1. Introduction
The spinal column consists of a series of intervertebral discs and vertebrae that originates at the base of the skull and extends to the coccyx. The complete structure is typically divided into five parts: the cervical region (C1-C7), the thoracic region (T1-T12), the lumbar region (L1-L5), the sacral region (S1-S5), and the coccygeal region (Co1-Co4/5). It provides flexibility and mobility for the body and gives protection to the spinal cord [
1].
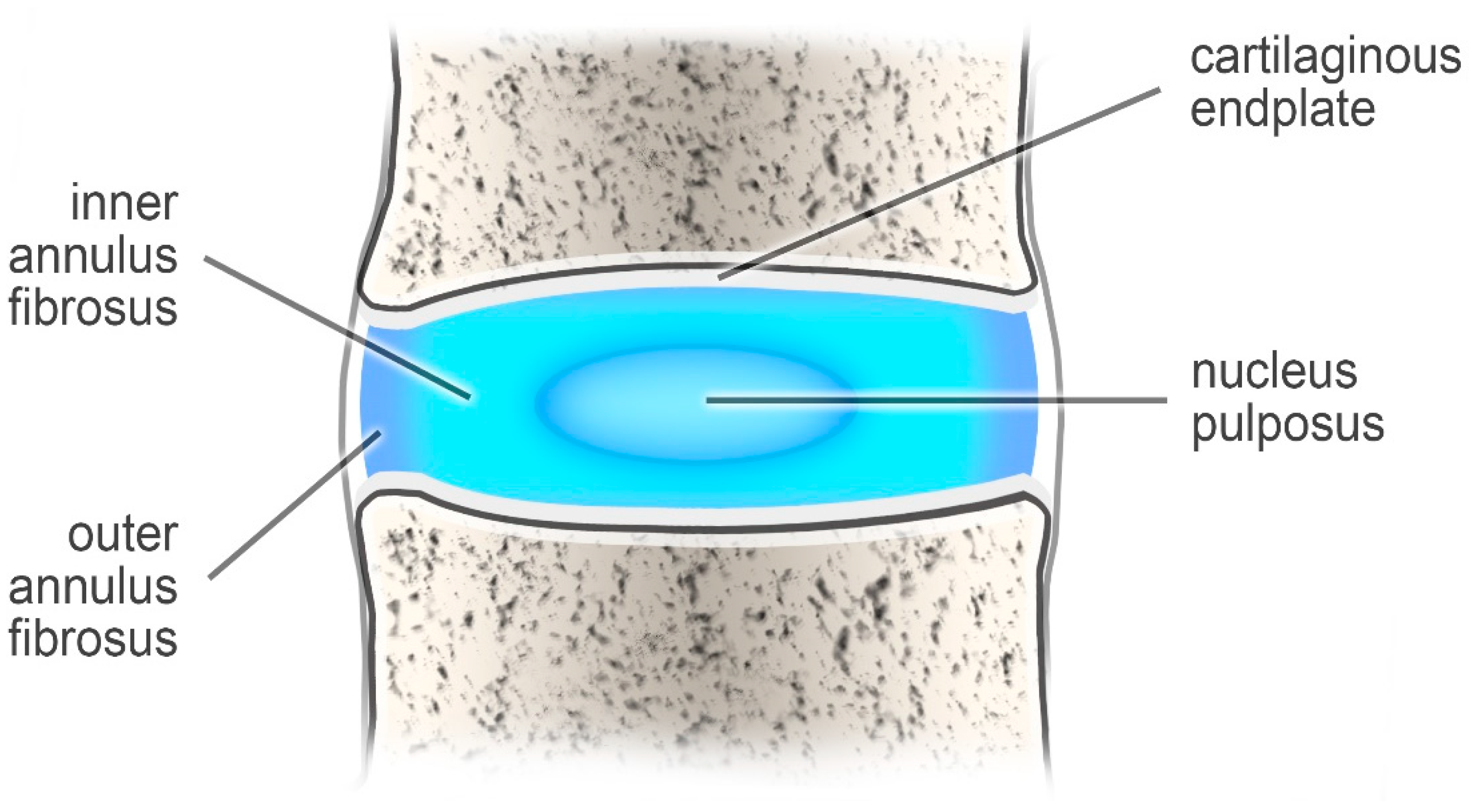
The intervertebral discs support the spinal column and behave like shock-absorbing cushions against the axial loading of the human body [
1]. These structures are built of two components: a nucleus pulposus (which is the inner part) and an outer annulus fibrosus (AF) [
2] (
Figure 1).
The nucleus pulposus, which consists of collagen (type II), makes up 20 percent of the dry weight of the disc. It also contains numerous proteoglycans that maintain hydrostatic pressure by retaining water. The other ingredients are water and chondrocyte-like cells. This composition helps the nucleus pulposus remain elastic, capable of absorbing compression, and flexible under stress forces [
2,
3].
In turn, the annulus fibrosus is formed from collagen type I, which forms a fibrous tissue around the nucleus pulposus. The annulus fibrosus is attached to the vertebral body by Sharpey fiber [
3].
Intervertebral disc pathology is a common name for various pathologies connected with intervertebral discs. This group includes degeneration of discs, disc-associated pain, disc herniation, and histology findings. Disc degeneration can be graded on MRI T2 spin-echo weighted images using a grading system (grade 1–5) proposed by Pfirmann [
4]. Grade 1 is represented by an inhomogeneous disc with bright hyperintense white signal intensity and normal disc height. In turn, grade 5 disc degeneration is characterized by an inhomogeneous disc with a hypointense black signal intensity. There is no clear distinction between the AF and NP, and the disc space is collapsed [
5]. There are also histological findings associated with disc degeneration. An increase in cell density (chondrocyte proliferation), the occurrence of granular changes, structural alterations with tears and clefts, and a severe increase in acid mucopolysaccharides (mucous degeneration) with dark-blue staining areas around clones of chondrocytes are the main characteristic features observed during histomorphological examination [
6]. Another pathology that can be included in intervertebral disc pathologies is low back pain. However, it may be caused not only by disc herniation, but also by infection, rheumatoid arthritis, fracture, osteoporosis, or tumors [
7].
Disc herniation is an intervertebral disc pathology that occurs when either part of the whole of the nucleus pulposus protrudes through the annulus fibrosus. The most common cause is chronic herniation, i.e., disc degeneration occurring due to reduced proteoglycan content, in which the nucleus pulposus is weakened due to a loss of hydration. In contrast, acute herniation can occur as a result of trauma, resulting in the nucleus pulposus extruding through a defect in the annulus fibrosus. Many genetic, mechanical, and behavioral factors are believed to be responsible [
8]. MRI is a first-choice examination method. It allows disc herniations to be classified according to their shape or extent and distinguishes three types: protrusion (largest diameter of the disc material projecting beyond the normal margin of this disc is narrower than the general width of the base), extrusion (characterized by a base length less than the height), and sequestration (a subtype of extrusion, which occurs when there is no connection between the herniated material and the intervertebral disc) [
9]. There is also a disc bulge (which involves more than 25% of the circumference), another kind of discopathy that MRI also reveals. It is important that the prevalence of disc bulge in asymptomatic individuals increases from 30% of those 20 years of age to 84% of those 80 years of age. In turn, more advanced disc herniations usually compress neural structure, which causes various types of pain [
10].
Due to the biomechanical forces in the flexible parts of the spinal column, the cervical and lumbar regions of spinal column are more susceptible to occurrence of disc herniation. Thoracic disc herniation is much less common [
8].
3. Factors Formerly and Today
Disk herniation is known to be caused by genetic, mechanical, and behavioral factors. However, even recently, this was not believed to be the case, with previous models of dysfunction being based on environmental and behavioral factors [
16]. These were later disproven [
17].
It was believed that the main causes of disc herniation were aging and mechanical insults, and that smoking status, aging, obesity, height, sciatic axial overloading, or occupation had the greatest influence on the presence of intervertebral disc pathology [
17]. Although both obesity and height can be classified as anthropometric measurements, the latter is rarely raised in studies [
18]. Even in 1999, the prevailing belief in many countries was that work-related mechanical factors like heavy physical loading, lifting, bending, and twisting played the main role in lumbar degeneration [
19].
Environmental, i.e., mechanical and behavioral, factors associated with occupation (physical workload, hard work, a working period longer than eight hours, and stress at work) are believed to be the major factors in intervertebral disc pathology [
20].
A number of studies from the last century focused solely on the environmental factors of intervertebral disc pathology, with no mention of the possible influence of genetic factors. For example, one study found that men who drove a motor vehicle for half or more of their working time were three times more vulnerable to acute lumbar herniation than other people; however, this study did not assume any possible genetic predisposition [
21]. Another study proposed that the main mechanism in the degeneration of the intervertebral disc was the nutritional decline of the central disc. This was suggested to lead to collection of waste products, the degeneration of matrix molecules, and a fall in pH, which may cause cell death [
22]. This is true, but later studies demonstrated that polymorphisms or mutations among genes associated with substances of connective tissue also accelerated the degeneration of intervertebral disc.
A 1992 study included the contribution of genetic factors; however, these were believed to play a lesser role than environmental factors such as age, gender, cigarette smoking, exposure to vehicular travel, and occupation [
23].
However, the use of new genetic and proteomic tools has allowed the effects of genetic variation to be researched. They made it possible to link genetic variations with the occurrence of different diseases and molecular-level processes to pathologies such as disc herniation [
17]. These new molecular studies cast doubt on the previous belief that environmental factors are the main (or only) cause of intervertebral disc pathologies. Later, in 1998, a number of gene alterations were found to be associated with disc degeneration [
24].
Over the years, the classical theory of aging and wear-and-tear evolved into a sophisticated model of a multiple-causative disease based on molecular and genetic alterations [
17,
25]. Polymorphisms or gene mutations were found to be associated with disc herniations in both humans and animals [
17,
25].
A number of new studies have confirmed that genetic anomalies are critical contributors to the initiation and further development of intervertebral disc pathologies [
17,
24,
26,
27,
28,
29].
The Twin Spine Study, a multinational research project that followed twins from Canada, Finland, and the United States, examined how occupational exposure, vibrational exposure, smoking, anthropomorphic characteristics, and genetics affect the occurrence of disc degeneration. The findings indicated that pairs of identical twins with different jobs, physical activity, and interests demonstrated similar levels of disc degeneration, suggesting that genetic factors have much larger influence on disc degeneration than expected [
16].
Another study of 75 monozygotic twin pairs that were examined twice, with five years in between measurements, found familial aggregation, i.e., genetic and environmental factors that were identical between twins, to explain 47% to 66% of the variance in progression of degenerative signs on lumbar MRI. In turn, environmental factors such as occupational physical loading and resistance training explained only 2% to 10% of the progression in the degenerative signs in lumbar MRIs. The main conclusion of this research was therefore that factors associated with genes play a dominant role in disc degeneration, while environmental factors only have a modest one [
30].
However, a 2009 study by Battié et al. of 152 monozygotic and 148 dizygotic male twin pairs [
31] indicated that genetic factors only have a partial influence on the development of disc degeneration, according to phenotype and spinal level. Estimates of heredity varied from 29% to 54%, depending on the phenotype and spinal level. In turn, disc narrowing and bulging had a mainly common genetic pathway [
31].
In addition, Videman and Battie reported that the influence of occupational risk factors and occupation on the occurrence some intervertebral disc diseases was also modest, especially in comparison with contribution of genetic factors [
19]. Kepler et al. [
29] reported that genetic factors account for up to 75% of individual susceptibility to intervertebral disc degeneration, and that environmental factors such as occupational exposure and smoking do not have the greatest impact on disc pathologies. In addition, a study of the effects of anthropometrics, lifting strength, and physical activities on disc degeneration found that occupational and leisure physical activity, body weight, and lifting strength have a low impact on occurrence of disc degeneration [
32].
A study of 63 patients under the age of 21 with confirmed lumbar disc herniation found 32% to have a positive family history. In contrast, the prevalence among the control group was only 7%, i.e., approximately one fifth of the group with a positive family history [
33]. Hence, genetic factors may play a significant role.
A recent systematic review about genetic predisposition to disc degeneration by Filho et al. found that genes play the main role in the occurrence of disc degeneration, and that environmental factors have less importance. Obesity, occupation, smoking status, diabetes, and alcohol consumption were described as aggravating factors. Hence, disc degeneration appears to be a multifactorial process, but with genetic factors having the greatest influence [
34].
A case-control study of risk factors for herniated disc in the lumbar region, including BMI, smoking index, level of education, occupation, socio-economic status, intensity of physical labor at home and work, and self-assessed limitation in physical activity, was performed on a population from Croatian Islands. Hereditary factors were found to be strongly associated with disc herniation; hence, such isolated populations appear to be valuable for detecting such effects due to decreased genetic and environmental variability. The findings indicated that a number of environmental factors, such as level of education, cardiovascular morbidity, smoking status, intensity of physical work, or socio-economic status, did not have a meaningful influence on the occurrence the lumbar intervertebral disc herniation in this population [
35].
Genetic factors appear to play the main role in pathology of diseases associated with disc degeneration, and they may be influenced by environmental factors such as spine injury, occupation, smoking, or aging [
29] In turn, environmental factors may only serve to accelerate this process of disc degeneration driven by genetic polymorphisms.
Although these findings provide a detailed picture of the etiology of intervertebral disc degeneration, there are still many unsolved issues connected with the magnitude and mechanism of genetic factors. In addition, the studies had various limitations that may have affected the correctness of some research results. For example, some studies used a small cohort, resulting in possible false-positive or false-negative errors.
In addition, to design genetic therapy, future studies should carefully examine the loci of genes connected with intervertebral disc degeneration. Such progress would be supported by progress in molecular biology and advancements in imaging technology, allowing faster diagnosis of intervertebral disc degeneration [
17].
Regarding individual risk factors influencing disc degeneration, both environmental and genetic factors interact in the formation of disc herniation. Although intervertebral disc disease has multiple causes, the key role appears to be played by changes in genes at the molecular level [
16,
17].
7. Statistics
One of the main intervertebral disc pathologies is disc herniation. Some studies suggest that the frequency of herniated discs is influenced by non-genetic factors (for example: age group, gender, occupation).
A study carried out by Cheng et al. [
75] investigated whether healthcare work is associated with increased occurrence of cervical disc herniation.
This result shows that physicians are more exposed to occurrence of cervical disc herniation than the reference group of non-healthcare providers. The same situation was observed in the case of non-physician HCPs and non-HCPs references, so we concluded that healthcare work is associated with an increased occurrence of cervical disc herniation (
Table 1).
Table 2 shows whether physicians are more vulnerable to this pathology than non-physician HCPs. There was also a breakdown by gender.
We observed that generally, physicians are more vulnerable to the occurrence of cervical disc herniation than non-physician HCPs. Unfortunately, these dependencies were not statistically significant. The same situation occurred in the stratification analysis of gender.
Cheng et al. [
75] also analyzed the occurrence of intervertebral cervical disc herniations among physicians according to specialization. The results are shown in
Table 3.
We concluded that orthopedists are most vulnerable to the occurrence of cervical disc herniation. Unfortunately, these statistics were not statistically significant.
As it mentioned above regarding risk factors, a heavy physical workload and occupations requiring harder effort are associated with a possibility of increased occurrence of disc herniation. Longer working hours may be the factor that causes physicians to be in a higher-risk group. This operating mode may lead to a poor posture and lifestyle, and it is a risk factor for the development of cervical disc herniation.
Young-Ki et al. [
76] carried out another study regarding the occurrence of cervical disc herniation and its association with age, gender, and health-insurance eligibility. However, we will only deal with the frequency distribution of this pathology between individual age groups and gender.
The number of patients increased with aging in the male and female groups. A small decrease was noticed between the 50–59 and 60–69 age groups in the female population. The frequency of cervical disc herniation was higher among female patients than male patients in every age group (
Table 4) [
76].
The next study was carried out by Tabesh et al. [
77]. Briefly, 269 patients (102 women and 167 men) underwent a straight leg-raising test. A positive result was observed in 182 patients (67.7%). A chi-square test showed that the gender distribution between the two groups was statistically significant (
p = 0.003). The distribution of the level of disc herniation between the two groups and the distribution of age between the two groups was also statistically significant (
p = 0.002 and
p <0.001, respectively). In these cases, the Fischer Exact test was used. The Mann–Whitney U-test showed that the distribution of severity between the two groups was also statistically significant (
p < 0.001). Considering gender, the vast majority of patients were men (62.5%). The largest group regarding age distribution was the 30–39 age group (
Table 5) [
65].
Another study [
76] showed the relationship between the occurrence of disc herniation and various factors. The number of patients with LDH in 100,000 individuals according to gender, age and health-insurance eligibility was studied and assigned to the appropriate groups. Given the results in
Table 6, we suggest that the number of patients increased with age. In addition, more male patients were found in the 20–39 age group. The situation was completely different among the surveyed population over 40 years of age. Lumbar disc herniation was more common in women [
76].
Another study carried out by Jong-myung et al. [
78] that was based on a national database in the Republic of Korea (ROK) from January 2008 to December 2016. The number of patients with diagnosed disc herniation increased from 2008 to 2012, and then fell. The patients were divided into nine age groups; the results of this research are presented in
Table 7 (percentage distribution)
Individuals were also divided according to gender. In 2008, there were higher numbers of lumbar disc herniation in female patients than in male patients in their 30s, and 1.6 times higher than patients in their 60s. In 2016, the crude incidence was also higher in female patients than male patients in the 40s group, and 1.5 times higher in the 60s group [
78].
Table 8 compares the results of the studies with regard to specific age groups.
These results show that the studies carried out by Young-Ki et al. [
76] and Jong-myung et al. [
78] had a similar frequency distribution in terms of age groups. Tabesh [
77] obtained another crude incidence of lumbar disc herniation.
Despite the fact that the above studies showed a relationship between the above-mentioned factors and disc herniation, it should be remembered that these are predisposing factors, the most important of which is their interaction with genetic factors (which play a main role in occurrence the intervertebral disc pathologies).