Multi-Organ Involvement in COVID-19: Beyond Pulmonary Manifestations
Abstract
1. Introduction
2. Clinical Presentation and Transmission of Coronavirus Disease 2019 (COVID-19)
3. Cutaneous Manifestations of Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2)
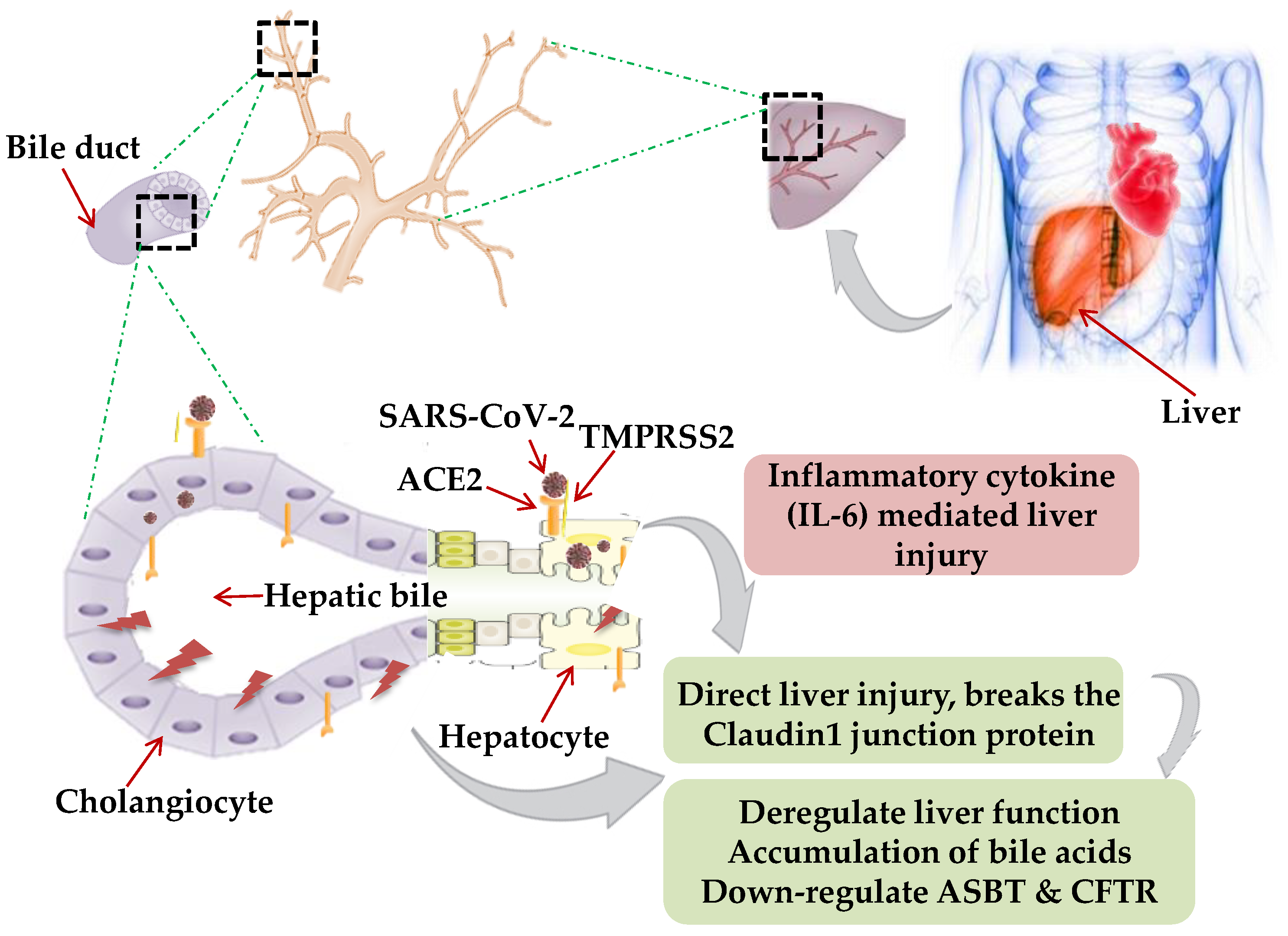
4. Hepatic Manifestations of SARS-CoV-2
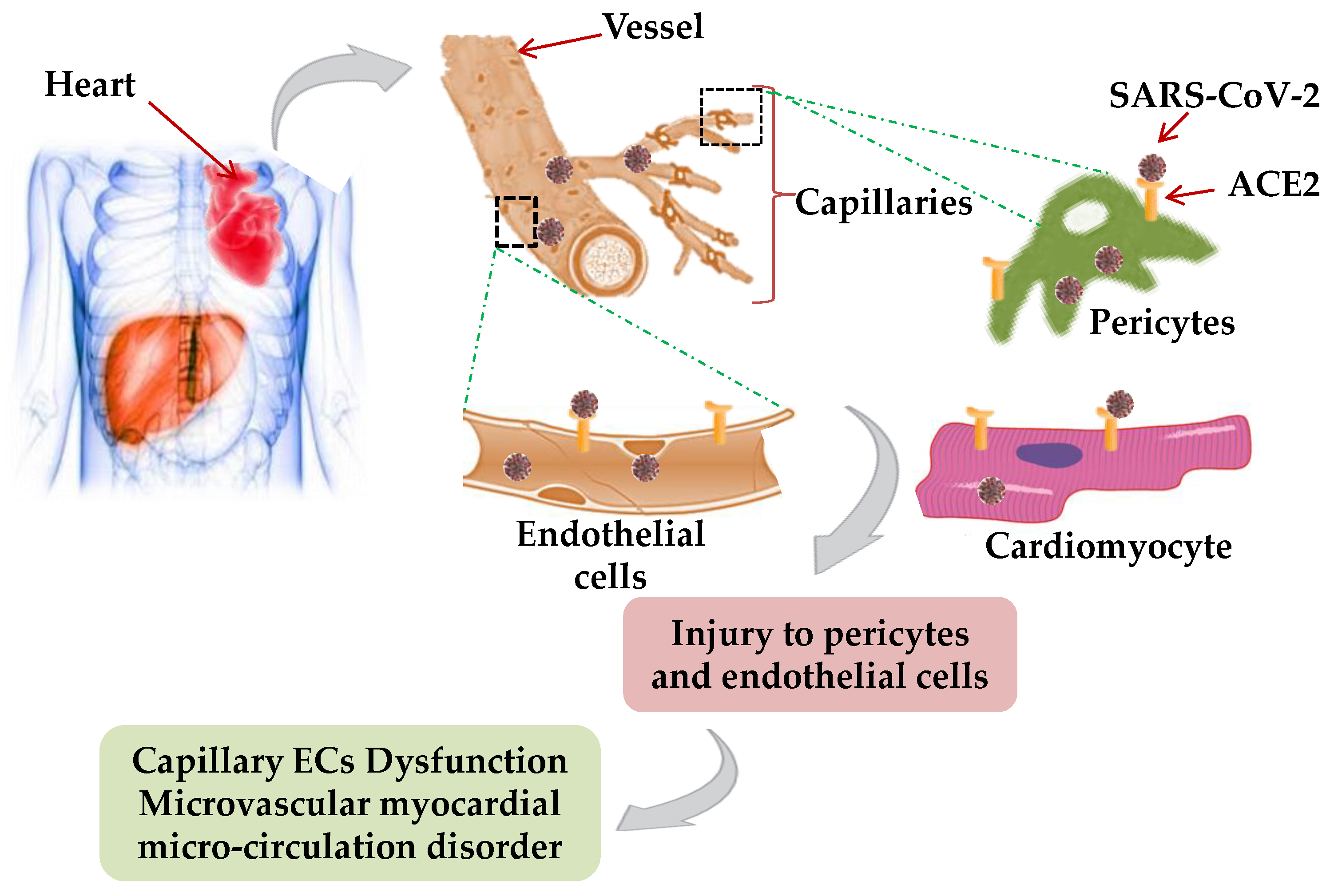
5. Cardiac Manifestations of SARS-CoV-2
Possible Mechanism of Cardiac Manifestations
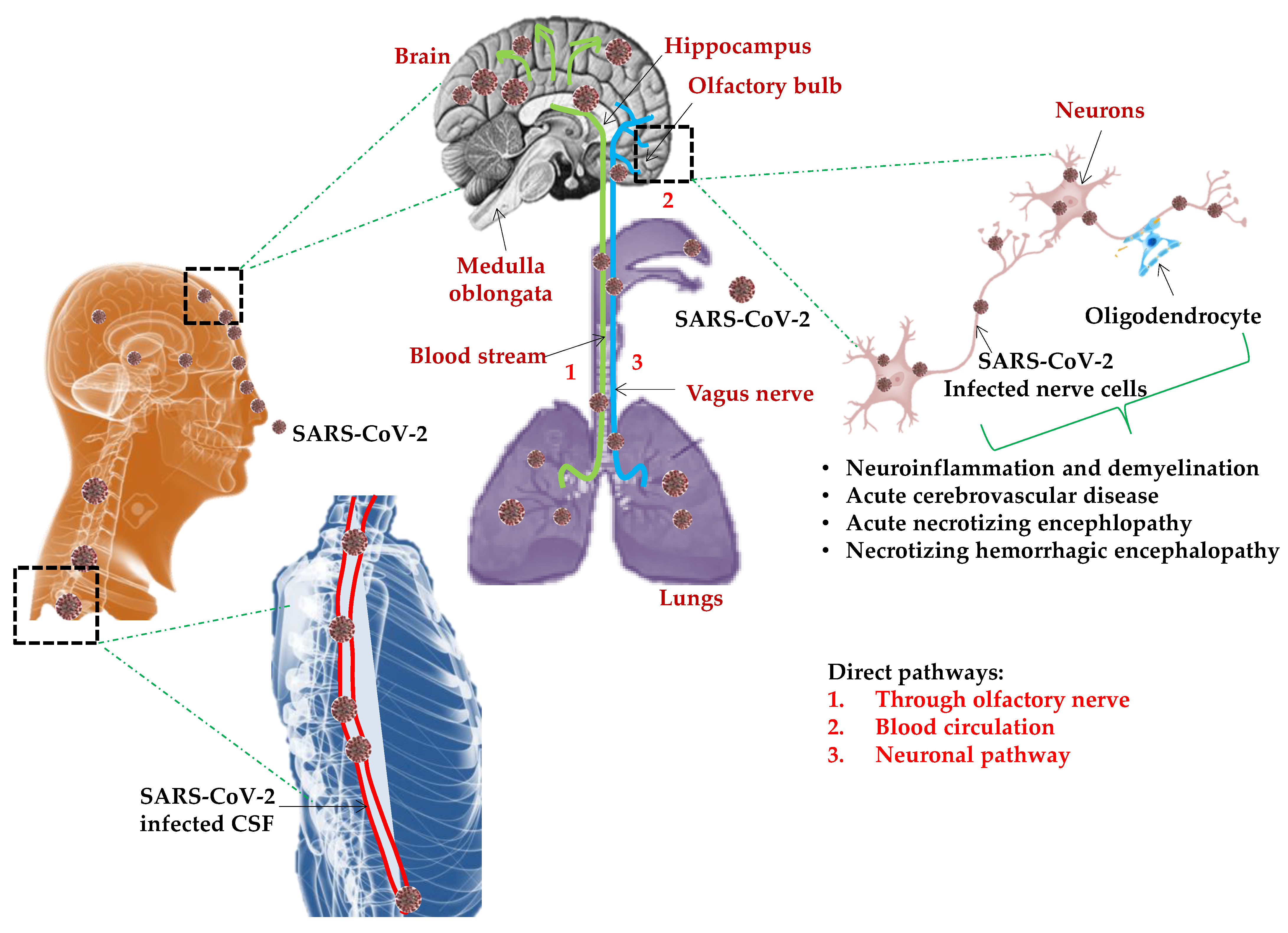
6. Neurological Manifestations of SARS-CoV-2
Mechanisms of Neurotropism and Neuroinvasion
7. Renal Manifestations
The Potential Mechanisms of Renal Manifestations
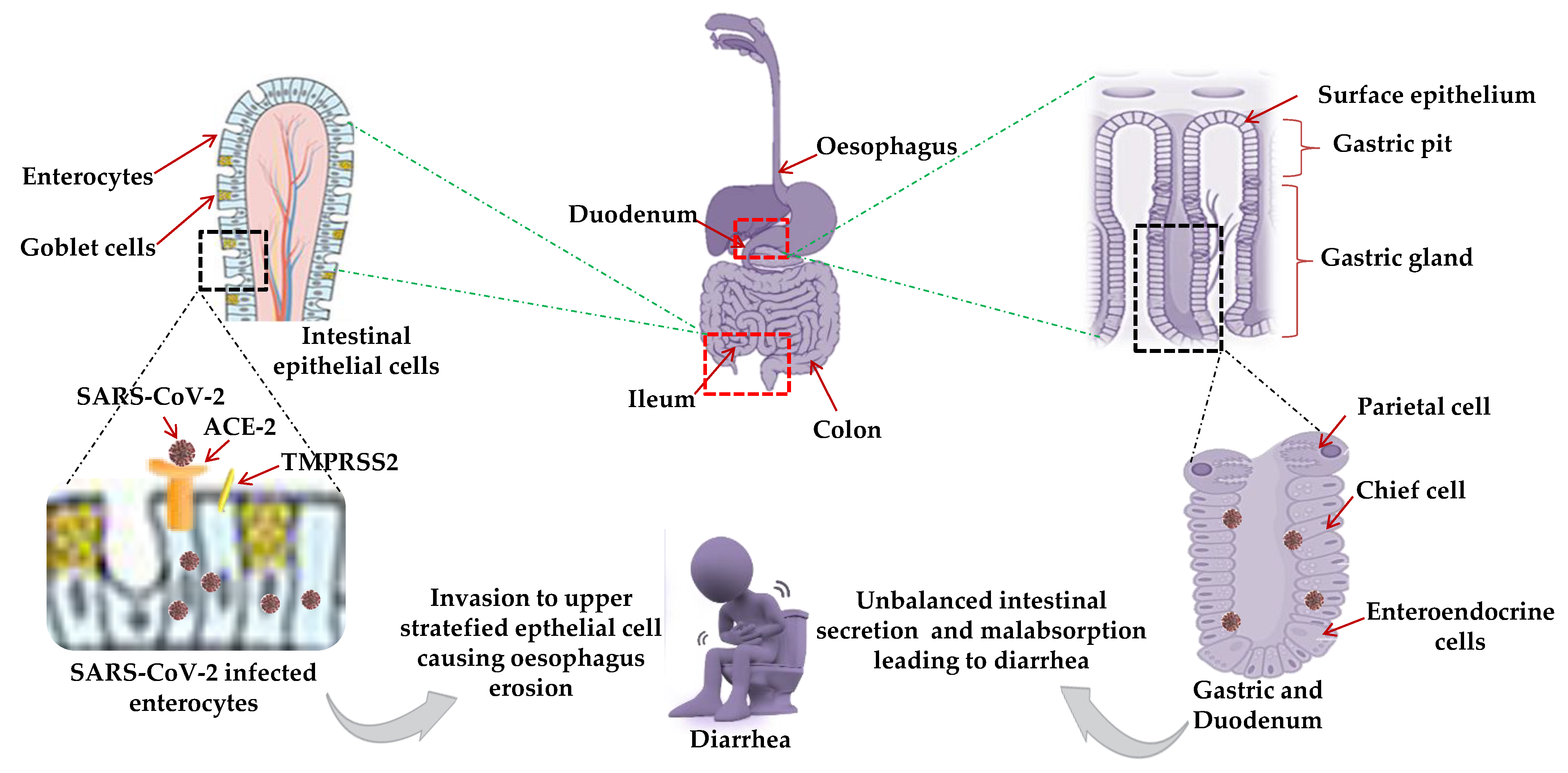
8. Gastrointestinal (GIT) Manifestations
Possible Mechanism for GIT Manifestations
9. Hematological Manifestations of SARS-CoV-2
10. Unusual Manifestations of SARS-CoV-2
11. COVID-19 in Immunocompromised Solid-Organ Transplant Recipients
12. Co-Infections with COVID-19: Viral, Bacterial, and Fungal
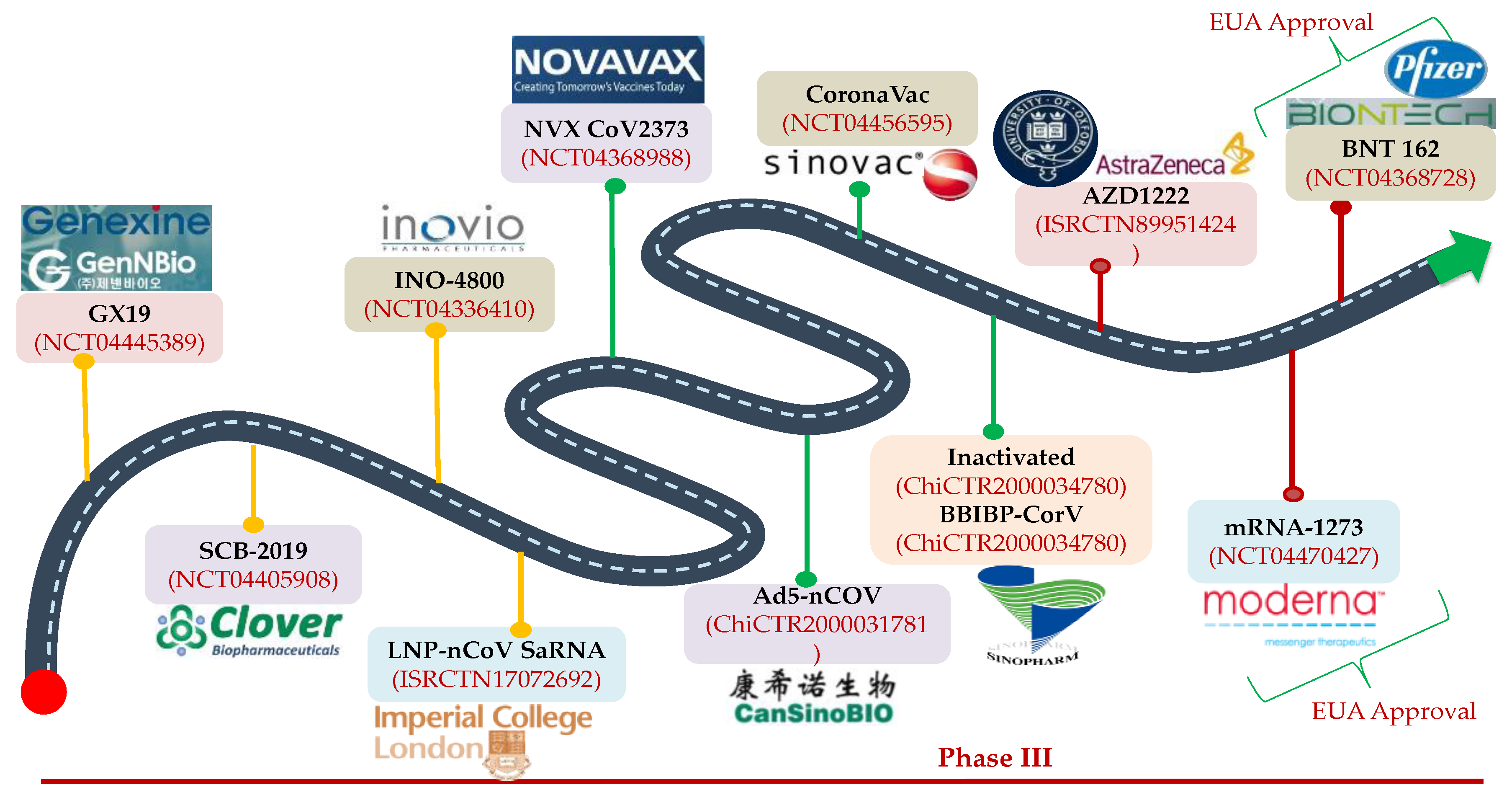
13. Advancements in Vaccine Research
14. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coronavirus Update (Live): 94,993,884 Cases and 2,031,875 Deaths from COVID-19 Virus Pandemic—Worldometer. Available online: https://www.worldometers.info/coronavirus/ (accessed on 17 January 2021).
- Cucinotta, D.; Vanelli, M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Koopmans, M.; Go, U.; Hamer, D.H.; Petrosillo, N.; Castelli, F.; Storgaard, M.; Khalili, S.A.; Simonsen, L. Comparing SARS-CoV-2 with SARS-CoV and influenza pandemics. Lancet Infect. Dis. 2020, 20, e238–e244. [Google Scholar] [CrossRef]
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Corman, V.M.; Muth, D.; Niemeyer, D.; Drosten, C. Hosts and Sources of Endemic Human Coronaviruses. In Advances in Virus Research; Academic Press Inc.: New York, NY, USA, 2018; Volume 100, pp. 163–188. [Google Scholar]
- Gralinski, L.E.; Baric, R.S. Molecular pathology of emerging coronavirus infections. J. Pathol. 2015, 235, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Neerukonda, S.N.; Katneni, U. A Review on SARS-CoV-2 Virology, Pathophysiology, Animal Models, and Anti-Viral Interventions. Pathogens 2020, 9, 426. [Google Scholar] [CrossRef]
- Xia, S.; Zhu, Y.; Liu, M.; Lan, Q.; Xu, W.; Wu, Y.; Ying, T.; Liu, S.; Shi, Z.; Jiang, S.; et al. Fusion mechanism of 2019-nCoV and fusion inhibitors targeting HR1 domain in spike protein. Cell. Mol. Immunol. 2020, 17, 765–767. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, Z.; Li, J.W.; Zhao, H.; Wang, G.Q. Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents 2020, 55. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases from the Chinese Center for Disease Control and Prevention. JAMA J. Am. Med. Assoc. 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA J. Am. Med. Assoc. 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Gandhi, M.; Yokoe, D.S.; Havlir, D.V. Asymptomatic Transmission, the Achilles’ Heel of Current Strategies to Control Covid-19. N. Engl. J. Med. 2020, 382, 2158–2160. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Pei, S.; Chen, B.; Song, Y.; Zhang, T.; Yang, W.; Shaman, J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2). Science 2020, 368, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Madjid, M.; Safavi-Naeini, P.; Solomon, S.D.; Vardeny, O. Potential Effects of Coronaviruses on the Cardiovascular System: A Review. JAMA Cardiol. 2020, 5, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Rothan, H.A.; Byrareddy, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020, 109. [Google Scholar] [CrossRef]
- Li, Y.C.; Bai, W.Z.; Hashikawa, T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020, 92, 552–555. [Google Scholar] [CrossRef]
- Yeo, C.; Kaushal, S.; Yeo, D. Enteric involvement of coronaviruses: Is faecal–oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol. Hepatol. 2020, 5, 335–337. [Google Scholar] [CrossRef]
- Sahin, A.R. 2019 Novel Coronavirus (COVID-19) Outbreak: A Review of the Current Literature. Eurasian J. Med. Oncol. 2020. [Google Scholar] [CrossRef]
- Tang, Y.W.; Schmitz, J.E.; Persing, D.H.; Stratton, C.W. Laboratory diagnosis of COVID-19: Current issues and challenges. J. Clin. Microbiol. 2020, 58. [Google Scholar] [CrossRef]
- Recalcati, S. Cutaneous manifestations in COVID-19: A first perspective. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e212–e213. [Google Scholar] [CrossRef]
- Guan, W.; Ni, Z.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Liu, L.; Shan, H.; Lei, C.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Fernandez-Nieto, D.; Ortega-Quijano, D.; Segurado-Miravalles, G.; Pindado-Ortega, C.; Prieto-Barrios, M.; Jimenez-Cauhe, J. Comment on: Cutaneous manifestations in COVID-19: A first perspective. Safety concerns of clinical images and skin biopsies. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e252–e254. [Google Scholar] [CrossRef] [PubMed]
- Marzano, A.V.; Genovese, G.; Fabbrocini, G.; Pigatto, P.; Monfrecola, G.; Piraccini, B.M.; Veraldi, S.; Rubegni, P.; Cusini, M.; Caputo, V.; et al. Varicella-like exanthem as a specific COVID-19–associated skin manifestation: Multicenter case series of 22 patients. J. Am. Acad. Dermatol. 2020, 83, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Joob, B.; Wiwanitkit, V. COVID-19 can present with a rash and be mistaken for dengue. J. Am. Acad. Dermatol. 2020, 82, e177. [Google Scholar] [CrossRef] [PubMed]
- Estébanez, A.; Pérez-Santiago, L.; Silva, E.; Guillen-Climent, S.; García-Vázquez, A.; Ramón, M.D. Cutaneous manifestations in COVID-19: A new contribution. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e250–e251. [Google Scholar] [CrossRef] [PubMed]
- Joob, B.; Wiwanitkit, V. Reply to: ‘Various forms of skin rash in COVID-19: Petechial rash in a patient with COVID-19 infection’. J. Am. Acad. Dermatol. 2020, 83, e143. [Google Scholar] [CrossRef]
- Cai, Q.; Huang, D.; Ou, P.; Yu, H.; Zhu, Z.; Xia, Z.; Su, Y.; Ma, Z.; Zhang, Y.; Li, Z.; et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy Eur. J. Allergy Clin. Immunol. 2020, 75, 1742–1752. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, X.; Qiu, Y.; Song, Y.; Feng, F.; Feng, J.; Song, Q.; Jia, Q. Clinical characteristics of 82 cases of death from COVID-19. PLoS ONE 2020, 15, e0235458. [Google Scholar] [CrossRef]
- Chai, X.; Hu, L.; Zhang, Y.; Han, W.; Lu, Z.; Ke, A.; Zhou, J.; Shi, G.; Fang, N.; Fan, J.; et al. Specific ACE2 expression in cholangiocytes may cause liver damage after 2019-nCoV infection. bioRxiv 2020. [Google Scholar] [CrossRef]
- Wen Seow, J.J.; Pai, R.; Mishra, A.; Shepherdson, E.; Lim Hon, T.K.; Goh Brian, K.P.; Chan Jerry, K.Y.; Chow Pierce, K.H.; Ginhoux, F.; DasGupta, R.; et al. scRNA-seq reveals ACE2 and TMPRSS2 expression in TROP2 + Liver Progenitor Cells: Implications in COVID-19 associated Liver Dysfunction. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhao, B.; Ni, C.; Gao, R.; Wang, Y.; Yang, L.; Wei, J.; Lv, T.; Liang, J.; Zhang, Q.; Xu, W.; et al. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids. Protein Cell 2020, 11, 771–775. [Google Scholar] [CrossRef]
- Zhang, C.; Shi, L.; Wang, F.S. Liver injury in COVID-19: Management and challenges. Lancet Gastroenterol. Hepatol. 2020, 5, 428–430. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Liu, J.; Liang, B.; Wang, X.; Wang, H.; Li, W.; Tong, Q.; Yi, J.; Zhao, L.; et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine 2020, 55. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; O’Kane, A.M.; Peng, H.; Bi, Y.; Motriuk-Smith, D.; Ren, J. SARS-CoV-2 and cardiovascular complications: From molecular mechanisms to pharmaceutical management. Biochem. Pharmacol. 2020, 178, 114114. [Google Scholar] [CrossRef] [PubMed]
- Bonow, R.O.; Fonarow, G.C.; O’Gara, P.T.; Yancy, C.W. Association of Coronavirus Disease 2019 (COVID-19) with Myocardial Injury and Mortality. JAMA Cardiol. 2020, 5, 751–753. [Google Scholar] [CrossRef]
- Inciardi, R.M.; Lupi, R.; Zaccone, G.; Italia, L.; Raffo, M.; Tomasoni, D.; Cani, D.S.; Cerini, M.; Farina, D.; Gavazzi, E.; et al. Cardiac Involvement in a Patient with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 819–824. [Google Scholar] [CrossRef]
- Han, H.; Xie, L.; Liu, R.; Yang, J.; Liu, F.; Wu, K.; Chen, L.; Hou, W.; Feng, Y.; Zhu, C. Analysis of heart injury laboratory parameters in 273 COVID-19 patients in one hospital in Wuhan, China. J. Med. Virol. 2020, 92, 819–823. [Google Scholar] [CrossRef]
- Vidovich, M.I. Transient Brugada-Like Electrocardiographic Pattern in a Patient with COVID-19. JACC Case Rep. 2020, 2, 1245–1249. [Google Scholar] [CrossRef]
- Sorgente, A.; Capulzini, L.; Brugada, P. The Known Into the Unknown. JACC Case Rep. 2020, 2, 1250–1251. [Google Scholar] [CrossRef]
- Chen, L.; Li, X.; Chen, M.; Feng, Y.; Xiong, C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020, 116, 1097–1100. [Google Scholar] [CrossRef]
- Zheng, Y.Y.; Ma, Y.T.; Zhang, J.Y.; Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020, 17, 259–260. [Google Scholar] [CrossRef] [PubMed]
- Bilimoria, J.; Singh, H. The Angiopoietin ligands and Tie receptors: Potential diagnostic biomarkers of vascular disease. J. Recept. Signal Transduct. 2019, 39, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Desforges, M.; Coupanec, A.L.; Dubeau, P.; Bourgouin, A.; Lajoie, L.; Dube, M.; Talbot, P.J. Human coronaviruses and other respiratory viruses: Underestimated opportunistic pathogens of the central nervous system? Viruses 2019, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Kabbani, N.; Olds, J.L. Does COVID19 Infect the Brain? If So, Smokers Might Be at a Higher Risk. Mol. Pharmacol. 2020, 97, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhang, M.; Wang, J.; Gao, J. Sars-Cov-2: Underestimated damage to nervous system. Travel Med. Infect. Dis. 2020, 36. [Google Scholar] [CrossRef]
- Poyiadji, N.; Shahin, G.; Noujaim, D.; Stone, M.; Patel, S.; Griffith, B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: Imaging features. Radiology 2020, 296, E119–E120. [Google Scholar] [CrossRef]
- Miller, E.H.; Namale, V.S.; Kim, C.; Dugue, R.; Waldrop, G.; Ciryam, P.; Chong, A.M.; Zucker, J.; Miller, E.C.; Bain, J.M.; et al. Cerebrospinal Analysis in Patients With COVID-19. Open Forum Infect. Dis. 2020, 7. [Google Scholar] [CrossRef]
- Wu, Y.; Xu, X.; Chen, Z.; Duan, J.; Hashimoto, K.; Yang, L.; Liu, C.; Yang, C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020, 87, 18–22. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients with Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef]
- Li, Y.; Li, M.; Wang, M.; Zhou, Y.; Chang, J.; Xian, Y.; Wang, D.; Mao, L.; Jin, H.; Hu, B. Acute cerebrovascular disease following COVID-19: A single center, retrospective, observational study. Stroke Vasc. Neurol. 2020, 5, 279–284. [Google Scholar] [CrossRef]
- Sharifi-Razavi, A.; Karimi, N.; Rouhani, N. COVID-19 and intracerebral haemorrhage: Causative or coincidental? New Microbes New Infect. 2020, 35, 100669. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, T.; Harii, N.; Goto, J.; Harada, D.; Sugawara, H.; Takamino, J.; Ueno, M.; Sakata, H.; Kondo, K.; Myose, N.; et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020, 94, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Ren, Y.; Lv, T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav. Immun. 2020, 88, 945–946. [Google Scholar] [CrossRef] [PubMed]
- Baig, A.M.; Khaleeq, A.; Ali, U.; Syeda, H. Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host-Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem. Neurosci. 2020, 11, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Portals of Viral Entry into the Central Nervous System. In The Blood-Brain Barrier in Health and Disease, Volume Two; CRC Press: Boca Raton, FL, USA, 2015; pp. 37–61.
- Saavedra, J.M. Brain angiotensin II: New developments, unanswered questions and therapeutic opportunities. Cell. Mol. Neurobiol. 2005, 25, 485–512. [Google Scholar] [CrossRef]
- Butowt, R.; Bilinska, K. SARS-CoV-2: Olfaction, Brain Infection, and the Urgent Need for Clinical Samples Allowing Earlier Virus Detection. ACS Chem. Neurosci. 2020. [Google Scholar] [CrossRef]
- Wan, S.; Yi, Q.; Fan, S.; Lv, J.; Zhang, X.; Guo, L.; Lang, C.; Xiao, Q.; Xiao, K.; Yi, Z.; et al. Characteristics of lymphocyte subsets and cytokines in peripheral blood of 123 hospitalized patients with 2019 novel coronavirus pneumonia (NCP). medRxiv 2020. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Pei, G.; Zhang, Z.; Peng, J.; Liu, L.; Zhang, C.; Yu, C.; Ma, Z.; Huang, Y.; Liu, Y.; Yao, Y.; et al. Renal involvement and early prognosis in patients with COVID-19 pneumonia. J. Am. Soc. Nephrol. 2020, 31, 1157–1165. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, R.; Wang, K.; Zhang, M.; Wang, Z.; Dong, L.; Li, J.; Yao, Y.; Ge, S.; Xu, G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020, 97, 829–838. [Google Scholar] [CrossRef]
- Gabarre, P.; Dumas, G.; Dupont, T.; Darmon, M.; Azoulay, E.; Zafrani, L. Acute kidney injury in critically ill patients with COVID-19. Intensive Care Med. 2020, 46, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhu, A.; Li, H.; Zheng, K.; Zhuang, Z.; Chen, Z.; Shi, Y.; Zhang, Z.; Chen, S.; Liu, X.; et al. Isolation of infectious SARS-CoV-2 from urine of a COVID-19 patient. Emerg. Microbes Infect. 2020, 9, 991–993. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.; Xu, S.B.; Lin, Y.X.; Tian, D.; Zhu, Z.Q.; Dai, F.H.; Wu, F.; Song, Z.G.; Huang, W.; Chen, J.; et al. Persistence and clearance of viral RNA in 2019 novel coronavirus disease rehabilitation patients. Chin. Med. J. 2020, 133, 1039–1043. [Google Scholar] [CrossRef]
- Puelles, V.G.; Lutgehetmann, M.; Lindenmeyer, M.T.; Sperhake, J.P.; Wong, M.N.; Allweiss, L.; Chilla, S.; Heinemann, A.; Wanner, N.; Liu, S.; et al. Multiorgan and Renal Tropism of SARS-CoV-2. N. Engl. J. Med. 2020, 383, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Diao, B.; Wang, C.; Wang, R.; Feng, Z.; Tan, Y.; Wang, H.; Wang, C.; Liu, L.; Liu, Y.; Wang, G.; et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. medRxiv 2020. [Google Scholar] [CrossRef]
- Wang, R.; Liao, C.; He, H.; Hu, C.; Wei, Z.; Hong, Z.; Zhang, C.; Liao, M.; Shui, H. COVID-19 in Hemodialysis Patients: A Report of 5 Cases. Am. J. Kidney Dis. 2020, 76, 141–143. [Google Scholar] [CrossRef]
- Wang, H. Maintenance Hemodialysis and COVID-19: Saving Lives With Caution, Care, and Courage. Kidney Med. 2020, 2, 365–366. [Google Scholar] [CrossRef]
- He, Q.; Mok, T.N.; Yun, L.; He, C.; Li, J.; Pan, J. Single-cell RNA sequencing analysis of human kidney reveals the presence of ACE2 receptor: A potential pathway of COVID-19 infection. Mol. Genet. Genom. Med. 2020, 8. [Google Scholar] [CrossRef]
- Lin, W.; Hu, L.; Zhang, Y.; Ooi, J.D.; Meng, T.; Jin, P.; Sing, X.; Peng, L.; Song, L.; Xiao, Z.; et al. Single-cell Analysis of ACE2 Expression in Human Kidneys and Bladders Reveals a Potential Route of 2019-nCoV Infection. bioRxiv 2020. [Google Scholar] [CrossRef]
- Fan, C.; Li, K.; Ding, Y.; Lu, W.; Wang, J. ACE2 Expression in Kidney and Testis May Cause Kidney and Testis Damage after 2019-nCoV Infection. MedRxiv 2020. [Google Scholar] [CrossRef]
- Husain-Syed, F.; Slutsky, A.S.; Ronco, C. Lung-kidney cross-talk in the critically ill patient. Am. J. Respir. Crit. Care Med. 2016, 194, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Leung, W.K.; To, K.F.; Chan, P.K.S.; Chan, H.L.Y.; Wu, A.K.L.; Lee, N.; Yuen, K.Y.; Sung, J.J.Y. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology 2003, 1251011–1251017. [Google Scholar] [CrossRef]
- Holshue, M.L.; DeBolt, C.; Lindquist, S.; Lofy, K.H.; Wiesman, J.; Bruce, H.; Spitters, C.; Ericson, K.; Wilkerson, S.; Tural, A.; et al. First Case of 2019 Novel Coronavirus in the United States. N. Engl. J. Med. 2002, 382, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Jiang, X.; Zhang, Z.; Huang, S.; Zhang, Z.; Fang, Z.; Gu, Z.; Gao, L.; Shi, H.; Mai, L.; et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut 2020, 69, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lou, J.; Bai, Y.; Wang, M. COVID-19 disease with positive fecal and negative pharyngeal and sputum viral tests. Am. J. Gastroenterol. 2020, 115, 790. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.-S.; Wang, X.; Niu, Y.R.; Ye, L.L.; Peng, W.B.; Wang, Z.H.; Yang, W.B.; Yang, B.H.; Zhang, J.C.; Ma, W.L.; et al. Clinical Characteristics of SARS-CoV-2 Infected Pneumonia with Diarrhea. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Pan, L.; Mu, M.; Yang, P.; Sun, Y.; Wang, R.; Yan, J.; Li, P.; Hu, B.; Wang, J.; Hu, C.; et al. Clinical Characteristics of COVID-19 Patients With Digestive Symptoms in Hubei, China. Am. J. Gastroenterol. 2020, 115, 766–773. [Google Scholar] [CrossRef]
- Jin, X.; Lian, J.S.; Hu, J.H.; Gao, J.; Zheng, L.; Zhang, Y.M.; Hao, S.R.; Jia, H.Y.; Cai, H.; Zhang, X.L.; et al. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut 2020, 69, 1002–1009. [Google Scholar] [CrossRef]
- Luo, S.; Zhang, X.; Xu, H. Don’t Overlook Digestive Symptoms in Patients With 2019 Novel Coronavirus Disease (COVID-19). Clin. Gastroenterol. Hepatol. 2020, 18, 1636–1637. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, C.; Tang, L.; Hong, Z.; Zhou, J.; Dong, X.; Yin, H.; Xiao, Q.; Tang, Y.; Qu, X.; et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol. Hepatol. 2020, 5, 434–435. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Zhu, B.; Liang, H.; Fang, C.; Gong, Y.; Guo, Q.; Sun, X.; Zhao, D.; Shen, J.; et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020, 26, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Elmunzer, B.J.; Spitzer, R.L.; Foster, L.D.; Merchant, A.A.; Howard, E.F.; Patel, V.A.; West, M.K.; Qayed, E.; Nustas, R.; Zakaria, A.; et al. Digestive Manifestations in Patients Hospitalized with COVID-19. Clin. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Muller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 581, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA J. Am. Med. Assoc. 2020, 323, 1843–1844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kang, Z.; Gong, H.; Xu, D.; Wang, J.; Li, Z.; Li, Z.; Cui, X.; Xiao, J.; Zhan, J.; et al. Digestive system is a potential route of COVID-19: An analysis of single-cell coexpression pattern of key proteins in viral entry process. Gut 2020, 69, 1010–1018. [Google Scholar] [CrossRef]
- Hashimoto, T.; Perlot, T.; Rehman, A.; Trichereay, J.; Ishiguro, H.; Paolino, M.; Sigl, V.; Hanada, T.; Hanada, R.; Lipinski, S.; et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature 2012, 487, 477–481. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Kruger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Liang, W.; Feng, Z.; Rao, S.; Xiao, C.; Xue, X.; Lin, Z.; Zhang, Q.; Qi, W. Diarrhoea may be underestimated: A missing link in 2019 novel coronavirus. Gut 2020, 69, 1141–1143. [Google Scholar] [CrossRef]
- Słomka, A.; Kowalewski, M.; Żekanowska, E. Coronavirus disease 2019 (COVID–19): A short review on hematological manifestations. Pathogens 2020, 9, 493. [Google Scholar] [CrossRef]
- Ludvigsson, J.F. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. Int. J. Paediatr. 2020, 109, 1088–1095. [Google Scholar] [CrossRef]
- Xu, P.; Zhou, Q.; Xu, J. Mechanism of thrombocytopenia in COVID-19 patients. Ann. Hematol. 2020, 99, 1205–1208. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Plebani, M.; Henry, B.M. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin. Chim. Acta 2020, 506, 2020. [Google Scholar] [CrossRef] [PubMed]
- Maier, C.L.; Truong, A.D.; Auld, S.C.; Polly, D.M.; Tanksley, C.L.; Duncan, A. COVID-19-associated hyperviscosity: A link between inflammation and thrombophilia? Lancet 2020, 395, 1758–1759. [Google Scholar] [CrossRef]
- Biswas, S.; Thakur, V.; Kaur, P.; Khan, A.; Kulshrestha, S.; Kumar, P. Blood clots in COVID-19 patients: Simplifying the curious mystery. Med. Hypotheses 2020, 146, 110371. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Wei, L.; Sun, S.; Xu, C.H.; Zhang, J.; Xu, Y.; Zhu, H.; Peh, S.C.; Korteweg, C.; McNutt, M.A.; Gu, J. Pathology of the thyroid in severe acute respiratory syndrome. Hum. Pathol. 2007, 38, 95–102. [Google Scholar] [CrossRef]
- Benbassat, C.A.; Olchovsky, D.; Tsvetov, G.; Shimon, I. Subacute thyroiditis: Clinical characteristics and treatment outcome in fifty-six consecutive patients diagnosed between 1999 and 2005. J. Endocrinol. Investig. 2007, 30, 631–635. [Google Scholar] [CrossRef]
- Chaux-Bodard, A.-G.; Deneuve, S.; Desoutter, A. Oral manifestation of Covid-19 as an inaugural symptom? J. Oral Med. Oral Surg. 2020, 26, 18. [Google Scholar] [CrossRef]
- Brancatella, A.; Ricci, D.; Viola, N.; Sgrò, D.; Santini, F.; Latrofa, F. Subacute Thyroiditis After Sars-COV-2 Infection. J. Clin. Endocrinol. Metab. 2020, 105, 2367–2370. [Google Scholar] [CrossRef]
- Oxley, T.J.; Mocco, J.; Majidi, S.; Kellner, C.P.; Shoirah, H.; Singh, I.P.; De Leacy, R.A.; Shigematsu, T.; Ladner, T.R.; Yaeger, K.A.; et al. Large-Vessel Stroke as a Presenting Feature of Covid-19 in the Young. N. Engl. J. Med. 2020, 382, e60. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Tsao, H.S.; Chason, H.M.; Fearon, D.M. Immune thrombocytopenia (ITP) in a pediatric patient positive for SARS-CoV-2. Pediatrics 2020, 146. [Google Scholar] [CrossRef]
- Zulfiqar, A.-A.; Lorenzo-Villalba, N.; Hassler, P.; Andrès, E. Immune Thrombocytopenic Purpura in a Patient with Covid-19. N. Engl. J. Med. 2020, 382, e43. [Google Scholar] [CrossRef]
- Nirenberg, M.S.; Del Mar Ruiz Herrera, M. Foot Manifestations in a COVID-19 Positive Patient: A Case Study. J. Am. Podiatr. Med. Assoc. 2020. [Google Scholar] [CrossRef]
- Arora, G.; Kassir, M.; Jafferany, M.; Galadari, H.; Lotti, T.; Satolli, F.; Sadoughifar, R.; Sitkowska, Z.; Goldust, M. The COVID-19 outbreak and rheumatologic skin diseases. Dermatol. Ther. 2020, 33. [Google Scholar] [CrossRef]
- Flammer, A.J.; Anderson, T.; Celermajer, D.S.; Creager, M.A.; Deanfield, J.; Ganz, P.; Hamburg, N.M.; Luscher, T.F.; Shechter, M.; Taddei, S.; et al. The assessment of endothelial function: From research into clinical practice. Circulation 2012, 126, 753–767. [Google Scholar] [CrossRef]
- Zhou, Y.; Fu, B.; Zheng, X.; Wang, D.; Zhao, C.; Qi, Y.; Sun, R.; Tian, Z.; Xu, X.; Wei, H. Aberrant pathogenic GM-CSF + T cells and inflammatory CD14+ CD16+ monocytes in severe pulmonary syndrome patients of a new coronavirus. Natl. Sci. Rev. 2020. [Google Scholar] [CrossRef]
- Danzi, G.B.; Loffi, M.; Galeazzi, G.; Gherbesi, E. Acute pulmonary embolism and COVID-19 pneumonia: A random association? Eur. Heart J. 2020, 41, 1858. [Google Scholar] [CrossRef]
- Gandolfini, I.; Delsante, M.; Fiaccadori, E.; Zaza, G.; Manenti, L.; Antoni, A.D.; Peruzzi, L.; Riella, L.V.; Cravedi, P.; Maggiore, U. COVID-19 in kidney transplant recipients. Am. J. Transpl. 2020, 20, 1941–1943. [Google Scholar] [CrossRef]
- Guillen, E.; Pineiro, G.J.; Revuelta, I.; Rodriguez, D.; Bodro, M.; Moreno, A.; Campistol, J.M.; Diekmann, F.; Ventura-Aguiar, P. Case report of COVID-19 in a kidney transplant recipient: Does immunosuppression alter the clinical presentation? Am. J. Transplant. 2002, 20, 1875–1878. [Google Scholar] [CrossRef]
- Hsu, J.J.; Gaynor, P.; Kamath, M.; Fan, A.; Al-Saffar, F.; Criz, D.; Nsair, A. COVID-19 in a high-risk dual heart and kidney transplant recipient. Am. J. Transplant. 2020, 20, 1911–1915. [Google Scholar] [CrossRef]
- Li, F.; Cai, J.; Dong, N. First cases of COVID-19 in heart transplantation from China. J. Heart Lung Transpl. 2020, 39, 496–497. [Google Scholar] [CrossRef]
- Cox, M.J.; Loman, N.; Bogaert, D.; O’Grady, J. Co-infections: Potentially lethal and unexplored in COVID-19. Lancet Microbe 2020, 1, e11. [Google Scholar] [CrossRef]
- Wang, H.; Ding, Y.; Li, X.; Yang, L.; Zhang, W.; Kang, W. Fatal Aspergillosis in a Patient with SARS Who Was Treated with Corticosteroids. N. Engl. J. Med. 2003, 349, 507–508. [Google Scholar] [CrossRef]
- Alanio, A.; Dellière, S.; Fodil, S.; Bretagne, S.; Mégarbane, B. Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Lancet Respir. Med. 2020, 8, e48–e49. [Google Scholar] [CrossRef]
- Koehler, P.; Cornely, A.A.; Bottiger, B.E.; Dusse, F.; Eichenauer, A.S.; Fuchs, F.; Hallek, M.; Jung, N.; Klein, F.; Persigehl, T.; et al. COVID-19 associated pulmonary aspergillosis. Mycoses 2002, 63, 528–534. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Wang, M.; Luo, L.; Bu, H.; Xia, H. One case of coronavirus disease 2019 (COVID-19) in a patient co-infected by HIV with a low CD4+ T-cell count. Int. J. Infect. Dis. 2020, 96, 148–150. [Google Scholar] [CrossRef]
- Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 18 January 2021).
| Case/Study | Symptoms (Neurological) |
|---|---|
| 79-year-old man (Positive-Oropharyngeal swab) [53] | Fever, acute loss of consciousness, and bilateral extensor plantar reflexes. Intracerebral hemorrhage in the right brain hemisphereIntraventricular and subarachnoid hemorrhage. |
| The first case of SARS-CoV-2 associated meningitis. Absence of SARS-CoV-2 RNA in nasopharyngeal swab.Positive in CSF [54] | Generalized fatigue and fever, transient generalized seizures, and neck stiffness. Convulsion accompanied by unconsciousness. Hyperintense signal in the right mesial temporal lobe in the brain along with significant paranasal sinusitis. |
| SARS-CoV-2 positive male with encephalitis, however, CSF is negative [55] | Fever, shortness of breath, lymphopenia, multiple subpleural ground-glass opacities in the chest, and myalgia. Progression of consciousness towards the confusion with signs of meningeal irritation. |
| Case/Study | Symptoms (Renal) |
|---|---|
| 85 COVID-19 positive patients Acute kidney injury (AKI) in 27% of patients [68] | High risk of AKI associated with age >60 years, coexisting hypertension, and coronary artery disease. Severe acute tubular injury with macrophage infiltration, and detection of viral antigen (6) |
| 701 hospitalized patients [63] | Proteinuria (44%), Haematuria (27%), and Acute kidney injury (3.2%) |
| 201 maintenance hemodialysis (MHD) patients. Five had COVID-19 pneumonia [69] | Diarrhea as common presenting symptoms (4/5), fever (3/5), fatigue (3/5), dyspnoea (2/5), and abdominal pain (2/5), lymphopenia (5/5), with ground-glass opacity. Fever, cough, and dyspnoea were absent. |
| 230 haemodialysis patients; 16.1% (37) diagnosed with COVID-19 [70] | Lymphopenia, lower levels of inflammatory cytokines, and mild clinical disease |
| Case/Study | Symptoms (GIT/others) |
|---|---|
| 26/84 COVID-19 patients with diarrhea [79] | Headache, myalgia, fatigue, cough, sputum production, nausea, vomiting |
| 204 COVID-19 patients GIT symptoms in 99 (48.5%) Cross-sectional study, China [80] | Anorexia (83.8%) Diarrhea (29.3%) |
| 651 COVID-19 patients 11.4% (74) patients with GI symptoms 28% lack respiratory symptoms [81] | Diarrhea, nausea, and vomiting |
| 95 COVID-19 patients 61.1% cases with GI symptoms 52.4% (22) positive faecal samples [77] | Diarrhoea (24.2%), Nausea (17.9%), Elevated transaminases (32.6%) |
| 1141 COVID-19 patients GIT symptoms in 16% of patients A retrospective study, China [82] | Anorexia, nausea, vomiting, diarrhea, abdominal pain |
| Manifestation Type | Case and Studies | Presentation/Symptoms |
|---|---|---|
| Oral | 45-year-old female; SARS-CoV-2 positive Nasopharyngeal swab (NPA) [101] | Irregular oral ulcer on the dorsal side of the tongue, painful inflammation of tongue papilla, macular erythematous lesion, vasculitis |
| Thyroid (Subacute thyroiditis) | 18-year-old female positive for SARS-CoV-2; (Oropharyngeal swab) [102] | Thyroid enlargement The bilateral and diffuse hypoechoic area around the neck |
| Large-vessel stroke | 5 SARS-CoV-2 positive patients (<50 years of age) [103] | New-onset symptoms of severe large-vessel ischemic stroke with the mean National Institutes of Health Stroke Scale (NIHSS) score of 17. Partial infarction of the right middle cerebral artery with a partially occlusive thrombus in the right carotid artery. Patchy ground-glass opacities in bilateral lung apices |
| Endothelitis | 71-year-old male renal transplant recipient with coronary artery disease and arterial hypertension diagnosed COVID-19 positive on post mortem analysis of transplanted kidney [104] | Viral inclusion structures in endothelial cells. Inflammatory cells and apoptotic bodies accumulation in the heart and lungs |
| Immune thrombocytopenia | 10-year-old SARS-CoV-2 positive patient with severe thrombocytopenia and wet purpura [105] 65-year-old SARS-CoV-2 positive woman with hypertension and autoimmune hypothyroidism [106] | Mouth: Development of purple lesions. Petechiae concentrated on lower extremities, chest and neck and ecchymoses in the popliteal regions and shins. Fatigue, fever, cough, and abdominal discomfort. Developed lower-extremity purpura, epistaxis, subarachnoid microhemorrhage, and intracranial hemorrhage |
| Pedo/axillary manifestation | 16-year-old COVID-19 positive female from Ciudad Real [107] 21% (7) critically ill COVID-19 patients with acromegaly [22] | Gait alteration of red-violet lesions, itching, pain with mild swelling of both feet, nausea, dizziness, headaches, and dry eyes. Neuropathic involvement of peripheral nervous system (neuralgia) Limb ischemic symptoms: acrops, cyanosis, plantar plaques, dry gangrene, and blood blisters on the feet and hands. Significant increase in D-dimer, fibrinogen, prothrombin time, hypercoagulation, and or disseminated intravascular coagulation causing micro thrombosis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thakur, V.; Ratho, R.K.; Kumar, P.; Bhatia, S.K.; Bora, I.; Mohi, G.K.; Saxena, S.K.; Devi, M.; Yadav, D.; Mehariya, S. Multi-Organ Involvement in COVID-19: Beyond Pulmonary Manifestations. J. Clin. Med. 2021, 10, 446. https://doi.org/10.3390/jcm10030446
Thakur V, Ratho RK, Kumar P, Bhatia SK, Bora I, Mohi GK, Saxena SK, Devi M, Yadav D, Mehariya S. Multi-Organ Involvement in COVID-19: Beyond Pulmonary Manifestations. Journal of Clinical Medicine. 2021; 10(3):446. https://doi.org/10.3390/jcm10030446
Chicago/Turabian StyleThakur, Vikram, Radha Kanta Ratho, Pradeep Kumar, Shashi Kant Bhatia, Ishani Bora, Gursimran Kaur Mohi, Shailendra K Saxena, Manju Devi, Dhananjay Yadav, and Sanjeet Mehariya. 2021. "Multi-Organ Involvement in COVID-19: Beyond Pulmonary Manifestations" Journal of Clinical Medicine 10, no. 3: 446. https://doi.org/10.3390/jcm10030446
APA StyleThakur, V., Ratho, R. K., Kumar, P., Bhatia, S. K., Bora, I., Mohi, G. K., Saxena, S. K., Devi, M., Yadav, D., & Mehariya, S. (2021). Multi-Organ Involvement in COVID-19: Beyond Pulmonary Manifestations. Journal of Clinical Medicine, 10(3), 446. https://doi.org/10.3390/jcm10030446










