Membrane of Plasma Rich in Growth Factors in Primary Pterygium Surgery Compared to Amniotic Membrane Transplantation and Conjunctival Autograft
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Outcome Measures
2.3. PRGF Preparation
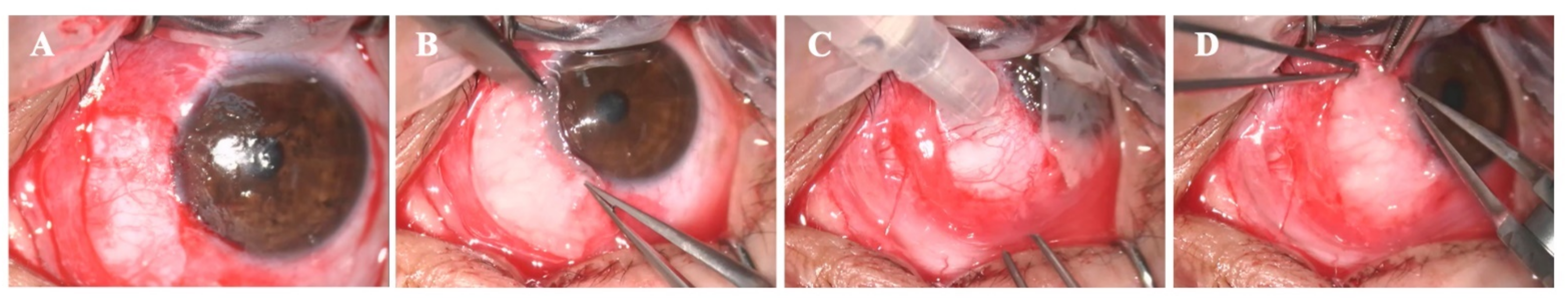
2.4. Surgical Procedures
2.5. Statistical Analysis
3. Results
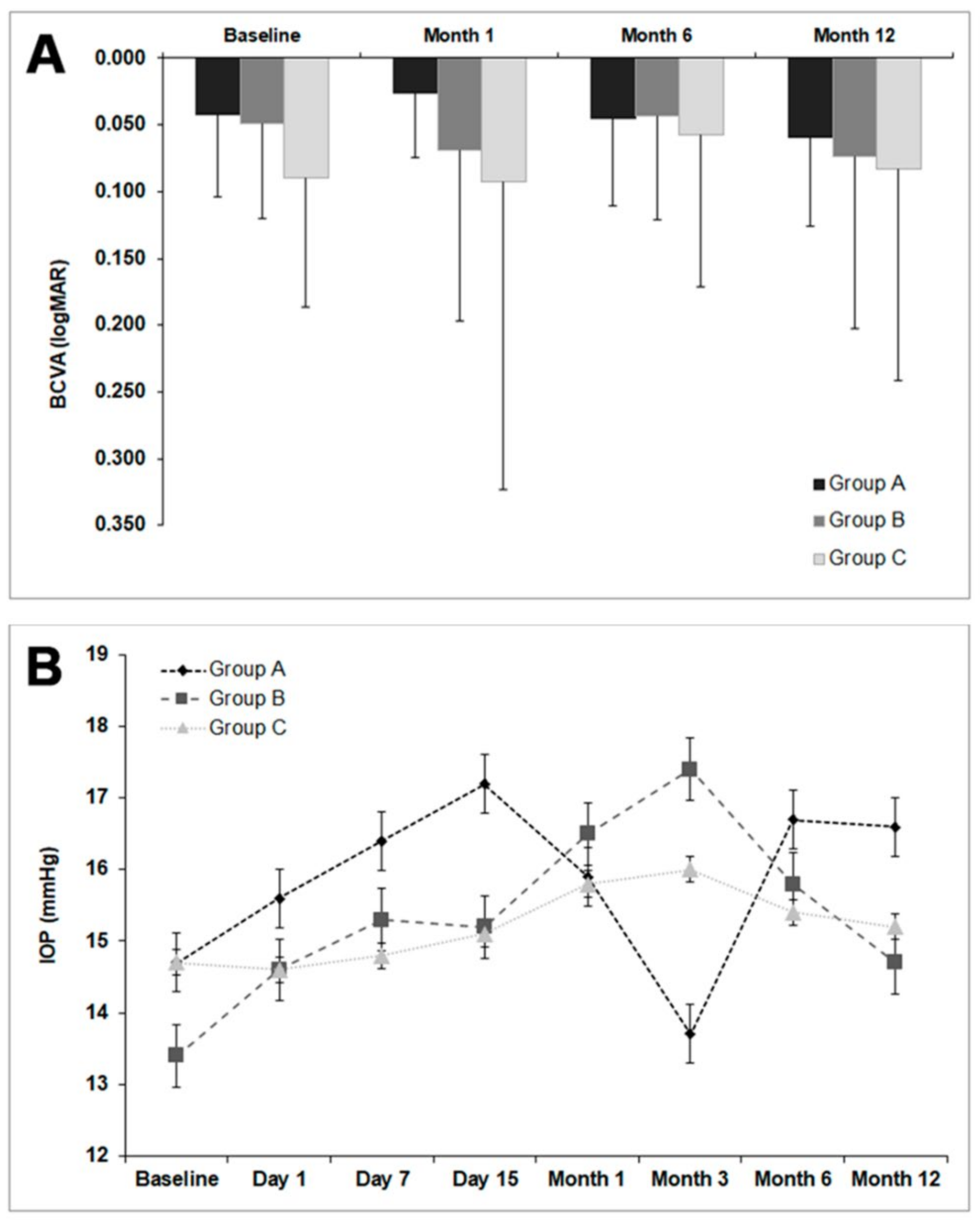
3.1. Visual Acuity and Intraocular Pressure
3.2. Pterygium Measurement (AS-OCT)
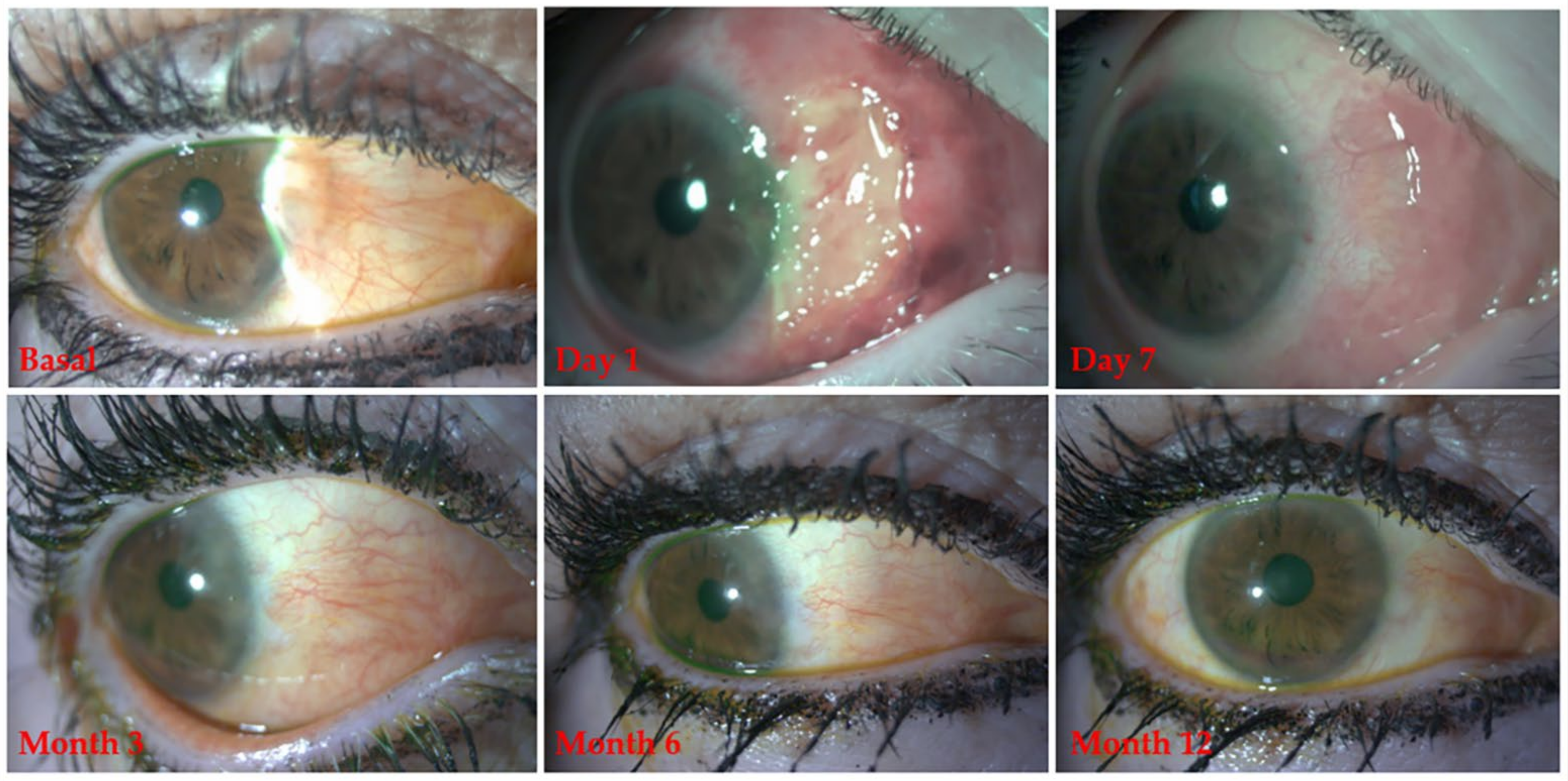
3.3. Pterygium Recurrence: Solomon Scale
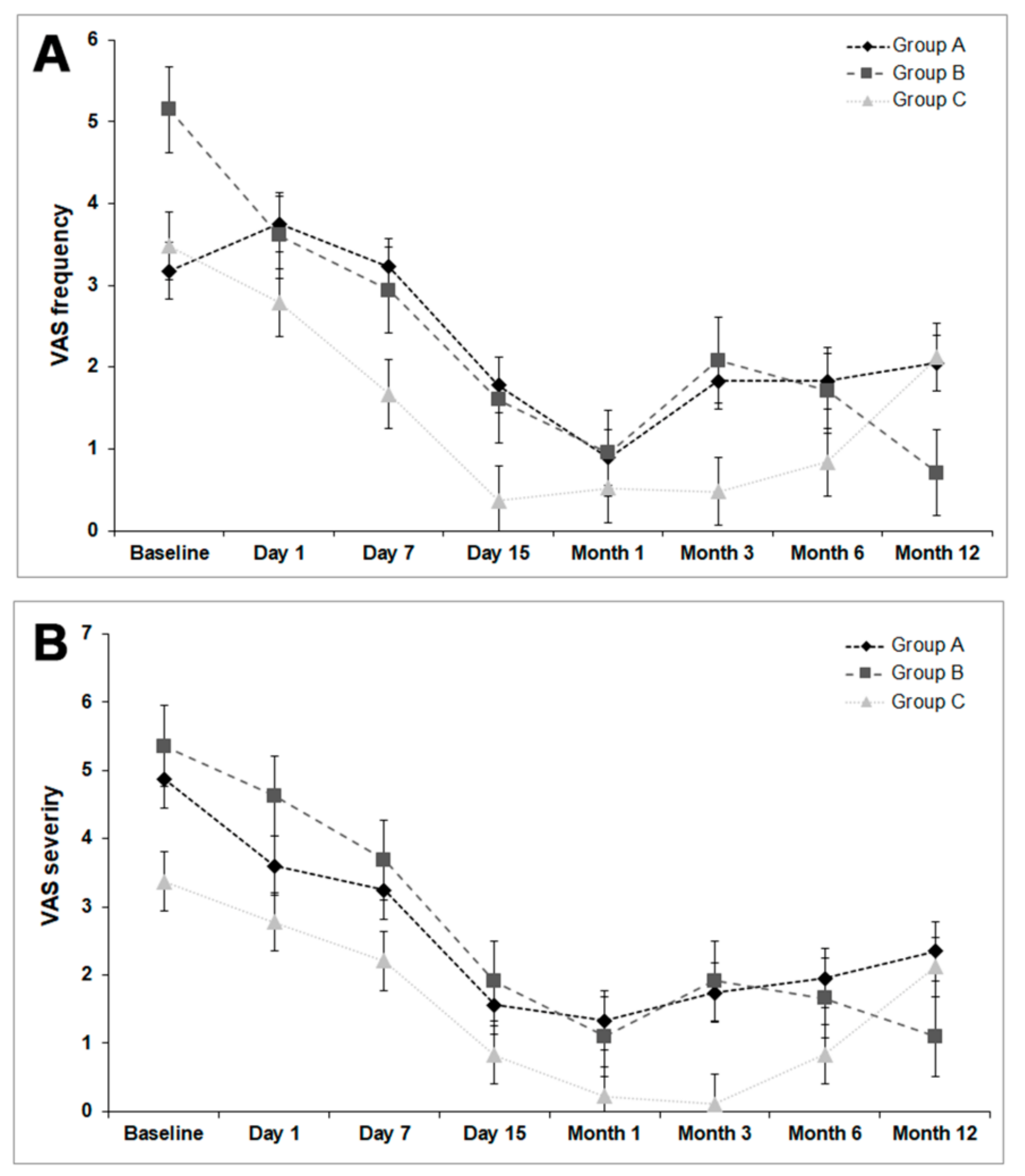
3.4. Ocular Surface Symptom Assessment (VAS and OSDI)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tan, D.T.; Chee, S.P.; Dear, K.B.; Lim, A.S. Effect of pterygium morphology on pterygium recurrence in a controlled trial comparing conjunctival autografting with bare sclera excision. Arch. Ophthalmol. 1997, 115, 1235–1240. [Google Scholar] [CrossRef]
- Solomon, A.S. Pterygium. Br. J. Ophthalmol. 2006, 90, 665–666. [Google Scholar] [CrossRef] [PubMed]
- Di Girolamo, N.; Coroneo, M.T.; Wakefield, D. UVB-elicited induction of MMP-1 expression in human ocular surface epithelial cells is mediated through the ERK1/2 MAPK-dependent pathway. Invest. Ophthalmol. Vis. Sci. 2003, 44, 4705–4714. [Google Scholar] [CrossRef]
- Nolan, T.M.; Di Girolamo, N.; Sachdev, N.H.; Hampartzoumian, T.; Coroneo, M.T.; Wakefield, D. The role of ultraviolet irradiation and heparin-binding epidermal growth factor-like growth factor in the pathogenesis of pterygium. Am. J. Pathol. 2003, 162, 567–574. [Google Scholar] [CrossRef]
- Tsai, Y.-Y.; Cheng, Y.-W.; Lee, H.; Tsai, F.-J.; Tseng, S.-H.; Lin, C.-L.; Chang, K.-C. Oxidative DNA damage in pterygium. Mol. Vis. 2005, 11, 71–75. [Google Scholar] [PubMed]
- Rezvan, F.; Khabazkhoob, M.; Hooshmand, E.; Yekta, A.; Saatchi, M.; Hashemi, H. Prevalence and risk factors of pterygium: A systematic review and meta-analysis. Surv. Ophthalmol. 2018, 63, 719–735. [Google Scholar] [CrossRef]
- Viso, E.; Gude, F.; Rodriguez-Ares, M.T. Prevalence of pinguecula and pterygium in a general population in Spain. Eye 2011, 25, 350–357. [Google Scholar] [CrossRef]
- Iradier, M.T. Cirugía del Pterigión. In Monografía de la Sociedad Española de Oftalmología; Macline: Madrid, Spain, 2006. [Google Scholar]
- Akinci, A.; Zilelioglu, O. Comparison of limbal-conjunctival autograft and intraoperative 0.02% mitomycin-C for treatment of primary pterygium. Int. Ophthalmol. 2007, 27, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Ozdamar, Y.; Mutevelli, S.; Han, U.; Ileri, D.; Onal, B.; Ilhan, O.; Karakaya, J.; Zilelioglu, O. A comparative study of tissue glue and vicryl suture for closing limbal-conjunctival autografts and histologic evaluation after pterygium excision. Cornea 2008, 27, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Ozer, A.; Yildirim, N.; Erol, N.; Yurdakul, S. Long-term results of bare sclera, limbal-conjunctival autograft and amniotic membrane graft techniques in primary pterygium excisions. Int. J. Ophthalmol. Z. Augenheilkd. 2009, 223, 269–273. [Google Scholar] [CrossRef]
- Benitez-Herreros, J.; Perez-Rico, C.; Montes-Mollon, M.A.; Gomez-San-Gil, Y.; Teus-Guezala, M.A. Endothelial cells analysis after intraoperative mitomycin-C adjuvant pterygium simple excision surgery: A pilot study. Arch. Soc. Esp. Oftalmol. 2010, 85, 11–15. [Google Scholar] [CrossRef]
- Koranyi, G.; Artzen, D.; Seregard, S.; Kopp, E.D. Intraoperative mitomycin C versus autologous conjunctival autograft in surgery of primary pterygium with four-year follow-up. Acta Ophthalmol. 2012, 90, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Mastropasqua, L.; Carpineto, P.; Ciancaglini, M.; Enrico Gallenga, P. Long term results of intraoperative mitomycin C in the treatment of recurrent pterygium. Br. J. Ophthalmol. 1996, 80, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Clearfield, E.; Muthappan, V.; Wang, X.; Kuo, I.C. Conjunctival autograft for pterygium. Cochrane Database Syst. Rev. 2016, 2, CD011349. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Sano, Y.; Kinoshita, S. Conjunctival inflammation induces Langerhans cell migration into the cornea. Curr. Eye Res. 2000, 21, 550–553. [Google Scholar] [CrossRef]
- Elwan, S.A.M. Comparison between sutureless and glue free versus sutured limbal conjunctival autograft in primary pterygium surgery. Saudi J. Ophthalmol. Off. J. Saudi Ophthalmol. Soc. 2014, 28, 292–298. [Google Scholar] [CrossRef]
- Kim, H.H.; Mun, H.J.; Park, Y.J.; Lee, K.W.; Shin, J.P. Conjunctivolimbal autograft using a fibrin adhesive in pterygium surgery. Korean J. Ophthalmol. 2008, 22, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A.; Pires, R.T.; Tseng, S.C. Amniotic membrane transplantation after extensive removal of primary and recurrent pterygia. Ophthalmology 2001, 108, 449–460. [Google Scholar] [CrossRef]
- Tseng, S.C.; Prabhasawat, P.; Barton, K.; Gray, T.; Meller, D. Amniotic membrane transplantation with or without limbal allografts for corneal surface reconstruction in patients with limbal stem cell deficiency. Arch. Ophthalmol. 1998, 116, 431–441. [Google Scholar] [CrossRef]
- Hanada, K.; Nishikawa, N.; Miyokawa, N.; Yoshida, A. Long-term outcome of amniotic membrane transplantation combined with mitomycin C for conjunctival reconstruction after ocular surface squamous neoplasia excision. Int. Ophthalmol. 2017, 37, 71–78. [Google Scholar] [CrossRef]
- Krysik, K.; Dobrowolski, D.; Wylęgała, E.A.; Lyssek-Boroń, A. Amniotic Membrane as a Main Component in Treatment Supporting Healing and Patch Grafts in Corneal Melting and Perforations. J. Ophthalmol. 2020, 2020, 4238919. [Google Scholar] [CrossRef]
- Ma, D.H.-K.; Wang, S.-F.; Su, W.-Y.; Tsai, R.J.-F. Amniotic membrane graft for the management of scleral melting and corneal perforation in recalcitrant infectious scleral and corneoscleral ulcers. Cornea 2002, 21, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Trinh, T.; Mimouni, M.; Mednick, Z.; Einan-Lifshitz, A.; Cohen, E.; Santaella, G.; Sorkin, N.; Slomovic, A. Outcomes of Ipsilateral Simple Limbal Epithelial Transplantation, Tenonectomy, Mitomycin and Amniotic Membrane Transplantation for Treatment of Recurrent Pterygium. Cornea 2020, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Koranyi, G.; Seregard, S.; Kopp, E.D. Cut and paste: A no suture, small incision approach to pterygium surgery. Br. J. Ophthalmol. 2004, 88, 911–914. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, V.; Rao, C.L.; Ganesh, S.; Brar, S. Visual outcome and efficacy of conjunctival autograft, harvested from the body of pterygium in pterygium excision. Clin. Ophthalmol. 2015, 9, 2285–2290. [Google Scholar] [CrossRef][Green Version]
- Pietrzak, W.S.; Eppley, B.L. Platelet rich plasma: Biology and new technology. J. Craniofac. Surg. 2005, 16, 1043–1054. [Google Scholar] [CrossRef]
- Anitua, E.; Andia, I.; Ardanza, B.; Nurden, P.; Nurden, A.T. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb. Haemost. 2004, 91, 4–15. [Google Scholar] [CrossRef]
- Freire, V.; Andollo, N.; Etxebarria, J.; Duran, J.A.; Morales, M.-C. In vitro effects of three blood derivatives on human corneal epithelial cells. Invest. Ophthalmol. Vis. Sci. 2012, 53, 5571–5578. [Google Scholar] [CrossRef] [PubMed]
- Weibrich, G.; Kleis, W.K.G.; Hitzler, W.E.; Hafner, G. Comparison of the platelet concentrate collection system with the plasma-rich-in-growth-factors kit to produce platelet-rich plasma: A technical report. Int. J. Oral Maxillofac. Implants 2005, 20, 118–123. [Google Scholar]
- Anitua, E. The use of plasma-rich growth factors (PRGF) in oral surgery. Pract. Proced. Aesthet. Dent. 2001, 13, 487–493. [Google Scholar]
- Cobos, R.; Aizpuru, F.; Parraza, N.; Anitua, E.; Orive, G. Effectiveness and efficiency of platelet rich plasma in the treatment of diabetic ulcers. Curr. Pharm. Biotechnol. 2015, 16, 630–634. [Google Scholar] [CrossRef]
- Sanchez, M.; Anitua, E.; Azofra, J.; Aguirre, J.J.; Andia, I. Intra-articular injection of an autologous preparation rich in growth factors for the treatment of knee OA: A retrospective cohort study. Clin. Exp. Rheumatol. 2008, 26, 910–913. [Google Scholar] [PubMed]
- Anitua, E.; Muruzabal, F.; Tayebba, A.; Riestra, A.; Perez, V.L.; Merayo-Lloves, J.; Orive, G. Autologous serum and plasma rich in growth factors in ophthalmology: Preclinical and clinical studies. Acta Ophthalmol. 2015, 93, e605-14. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Nurden, P.; Prado, R.; Nurden, A.T.; Padilla, S. Autologous fibrin scaffolds: When platelet- and plasma-derived biomolecules meet fibrin. Biomaterials 2019, 192, 440–460. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Muruzabal, F.; de la Fuente, M.; Merayo, J.; Duran, J.; Orive, G. Plasma Rich in Growth Factors for the Treatment of Ocular Surface Diseases. Curr. Eye Res. 2016, 41, 875–882. [Google Scholar] [CrossRef]
- Merayo-Lloves, J.; Sanchez, R.M.; Riestra, A.C.; Anitua, E.; Begona, L.; Orive, G.; Fernandez-Vega, L. Autologous Plasma Rich in Growth Factors Eyedrops in Refractory Cases of Ocular Surface Disorders. Ophthalmic Res. 2015, 55, 53–61. [Google Scholar] [CrossRef]
- Anitua, E.; de la Fuente, M.; Muruzabal, F.; Riestra, A.; Merayo-Lloves, J.; Orive, G. Plasma rich in growth factors (PRGF) eye drops stimulates scarless regeneration compared to autologous serum in the ocular surface stromal fibroblasts. Exp. Eye Res. 2015, 135, 118–126. [Google Scholar] [CrossRef]
- Anitua, E.; de la Fuente, M.; Muruzábal, F.; Merayo-Lloves, J. Short- and Long-Term Stability of Plasma Rich in Growth Factors Eye Drops. Cornea 2021, 40, 107–112. [Google Scholar] [CrossRef]
- Rodriguez-Agirretxe, I.; Freire, V.; Muruzabal, F.; Orive, G.; Anitua, E.; Diez-Feijoo, E.; Acera, A. Subconjunctival PRGF Fibrin Membrane as an Adjuvant to Nonpenetrating Deep Sclerectomy: A 2-Year Pilot Study. Ophthalmic Res. 2018, 59, 45–52. [Google Scholar] [CrossRef]
- Sanchez-Avila, R.M.; Merayo-Lloves, J.; Riestra, A.C.; Berisa, S.; Lisa, C.; Sanchez, J.A.; Muruzabal, F.; Orive, G.; Anitua, E. Plasma rich in growth factors membrane as coadjuvant treatment in the surgery of ocular surface disorders. Medicine 2018, 97, e0242. [Google Scholar] [CrossRef]
- Prabhasawat, P.; Barton, K.; Burkett, G.; Tseng, S.C. Comparison of conjunctival autografts, amniotic membrane grafts, and primary closure for pterygium excision. Ophthalmology 1997, 104, 974–985. [Google Scholar] [CrossRef]
- Ghanavati, S.Z.; Shousha, M.A.; Betancurt, C.; Perez, V.L. Combined conjunctival autograft and overlay amniotic membrane transplantation; A novel surgical treatment for pterygium. J. Ophthalmic Vis. Res. 2014, 9, 399–403. [Google Scholar] [CrossRef]
- Schiffman, R.M.; Christianson, M.D.; Jacobsen, G.; Hirsch, J.D.; Reis, B.L. Reliability and validity of the Ocular Surface Disease Index. Arch. Ophthalmol. 2000, 118, 615–621. [Google Scholar] [CrossRef]
- Anitua, E.; Muruzabal, F.; De la Fuente, M.; Merayo-Lloves, J.; Orive, G. Effects of heat-treatment on plasma rich in growth factors-derived autologous eye drop. Exp. Eye Res. 2014, 119, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Fan, G. Stem cell-based therapy of corneal epithelial and endothelial diseases. Regen. Med. 2015, 10, 495–504. [Google Scholar] [CrossRef]
- Samoila, O.; Gocan, D. Clinical Outcomes From Cultivated Allogenic Stem Cells vs. Oral Mucosa Epithelial Transplants in Total Bilateral Stem Cells Deficiency. Front. Med. 2020, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, M.E.; Avanzini, M.A.; Perotti, C.; Cometa, A.M.; Moretta, A.; Lenta, E.; Del Fante, C.; Novara, F.; de Silvestri, A.; Amendola, G.; et al. Optimization of in vitro expansion of human multipotent mesenchymal stromal cells for cell-therapy approaches: Further insights in the search for a fetal calf serum substitute. J. Cell. Physiol. 2007, 211, 121–130. [Google Scholar] [CrossRef]
- Fernandez-Buenaga, R.; Aiello, F.; Zaher, S.S.; Grixti, A.; Ahmad, S. Twenty years of limbal epithelial therapy: An update on managing limbal stem cell deficiency. BMJ Open Ophthalmol. 2018, 3, e000164. [Google Scholar] [CrossRef]
- Coquelin, L.; Fialaire-Legendre, A.; Roux, S.; Poignard, A.; Bierling, P.; Hernigou, P.; Chevallier, N.; Rouard, H. In vivo and in vitro comparison of three different allografts vitalized with human mesenchymal stromal cells. Tissue Eng. Part A 2012, 18, 1921–1931. [Google Scholar] [CrossRef]
- Anitua, E.; Zalduendo, M.M.; Alkhraisat, M.H.; Orive, G. Release kinetics of platelet-derived and plasma-derived growth factors from autologous plasma rich in growth factors. Ann. Anat. 2013, 195, 461–466. [Google Scholar] [CrossRef]
- Anitua, E.; Sanchez, M.; Merayo-Lloves, J.; De la Fuente, M.; Muruzabal, F.; Orive, G. Plasma rich in growth factors (PRGF-Endoret) stimulates proliferation and migration of primary keratocytes and conjunctival fibroblasts and inhibits and reverts TGF-beta1-Induced myodifferentiation. Invest. Ophthalmol. Vis. Sci. 2011, 52, 6066–6073. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Q.; Xiang, M.; Zou, H.; Liu, B.; Zhou, H.; Han, Z.; Fu, Z.; Zhang, Z.; Wang, H. Bulbar conjunctival thickness measurements with optical coherence tomography in healthy chinese subjects. Invest. Ophthalmol. Vis. Sci. 2013, 54, 4705–4709. [Google Scholar] [CrossRef] [PubMed]
- Ozgurhan, E.B.; Kara, N.; Bozkurt, E.; Gencer, B.; Yuksel, K.; Demirok, A. Comparison of conjunctival graft thickness after primary and recurrent pterygium surgery: Anterior segment optical coherence tomography study. Indian J. Ophthalmol. 2014, 62, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Heher, P.; Mühleder, S.; Mittermayr, R.; Redl, H.; Slezak, P. Fibrin-based delivery strategies for acute and chronic wound healing. Adv. Drug Deliv. Rev. 2018, 129, 134–147. [Google Scholar] [CrossRef]
- Nampei, K.; Oie, Y.; Kiritoshi, S.; Morota, M.; Satoh, S.; Kawasaki, S.; Nishida, K. Comparison of ocular surface squamous neoplasia and pterygium using anterior segment optical coherence tomography angiography. Am. J. Ophthalmol. Case Rep. 2020, 20, 100902. [Google Scholar] [CrossRef] [PubMed]
- Lozano García, I.; Romero Caballero, M.D.; Sellés Navarro, I. High resolution anterior segment optical coherence tomography for differential diagnosis between corneo-conjunctival intraepithelial neoplasia and pterygium. Arch. Soc. Esp. Oftalmol. 2020, 95, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Gris, O.; del Campo, Z.; Wolley-Dod, C.; Güell, J.L.; Bruix, A.; Calatayud, M.; Adán, A. Amniotic membrane implantation as a therapeutic contact lens for the treatment of epithelial disorders. Cornea 2002, 21, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Duffield, J.S.; Lupher, M.; Thannickal, V.J.; Wynn, T.A. Host responses in tissue repair and fibrosis. Annu. Rev. Pathol. 2013, 8, 241–276. [Google Scholar] [CrossRef]
- Maltseva, O.; Folger, P.; Zekaria, D.; Petridou, S.; Masur, S.K. Fibroblast growth factor reversal of the corneal myofibroblast phenotype. Invest. Ophthalmol. Vis. Sci. 2001, 42, 2490–2495. [Google Scholar] [PubMed]
- Netto, M.V.; Mohan, R.R.; Sinha, S.; Sharma, A.; Dupps, W.; Wilson, S.E. Stromal haze, myofibroblasts, and surface irregularity after PRK. Exp. Eye Res. 2006, 82, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Muruzabal, F.; Alcalde, I.; Merayo-Lloves, J.; Orive, G. Plasma rich in growth factors (PRGF-Endoret) stimulates corneal wound healing and reduces haze formation after PRK surgery. Exp. Eye Res. 2013, 115, 153–161. [Google Scholar] [CrossRef]
- Chu, W.K.; Choi, H.L.; Bhat, A.K.; Jhanji, V. Pterygium: New insights. Eye 2020, 34, 1047–1050. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Xing, Y.; Zhao, Q.; Zeng, S.; Yang, J.; Du, L. Application of Platelet-rich Fibrin Grafts Following Pterygium Excision. Int. J. Clin. Pract. 2021, e14560. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Zalduendo, M.M.; Prado, R.; Alkhraisat, M.H.; Orive, G. Morphogen and proinflammatory cytokine release kinetics from PRGF-Endoret fibrin scaffolds: Evaluation of the effect of leukocyte inclusion. J. Biomed. Mater. Res. Part A 2015, 103, 1011–1020. [Google Scholar] [CrossRef] [PubMed]

| Country | Group A n (%) | Group B n (%) | Group C n (%) | Total n (%) |
|---|---|---|---|---|
| Ecuador | 3 (6.1) | 7 (14.3) | 5 (10.2) | 15 (30.6) |
| Spain * | 1 (2.0) | 7 (14.3) | 2 (4.1) | 10 (20.4) |
| Nicaragua | 3 (6.1) | 3 (6.1) | - | 6 (12.2) |
| Colombia | 2 (4.1) | 2 (4.1) | - | 4 (8.2) |
| Peru | 1 (2.0) | 1 (2.0) | 1 (2.0) | 3 (6.1) |
| Argelia | - | 2 (4.1) | - | 2 (4.1) |
| Brazil | 1 (2.0) | - | - | 1 (2.0) |
| Honduras | - | 1 (2.0) | - | 1 (2.0) |
| Romania | - | 1 (2.0) | - | 1 (2.0) |
| Senegal | 1 (2.0) | - | - | 1 (2.0) |
| Uruguay | - | 1 (2.0) | - | 1 (2.0) |
| Venezuela | - | - | 1 (2.0) | 1 (2.0) |
| Bolivia | - | - | 1 (2.0) | 1 (2.0) |
| France | 1 (2.0) | - | - | 1 (2.0) |
| Dominican Republic | - | 1 (2.0) | - | 1 (2.0) |
| Overall | 13 (26.5) | 26 (53.1) | 10 (20.4) | 49 (100) |
| Group A | Group B | Group C | p-Value | |
|---|---|---|---|---|
| Pacients, n (%) | 13 (26.5) | 26 (53.1) | 10 (20.4) | - |
| Age, mean ± SD (range) | 44.4 ± 12.9 (33.0–78.0) | 47.5 ± 14.0 (31.0–77.0) | 47.2 ± 14.0 (31.0–82.0) | 0.791 |
| Gender, M (%) | 8 (61.5) | 14 (53.8) | 4 (40.0) | 0.593 |
| Race * | ||||
| Amerindian, n (%) | 8 (61.5) | 12 (46.2) | 8 (80.0) | 0.235 |
| African, n (%) | 1 (7.7) | 3 (11.5) | 0 (0.0) | |
| European, n (%) | 4 (30.8) | 11 (42.3) | 2 (20.0) | |
| Evolution time of the pterygium, mean ± SD (range) | 8.2 ± 4.6 (1.0–16.0) | 6.1 ± 5.9 (1.0–30.0) | 7.5 ± 5.5 (2.0–20.0) | 0.503 |
| Residence time in Zaragoza, mean ± SD (range) | 16.2 ± 12.9 (1.0-–50.0) | 20.2 ± 22.1 (0.0–75.0) | 23.8 ± 23.4 (6.0–82.0) | 0.668 |
| Hours of sun exposure per day, mean ± SD (range) | 2.52 ± 3.62 (0.0–10.0) | 3.92 ± 3.39 (0.0–10.0) | 2.29 ± 2.57 (0.0–8.0) | 0.306 |
| Sun protection * | ||||
| None, n (%) | 5 (38.5) | 14 (53.8) | 3 (30.0) | 0.429 |
| Hat, n (%) | 1 (7.7) | 4 (15.4) | 1 (10.0) | |
| UV filter glasses + Hat, n (%) | 1 (7.7) | 2 (7.7) | 2 (20.0) | |
| UV filter glasses (occasional), n (%) | 1 (7.7) | 0 (0.0) | 0 (0.0) | |
| UV filter glasses (usually), n (%) | 5 (38.5) | 4 (15.4) | 4 (40.0) |
| Group A Mean ± SD (µm) | Group B Mean ± SD (µm) | Group C Mean ± SD (µm) | p-Value | ||
|---|---|---|---|---|---|
| Thickness next to limbus | Baseline | 471 ± 143 | 478 ± 163 | 424 ± 110 | 0.767 |
| Horizontal size from limbus | 2862 ± 964 | 3469 ± 1594 | 2387 ± 988 | 0.106 | |
| Total horizontal size | 7755 ± 2152 | 7482 ± 2817 | 7971 ± 1878 | 0.876 | |
| Vertical size in limbus | 6206 ± 1236 | 6010 ± 1666 | 5343 ± 2038 | 0.461 | |
| Graft central thickness | Day 1 | 611 ± 216 | 455 ± 240 | 412 ± 207 | 0.103 |
| Day 7 | 620 ± 257 ** | 359 ± 271 | 452 ± 155 | 0.020 | |
| Day 15 | 356 ± 141 | 316 ± 286 | 337 ± 119 | 0.362 | |
| Month 1 | 324 ± 131 | 207 ± 163 | 271 ± 114 | 0.089 | |
| Month 3 | 252 ± 147 ** | 84 ± 148 | 231 ± 147 # | 0.002 | |
| Month 6 | 229 ± 90 ** | 102 ± 124 | 183 ± 45 | 0.011 | |
| Month 12 | 151 ± 65 | 192 ± 109 | 190 ± 183 | 0.503 | |
| Graft thickness next to limbus | Day 1 | 459 ± 184 | 499 ± 417 | 412 ± 139 | 0.929 |
| Day 7 | 460 ± 232 * | 248 ± 171 | 430 ± 183 | 0.005 | |
| Day 15 | 365 ± 159 | 250 ± 193 | 274 ± 79 | 0.163 | |
| Month 1 | 314 ± 114 | 259 ± 178 | 273 ± 109 | 0.770 | |
| Month 3 | 250 ± 109 ** | 79 ± 118 | 230 ± 185 # | 0.002 | |
| Month 6 | 207 ± 60 * | 96 ± 115 | 188 ± 98 | 0.036 | |
| Month 12 | 200 ± 91 | 197 ± 115 | 116 ± 37 | 0.226 | |
| Graft horizontal size | Day 1 | 6499 ± 2192 | 5949 ± 2336 | 5645 ± 2047 | 0.673 |
| Day 7 | 5813 ± 1894 * | 3639 ± 2554 | 4784 ± 945 | 0.044 | |
| Day 15 | 5287 ± 2411 | 3314 ± 2523 | 5983 ± 1028 | 0.059 | |
| Month 1 | 4079 ± 1309 ** | 1048 ± 1829 | 3903 ± 903 # | 0.001 | |
| Month 3 | 4598 ± 1492 **‡ | 1433 ± 2077 | 2626 ± 2346 | 0.003 | |
| Month 6 | 3809 ± 1396 ** | 1433 ± 1908 | 2176 ± 1928 | 0.009 | |
| Month 12 | 4815 ± 1426 * | 1394 ± 1456 | 2299 ± 1970 | 0.003 | |
| Graft vertical size | Day 1 | 7356 ± 2322 | 7054 ± 2713 | 7882 ± 1153 | 0.651 |
| Day 7 | 7140 ± 2266 | 5230 ± 3640 | 6476 ± 1446 | 0.355 | |
| Day 15 | 6298 ± 1619 | 5106 ± 3559 | 5983 ± 1097 | 0.442 | |
| Month 1 | 6881 ± 959 ** | 4018 ± 3071 | 5320 ± 1906 | 0.007 | |
| Month 3 | 6951 ± 1699 **‡‡ | 1649 ± 2450 | 3728 ± 2259 | 0.000 | |
| Month 6 | 5926 ± 1274 ** | 1921 ± 2138 | 4762 ± 2507 # | 0.000 | |
| Month 12 | 5653 ± 824 *‡ | 2374 ± 2457 | 2951 ± 1702 | 0.019 |
| Month 1 Mean ± SD | Month 3 Mean ± SD | Month 6 Mean ± SD | Month 12 Mean ± SD | |
|---|---|---|---|---|
| Group A | 1.00 ± 0.00 * | 1.08 ± 0.29 * | 1.10 ± 0.32 ‡ | 1.00 ± 0.00 |
| Group B | 1.55 ± 0.76 | 2.06 ± 0.87 | 1.88 ± 0.96 | 1.91 ± 1.04 |
| Group C | 1.11 ± 0.33 | 2.00 ± 1.22 | 2.43 ± 1.13 | 2.17 ± 0.98 |
| Baseline Mean ± SD | Day 7 Mean ± SD | Day 15 Mean ± SD | Month 1 Mean ± SD | Month 3 Mean ± SD | Month 6 Mean ± SD | Month 12 Mean ± SD | |
|---|---|---|---|---|---|---|---|
| Group A | 36.82 ± 26.00 | 36.45 ± 23.24 | 23.87 ± 18.61 | 19.60 ± 21.95 | 22.85 ± 22.85 | 31.31 ± 24.70 | 41.48 ± 27.34 |
| Group B | 33.92 ± 26.18 | 42.39 ± 25.60 | 26.62 ± 25.23 | 22.41 ± 21.76 | 20.04 ± 23.11 | 19.93 ± 23.46 | 20.68 ± 24.65 |
| Group C | 37.12 ± 26.18 | 38.85 ± 30.99 | 12.62 ± 14.79 | 5.72 ± 6.68 | 8.59 ± 14.51 | 7.87 ‡ ± 13.97 | 5.90 ‡ ± 9.72 |
| Baseline Mean (Range) | Month 12 Mean (Range) | p-Value | ||
|---|---|---|---|---|
| Group A | Light sensitivity | 2.08 (0–4) | 2.00 (0–4) | 0.41 |
| Grit feeling | 2.15 (0–4) | 1.50 (0–4) | 0.34 | |
| Eye pain | 1.00 (0–4) | 1.17 (0–4) | 1.00 | |
| Blurry vision | 0.77 (0–3) | 1.00 (0–2) | 0.49 | |
| Bad vision | 0.69 (0–3) | 1.00 (0–4) | 0.59 | |
| Read | 1.08 (0–4) | 2.50 (0–4) | 0.24 | |
| Night driving | 0.88 (0–4) | 0.40 (0–2) | 0.32 | |
| Use of computer or screen | 0.92 (0–4) | 1.33 (0–4) | 0.85 | |
| Watch TV | 1.08 (0–4) | 1.50 (0–4) | 0.56 | |
| Wind | 2.38 (0–4) | 2.83 (0–4) | 0.79 | |
| Very dry environments | 2.46 (0–4) | 2.50 (0–4) | 1.00 | |
| Air conditioning | 1.92 (0–4) | 1.83 (0–4) | 0.79 | |
| Total OSDI score | 36.82 (0–93) | 41.48 (6–61) | 0.35 | |
| Group B | Light sensitivity | 1.57 (0–4) | 0.92 (0–3) | ≤0.01 * |
| Grit feeling | 1.78 (0–4) | 0.83 (0–3) | 0.02 * | |
| Eye pain | 0.91 (0–4) | 0.67 (0–3) | ≤0.01 * | |
| Blurry vision | 0.83 (0–3) | 1.80 (0–4) | 0.19 | |
| Bad vision | 0.48 (0–3) | 0.73 (0–3) | 0.16 | |
| Read | 1.30 (0–4) | 0.92 (0–4) | 0.06 | |
| Night driving | 0.50 (0–4) | 0.40 (0–3) | 1.00 | |
| Use of computer or screen | 1.48 (0–4) | 0.50 (0–3) | 0.04 * | |
| Watch TV | 1.22 (0–4) | 0.83 (0–4) | 0.03 * | |
| Wind | 2.65 (0–4) | 1.50 (0–4) | ≤0.01 * | |
| Very dry environments | 1.78 (0–4) | 0.75 (0–3) | 0.06 | |
| Air conditioning | 1.25 (0–4) | 0.58 (0–3) | 0.42 | |
| Total OSDI score | 33.9 (0–77) | 20.7 (0–66) | ≤0.01 * | |
| Group C | Light sensitivity | 1.50 (0–4) | 0.33 (0–1) | 0.11 |
| Grit feeling | 2.30 (0–4) | 0.67 (0–3) | 0.06 | |
| Eye pain | 0.70 (0–3) | 0.0 (0–0) | 0.32 | |
| Blurry vision | 1.60 (0–4) | 0.17 (0–1) | 0.03 * | |
| Bad vision | 1.60 (0–4) | 0.17 (0–1) | 0.03 * | |
| Read | 1.00 (0–4) | 0.17 (0–1) | 1.00 | |
| Night driving | 1.00 (0–4) | 0.00 (0–0) | 1.00 | |
| Use of computer or screen | 0.78 (0–4) | 0.20 (0–1) | 0.32 | |
| Watch TV | 0.40 (0–4) | 0.17 (0–1) | 0.32 | |
| Wind | 2.40 (0–4) | 0.50 (0–2) | 0.07 | |
| Very dry environments | 2.40 (0–4) | 0.17 (0–1) | 0.10 | |
| Air conditioning | 1.50 (0–4) | 0.17 (0–1) | 0.10 | |
| Total OSDI score | 37.1 (6–75) | 5.9 (0.25) | 0.03 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Idoipe, M.; de la Sen-Corcuera, B.; Sánchez-Ávila, R.M.; Sánchez-Pérez, C.; Satué, M.; Sánchez-Pérez, A.; Orive, G.; Muruzabal, F.; Anitua, E.; Pablo, L. Membrane of Plasma Rich in Growth Factors in Primary Pterygium Surgery Compared to Amniotic Membrane Transplantation and Conjunctival Autograft. J. Clin. Med. 2021, 10, 5711. https://doi.org/10.3390/jcm10235711
Idoipe M, de la Sen-Corcuera B, Sánchez-Ávila RM, Sánchez-Pérez C, Satué M, Sánchez-Pérez A, Orive G, Muruzabal F, Anitua E, Pablo L. Membrane of Plasma Rich in Growth Factors in Primary Pterygium Surgery Compared to Amniotic Membrane Transplantation and Conjunctival Autograft. Journal of Clinical Medicine. 2021; 10(23):5711. https://doi.org/10.3390/jcm10235711
Chicago/Turabian StyleIdoipe, Miriam, Borja de la Sen-Corcuera, Ronald M. Sánchez-Ávila, Carmen Sánchez-Pérez, María Satué, Antonio Sánchez-Pérez, Gorka Orive, Francisco Muruzabal, Eduardo Anitua, and Luis Pablo. 2021. "Membrane of Plasma Rich in Growth Factors in Primary Pterygium Surgery Compared to Amniotic Membrane Transplantation and Conjunctival Autograft" Journal of Clinical Medicine 10, no. 23: 5711. https://doi.org/10.3390/jcm10235711
APA StyleIdoipe, M., de la Sen-Corcuera, B., Sánchez-Ávila, R. M., Sánchez-Pérez, C., Satué, M., Sánchez-Pérez, A., Orive, G., Muruzabal, F., Anitua, E., & Pablo, L. (2021). Membrane of Plasma Rich in Growth Factors in Primary Pterygium Surgery Compared to Amniotic Membrane Transplantation and Conjunctival Autograft. Journal of Clinical Medicine, 10(23), 5711. https://doi.org/10.3390/jcm10235711






