renin-aldosterone system and vasoactive peptides in Type 2 Diabetes: The KORA F4 study. Atherosclerosis 2016, 252, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Elmas, E.; Lang, S.; Dempfle, C.E.; Kälsch, T.; Papassotiriou, J.; Morgenthaler, N.G.; Borggrefe, M.; Brueckmann, M. Diagnostic performance of mid-regional pro-adrenomedullin as an analyte for the exclusion of left ventricular dysfunction. Int. J. Cardiol. 2008, 128, 107–111. [Google Scholar] [CrossRef]
- Elmas, E.; Tülümen, E.; Liebe, V.; Rudic, B.; Lang, S.; Akin, İ.; Schimpf, R.; Borggrefe, M. Mid-Regional pro-Adrenomedullin and N-Terminal pro B-Type natriuretic peptide predict the recurrence of atrial fibrillation after cryoballoon pulmonary vein isolation. Int. J. Cardiol. 2016, 203, 369–371. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Gottsäter, A.; Engström, G.; Melander, O.; Zarrouk, M.; Nilsson, P.M.; Smith, J.G. Circulating midregional proadrenomedullin and risk of incident abdominal aortic aneurysm: A prospective longitudinal cohort study. Angiology 2018, 69, 333–338. [Google Scholar] [CrossRef]
- Baldenhofer, G.; Laule, M.; Mockel, M.; Sanad, W.; Knebel, F.; Dreger, H.; Leonhardt, F.; Sander, M.; Grubitzsch, H.; Baumann, G.; et al. Mid-Regional pro-Adrenomedullin (MR-ProADM) and Mid-Regional pro-Atrial Natriuretic Peptide (MR-ProANP) in severe aortic valve stenosis: Association with outcome after Transcatheter Aortic Valve Implantation (TAVI). Clin. Chem. Lab. Med. 2017, 55, 275–283. [Google Scholar] [CrossRef]
- Nishikimi, T.; Karasawa, T.; Inaba, C.; Ishimura, K.; Tadokoro, K.; Koshikawa, S.; Yoshihara, F.; Nagaya, N.; Sakio, H.; Kangawa, K.; et al. Effects of long-term intravenous administration of Adrenomedullin (AM) Plus HANP therapy in acute decompensated heart failure. Circ. J. 2009, 73, 892–898. [Google Scholar] [CrossRef]
- Nagaya, N.; Satoh, T.; Nishikimi, T.; Uematsu, M.; Furuichi, S.; Sakamaki, F.; Oya, H.; Kyotani, S.; Nakanishi, N.; Goto, Y.; et al. Hemodynamic, renal, and hormonal effects of adrenomedullin infusion in patients with congestive heart failure. Circulation 2000, 101, 498–503. [Google Scholar] [CrossRef]
- Parameswaran, N.; Nowak, W.; Hall, C.S.; Sparks, H.V.; Spielman, W.S. Cellular and molecular actions of adrenomedullin in glomerular mesangial cells. Peptides 2001, 22, 1919–1924. [Google Scholar] [CrossRef]
- Nishikimi, T. Adrenomedullin in the kidney-renal physiological and pathophysiological roles. Curr. Med. Chem. 2007, 14, 1689–1699. [Google Scholar] [CrossRef] [PubMed]
- Ebara, T.; Miura, K.; Okumura, M.; Matsuura, T.; Kim, S.; Yukimura, T.; Iwao, H. Effect of adrnomedullin on renal hemodynamics and functions in dogs. Eur. J. Pharmacol. 1994, 263, 69–73. [Google Scholar] [CrossRef]
- Jougasaki, M.; Wei, C.M.; Aarhus, L.L.; Heublein, D.M.; Sandberg, S.M.; Burnett, J.C. Renal localization and actions of adrenomedullin: A natriuretic peptide. Am. J. Physiol. -Ren. Physiol. 1995, 268, F657–F663. [Google Scholar] [CrossRef]
- Vari, R.C.; Adkins, S.D.; Samson, W.K. Renal effects of adrenomedullin in the rat. Proc. Soc. Exp. Biol Med. 1996, 211, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Majid, D.S.; Kadowitz, P.J.; Coy, D.H.; Navar, L.G. Renal responses to intra-arterial administration of adrenomedullin in dogs. Am. J. Physiol. Ren. Physiol. 1996, 270, F200–F205. [Google Scholar] [CrossRef]
- Owada, A.; Nonoguchi, H.; Terada, Y.; Marumo, F.; Tomita, K. Microlocalization and effects of adrenomedullin in nephron segments and in mesangial cells of the rat. Am. J. Physiol. Ren. Physiol. 1997, 272, F691–F697. [Google Scholar] [CrossRef]
- Jensen Boye, L.; Krämer Bernhard, K. Kurtz armin adrenomedullin stimulates renin release and renin mrna in mouse juxtaglomerular granular cells. Hypertension 1997, 29, 1148–1155. [Google Scholar] [CrossRef]
- Leclerc, M.; Brunette, M.G. The paradoxical effect of adrenomedullin on na+ transport by the renal distal tubule luminal membrane. Mol. Cell. Endocrinol. 2000, 164, 159–167. [Google Scholar] [CrossRef]
- Dieplinger, B.; Mueller, T.; Kollerits, B.; Struck, J.; Ritz, E.; von Eckardstein, A.; Haltmayer, M.; Kronenberg, F. Pro-A-Type natriuretic peptide and pro-adrenomedullin predict progression of chronic kidney disease: The MMKD study. Kidney Int. 2009, 75, 408–414. [Google Scholar] [CrossRef]
- Nagata, D.; Hirata, Y.; Suzuki, E.; Kakoki, M.; Hayakawa, H.; Goto, A.; Ishimitsu, T.; Minamino, N.; Ono, Y.; Kangawa, K.; et al. Hypoxia-Induced adrenomedullin production in the kidney. Kidney Int. 1999, 55, 1259–1267. [Google Scholar] [CrossRef] [PubMed]
- Nishimatsu, H.; Hirata, Y.; Shindo, T.; Kurihara, H.; Kakoki, M.; Nagata, D.; Hayakawa, H.; Satonaka, H.; Sata, M.; Tojo, A.; et al. Role of endogenous adrenomedullin in the regulation of vascular tone and ischemic renal injury: Studies on transgenic/knockout mice of adrenomedullin gene. Circ. Res. 2002, 90, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Kakareko, K.; Rydzewska-Rosołowska, A.; Rygasiewicz, K.; Siemiątkowski, A.; Koc-Żórawska, E.; Zbroch, E.; Hryszko, T. Prognostic value of mid-regional proadrenomedullin in critically Ill patients. Pol. Arch. Intern. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zheng, X.; Wang, H.; Wang, S.; Yu, K.; Wang, C. The value of plasma pro-enkephalin and adrenomedullin for the prediction of sepsis-associated acute kidney injury in critically Ill patients. Crit. Care 2020, 24. [Google Scholar] [CrossRef] [PubMed]
- Kuo, M.-C.; Kuo, H.-T.; Chiu, Y.-W.; Chang, J.-M.; Guh, J.-Y.; Lai, Y.-H.; Chen, H.-C. Decreased synthesis of glomerular adrenomedullin in patients with IgA nephropathy. J. Lab. Clin. Med. 2005, 145, 233–238. [Google Scholar] [CrossRef]
- Kubo, A.; Kurioka, H.; Minamino, N.; Nishitani, Y.; Sato, H.; Nishino, T.; Iwano, M.; Shiiki, H.; Kangawa, K.; Matsuo, H.; et al. Plasma and urinary levels of adrenomedullin in chronic glomerulonephritis patients with proteinuria. NEF 1998, 80, 227–230. [Google Scholar] [CrossRef]
- Mak, A.; Cheung, B.M.Y.; Mok, C.C.; Leung, R.; Lau, C.S. Adrenomedullin—A potential disease activity marker and suppressor of nephritis activity in systemic lupus erythematosus. Rheumatology 2006, 45, 1266–1272. [Google Scholar] [CrossRef][Green Version]
- Artunc, F.; Nowak, A.; Mueller, C.; Breidthardt, T.; Twerenbold, R.; Wagner, R.; Peter, A.; Haering, H.-U.; Ebmeyer, S.; Friedrich, B. Plasma Concentrations of the Vasoactive Peptide Fragments Mid-Regional Pro-Adrenomedullin, C-Terminal Pro-Endothelin 1 and copeptin in hemodialysis patients: Associated factors and prediction of mortality. PLoS ONE 2014, 9, e86148. [Google Scholar] [CrossRef]
- Gouya, G.; Sturm, G.; Lamina, C.; Zitt, E.; Freistätter, O.; Struck, J.; Wolzt, M.; Knoll, F.; Lins, F.; Lhotta, K.; et al. The association of mid-regional pro-adrenomedullin and mid-regional pro-atrial natriuretic peptide with mortality in an incident dialysis cohort. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Yoshihara, F.; Ernst, A.; Morgenthaler, N.G.; Horio, T.; Nakamura, S.; Nakahama, H.; Nakata, H.; Bergmann, A.; Kangawa, K.; Kawano, Y. Midregional proadrenomedullin reflects cardiac dysfunction in haemodialysis patients with cardiovascular disease. Nephrol Dial. Transpl. 2007, 22, 2263–2268. [Google Scholar] [CrossRef][Green Version]
- Toepfer, M.; Schlosshauer, M.; Sitter, T.; Burchardi, C.; Behr, T.; Schiffl, H. Effects of hemodialysis on circulating adrenomedullin concentrations in patients with end-stage renal disease. BPU 1998, 16, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Itoh, H.; Katagiri, F.; Sato, F.; Kawasaki, K.; Sato, Y.; Sato, Y.; Mimata, H.; Takeyama, M. Significant decrease in plasma midregional proadrenomedullin level in patients with end-stage renal disease after living kidney transplantation. Peptides 2013, 43, 102–104. [Google Scholar] [CrossRef]
- Suzuki, Y.; Itoh, H.; Katagiri, F.; Sato, F.; Kawasaki, K.; Sato, Y.; Sato, Y.; Mimata, H.; Takeyama, M. Relationship between plasma mid-regional pro-adrenomedullin level and resistance to antihypertensive therapy in stable kidney transplant recipients. Peptides 2013, 48, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Sudoh, T.; Kangawa, K.; Minamino, N.; Matsuo, H. A new natriuretic peptide in porcine brain. Nature 1988, 332, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Das Sandeep, R.; Drazner, M.H.; Dries, D.L.; Vega, G.L.; Stanek, H.G.; Abdullah, S.M.; Canham, R.M.; Chung, A.K.; Leonard, D.; Wians, F.H.; et al. Impact of body mass and body composition on circulating levels of natriuretic peptides. Circulation 2005, 112, 2163–2168. [Google Scholar] [CrossRef]
- Redfield, M.M.; Rodeheffer, R.J.; Jacobsen, S.J.; Mahoney, D.W.; Bailey, K.R.; Burnett, J.C. Plasma brain natriuretic peptide concentration: Impact of age and gender. J. Am. Coll. Cardiol. 2002, 40, 976–982. [Google Scholar] [CrossRef]
- Raymond, I.; Groenning, B.A.; Hildebrandt, P.R.; Nilsson, J.C.; Baumann, M.; Trawinski, J.; Pedersen, F. The influence of age, sex and other variables on the plasma level of n-terminal pro brain natriuretic peptide in a large sample of the general population. Heart 2003, 89, 745–751. [Google Scholar] [CrossRef]
- Cataliotti, A.; Malatino, L.S.; Jougasaki, M.; Zoccali, C.; Castellino, P.; Giacone, G.; Bellanuova, I.; Tripepi, R.; Seminara, G.; Parlongo, S.; et al. Circulating natriuretic peptide concentrations in patients with end-stage renal disease: Role of brain natriuretic peptide as a biomarker for ventricular remodeling. Mayo Clin. Proc. 2001, 76, 1111–1119. [Google Scholar] [CrossRef]
- Anwaruddin, S.; Lloyd-Jones, D.M.; Baggish, A.; Chen, A.; Krauser, D.; Tung, R.; Chae, C.; Januzzi, J.L. Renal function, congestive heart failure, and amino-terminal pro-brain natriuretic peptide measurement: Results from the ProBNP investigation of dyspnea in the emergency department (PRIDE) study. J. Am. Coll. Cardiol. 2006, 47, 91–97. [Google Scholar] [CrossRef]
- Cardarelli, R.; Lumicao, T.G. B-Type natriuretic peptide: A review of its diagnostic, prognostic, and therapeutic monitoring value in heart failure for primary care physicians. J. Am. Board Fam. Pr. 2003, 16, 327–333. [Google Scholar] [CrossRef]
- Cowie, M.R.; Struthers, A.D.; Wood, D.A.; Coats, A.J.; Thompson, S.G.; Poole-Wilson, P.A.; Sutton, G.C. Value of natriuretic peptides in assessment of patients with possible new heart failure in primary care. Lancet 1997, 350, 1349–1353. [Google Scholar] [CrossRef]
- Hartmann, F.; Packer, M.; Coats, A.J.S.; Fowler, M.B.; Krum, H.; Mohacsi, P.; Rouleau, J.L.; Tendera, M.; Castaigne, A.; Anker, S.D.; et al. Prognostic impact of plasma n-terminal pro–brain natriuretic peptide in severe chronic congestive heart failure. Circulation 2004, 110, 1780–1786. [Google Scholar] [CrossRef] [PubMed]
- Eurlings, L.W.; Wijk, S.S.; van Kraaij, D.J.W.; van Kimmenade, R.; Meeder, J.G.; Kamp, O.; van Dieijen-Visser, M.P.; Tijssen, J.G.P.; Rocca, H.-P.B.-L.; Pinto, Y.M. Risk stratification with the use of serial N-terminal Pro–B-Type natriuretic peptide measurements during admission and early after discharge in heart failure patients: Post hoc analysis of the PRIMA study. J. Card. Fail. 2014, 20, 881–890. [Google Scholar] [CrossRef] [PubMed]
- McLellan, J.; Heneghan, C.J.; Perera, R.; Clements, A.M.; Glasziou, P.P.; Kearley, K.E.; Pidduck, N.; Roberts, N.W.; Tyndel, S.; Wright, F.L.; et al. B-type natriuretic peptide-guided treatment for heart failure. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.S.J.; Chan, S.-P.; Liew, O.-W.; Chong, J.P.C.; Leong, G.K.T.; Yeo, D.P.S.; Ong, H.-Y.; Jaufeerally, F.; Yap, J.; Sim, D.; et al. Atrial fibrillation and the prognostic performance of biomarkers in heart failure. Clin. Chem. 2021, 67, 216–226. [Google Scholar] [CrossRef]
- Fabris, E.; ten Berg, J.M.; Hermanides, R.S.; Ottervanger, J.P.; Dambrink, J.H.E.; Gosselink, A.M.; Sinagra, G.; Koopmans, P.C.; Giannitsis, E.; Hamm, C.; et al. NT-ProBNP Level before primary PCI and risk of poor myocardial reperfusion: Insight from the On-TIME II trial. Am. Heart J. 2021, 233, 78–85. [Google Scholar] [CrossRef]
- Nicoli, C.D.; Plante, T.B.; Long, D.L.; Judd, S.E.; McClure, L.A.; Arora, P.; Cushman, M. N-Terminal Pro-B-Type natriuretic peptide and longitudinal risk of hypertension. Am. J. Hypertens. 2020. [Google Scholar] [CrossRef]
- Richards, A.M.; Nicholls, M.G.; Yandle, T.G.; Frampton, C.; Espiner, E.A.; Turner, J.G.; Buttimore, R.C.; Lainchbury, J.G.; Elliott, J.M.; Ikram, H.; et al. Plasma N-Terminal pro-Brain natriuretic peptide and adrenomedullin: New neurohormonal predictors of left ventricular function and prognosis after myocardial infarction. Circulation 1998, 97, 1921–1929. [Google Scholar] [CrossRef]
- Daggubati, S.; Parks, J.R.; Overton, R.M.; Cintron, G.; Schocken, D.D.; Vesely, D.L. Adrenomedullin, endothelin, neuropeptide y, atrial, brain, and C-natriuretic prohormone peptides compared as early heart failure indicators. Cardiovasc. Res. 1997, 36, 246–255. [Google Scholar] [CrossRef]
- Fraty, M.; Velho, G.; Gand, E.; Fumeron, F.; Ragot, S.; Sosner, P.; Mohammedi, K.; Gellen, B.; Saulnier, P.-J.; Halimi, J.-M.; et al. Prognostic value of plasma MR-ProADM vs NT-ProBNP for heart failure in people with Type 2 Diabetes: The surdiagene prospective study. Diabetologia 2018, 61, 2643–2653. [Google Scholar] [CrossRef]
- Morbach, C.; Marx, A.; Kaspar, M.; Güder, G.; Brenner, S.; Feldmann, C.; Störk, S.; Vollert, J.O.; Ertl, G.; Angermann, C.E.; et al. Prognostic potential of midregional pro-adrenomedullin following decompensation for systolic heart failure: Comparison with cardiac natriuretic peptides. Eur. J. Heart Fail. 2017, 19, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
| Author | Population | Organ | Effect |
|---|---|---|---|
| Lippton (1994) [14] | rat | pulmonary vascular | decreased lobar arterial pressure highest doses mildly decreased systemic arterial pressure |
| Lainchbury (1997) [15] | human | vascular | reduced mean arterial pressure reduced systolic arterial pressure reduced diastolic arterial pressure |
| Parkes (1997) [16] | sheep | vascular, heart | vasodilation increased cardiac contractility and cardiac output hypotensive actions |
| Ikenouchi (1997) [17] | rabbit | cardiomyocytes | negative inotropic effect |
| Yoshimoto (1998) [18] | rat, pig | rat aorta, pig coronary artery | vasodilation |
| Sait (1998) [19] | rat | brain ventricular | induced hypertension, tachycardia central regulation of the cardiovascular system |
| Szokodi (1998) [20] | rat | heart | enhanced cardiac contractility |
| Ihara (2000) [21] | rat | papillary muscles | positive inotropic effects increased intracellular contents of cAMP |
| Saetrum Opgaard (2000) [22] | human | cardiomyocytes | no positive or negative inotropic actions |
| Author | Population | Organ | Effect |
|---|---|---|---|
| Ebara (1994) [54] | dog | kidney | vasodilatation increased urine flow increased urinary excretion of sodium and potassium with no effect onrenin activity |
| Jougasaki (1995) [55] | dog | kidney | increased GFR decrease in distal tubular sodium reabsorption, natriuretic effect, diuretic effect |
| Hirata (1995) [10] | rat | kidney | increased diameters of both afferent and efferent arterioles reduction in BP vasodilatation |
| Vari (1996) [56] | rat | kidney | minimal hypotensive effect increased renal plasma flow natiuretic effect |
| Majid (1996) [57] | dog | kidney | decreased renal vascular resistance increased renal blood flow no change ofGFR regulation ofwater and electrolyte excretion |
| Owada (1997) [58] | rat | nephron, kidney | increased GFR diureticeffect |
| Jensen (1997) [59] | rat | Juxtaglomerulargranularcells, kidney | stimulatory effect on renin secretion |
| Leclerc (2000) [60] | human | proximal and distal tubule, kidney | natriuretic effect |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czajkowska, K.; Zbroch, E.; Bielach-Bazyluk, A.; Mitrosz, K.; Bujno, E.; Kakareko, K.; Rydzewska-Rosolowska, A.; Hryszko, T. Mid-Regional Proadrenomedullin as a New Biomarker of Kidney and Cardiovascular Diseases—Is It the Future? J. Clin. Med. 2021, 10, 524. https://doi.org/10.3390/jcm10030524
Czajkowska K, Zbroch E, Bielach-Bazyluk A, Mitrosz K, Bujno E, Kakareko K, Rydzewska-Rosolowska A, Hryszko T. Mid-Regional Proadrenomedullin as a New Biomarker of Kidney and Cardiovascular Diseases—Is It the Future? Journal of Clinical Medicine. 2021; 10(3):524. https://doi.org/10.3390/jcm10030524
Chicago/Turabian StyleCzajkowska, Katarzyna, Edyta Zbroch, Angelika Bielach-Bazyluk, Katarzyna Mitrosz, Elzbieta Bujno, Katarzyna Kakareko, Alicja Rydzewska-Rosolowska, and Tomasz Hryszko. 2021. "Mid-Regional Proadrenomedullin as a New Biomarker of Kidney and Cardiovascular Diseases—Is It the Future?" Journal of Clinical Medicine 10, no. 3: 524. https://doi.org/10.3390/jcm10030524
APA StyleCzajkowska, K., Zbroch, E., Bielach-Bazyluk, A., Mitrosz, K., Bujno, E., Kakareko, K., Rydzewska-Rosolowska, A., & Hryszko, T. (2021). Mid-Regional Proadrenomedullin as a New Biomarker of Kidney and Cardiovascular Diseases—Is It the Future? Journal of Clinical Medicine, 10(3), 524. https://doi.org/10.3390/jcm10030524







