Artificial Intelligence: A Shifting Paradigm in Cardio-Cerebrovascular Medicine
Abstract
1. Introduction
2. Methods
3. Results
3.1. Application of AI in Pre-Diagnosis Modeling: Primary Prevention
- (a)
- Risk Estimation
- (b)
- Clustering and Patient Profiling Before Event
- (c)
- Care Gap Identification and Personalized Prevention
3.2. Application of Computational Algorithms in Diagnosis and Acute Phase Treatment
- (a)
- Emergency Medical Services (EMS) Proper Referral
- (b)
- Acute Diagnosis
- (c)
- Acute Imaging
- (d)
- Triaging and Acute Treatment
3.3. Application of AI in Post-Diagnosis Outcome Prediction and Secondary Prevention
- (a)
- Personalized Treatment
- (b)
- Outcome Prediction
3.4. Application of AI in Rehabilitation
- (a)
- Personalized Treatment
- (b)
- Outcome Prediction
| Ref., Year—Category ** | Study Details | Sample Size | Algorithms |
|---|---|---|---|
| AI and Risk Stratification Modeling | |||
| [10], 2017—1a | Location: United Kingdom Aim: Predicting the first CVD event over 10-years and comparing that with the American College of Cardiology guidelines. Variables: Routine clinical data from family practices Strengths: Prospective; large sample size Limitations: Unbalanced dataset Findings: Highest achieving algorithm was NN: AUC 0.76, predicted 4998/7404 cases (sensitivity 67.5%, PPV 18.4%) and 53,458/75,585 non-cases (specificity 70.7%, NPV 95.7%), correctly predicting 7.6% more patients than the established algorithm | 378,256 | RF, LR, GBM, NN |
| [12], 2017—1a | Location: United States Aim: Predict six cardiovascular outcomes in comparison to standard risk scores. Variables: 735 variables from imaging and non-invasive tests, questionnaires, and biomarker panels Strengths: Prospective; included participants from the MESA (Multi-Ethnic Study of Atherosclerosis) [22]; 12-year follow-up; four ethnicities Limitations: Potential cause for biases due to imputation procedure Findings: Age was the most important predictor for all-cause mortality. Fasting glucose levels and carotid ultrasonography measures were important predictors of stroke. CAC was the most important predictor of coronary heart disease and all atherosclerotic cardiovascular disease combined outcomes. Left ventricular structure and function and cardiac troponin-T were among the top predictors for incident heart failure. Creatinine, age, and ankle-brachial index were among the top predictors of AF. TNF-α and IL-2 soluble receptors and NT-proBNP levels were important across all outcomes. Notable facts: ML in conjunction with deep phenotyping improves prediction accuracy in cardiovascular event prediction in an initially asymptomatic population. | 6814 | RF |
| [11], 2019—1a, 1b | Location: United States Aim: Predicting of long-term risk of MI and cardiac death in asymptomatic subjects by integrating clinical parameters with CAC, and automated EAT quantification. Variables: Clinical co-variates, lipid panel, risk factors, CAC, aortic calcium, and automated EAT measures Strengths: Prospective; subjects from EISNER trial [23]; 14.5 years follow-up Limitations: Unbalanced data Findings: AUC 0.82; Subjects with a higher ML score had high hazard of suffering events (HR: 10.38, p < 0.001); the relationships persisted in multivariable analysis including ASCVD-risk and CAC measures (HR: 2.94, p = 0.005). Age, ASCVD-risk, and CAC were prognostically important for both genders. Notable facts: ML used to integrate clinical and quantitative imaging-based variables significantly improves prediction of MI and cardiac death. | 1912 | XGBoost |
| [14], 2017—>1a | Location: China Aim: Identifying the association between the clinical reference range of serum HbA1c and TSH, and the risk of CAD in non-diabetic and euthyroid patients. Variables: HbA1c and TSH levels Strengths: Prospective; 10-year follow-up Limitations: Small sample size Findings: Baseline HbA1c and TSH within the reference range were positively associated with CAD risk. No correlation and interaction between the baseline HbA1c and TSH for the development of CAD. The combination of these baselines showed sensitivity of 87.2%, specificity of 92.7%, and accuracy of 92.3% for identifying the participants who will later develop CAD. | 538 | SVM |
| [107], 2018—1a | Location: Lebanon Aim: Comparing ANN-based prediction models to the other risk models being used in practice (the Diamond–Forrester and the Morise models). Variables: Imaging-based stress test measures Strengths: Prospective Limitations: Small sample size Findings: Compared to other models, the ANN model had higher discriminatory power (DP) (1.61) for predicting ischemia, 98% negative predictive value, 91% sensitivity, 65% specificity, 26% positive predictive value, and a potential 59% reduction of non-invasive imaging. | 486 | ANN |
| [28], 2018—1b, 3a | Location: United Kingdom, Italy, Norway Aim: Discriminating between healthy and HFpEF subjects with impaired functional reserve and identifying new descriptors to better characterize HFpEF syndrome using basal myocardial long-axis velocity patterns at rest and exercise. Variables: Left ventricular long-axis myocardial velocity patterns Strengths: Prospective, 6–60 months survival analysis Limitations: Confounding effects (age, gender) not studied, small sample size Findings: ML-diagnostic zones differed for age, body mass index, six-minute walk distance, B-type natriuretic peptide, and left ventricular mass index. Correlation with diagnosis was 72.6%; ML identified 6% of healthy controls as HFpEF. Blinded reinterpretation of imaging from subjects with discordant clinical and ML diagnoses revealed abnormalities not included in diagnostic criteria. | 156 | Clustering |
| [71], 2015—1b, 3a | Location: United States Aim: Identify phenotypically distinct HFpEF categories. Variables: Clinical, laboratory, ECG, and echocardiographic phenotyping (phenomapping) Strengths: Prospective Findings: Phenomapping classified study participants into three risk-stratified groups. Notable facts: A novel classification of HFpEF using phenomapping that can define therapeutically homogeneous patient subclasses. | 397 | Clustering |
| [16], 2019—1a, 3a, 4b | Location: United States Aim: Predicting survival after echocardiography. Variables: 90 cardiovascular-relevant ICD-10 codes, age, sex, height, weight, heart rate, blood pressures, LDL, HDL, smoking, physician-reported EF, 57 echocardiographic measurements Strengths: Large sample size Limitations: Retrospective, model derivation from EHR data missing important variables Findings: Overall AUC > 0.82 over common clinical risk scores. RF outperformed LR. RF including all echocardiographic measurements yielded the highest prediction accuracy. Ten variables needed to achieve 96% maximum prediction accuracy, six from echocardiography. | 171,510 | RF |
| [17], 2019—3a, 3b | Location: United States Aim: Using ML to develop a model of vessel features to discriminate between patients with and without subsequent death or cardiovascular events and comparing to CAD-RADS. Variables: Four CTA features for each of the sixteen coronary segments Strengths: Comparing four different ML methods Limitations: Low MI incidence leading to possible misclassification bias Findings: ML all-cause mortality AUC = 0.77; ML CAD deaths AUC = 0.85. For starting statin therapy (NNT = 45), use of ML score ensures 93% of patients with events will be administered the drug; compared to 69% with CAD-RADS. Notable facts: Compared to CAD-RADS, ML better discriminated patients who subsequently experienced an adverse event from those who did not. | 6892 | Best models: bootstrap-aggregated DTE, KNN, |
| [13], 2018—1a, 1c | Location: United States Aim: Developing a risk calculator for CAD incidence to aid initiation of statin therapy. Variables: Same as ACC/AHA risk calculator Strengths: Model training by 13-year follow-up data from MESA cohort [22] and validation by FLEMENGHO cohort [108] Limitations: Retrospective Findings: ML Risk Calculator recommended only 11.4% to take statin, and only 14.4% of “Hard CVD” events occurred in those not recommended statin, resulting in sensitivity 0.86, specificity 0.95, and AUC 0.92. Notable facts: ML Risk Calculator outperformed the ACC/AHA Risk Calculator by recommending less drug therapy yet missing fewer events. | 10,291 | SVM |
| [109], 2019—1a, 1b | Location: Iran Aim: Compare ANN and SVM algorithms for predicting CAD. Variables: 25 variables affecting CAD including laboratory values Strengths: Data collected from three hospitals Limitations: Retrospective; no detail provided regarding missingness, or lack thereof Findings: SVM model had higher AUC, higher sensitivity, higher Hosmer–Lemeshow test’s result and lower MAPE compared to ANN. Variables affecting CAD yielded better goodness of fit in SVM model and provided more accurate result than ANN. | 1324 | ANN, SVM |
| AI-enabled Diagnostic Studies | |||
| [76], 2016—3b | Location: Multi-national Aim: Predicting five-year all-cause mortality in patients undergoing CCTA and comparing to existing prediction algorithms. Variables: 25 clinical and 44 CCTA parameters, SSS, SIS, DI, number of segments with non-calcified, mixed or calcified plaques, age, sex, gender, standard cardiovascular risk factors, and FRS Strengths: Data from CONFIRM registry [110]; large sample size Limitations: Selection bias; only LogitBoost was evaluated for efficacy. Findings: ML exhibited a higher area-under-curve compared with the FRS or CCTA severity scores alone (SSS, SIS, DI) for predicting all-cause mortality (ML: 0.79 vs. FRS: 0.61, SSS: 0.64, SIS: 0.64, DI: 0.62; p < 0.001). Notable facts: ML combining clinical and CCTA data was found to predict five-year all-cause mortality significantly better than existing clinical or CCTA metrics alone. | 10,030 | LogitBoost |
| [77], 2019—3a, 3b | Location: Korea Aim: Developing an angiography-based supervised ML algorithm with five-fold cross-validation to classify coronary lesions based on fractional flow reserve (≤0.80 vs. >0.80). Variables: 24 computed angiographic features based on the diameter plot and four clinical features (age, sex, body surface area, and involve segment) Strengths: Randomized controlled trial; external validation in 79 patients Limitations: Data, analytic methods, and study materials not available to other researchers; model limited to left main disease, side branch, and diffuse and tandem lesions Findings: ML model predicted fractional flow reserve ≤ 0.80 with overall diagnostic accuracy of 78% (AUC = 0.84). Using 12 main angiography features, the ML predicted fractional flow reserve ≤ 0.80 in the test set with sensitivity of 84%, specificity of 80%, and overall accuracy of 82% (AUC = 0.87). The averaged diagnostic accuracy in bootstrap replicates was 81% (AUC = 0.87). External validation showed accuracy of 85% (AUC = 0.87). | 1501 | XGBoost |
| [39], 2017—2a, 2b | Location: Canada Aim: Automating the diagnosis of STEMI at the time of first contact with healthcare system and pre-hospital CCL activation. Variables: ECG reading data Limitations: Retrospective analysis of real-time automated diagnosis; only ECG data used; small sample size Findings: Algorithm modification resulted in a 42% relative decrease in the rate of inappropriate activations (12% vs. 7%) without a significant effect on treatment delay. | 466 | Automated STEMI diagnosis and “physician-less” CCL activation |
| [41], 2019—2b, 2c, 2e | Location: Japan Aim: Making an AI prediction model for the need for urgent revascularization from 12-lead ECG in patients presenting with chest pain in the ER. Variables: ECG reading data Limitations: Retrospective; only ECG data used, small sample size Findings: Predictive value of the c-statistics 0.88 (95% CI 0.84–0.93) for detecting patients who required urgent revascularization. | 362 | LSTM |
| AI in Outcome Prediction/Prognosis | |||
| [89], 2017—3b | Location: United Kingdom Aim: Predicting patient survival in pulmonary hypertension using 3D patterns of systolic cardiac motion. Variables: Conventional imaging; hemodynamic, functional, and clinical markers; 3D motion pattern of right ventricle Strengths: Prospective Limitations: Limited patient selection including non-congenital cases of PH. Model trained to measure excursion rather than contractility. Findings: Survival prediction AUC 0.73; difference in median survival time between high- and low-risk groups was 13.8 years. | 256 | Supervised ML using nested multivariable risk prediction |
| [111], 2019—3a | Location: United States Aim: Testing generalizability and precision in imaging biomarker analysis by comparing scan:rescan data. Variables: MR-measured left ventricular chamber volumes, mass, and ejection fraction Strengths: Prospective Limitations: Data from five institutions, but scans performed at the same institution; one-week interval between scans limited the ability to assess long-term changes Findings: Expert, trained junior, and automated scan:rescan precision were similar (coefficient of variation 6.1 vs. 8.8). Automated analysis was 186× faster than humans. | 110 | CNN |
| [82], 2017—3b | Location: Sweden Aim: Predicting two-year survival vs. non-survival after first MI. Variables: 39 survival predictors Strengths: Large sample size Limitations: Retrospective Findings: SVM had the highest performance (AUC = 0.845, PPV = 0.280, NPV = 0.966) outperforming Boosted C5.0 (AUC = 0.841), but not significantly higher than LR or RF. Models converged to the point of algorithm indifference with increased sample size and predictors. | 51,943 | SVM, RF, LR, Boosted C5.0 |
| [86], 2018—3b, 4b | Location: Sweden Aim: Using mixture of supervised and unsupervised approach to predict outcome and identify distinct phenotypes of heart failure. Variables: Demographic, clinical, laboratory, and medication data Strengths: Large sample size Limitations: Retrospective Findings: RF demonstrated excellent calibration and discrimination for survival (C-statistic = 0.83) whereas LVEF did not (C-statistic = 0.52). Cluster analysis using the eight highest predictive variables identified four clinically relevant subgroups of HF with marked differences in one-year survival. | 44,886 | RF, K-means clustering |
| [79], 2017—3b | Location: United States Aim: Modeling all-cause in-hospital mortality in women admitted with STEMI. Variables: 11 variables for LR; 32 variables for full RF model; 17 variables for reduced RF model Strengths: Model validation using external cohort of 13,361 patients Limitations: Retrospective; class imbalance (in-hospital mortality in 11% of patients) Findings: Internal validation C-index was 0.84, 0.81, and 0.80 for the LR, full, and reduced RF models, respectively. External validation C-index was 0.84, 0.85, and 0.81 for year 2011, and 0.82, 0.81, and 0.81 for the year 2013 for the LR, full, and reduced RF models, respectively. Notable facts: RF was comparable to LR in predicting in-hospital mortality in women with STEMI. | 12,047 | LR and RF |
| [84], 2019—3b | Location: Korea Aim: DL-based risk stratifying mortality of patients with acute MI. Variables: Initial demographic and laboratory data Strengths: Large sample size; data from the Korean working group of myocardial infarction registry (network of 59 hospitals) Limitations: Retrospective Findings: AUC for STEMI = 0.905. AUC for NSTEMI = 0.870. DL predicted 30.9% of patients more accurately than conventional scores. During the six-month follow-up, the DL-defined high-risk group had a significantly higher mortality rate than the low-risk group (17.1% vs. 0.5%). | 22,875 | DL, LR, RF |
| [58], 2019—2d, 3b | Location: China Aim: Identify in-hospital cardiac arrest in hospitalized patients with acute coronary syndrome. Variables: Seven explanatory variables: VitalPAC Early Warning Score (ViEWS), fatal arrhythmia, Killip class, cardiac troponin I, blood urea nitrogen, age, and diabetes Limitations: Possibility of selection bias Findings: Sensitivity = 0.762; Specificity = 0.882; AUC = 0.844; a 10-fold cross-validated risk estimate = 0.198; optimism-corrected AUC = 0.823. Notable facts: The developed DT model may provide healthcare workers with a practical bedside tool and could positively impact decision-making in deteriorating patients with ACS. | 656 | DT |
| [78], 2019—3b | Location: United States Aim: Identify patients at risk of death or CHF rehospitalization after PCI. Variables: 52 features at admission to predict in-hospital mortality; 358 features at discharge to predict CHF readmission Strengths: Large sample size Limitations: Retrospective; high missingness level in certain features causing high data sparsity Findings: RF prediction of in-hospital mortality AUC = 0.925. RF outperformed LR for predicting 30-day CHF readmission (AUC: 0.90 vs. 0.85) and 180-day cardiovascular death (AUC: 0.88 vs. 0.81). | 11,709 | RF |
| [88], 2019—3b | Location: Korea Aim: Developing and validating a deep-learning-based AI algorithm for predicting mortality of acute HF. Variables: Demographics, treatment and medication, laboratory, ECG and echocardiography findings, final diagnosis, clinical outcome during hospital stay, and 12-month prognosis Strengths: Multi-center study; large sample size Limitations: Retrospective Findings: AUC of the DL was 0.880 for predicting in-hospital mortality, which outperformed other machine learning models. For predicting 12- and 36-month endpoints, DL had an AUC of 0.782 and 0.813, respectively. During the 36-month follow-up, the high-risk group, defined by the DL, had a significantly higher mortality rate than the low-risk group. | 6924 | DNN, RF, LR, SVM, BN |
| [53], 2019—2c, 2d | Location: Korea Aim: Using ML to predict ACS requiring revascularization in patients presenting with early-stage angina-like symptoms. Variables: 20 features relevant to ACS Strengths: Large sample size Limitations: Retrospective; inaccuracy in checking the vulnerable plaque burden of all coronary arteries Findings: AUC = 0.860 for the prediction of ACS requiring revascularization. A reliable prediction of 2.60% of non-ACS patients was made with a specificity of 1.0 to only receive medical therapy. | 5882 | SVM, LDA |
| [87], 2019—3b | Location: United States Aim: Using a ML algorithm to predict mortality in HF patients. Variables: Eight variables: diastolic blood pressure, creatinine, blood urea nitrogen, hemoglobin, white blood cell count, platelets, albumin, and red blood cell distribution width Strengths: Large sample size Limitations: Retrospective; selection bias due to excluding significant number of patients with missingness Findings: The risk score developed by DT accurately discriminated between low and high-risk of death with an AUC of 0.88. External validation in two separate HF populations gave AUCs of 0.84 and 0.81. | 5822 | DT |
| [83], 2019—3b | Location: United Kingdom Aim: Predicting long-term mortality after ACS using laboratory values. Variables: Hematological indices and inflammation markers Strengths: Large sample size Limitations: Imputation for the ML was performed using mean of all observations, the latter is typically not ideal since missing in EHR data tend to be not-at-random Findings: The model achieved a c-statistic of 0.89 for in-hospital mortality. C-statistic was 0.77 for six-month mortality. Red cell distribution width (HR 1.23) and neutrophil to lymphocyte ratio (HR 1.08) showed independent association with all-cause mortality in multivariable Cox regression. | 5053 | XGBoost |
| [85], 2019—3b | Location: China Aim: Developing a DL model to predict major adverse cardiac events after ACS. Variables: 232 static feature types and 2194 dynamic feature types. Strengths: Large sample size; comparison to previous models Limitations: Retrospective; missing values (up to 30%) were imputed using median of all the observations; variables with more than 30% missing were excluded Findings: The best model presented had an AUC of 0.713 and an accuracy of 0.764. Notable facts: The proposed model adapted to leverage dynamic treatment information in EHR data boosted the performance of major adverse cardiac event prediction for ACS. | 2930 | RNN |
| [80], 2017—3b | Location: Israel Aim: Predicting mortality at 30-days in STEMI patients and to compare these to the conventional validated risk scores. Variables: 54 variables; performance of most models plateaued with 15 variables Strengths: Large sample size Limitations: Retrospective Findings: ML models AUC range: 0.64 to 0.91. The best models had similar or better performance compared to standard scoring methods. Top predictors were creatinine, Killip class on admission, blood pressure, glucose level, and age. Notable facts: The algorithms selected showed competence in prediction across an increasing number of variables. | 2782 | NB, DT, LR, rules-based classification tree, RF, Adaptive Boosting |
| [112], 2018—3a | Location: Canada Aim: Assessing the prognostication of NN in HF patients using CPET data as opposed to using summary indicators alone. Variables: Detailed CPET data Strengths: Using various ML models Limitations: Retrospective Findings: NN incorporating breath-by-breath data achieved the best performance (AUC = 0.842). All models outperformed summary indices (AUC ≤ 0.800). When compared with the CPET risk score (AUC = 0.759), the top-performing model obtained a net reclassification index of 4.9%. Notable facts: The current practice of considering summary indices in isolation fails to realize the full value of CPET data. Higher data resolution leads to improved prediction. | 1434 | LASSO, NN |
| [81], 2020—3b | Location: China Aim: Using ML to predict one-year mortality rate of anterior STEMI patients and comparing to conventional risk scores. Variables: 59 features; including all features as opposed to top 20 provided better performance Strengths: Using six different ML algorithms Limitations: Retrospective Findings: AUC of ML models ranged from 0.709 to 0.942. XGBoost achieved the highest accuracy (92%), specificity (99%) and f1 score (0.72) for predictions with the full variable model. After feature selection, XGBoost still obtained the highest accuracy (93%), specificity (99%) and f1 score (0.73). | 1244 | NB, LR, KNN, DT, RF and XGBoost |
| [105], 2019—4b | Location: United States Aim: Using ML on EHR data to predict CRT outcome. Variables: Demographics, laboratory values, medications, clinical characteristics, and past health services utilization, bigrams (i.e., two-word sequences) in EHR data Strengths: Comparing various ML models Limitations: No distinction between the type of CRT implant. Findings: The final model identified 26% of patients having a reduced benefit from the CRT device at a PPV of 79% (model performance: Fβ (β = 0.1): 77%; recall 0.26; precision 0.79; accuracy 0.65). Notable facts: A ML model that leveraged readily available EHR data and clinical notes identified a subset of CRT patients who may not benefit from CRT before the procedure. | 990 | LR, SVM, RF and GBM |
| [113], 2019—1a | Location: Japan Aim: Assessing stroke risk by ML using integrated risk factors. Variables: 47 features comprised of 13 conventional risk factors and 34 carotid ultrasound image-based phenotypes (carotid intima-media thickness, carotid plaque and carotid artery stenosis) Strengths: Using integrated risk factors Limitations: Retrospective; small sample size; data imbalance (12 high-risk patients vs. 190 low-risk patients) Findings: ML with integrated risk factors (AUC = 0.80) showed an improvement of ~18% against conventional ML (AUC = 0.68). Notable facts: ML model integrated with the event-equivalent gold standard as percentage stenosis is powerful and offers low cost and high-performance stroke risk assessment. | 202 | RF |
| AI in Treatment Strategies | |||
| [99], 2018—3a, 4a | Location: Multi-national Aim: Using ML to phenotypically classify a heterogeneous HF cohort and aid in optimizing the rate of responders to specific therapies. Variables: 50 variables including clinical parameters, biomarker values, and measures of left and right ventricular structure and function Strengths: Data from MADIT-CRT trial [114]; randomized cohort Limitations: Possibility of selection bias; results confined to a selected population of HF patients enrolled in a clinical trial with robust inclusion/exclusion criteria Findings: Four phenogroups identified, significantly different in the primary outcome occurrence. Two phenogroups included a higher proportion of known clinical characteristics predictive of CRT response and were associated with a substantially better treatment effect of CRT-D on the primary outcome (HR = 0.35 and HR = 0.36) than observed in the other groups. Notable facts: By integrating clinical parameters and full heart cycle imaging data, unsupervised ML can provide a clinically meaningful classification of a phenotypically heterogeneous HF cohort and might aid in optimizing the rate of responders to specific therapies. | 1106 | Multiple Kernel Learning, K-means clustering |
| [104], 2019—4b | Location: United States Aim: Develop and compare ML models to predict response to CRT. Variables: Nine variables; QRS morphology, QRS duration, New York Heart Association classification, left ventricular ejection fraction and end-diastolic diameter, sex, ischemic cardiomyopathy, AF, and epicardial left ventricular lead Strengths: Multi-center study comparing various ML models Limitations: Retrospective Findings: The best ML model was a naïve Bayes classifier. On the testing cohort, ML demonstrated better response prediction than guidelines (AUC 0.70 vs. 0.65) and greater discrimination of event-free survival (concordance index, 0.61 vs. 0.56). The fourth quartile of the ML model had the greatest risk of reaching the composite end point, whereas the first quartile had the least (hazard ratio, 0.34). | 925 | Supervised ML |
| [106], 2018—4b | Location: United States Aim: Using ML to predict all-cause mortality or heart failure hospitalization 12 months post-CRT. Variables: 45 features: demographics, physical characteristics, heart failure, LV assessment, ECG, medical history, medication class Strengths: Used data from COMPANION trial [115] Limitations: Possibility of selection bias; only class III and IV HF patients enrolled with specific inclusion/exclusion criteria Findings: RF model produced quartiles of patients with an eight-fold difference in survival between those with the highest and lowest predicted probability for events (hazard ratio, 7.96). The model discriminated the risk of the composite end point of all-cause mortality or heart failure hospitalization better than conventional methods. | 1076 | Multiple models with RF producing best results |
| AI-enabled Diagnostic Imaging Studies | |||
| [24], 2018—1b | Location: United States Aim: Determining the diagnostic performance of cPSTA in assessing CAD in patients presenting with chest pain who had been referred by their physician for coronary angiography. Variables: cPSTA recorded signals Strengths: Prospective Limitations: Small sample size Findings: The machine-learned algorithm had a sensitivity of 92% and specificity of 62% on blind testing in the verification cohort. The NPV was 96%. Notable facts: Resting cPSTA may have comparable diagnostic utility to functional tests currently used to assess CAD without requiring cardiac stress (exercise or pharmacological) or exposure of the patient to radioactivity. | 606 | Elastic net |
| [25], 2018—1b, 3a | Location: Multi-national Aim: Predicting lesion-specific ischemia by invasive FFR using an integrated ML ischemia risk score from quantitative plaque measures from CCTA. Variables: Quantitative CTA data: stenosis, NCP, low-density NCP (LD-NCP), calcified and total plaque volumes, contrast density difference (maximum difference in luminal attenuation per unit area) and plaque length Strengths: Multi-center data from NXT trial [116] Limitations: Small sample size; plaque findings were not confirmed by invasive intravascular ultrasound Findings: Information gain for predicting ischemia was highest for contrast density difference (0.172), followed by LD-NCP (0.125), NCP (0.097), and total plaque volumes (0.092). ML had higher AIUC (0.84) than individual CTA measures, including stenosis (0.76), LD-NCP volume (0.77), total plaque volume (0.74) and pre-test likelihood of CAD (0.63). | 254 | LogitBoost |
| [15], 2020—1a | Location: Multi-national Aim: Evaluate the prognostic value of fully automated DL-based EAT volume and attenuation quantified from non-contrast cardiac CT. Variables: Non-contrast cardiac CT scan data, inflammatory biomarkers Strengths: Data from the EISNER trial [23] Limitations: Long-term follow-up not obtained Findings: Increased EAT volume and decreased EAT attenuation were independently associated with MACE. CAD risk score, CAC, and EAT volume were associated with increased risk of MACE (hazard ratio: 1.03, 1.25, and 1.35). EAT attenuation was inversely associated with MACE (hazard ratio: 0.83, Harrell C statistic: 0.76). MACE risk progressively increased with EAT volume ≥ 113 cm3 and CAC ≥ 100 AU; highest in subjects with both. EAT volume correlated with inflammatory biomarkers; EAT attenuation inversely related to inflammatory biomarkers. | 2068 | DL |
| [117], 2018—1a | Location: Multi-national Aim: Investigating whether a ML score, using only plaque stenosis and composition information from the 16 coronary segments, has better predictive accuracy compared to the traditional CCTA based risk scores. Variables: 16 segment based coronary stenosis (0%, 1–24%, 25–49%, 50–69%, 70–99% and 100%) and composition (calcified, mixed and non-calcified plaque) derived from CCTA Strengths: Data from CONFIRM registry [110] Findings: ML-based approach showed better AUC for event discrimination (0.771) vs. other scores (ranging from 0.685 to 0.701). Improved risk stratification was the result of down-classification of risk among patients that did not experience events (non-events). | 8844 | XGBoost |
| [26], 2018—1b, 1c, 2c | Location: Multi-national Aim: Evaluating DL-based automatic prediction of obstructive disease from MPI, compared with TPD. Variables: MPI recorded data Strengths: Multi-center study Limitations: Retrospective; degree of stenosis from invasive angiography was interpreted visually Findings: AUC for DL was higher than for TPD (per patient: 0.80 vs. 0.78; per-vessel: 0.76 vs. 0.73). Sensitivity per patient improved from 79.8% (TPD) to 82.3% (DL), and per-vessel sensitivity improved from 64.4% (TPD) to 69.8% (DL). | 1018 | DCNN |
| [52], 2018—2c, 2d | Location: United States Aim: Evaluating the effectiveness of using Computer-Aided Diagnosis in the triage of low to intermediate risk emergency chest pain patients with CCTA. Variables: Data from 64 and 320 slice CCTA scanners Strengths: Looking at 30-day outcome Limitations: Retrospective Findings: Sensitivity: 85%; specificity: 50.6% and 56.5% for the 64 and 320 slice scanners. NPV: 97.8 and 97.1 for the 64 and 320 slice scanners. AUC: 0.6794 and 0.7097 for the 64 and 320 slice scanners. Software unable to read 18% of the cases. | 923 | Computer aided diagnosis software |
| [118], 2018—2c | Location: Multi-national Aim: Improving diagnostic performance of CTA to potentially reducing the number of unnecessary referrals for invasive coronary angiography. Variables: 28 variables from CTA data Strengths: Multi-center Limitations: Retrospective; possibility of selection bias due to the inclusion of patients with the disease only Findings: ML-FFR (AUC = 0.84) and CFD-FFR (AUC = 0.84) outperformed visual CTA (AUC = 0.69). Per-vessel and per-patient diagnostic accuracy improved 78% and 85%, respectively. ML-FFR correctly reclassified 73% of false-positive CTA results. Notable facts: On-site ML-FFR improves the performance of CTA by correctly reclassifying hemodynamically nonsignificant stenosis and performs equally well as CFD-FFR. | 351 | NN |
| [27], 2017—1b, 1c | Location: United States Aim: Evaluating the incremental benefit of ML-powered resting myocardial CTP over coronary CT stenosis for predicting ischemia Variables: CCTA and FFR data Strengths: Data from DeFACTO study [119] Limitations: Small sample size Findings: Accuracy, sensitivity, specificity, PPV, and NPV of resting CTP were 68.3%, 52.7%, 84.6%, 78.2%, and 63.0%, respectively, for predicting ischemia. Addition of resting CTP improved discrimination (AUC = 0.75) and reclassification (net reclassification improvement: 0.52) of ischemia compared to CT stenosis alone (AUC = 0.68). Notable facts: The addition of resting CTP analysis acquired from ML techniques may improve the predictive utility of significant ischemia over coronary stenosis. | 252 | Gradient boosting classifier |
| Ref., Year—Category ** | Study Details | Sample Size | Algorithms |
|---|---|---|---|
| AI and Risk Stratification Modeling | |||
| [18], 2019—1a | Location: China Aim: Proposed a new feature selection method to select important risk factors for detecting ischemic stroke. Variables: 24 blood test features and four demographic features Limitations: Single-center study Findings: Top nine features selected. Sensitivity: 82.7%, specificity: 80.4%, classification accuracy: 81.5%, Youden index: 0.63. | 792 | Weighting and ranking-based hybrid feature selection (WRHFS) |
| [19], 2017—1a | Location: China Aim: Build 2-year thromboembolism prediction models for AF patients, Variables: Chinese AF Registry data Strengths: Large dataset, two-year follow-up Limitations: Retrospective; design of the preprocessing and imputation strategy could lead to bias results and model overfitting Findings: AUC: 0.71–0.74. Notable facts: Model superior to previous thromboembolism prediction models. | 3535 | LR, Cox, NB, CART, RF |
| [20], 2018—1a | Location: China Aim: Build one-year ischemic stroke prediction models for AF patients. Variables: Chinese AF Registry data Strengths: Large dataset Limitations: Retrospective; highly imbalanced dataset (3.8% rate of stroke at one-year) Findings: AUC: 0.714. Notable facts: Boots-wrapper can balance model discrimination and statistical significance of features for developing AF stroke prediction models. | 3736 | Bootstrap-based wrapper for feature selection |
| [21], 2019—1a | Location: Taiwan Aim: Develop a predictive model to estimate three-year risk of ischemic stroke in the general population. Variables: Insurance claim data Strengths: Large sample size; model maintained high predictability five years after being developed. Limitations: Retrospective Findings: AUC: 0.920 (95% CI, 0.908–0.932) in testing dataset 1 and 0.925 (95% CI, 0.914–0.937) in testing dataset 2. Sensitivity and specificity were 80.3–92.5% and 79.8–87.5% for testing dataset 1; 83.7–91.8% and 79.9–87.5% for testing dataset 2. Notable facts: DNN algorithm is capable of obtaining a high performing model for assessment of ischemic stroke risk. | 840,487 | DNN |
| [93], 2019—3b | Location: China Aim: Identify the ischemic stroke readmission risk factors and establish a 90-day readmission prediction model for first-time ischemic stroke patients. Variables: Clinical data Strengths: Compared predictions at various follow-up periods Limitations: Retrospective; imputation of missing values is not discussed; dataset highly imbalanced (8.6% readmission rate) Findings: Standard AUC: 0.782 (0.729–0.834); best time-dependent AUC : 0.808 in 54 days. Notable facts: XGboost model obtained a better risk prediction for 90-day readmission for first-time ischemic stroke patients than the LR model. | 6070 | XGBoost, LR |
| AI-enabled Diagnostic Studies | |||
| [42], 2017—2b, 2d | Location: USA Aim: Recognize acute cerebral ischemia and differentiate that from stroke mimics at the initial examination. Variables: Clinical data Strengths: Prospective; ten-fold cross-validation Limitations: Stroke subtypes not classified Findings: Sensitivity: 80.0% (95% CI, 71.8–86.3); specificity: 86.2% (95% CI, 78.7–91.4); median precision: 92% (95% CI, 88.7–95.3). Notable facts: ANN can be an effective tool to recognize ACI and differentiate it from stroke mimics at the initial examination. | 260 | ANN |
| [49], 2019—2b | Location: Korea Aim: Detecting stroke and modeling mortality; stroke definition based on ICD code. Variables: Gender, age, type of insurance, admission type, brain surgery required, region, LOS, hospital location, number of hospital beds, stroke type, and CCI Strengths: Large sample size Limitations: Retrospective Findings: AUC: 83.48%. Notable facts: A scaled PCA/deep neural network approach can be used by both patients and doctors to prescreen for possible stroke. | 15,099 | PCA, DNN, RF, GNB, KNNC, SVM, ADB |
| [45], 2019—2b, 2c | Location: Sweden Aim: Detecting intracranial bleeding using simulated microwave transmission data, leveraging numerical simulation based on 3D finite-difference time-domain modeling. Variables: Computational model from an anatomical tissue of a human head; bleeding model is simplified representation of intracranial bleeding (resembling acute phase) Strengths: Simulated cohort Limitations: Retrospective Findings: With a sample size that approached 1000 subjects, classification results characterized as AUC > 0.9. Notable facts: Results indicate very high sensitivity and specificity. | Synthetic cohort | BC |
| [94], 2019—3b | Location: China Aim: Identifying high-risk TIA or minor stroke patients (recurrent ischemic stroke within one year). Variables: Demographics, clinical and imaging data Strengths: Patients with stroke or TIA mimics were excluded Limitations: Retrospective; downsampling the majority class applied to address data imbalance Findings: ANN median sensitivity: 75%; specificity: 75%; accuracy: 75%; c statistic: 0.77. Notable facts: ANN model outperformed SVM and Naïve Bayes. | 451 | ANN, SVM, NB |
| [46], 2019—2b, 2c | Location: USA Aim: Detecting acute intracranial hemorrhage on head CT scans using DL. Variables: CT scan data Strengths: Large sample size Limitations: Retrospective Findings: AUC: 0.991 ± 0.006. Notable facts: Demonstrated end-to-end network that performs joint classification and segmentation with examination-level classification comparable to experts, in addition to robust localization of abnormalities. | 4596 | FCN |
| [60], 2018—2d | Location: USA Aim: Classifying acute ischemic stroke onset time. Variables: MRI features Strengths: Extracted hidden representations from the MR perfusion-weighted images Limitations: Retrospective; possibly selection bias due to missingness; only ~10% of patients had sufficient information to be included in the study Findings: AUC: 0.68. Notable facts: Classification significantly improved over current clinical methods, demonstrating the potential benefit of using ML methods in TSS classification. | 105 | FLIRT, SMR, SVM, RF, GBRT |
| AI in Outcome Prediction/Prognosis | |||
| [96], 2018—3a, 3b | Location: USA Aim: Developing and validating model for delayed cerebral ischemia after subarachnoid hemorrhage. Variables: Age, sex, Hunt-Hess grade, modified Fisher Scale (mFS), and Glasgow Coma Scale (GCS) Strengths: Prospective Limitations: Possibility of selection bias; patients with missingness excluded Findings: Standard grading scale (mFS): AUC 0.58; combined demographics and grading scales: AUC 0.60; random kernel derived physiologic features: AUC 0.74; combined baseline and physiologic features with redundant feature reduction: AUC 0.77. | 488 | PLS, linear & kernel SVM |
| [91], 2019—3b | Location: Korea Aim: Predict the three-month outcomes (mRS) in ischemic stroke patients. Variables: Clinical data Strengths: Large sample size Limitations: Retrospective Findings: DNN AUC was significantly higher than that of the ASTRAL score (0.888 vs. 0.839; p < 0.001) when 38 variables were used. When only the six variables from the ASTRAL score were used in the ML models, there was no significant difference in performance. | 2604 | DNN, RF, LR |
| [95], 2018—3a, 3b | Location: Netherlands Aim: Predicting the outcome of endovascular treatment for acute ischemic stroke. Variables: 53 baseline variables and 30 treatment variables Strengths: Large sample size Limitations: Retrospective Findings: Range mean AUC = 0.88–0.91 with a negligible difference of mean AUC (0.01; 95%CI: 0.00–0.01) between best performing ML algorithm (RF) and best performing LR model. Notable facts: In large vessel occlusion patients, ML did not outperform LR models in predicting reperfusion and three-month functional independence after endovascular treatment. Radiological outcome was more difficult to predict than clinical outcome at time of admission. | 1383 | Super Learner (ensemble method), RF, SVM, ANN |
| [92], 2019—3b | Location: Switzerland Aim: Predicting the outcome (mRS > 2) at 90 days in patients with acute ischemic stroke. Variables: Biomarkers available at admission, NIHSS score Limitations: Retrospective Findings: XGB and GBM AUC = 0.746 and 0.748; improved after adding NIHSS and feature selection to 0.884 and 0.877, respectively. Notable facts: DT-based GBMs can predict the recovery outcome of stroke patients at admission with a high AUC. | 512 | XGB, GBM |
| [120], 2018—3a | Location: China Aim: Identifying a neurological deterioration prognostic model, based on dehydration equations. Variables: age, sex, laboratory values, and vascular risk factor data Strengths: Feature selection by the Boruta algorithm Limitations: Retrospective Findings: After decreasing the number of variables from 18 to 5, the specificity of test samples for the SVM prediction model increased from 44.1% to 89.4%, and the AUC increased from 0.700 to 0.927. Notable facts: SVM algorithms can be used to establish a prediction model for dehydration-associated ND, with good classification results. | 382 | SVM |
| [100], 2018—4a, 4b | Location: Taiwan Aim: Prediction of Barthel index (BI) status at discharge to optimize care of post-stroke patients. Variables: 15 rehabilitation assessments variables Limitations: Retrospective; patients were excluded (43) due to incomplete data; ratio of men to women was 2:1 Findings: LR and RF algorithms performed higher (AUC = 0.79) than SVM (AUC = 0.77). Mean absolute error of SVM and LR in predicting BI at discharge were 9.86 and 9.95, respectively. Notable facts: The proposed ML-based method provides a promising and practical computer-assisted decision-making tool for predicting ADL in clinical practice. | 313 | SVM, RF, LR |
| AI in Treatment Strategies | |||
| [35], 2018—1a, 1b, 1c | Location: Switzerland Aim: Investigating whether a CDS tool for stroke prevention integrated in EHR could improve adherence to guidelines in patients with AF in a PCP setting. Strengths: Randomized clinical trial; the analysis was carried out in a catchment area with high baseline adherence rate Findings: No difference observed in the incidence of stroke, TIA, or systemic thromboembolism in CDS group vs. control group. CDS group had a lower incidence of significant bleeding. | 444,347 | CDS system |
| [59], 2019—2d | Location: USA Aim: Develop a regression tree model predict 90-day modified Rankin Scale (mRS) scores to aid with ET. Variables: Elderly patients defined as ≥ 80 years of age Strengths: Retrospective and prospective; the model validated using an independent prospective cohort (36) of patients presenting to the same institution Limitations: Small sample size Findings: Sensitivity: 89.36%; specificity: 89.66%; AUC: 0.952. Notable facts: Algorithm is useful to determine which patients to exclude from ET, and has been implemented in an online calculator for public use. | 110 | Regression tree |
| AI-enabled Diagnostic Imaging Studies | |||
| [47], 2018—2b, 2c, 2d | Location: USA Aim: Detecting and quantifying intraparenchymal, epidural, subdural and subarachnoid hemorrhages on non-contrast CT (NCCT) and estimating hemorrhage volume. Variables: Training set: 10,159 NCCT examinations, 901 of which contained hemorrhage. Testing set: 682 prospective NCCT examinations, 82 of which contained hemorrhage Strengths: Retrospective and prospective evaluation Findings: Hemorrhage detection accuracy: 0.970, AUC: 0.981, sensitivity: 0.951, specificity: 0.973, PPV: 0.829, and NPV: 0.993. Dice scores for intraparenchymal hemorrhage: 0.931, epidural/subdural hemorrhage: 0.863. SAH: 0.772. | 10,841 | CNN |
| [55], 2019—2c, 2d | Location: International Aim: Segmentation and phenotyping of acute ischemic lesions on MRI. Variables: MRI data Strengths: Single-center cohort: 267; MRI-GENIE cohort (from 12 international centers from the Stroke Genetics Network): 3301 Limitations: Retrospective Findings: No algorithm-specific results reported. Automated and manual lesion volumes were statistically correlated. Notable facts: Deep learning algorithms trained on diverse data can be successfully used for segmentation of clinical diffusion-weighted MRI lesions. | 3568 | CNN |
| [48], 2019—2b, 2c | Location: China Aim: Detecting ICH and subtypes (cerebral, parenchymal, intraventricular, subdural, epidural, and subarachnoid) in NCCT. Variables: CT scan image slices data Strengths: Multi-institutional Limitations: Retrospective; prevalence of ICH (65%) was higher than that in a real clinical setting; limited number of cases in some subtypes (case/control ratio of 1:14); comparison was made with junior radiology trainees Findings: AUC (detecting ICH): 0.98; AUC (detecting subtype): 0.8. | 2836 | CNN-RNN |
| [56], 2019—2c, 3b | Location: China Aim: Predicting hematoma expansion in patients with spontaneous ICH. Variables: Fibrinogen level, sex, Glasgow Coma Score, time to initial CT scan, black hole sign, blend sign, satellite sign, midline shift, and baseline hematoma volume Strengths: Large sample size Limitations: Retrospective Findings: Sensitivity: 81.3%; specificity: 84.8%; accuracy of 83.3%; AUC: 0.89. Notable facts: Potential utility in institutions where CTA is limited. | 1157 | SVM |
| [29], 2019—1b, 1c | Location: Japan Aim: Detecting cerebral aneurysms at time-of-flight MR angiography. Variables: MRA image data Limitations: Retrospective; variable number of training samples per aneurysm location Findings: Sensitivity: 91–93% Notable facts: The model improved aneurysm detection by 4.8–13% compared with the initial reports. | 748 | DL (ResNet) |
| [44], 2019—2b, 2c | Location: USA Aim: Using an automated algorithm to detect intracranial LVO on CTA. Variables: CTA image data Limitations: Retrospective Findings: Sensitivity: 92–94%, NPV: 97–98%; specificity 0.76–0.81. Notable facts: RAPID CTA can be used in the emergent setting as a screening tool to alert radiologists. | 477 | RAPID CTA |
| [43], 2019—2b, 2c | Location: USA Aim: Identifying LVO and ischemic core volume in patients using CTA. Variables: CTA image data Strengths: Comparison with RAPID CTA Limitations: Retrospective; 338 patients excluded mainly due to imaging artifacts/quality Findings: AUC (LVO detection): 0.88; AUC (Ischemic core detection ≤ 30 mL): 0.88; AUC (Ischemic core detection ≤ 50 mL): 0.90; AUC (early time window): 0.90; AUC (late time window): 0.91. Notable facts: CTA has the required information for neuroimaging evaluation of endovascular therapy with potential to be automated by ML. | 297 | CNN (DeepSymNet) |
| [90], 2018—3b | Location: Denmark Aim: Use deep learning to identify and combine acute imaging features of ischemic stroke to predict lesion volume. Variables: MRI data Strengths: Comparing different CNNs Limitations: Retrospective; no control group; model is potentially biased toward infarct overestimation Findings: AUC: 0.88 ± 0.12. Notable facts: CNN improved prediction accuracy over current methods. | 222 | CNN |
| [54], 2017—2c | Location: USA Aim: Distinguishing between hyperacute ischemic lesions and their corresponding contralateral brain tissue in NCCT Variables: CT image data Limitations: Retrospective; used contralateral hemisphere as control possibly capturing old ischemic lesions. Findings: AUC: 0.82. Notable facts: Optimal texture features provided to distinguish between hyperacute ischemic lesions and their corresponding contralateral brain tissue in NCCT. | 139 | SVM, Decision trees, AdaBoost |
| [57], 2019—2c | Location: USA and Australia Aim: Predicting ischemic core on CT perfusion image. Variables: CT image data Strengths: Included patients who underwent back-to-back CT perfusion imaging and MRI Limitations: Retrospective; possibly overestimating the ischemic core volume (due to the dependency on the arbitrary subregion of the brain) Findings: AUC (ischemic core prediction): 0.85–0.87; sensitivity (ischemic core prediction): 0.90–0.91; specificity (ischemic core prediction): 0.62–0.65; maximal Dice coefficient: 0.48. Notable facts: ANN accurately integrates clinical and CT perfusion imaging data to predict ischemic core. | 128 | ANN |
4. Other Applications of AI
4.1. Clinical Trials in the AI-Era
4.2. AI at Physicians’ Fingertips—Implication and Future Directions
4.3. Health Disparity and Implicit Bias
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Noorbakhsh-Sabet, N.; Zand, R.; Zhang, Y.; Abedi, V. Artificial Intelligence Transforms the Future of Health Care. Am. J. Med. 2019, 132, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Krittanawong, C.; Zhang, H.; Wang, Z.; Aydar, M. Artificial Intelligence in Precision Cardiovascular Medicine. J. Am. Coll. Cardiol. 2017, 691, 2657–2664. [Google Scholar] [CrossRef] [PubMed]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2013, 380, 2095–2128. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart disease and stroke statistics–2018 update: A report from the American Heart Association. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef] [PubMed]
- Rowley, W.R.; Bezold, C.; Arikan, Y.; Byrne, E.; Krohe, S. Diabetes 2030: Insights from Yesterday, Today, and Future Trends. Popul. Health Manag. 2017, 20, 6–12. [Google Scholar] [CrossRef]
- Leischik, R.; Dworrak, B.; Strauss, M.; Przybylek, B.; Dworrak, T.; Schöne, D.; Horlitz, M.; Mügge, A. Plasticity of Health. Ger. J. Med. 2016, 1, 1–17. [Google Scholar]
- Sharma, V.; Sharma, V.; Shahjouei, S.; Li, J.; Chaudhary, D.; Khan, A.; Wolk, D.M.; Zand, R.; Abedi, V. At the Intersection of Gut Microbiome and Stroke: A Systematic Review of the Literature. Front. Neurol. 2021, 12, 729399. [Google Scholar] [CrossRef] [PubMed]
- Lv, S.; Wang, Y.; Zhang, W.; Shang, H. Trimethylamine oxide: A potential target for heart failure therapy. Heart 2021. [Google Scholar] [CrossRef] [PubMed]
- Collins, G.S.; Reitsma, J.B.; Altman, D.G.; Moons, K.G.M. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD statement. BMJ 2015, 4, g7594. [Google Scholar] [CrossRef]
- Weng, S.F.; Reps, J.; Kai, J.; Garibaldi, J.M.; Qureshi, N. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS ONE 2017, 12, e0174944. [Google Scholar] [CrossRef]
- Commandeur, F.C.; Slomka, P.J.; Goeller, M.; Chen, X.; Cadet, S.; Razipour, A.; Gransar, H.; Cantu, S.; Miller, R.; Rozanski, A.; et al. Machine learning to predict the long-term risk of myocardial infarction and cardiac death based on clinical risk, coronary calcium, and epicardial adipose tissue: A prospective study. Cardiovasc. Res. 2019, 40, ehz747-0002. [Google Scholar] [CrossRef] [PubMed]
- Ambale-Venkatesh, B.; Yang, X.; Wu, C.O.; Liu, K.; Hundley, W.G.; McClelland, R.; Gomes, A.S.; Folsom, A.R.; Shea, S.; Guallar, E.; et al. Cardiovascular Event Prediction by Machine Learning. Circ. Res. 2017, 121, 1092–1101. [Google Scholar] [CrossRef]
- Kakadiaris, I.A.; Vrigkas, M.; Yen, A.A.; Kuznetsova, T.; Budoff, M.; Naghavi, M. Machine learning outperforms ACC/AHA CVD risk calculator in MESA. J. Am. Heart Assoc. 2018, 72, e009476. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cui, Y.; Zhu, Y.; Yan, H.; Xu, W. Association of high normal HbA1c and TSH levels with the risk of CHD: A 10-year cohort study and SVM analysis. Sci. Rep. 2017, 7, 45406. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, E.; McElhinney, P.A.; Commandeur, F.; Chen, X.; Cadet, S.; Goeller, M.; Razipour, A.; Gransar, H.; Cantu, S.; Miller, R.J.; et al. Deep Learning–Based Quantification of Epicardial Adipose Tissue Volume and Attenuation Predicts Major Adverse Cardiovascular Events in Asymptomatic Subjects. Circ. Cardiovasc. Imaging 2020, 13, e009829. [Google Scholar] [CrossRef]
- Samad, M.D.; Ulloa, A.; Wehner, G.J.; Jing, L.; Hartzel, D.; Good, C.W.; Williams, B.A.; Haggerty, C.M.; Fornwalt, B.K. Predicting Survival from Large Echocardiography and Electronic Health Record Datasets: Optimization with Machine Learning. JACC Cardiovasc. Imaging 2019, 12, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.M.; Johnson, H.E.; Zhao, Y.; Dowe, D.A.; Staib, L.H. Scoring of Coronary Artery Disease Characteristics on Coronary CT Angiograms by Using Machine Learning. Radiology 2019, 292, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, Y.; Zhang, D.; Song, W. A Stroke Risk Detection: Improving Hybrid Feature Selection Method. J. Med. Internet Res. 2019, 21, e12437. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, H.; Du, X.; Zhang, P.; Hu, G.; Xie, G.; Guo, S.; Xu, M.; Xie, X. Integrated Machine Learning Approaches for Predicting Ischemic Stroke and Thromboembolism in Atrial Fibrillation. AMIA Annu. Symp. Proc. 2017, 2016, 799–807. [Google Scholar]
- Li, X.; Sun, Z.; Du, X.; Liu, H.; Hu, G.; Xie, G. Bootstrap-based Feature Selection to Balance Model Discrimination and Predictor Significance: A Study of Stroke Prediction in Atrial Fibrillation. AMIA Annu. Symp. 2017, 2017, 1130–1139. [Google Scholar]
- Hung, C.-Y.; Lin, C.-H.; Lan, T.-H.; Peng, G.-S.; Lee, C.-C. Development of an intelligent decision support system for ischemic stroke risk assessment in a population-based electronic health record database. PLoS ONE 2019, 14, e0213007. [Google Scholar] [CrossRef] [PubMed]
- Bild, D.E.; Bluemke, D.; Burke, G.L.; Detrano, R.; Roux, A.V.D.; Folsom, A.R.; Greenland, P.; Jacobs, D.R., Jr.; A Kronmal, R.; Liu, K.; et al. Multi-Ethnic Study of Atherosclerosis: Objectives and design. Am. J. Epidemiol. 2002, 156, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Rozanski, A.; Gransar, H.; Shaw, L.J.; Kim, J.; Miranda-Peats, L.; Wong, N.D.; Rana, J.S.; Orakzai, R.; Hayes, S.W.; Friedman, J.D.; et al. Impact of coronary artery calcium scanning on coronary risk factors and downstream testing the EISNER (Early Identification of Subclinical Atherosclerosis by Noninvasive Imaging Research) prospective randomized trial. J. Am. Coll. Cardiol. 2011, 575, 1622–1632. [Google Scholar] [CrossRef] [PubMed]
- Stuckey, T.D.; Gammon, R.S.; Goswami, R.; Depta, J.P.; Steuter, J.A.; Meine, F.J., 3rd; Roberts, M.C.; Singh, N.; Ramchandani, S.; Burton, T.; et al. Cardiac Phase Space Tomography: A novel method of assessing coronary artery disease utilizing machine learning. PLoS ONE 2018, 13, e0198603. [Google Scholar]
- Dey, D.; Gaur, S.; Ovrehus, K.A.; Slomka, P.J.; Betancur, J.; Goeller, M.; Hell, M.M.; Gransar, H.; Berman, D.S.; Achenbach, S.; et al. Integrated prediction of lesion-specific ischaemia from quantitative coronary CT angiography using machine learning: A multicentre study. Eur. Radiol. 2018, 28, 2655–2664. [Google Scholar] [CrossRef] [PubMed]
- Betancur, J.; Commandeur, F.; Motlagh, M.; Sharir, T.; Einstein, A.J.; Bokhari, S.; Fish, M.B.; Ruddy, T.D.; Kaufmann, P.; Sinusas, A.J.; et al. Deep Learning for Prediction of Obstructive Disease from Fast Myocardial Perfusion SPECT: A Multicenter Study. JACC Cardiovasc. Imaging 2018, 111, 1654–1663. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Lee, J.H.; Rizvi, A.; Gransar, H.; Baskaran, L.; Schulman-Marcus, J.; Hartaigh, B.Ó.; Lin, F.Y.; Min, J.K. Incremental role of resting myocardial computed tomography perfusion for predicting physiologically significant coronary artery disease: A machine learning approach. J. Nucl. Cardiol. 2018, 25, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Martinez, S.; Duchateau, N.; Erdei, T.; Kunszt, G.; Aakhus, S.; Degiovanni, A.; Marino, P.; Carluccio, E.; Piella, G.; Fraser, A.G.; et al. Machine learning analysis of left ventricular function to characterize heart failure with preserved ejection fraction. Circ. Cardiovasc. Imaging 2018, 11, e007138. [Google Scholar] [CrossRef] [PubMed]
- Ueda, D.; Yamamoto, A.; Nishimori, M.; Shimono, T.; Doishita, S.; Shimazaki, A.; Katayama, Y.; Fukumoto, S.; Choppin, A.; Shimahara, Y.; et al. Deep Learning for MR Angiography: Automated Detection of Cerebral Aneurysms. Radiology 2018, 290, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Cammarota, G.; Ianiro, G.; Ahern, A.; Carbone, C.; Temko, A.; Claesson, M.J.; Gasbarrini, A.; Tortora, G. Gut microbiome, big data and machine learning to promote precision medicine for cancer. Nat. Rev. Gastroenterol. Hepatol. 2020, 170, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, S.L.; Nir, G.; Salcudean, S.E. A new era: Artificial intelligence and machine learning in prostate cancer. Nat. Rev. Urol. 2019, 16, 391–403. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.; Churpek, M.M.; Calfee, C.S. Machine Learning Classifier Models Can Identify Acute Respiratory Distress Syndrome Phenotypes Using Readily Available Clinical Data. Am. J. Respir. Crit. Care Med. 2020, 202, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Tsoi, K.K.F.; Chan, N.B.; Yiu, K.K.L.; Poon, S.K.S.; Lin, B.; Ho, K. Machine Learning Clustering for Blood Pressure Variability Applied to Systolic Blood Pressure Intervention Trial (SPRINT) and the Hong Kong Community Cohort. Hypertension 2020, 76, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Fleuren, L.M.; Klausch, T.L.T.; Zwager, C.L.; Schoonmade, L.; Guo, T.; Roggeveen, L.F.; Swart, E.L.; Girbes, A.R.J.; Thoral, P.; Ercole, A.; et al. Machine learning for the prediction of sepsis: A systematic review and meta-analysis of diagnostic test accuracy. Intensive Care Med. 2020, 46, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, L.O.; Nilsson, S.; Bång, M.; Nilsson, L.; Charitakis, E.; Janzon, M. A clinical decision support tool for improving adherence to guidelines on anticoagulant therapy in patients with atrial fibrillation at risk of stroke: A cluster-randomized trial in a Swedish primary care setting (the CDS-AF study). PLoS Med. 2018, 15, e1002528. [Google Scholar] [CrossRef] [PubMed]
- Danchin, N.; Popovic, B.; Puymirat, E.; Goldstein, P.; Belle, L.; Cayla, G.; Roubille, F.; Lemesle, G.; Ferrières, J.; Schiele, F.; et al. Five-year outcomes following timely primary percutaneous intervention, late primary percutaneous intervention, or a pharmaco-invasive strategy in ST-segment elevation myocardial infarction: The FAST-MI programme. Eur. Heart J. 2020, 41, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, B.D.; Dhindsa, H.S.; Roe, M.T.; Chen, A.Y.; Jollis, J.G.; Kontos, M.C. Relationship of the Distance Between Non-PCI Hospitals and Primary PCI Centers, Mode of Transport, and Reperfusion Time Among Ground and Air Interhospital Transfers Using NCDR’s ACTION Registry-GWTG. Circ. Cardiovasc. Interv. 2014, 7, 797–805. [Google Scholar] [CrossRef]
- Peretz, S.; Raphaeli, G.; Borenstein, N.; Leker, R.R.; Brauner, R.; Horev, A.; E Cohen, J.; Telman, G.; Halevi, H.; Tanne, D. Effect of time from onset to endovascular therapy on outcomes: The National Acute Stroke Israeli (NASIS)-REVASC registry. J. Neurointerv. Surg. 2020, 12, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Potter, B.J.; Matteau, A.; Mansour, S.; Naim, C.; Riahi, M.; Essiambre, R.; Montigny, M.; Sareault, I.; Gobeil, F. Sustained Performance of a ‘Physicianless’ System of Automated Prehospital STEMI Diagnosis and Catheterization Laboratory Activation. Can. J. Cardiol. 2017, 33, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Lyon, A.; Mincholé, A.; Martínez, J.; Laguna, P.; Rodriguez, B. Computational techniques for ECG analysis and interpretation in light of their contribution to medical advances. J. R. Soc. Interface 2018, 1538, 20170821. [Google Scholar] [CrossRef] [PubMed]
- Goto, S.; Kimura, M.; Katsumata, Y.; Goto, S.; Kamatani, T.; Ichihara, G.; Ko, S.; Sasaki, J.; Fukuda, K.; Sano, M. Artificial intelligence to predict needs for urgent revascularization from 12-leads electrocardiography in emergency patients. PLoS ONE 2019, 14, e0210103. [Google Scholar]
- Abedi, V.; Goyal, N.; Tsivgoulis, G.; Hosseinichimeh, N.; Hontecillas, R.; Bassaganya-Riera, J.; Elijovich, L.; Metter, J.E.; Alexandrov, A.W.; Liebeskind, D.S.; et al. Novel Screening Tool for Stroke Using Artificial Neural Network. Stroke 2017, 48, 1678–1681. [Google Scholar] [CrossRef]
- Sheth, S.A.; Lopez-Rivera, V.; Barman, A.; Grotta, J.C.; Yoo, A.J.; Lee, S.; Inam, M.E.; Savitz, S.I.; Giancardo, L. Machine Learning-Enabled Automated Determination of Acute Ischemic Core from Computed Tomography Angiography. Stroke 2019, 501, 3093–3100. [Google Scholar] [CrossRef] [PubMed]
- Amukotuwa, S.A.; Straka, M.; Smith, H.; Chandra, R.; Dehkharghani, S.; Fischbein, N.J.; Bammer, R. Automated Detection of Intracranial Large Vessel Occlusions on Computed Tomography Angiography. Stroke 2019, 500, 2790–2798. [Google Scholar] [CrossRef] [PubMed]
- Fhager, A.; Candefjord, S.; Elam, M.; Persson, M. 3D Simulations of Intracerebral Hemorrhage Detection Using Broadband Microwave Technology. Sensors 2019, 196, 3482. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.; Häne, C.; Mukherjee, P.; Malik, J.; Yuh, E.L. Expert-level detection of acute intracranial hemorrhage on head computed tomography using deep learning. Proc. Natl. Acad. Sci. USA 2019, 1165, 22737–22745. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.D.; Kuoy, E.; Grinband, J.; Weinberg, B.D.; Thompson, M.; Homo, R.; Chen, J.; Abcede, H.; Shafie, M.; Sugrue, L.; et al. Hybrid 3D/2D Convolutional Neural Network for Hemorrhage Evaluation on Head CT. AJNR. Am. J. Neuroradiol. 2018, 39, 1609–1616. [Google Scholar] [CrossRef]
- Ye, H.; Gao, F.; Yin, Y.; Guo, D.; Zhao, P.; Lu, Y.; Wang, X.; Bai, J.; Cao, K.; Song, Q.; et al. Precise diagnosis of intracranial hemorrhage and subtypes using a three-dimensional joint convolutional and recurrent neural network. Eur. Radiol. 2019, 291, 6191–6201. [Google Scholar] [CrossRef] [PubMed]
- Cheon, S.; Kim, J.; Lim, J. The Use of Deep Learning to Predict Stroke Patient Mortality. Int. J. Environ. Res. Public Health 2019, 161, 1876. [Google Scholar] [CrossRef]
- Abedi, V.; Avula, V.; Razavi, S.-M.; Shreya, B.; Chaudhary, D.; Shahjouei, S.; Wang, M.; Griessenauer, C.J.; Li, J.; Zand, R. Predicting short and long-term mortality after acute ischemic stroke using EHR. J. Neurol. Sci. 2021, 427, 117560. [Google Scholar] [CrossRef]
- Stanciu, A.; Banciu, M.; Sadighi, A.; Marshall, K.A.; Holland, N.R.; Abedi, V.; Zand, R. A predictive analytics model for differentiating between transient ischemic attacks (TIA) and its mimics. BMC Med. Inform. Decis. Mak. 2020, 20, 112. [Google Scholar] [CrossRef] [PubMed]
- alamir, M.A.; Noack, P.; Jang, K.H.; Moore, J.A.; Goldberg, R.; Poon, M. Computer-aided analysis of 64- and 320-slice coronary computed tomography angiography: A comparison with expert human interpretation. Int. J. Cardiovasc. Imaging 2018, 34, 1473–1483. [Google Scholar] [CrossRef]
- Noh, Y.K.; Park, J.Y.; Choi, B.G.; Kim, K.E.; Rha, S.W. A Machine Learning-Based Approach for the Prediction of Acute Coronary Syndrome Requiring Revascularization. J. Med. Syst. 2019, 43, 253. [Google Scholar] [CrossRef] [PubMed]
- Peter, R.; Korfiatis, P.; Blezek, D.; Beitia, A.O.; Stepan-Buksakowska, I.; Horinek, D.; Flemming, K.D.; Erickson, B.J. A quantitative symmetry-based analysis of hyperacute ischemic stroke lesions in noncontrast computed tomography. Med. Phys. 2017, 44, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Wu, O.; Winzeck, S.; Giese, A.-K.; Hancock, B.L.; Etherton, M.R.; Bouts, M.J.; Donahue, K.; Schirmer, M.D.; Irie, R.E.; Mocking, S.J.; et al. Big Data Approaches to Phenotyping Acute Ischemic Stroke Using Automated Lesion Segmentation of Multi-Center Magnetic Resonance Imaging Data. Stroke 2019, 50, 1734–1741. [Google Scholar] [CrossRef]
- Liu, J.; Xu, H.; Chen, Q.; Zhang, T.; Sheng, W.; Huang, Q.; Song, J.; Huang, D.; Lan, L.; Li, Y.; et al. Prediction of hematoma expansion in spontaneous intracerebral hemorrhage using support vector machine. EBioMedicine 2019, 43, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Kasasbeh, A.S.; Christensen, S.; Parsons, M.W.; Campbell, B.; Albers, G.W.; Lansberg, M.G. Artificial Neural Network Computer Tomography Perfusion Prediction of Ischemic Core. Stroke 2019, 50, 1578–1581. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wu, T.T.; Yang, D.L.; Guo, Y.S.; Liu, P.C.; Chen, Y.; Xiao, L.P. Decision tree model for predicting in-hospital cardiac arrest among patients admitted with acute coronary syndrome. Clin. Cardiol. 2019, 421, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Alawieh, A.; Zaraket, F.; Alawieh, M.B.; Chatterjee, A.R.; Spiotta, A. Using machine learning to optimize selection of elderly patients for endovascular thrombectomy. J. Neurointerv. Surg. 2019, 11, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.C.; Speier, W.; El-Saden, S.; Arnold, C.W. Classifying Acute Ischemic Stroke Onset Time using Deep Imaging Features. AMIA Annu. Symp. Proc. 2018, 2017, 892–901. [Google Scholar] [PubMed]
- Abedi, V.; Khan, A.; Chaudhary, D.; Misra, D.; Avula, V.; Mathrawala, D.; Kraus, C.; Marshall, K.; Chaudhary, N.; Li, X.; et al. Using artificial intelligence for improving stroke diagnosis in emergency departments: A practical framework. Ther. Adv. Neurol. Disord. 2020, 13, 175628642093896. [Google Scholar] [CrossRef] [PubMed]
- Ong, C.J.; Orfanoudaki, A.; Zhang, R.; Caprasse, F.P.M.; Hutch, M.; Ma, L.; Fard, D.; Balogun, O.; Miller, M.I.; Minnig, M.; et al. Machine learning and natural language processing methods to identify ischemic stroke, acuity and location from radiology reports. PLoS ONE 2020, 15, e0234908. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.; Oh, E.; Naidech, A.; Kording, K.; Prabhakaran, S. Automating Ischemic Stroke Subtype Classification Using Machine Learning and Natural Language Processing. J. Stroke Cerebrovasc. Dis. 2019, 28, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Adams, H.P., Jr.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E., 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Keerthana, S.; Sathiyakumari, K. Brain Stroke Segmentation using Fuzzy C-Means Clustering. Int. J. Comput. Appl. 2016, 154, 26–30. [Google Scholar] [CrossRef]
- Dey, D.; Slomka, P.J.; Leeson, P.; Comaniciu, D.; Shrestha, S.; Sengupta, P.P.; Marwick, T.H. Artificial Intelligence in Cardiovascular Imaging: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 154, 26–30. [Google Scholar]
- Ernande, L.; Audureau, E.; Jellis, C.L.; Bergerot, C.; Henegar, C.; Sawaki, D.; Czibik, G.; Volpi, C.; Canoui-Poitrine, F.; Thibault, H.; et al. Clinical Implications of Echocardiographic Phenotypes of Patients with Diabetes Mellitus. J. Am. Coll. Cardiol. 2017, 704, 1704–1716. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.; Deo, R.C.; Aguilar, F.G.; Selvaraj, S.; Martinez, E.E.; Beussink-Nelson, M.L.; Kim, K.-Y.A.; Irvin, M.R.; Tiwari, H.; Rao, D.C.; et al. Phenomapping for the Identification of Hypertensive Patients with the Myocardial Substrate for Heart Failure with Preserved Ejection Fraction. J. Cardiovasc. Transl. Res. 2017, 10, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, M.C.; Omar, A.M.S.; Narula, S.; Kulkarni, H.; Narula, J.; Sengupta, P.P. Phenotypic Clustering of Left Ventricular Diastolic Function Parameters. JACC Cardiovasc. Imaging 2019, 12, 1149–1161. [Google Scholar] [CrossRef] [PubMed]
- Pecková, M.; Charvat, J.; Schuck, O.; Hill, M.; Svab, P.; Horackova, M. The Association between Left Ventricular Diastolic Function and a Mild-to-Moderate Decrease in Glomerular Filtration Rate in Patients with Type 2 Diabetes Mellitus. J. Int. Med. Res. 2011, 39, 2178–2186. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.J.; Katz, D.; Selvaraj, S.; Burke, M.A.; Yancy, C.W.; Gheorghiade, M.; Bonow, R.O.; Huang, C.-C.; Deo, R.C. Phenomapping for novel classification of heart failure with preserved ejection fraction. Circulation 2015, 131, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Y.; Schlueter, D.J.; Wu, P.; Kerchberger, V.; Rosenbloom, S.T.; Wells, Q.S.; Feng, Q.; Denny, J.C.; Wei, W.-Q. Detecting time-evolving phenotypic topics via tensor factorization on electronic health records: Cardiovascular disease case study. J. Biomed. Inform. 2019, 98, 103270. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Pencina, M.J.; Schulte, P.J.; O’Brien, E.; Whellan, D.J.; Piña, I.L.; Kitzman, D.W.; Lee, K.L.; O’Connor, C.M.; Felker, G. Clinical implications of chronic heart failure phenotypes defined by cluster analysis. J. Am. Coll. Cardiol. 2014, 647, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Schulam, P.; Wigley, F.; Saria, S. Clustering longitudinal clinical marker trajectories from electronic health data: Applications to phenotyping and endotype discovery. In Proceedings of the National Conference on Artificial Intelligence, Austin, TX, USA, 25–30 January 2015. [Google Scholar]
- Panahiazar, M.; Taslimitehrani, V.; Pereira, N.L.; Pathak, J. Using EHRs for Heart Failure Therapy Recommendation Using Multidimensional Patient Similarity Analytics. Stud. Health Technol. Inform. 2015, 210, 369–373. [Google Scholar] [PubMed]
- Motwani, M.; Dey, D.; Berman, D.S.; Germano, G.; Achenbach, S.; Al-Mallah, M.; Andreini, D.; Budoff, M.J.; Cademartiri, F.; Callister, T.Q.; et al. Machine learning for prediction of all-cause mortality in patients with suspected coronary artery disease: A 5-year multicentre prospective registry analysis. Eur. Heart J. 2017, 38, 500–507. [Google Scholar] [CrossRef]
- Cho, H.; Lee, J.-G.; Kang, S.-J.; Kim, W.-J.; Choi, S.-Y.; Ko, J.; Min, H.-S.; Choi, G.-H.; Kang, D.-Y.; Lee, P.H.; et al. Angiography-Based Machine Learning for Predicting Fractional Flow Reserve in Intermediate Coronary Artery Lesions. J. Am. Heart Assoc. 2019, 8, e011685. [Google Scholar] [CrossRef]
- Zack, C.J.; Senecal, C.; Kinar, Y.; Metzger, Y.; Bar-Sinai, Y.; Widmer, R.J.; Lennon, R.; Singh, M.; Bell, M.R.; Lerman, A.; et al. Leveraging Machine Learning Techniques to Forecast Patient Prognosis After Percutaneous Coronary Intervention. JACC Cardiovasc. Interv. 2019, 124, 1304–1311. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, H.; Elgendy, I.Y.; Segal, R.; Bavry, A.A.; Bian, J. Risk prediction model for in-hospital mortality in women with ST-elevation myocardial infarction: A machine learning approach. Heart Lung 2017, 46, 405–411. [Google Scholar] [CrossRef]
- Shouval, R.; Hadanny, A.; Shlomo, N.; Iakobishvili, Z.; Unger, R.; Zahger, D.; Alcalai, R.; Atar, S.; Gottlieb, S.; Matetzky, S.; et al. Machine learning for prediction of 30-day mortality after ST elevation myocardial infraction: An Acute Coronary Syndrome Israeli Survey data mining study. Int. J. Cardiol. 2017, 246, 7–13. [Google Scholar] [CrossRef]
- Li, Y.-M.; Jiang, L.-C.; He, J.-J.; Jia, K.-Y.; Peng, Y.; Chen, M. Machine Learning to Predict the 1-Year Mortality Rate After Acute Anterior Myocardial Infarction in Chinese Patients. Ther. Clin. Risk Manag. 2020, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wallert, J.; Tomasoni, M.; Madison, G.; Held, C. Predicting two-year survival versus non-survival after first myocardial infarction using machine learning and Swedish national register data. BMC Med. Inform. Decis. Mak. 2017, 17, 99. [Google Scholar] [CrossRef] [PubMed]
- Pieszko, K.; Hiczkiewicz, J.; Budzianowski, P.; Budzianowski, J.; Rzeźniczak, J.; Pieszko, K.; Burchardt, P. Predicting Long-Term Mortality after Acute Coronary Syndrome Using Machine Learning Techniques and Hematological Markers. Dis. Markers 2019, 2019, 9056402. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.-M.; Jeon, K.-H.; Kim, H.M.; Kim, M.J.; Lim, S.; Kim, K.-H.; Song, P.S.; Park, J.; Choi, R.K.; Oh, B.-H. Deep-learning-based risk stratification for mortality of patients with acute myocardial infarction. PLoS ONE 2019, 140, e0224502. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Sun, Z.; Dong, W.; Huang, Z. Utilizing dynamic treatment information for MACE prediction of acute coronary syndrome. BMC Med. Inform. Decis. Mak. 2019, 19, 5. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Lund, L.H.; Rao, P.; Ghosh, R.; Warier, P.; Vaccaro, B.; Dahlström, U.; O’connor, C.M.; Felker, G.M.; Desai, N.R. Machine Learning Methods Improve Prognostication, Identify Clinically Distinct Phenotypes, and Detect Heterogeneity in Response to Therapy in a Large Cohort of Heart Failure Patients. J. Am. Heart Assoc. 2018, 7, e008081. [Google Scholar] [CrossRef] [PubMed]
- Adler, E.D.; Voors, A.A.; Klein, L.; Macheret, F.; Braun, O.O.; Urey, M.A.; Zhu, W.; Sama, I.; Tadel, M.; Campagnari, C.; et al. Improving risk prediction in heart failure using machine learning. Eur. J. Heart Fail. 2019, 22, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.M.; Kim, K.H.; Jeon, K.H.; Lee, S.E.; Lee, H.Y.; Cho, H.J.; Choi, J.O.; Jeon, E.S.; Kim, M.S.; Kim, J.J.; et al. Artificial intelligence algorithm for predicting mortality of patients with acute heart failure. PLoS ONE 2019, 14, e0219302. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.-M.; Kim, K.-H.; Jeon, K.-H.; Lee, S.E.; Lee, H.-Y.; Cho, H.-J.; Choi, J.O.; Jeon, E.-S.; Kim, M.-S.; Kim, J.-J.; et al. Machine Learning of Three-dimensional Right Ventricular Motion Enables Outcome Prediction in Pulmonary Hypertension: A Cardiac MR Imaging Study. Radiology 2017, 283, 381–390. [Google Scholar]
- Anne, N.; Bo, H.M.; Anna, T.; Kim, M. Prediction of Tissue Outcome and Assessment of Treatment Effect in Acute Ischemic Stroke Using Deep Learning. Stroke 2018, 49, 1394–1401. [Google Scholar]
- Heo, J.; Yoon, J.G.; Park, H.; Kim, Y.D.; Nam, H.S.; Heo, J.H. Machine Learning–Based Model for Prediction of Outcomes in Acute Stroke. Stroke 2019, 50, 1263–1265. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Jiang, B.; Gong, E.; Li, Y.; Zhu, G.; Michel, P.; Wintermark, M.; Zaharchuk, G. Use of gradient boosting machine learning to predict patient outcome in acute ischemic stroke on the basis of imaging, demographic, and clinical information. Am. J. Roentgenol. 2019, 22, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Yang, X.; Huang, H.; Peng, C.; Ge, Y.; Wu, H.; Wang, J.; Xiong, G.; Yi, Y. Extreme Gradient Boosting Model Has a Better Performance in Predicting the Risk of 90-Day Readmissions in Patients with Ischaemic Stroke. J. Stroke Cerebrovasc. Dis. 2019, 282, 104441. [Google Scholar] [CrossRef]
- Chan, K.L.; Leng, X.; Zhang, W.; Dong, W.; Qiu, Q.; Yang, J.; Soo, Y.; Wong, K.S.; Leung, T.W.; Liu, J. Early Identification of High-Risk TIA or Minor Stroke Using Artificial Neural Network. Front. Neurol. 2019, 10, 171. [Google Scholar] [CrossRef] [PubMed]
- Van Os, H.J.A.; Ramos, L.A.; Hilbert, A.; van Leeuwen, M.; Van Walderveen, M.A.A.; Kruyt, N.D.; Dippel, D.W.J.; Steyerberg, E.W.; Van Der Schaaf, I.C.; Lingsma, H.F.; et al. Predicting outcome of endovascular treatment for acute ischemic stroke: Potential value of machine learning algorithms. Front. Neurol. 2018, 9, 784. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Megjhani, M.; Frey, H.-P.; Grave, E.; Wiggins, C.; Terilli, K.L.; Roh, D.J.; Velázquez, Á.; Agarwal, S.; Connolly, E.S.; et al. Predicting delayed cerebral ischemia after subarachnoid hemorrhage using physiological time series data. J. Clin. Monit. Comput. 2019, 33, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Broderick, J.; Adeoye, O.; Elm, J. Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke 2017, 48, 2007–2012. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, C.; Desai, N.R.; Mulder, H.; Alhanti, B.; Wilson, F.P.; Fiuzat, M.; Felker, G.M.; Piña, I.L.; O’Connor, C.M.; Lindenfeld, J.; et al. Clinical Implications of the New York Heart Association Classification. J. Am. Heart Assoc. 2019, 83, e014240. [Google Scholar] [CrossRef]
- Cikes, M.; Sanchez-Martinez, S.; Claggett, B.; Duchateau, N.; Piella, G.; Butakoff, C.; Pouleur, A.C.; Knappe, D.; Biering-Sørensen, T.; Kutyifa, V.; et al. Machine learning-based phenogrouping in heart failure to identify responders to cardiac resynchronization therapy. Eur. J. Heart Fail. 2019, 21, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.-Y.; Chen, C.-H.; Tseng, Y.-J.; Tsai, Y.-T.; Chang, C.-Y.; Wang, H.-Y.; Chen, C.-K. Predicting post-stroke activities of daily living through a machine learning-based approach on initiating rehabilitation. Int. J. Med. Inform. 2018, 111, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Shields, G.E.; Wells, A.; Doherty, P.; Heagerty, A.; Buck, D.; Davies, L.M. Cost-effectiveness of cardiac rehabilitation: A systematic review. Heart 2018, 1047, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Rajsic, S.; Gothe, H.; Borba, H.H.; Sroczynski, G.; Vujicic, J.; Toell, T.; Siebert, U. Economic burden of stroke: A systematic review on post-stroke care. Eur. J. Health Econ. 2019, 20, 107–134. [Google Scholar] [CrossRef]
- Goetz, L.H.; Schork, N.J. Personalized medicine: Motivation, challenges, and progress. Fertil. Steril. 2018, 109, 952–963. [Google Scholar] [CrossRef] [PubMed]
- Feeny, A.K.; Rickard, J.; Patel, D.; Toro, S.; Trulock, K.M.; Park, C.J.; LaBarbera, M.A.; Varma, N.; Niebauer, M.J.; Sinha, S.; et al. Machine Learning Prediction of Response to Cardiac Resynchronization Therapy: Improvement Versus Current Guidelines. Circ. Arrhythm. Electrophysiol. 2019, 12, e007316. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.-Y.; Santus, E.; Forsyth, A.W.; Malhotra, D.; Haimson, J.; Chatterjee, N.A.; Kramer, D.B.; Barzilay, R.; Tulsky, J.A.; Lindvall, C. Can machine learning improve patient selection for cardiac resynchronization therapy? PLoS ONE 2019, 140, e0222397. [Google Scholar] [CrossRef]
- Kalscheur, M.M.; Kipp, R.T.; Tattersall, M.C.; Mei, C.; Buhr, K.A.; DeMets, D.L.; Field, M.E.; Eckhardt, L.L.; Page, C.D. Machine Learning Algorithm Predicts Cardiac Resynchronization Therapy Outcomes: Lessons from the COMPANION Trial. Circ. Arrhythm. Electrophysiol. 2018, 11, e005499. [Google Scholar] [CrossRef] [PubMed]
- Isma’Eel, H.A.; Sakr, G.E.; Serhan, M.; Lamaa, N.; Hakim, A.; Cremer, P.C.; Jaber, W.A.; Garabedian, T.; Elhajj, I.; Abchee, A.B. Artificial neural network-based model enhances risk stratification and reduces non-invasive cardiac stress imaging compared to Diamond-Forrester and Morise risk assessment models: A prospective study. J. Nucl. Cardiol. Off. Publ. Am. Soc. Nucl. Cardiol. 2018, 25, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Stolarz-Skrzypek, K.; Kuznetsova, T.; Thijs, L.; Tikhonoff, V.; Seidlerová, J.; Richart, T.; Jin, Y.; Olszanecka, A.; Malyutina, S.; Casiglia, E.; et al. Fatal and nonfatal outcomes, incidence of hypertension, and blood pressure changes in relation to urinary sodium excretion. JAMA 2011, 3057, 1777–1785. [Google Scholar] [CrossRef]
- Ayatollahi, H.; Gholamhosseini, L.; Salehi, M. Predicting coronary artery disease: A comparison between two data mining algorithms. BMC Public Health 2019, 19, 448. [Google Scholar] [CrossRef] [PubMed]
- Min, J.K.; Dunning, A.; Lin, F.Y.; Achenbach, S.; Al-Mallah, M.H.; Berman, D.S.; Budoff, M.J.; Cademartiri, F.; Callister, T.Q.; Chang, H.-J.; et al. Rationale and design of the CONFIRM (COronary CT Angiography EvaluatioN for Clinical Outcomes: An InteRnational Multicenter) Registry. J. Cardiovasc. Comput. Tomogr. 2011, 5, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Bhuva, A.N.; Bai, W.; Lau, C.; Davies, R.H.; Ye, Y.; Bulluck, H.; McAlindon, E.; Culotta, V.; Swoboda, P.; Captur, G.; et al. A Multicenter, Scan-Rescan, Human and Machine Learning CMR Study to Test Generalizability and Precision in Imaging Biomarker Analysis. Circ. Cardiovasc. Imaging 2019, 120, e009214. [Google Scholar] [CrossRef] [PubMed]
- Hearn, J.; Ross, H.J.; Mueller, B.; Fan, C.-P.; Crowdy, E.; Duhamel, J.; Walker, M.; Alba, A.C.; Manlhiot, C. Neural Networks for Prognostication of Patients with Heart Failure. Circ. Heart Fail. 2018, 11, e005193. [Google Scholar] [CrossRef]
- Jamthikar, A.; Gupta, D.; Khanna, N.N.; Saba, L.; Araki, T.; Viskovic, K.; Suri, H.S.; Gupta, A.; Mavrogeni, S.; Turk, M.; et al. A low-cost machine learning-based cardiovascular/stroke risk assessment system: Integration of conventional factors with image phenotypes. Cardiovasc. Diagn. Ther. 2019, 9, 420–430. [Google Scholar] [CrossRef] [PubMed]
- Bristow, M.R.; Saxon, L.A.; Boehmer, J.; Krueger, S.; Kass, D.A.; De Marco, T.; Carson, P.; Dicarlo, L.; DeMets, D.; White, B.G.; et al. Cardiac-Resynchronization Therapy for the Prevention of Heart-Failure Events. N. Engl. J. Med. 2009, 3614, 1329–1338. [Google Scholar]
- Bristow, M.R.; Saxon, L.A.; Boehmer, J.; Krueger, S.; Kass, D.A.; De Marco, T.; Carson, P.; Dicarlo, L.; DeMets, D.; White, B.G.; et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N. Engl. J. Med. 2004, 3501, 2140–2150. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, B.L.; Leipsic, J.; Gaur, S.; Seneviratne, S.; Ko, B.S.; Ito, H.; Jensen, J.M.; Mauri, L.; De Bruyne, B.; Bezerra, H.; et al. Diagnostic Performance of Noninvasive Fractional Flow Reserve Derived from Coronary Computed Tomography Angiography in Suspected Coronary Artery Disease. J. Am. Coll. Cardiol 2014, 632, 1145–1155. [Google Scholar] [CrossRef]
- van Rosendael, A.R.; Maliakal, G.; Kolli, K.K.; Beecy, A.; Al’Aref, S.J.; Dwivedi, A.; Singh, G.; Panday, M.; Kumar, A.; Ma, X.; et al. Maximization of the usage of coronary CTA derived plaque information using a machine learning based algorithm to improve risk stratification; insights from the CONFIRM registry. J. Cardiovasc. Comput. Tomogr. 2018, 12, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Coenen, A.; Kim, Y.H.; Kruk, M.; Tesche, C.; De Geer, J.; Kurata, A.; Lubbers, M.L.; Daemen, J.; Itu, L.; Rapaka, S.; et al. Diagnostic Accuracy of a Machine-Learning Approach to Coronary Computed Tomographic Angiography–Based Fractional Flow Reserve. Circ. Cardiovasc. Imaging 2018, 11, e007217. [Google Scholar] [CrossRef] [PubMed]
- Min, J.K.; Leipsic, J.; Pencina, M.J.; Berman, D.S.; Koo, B.-K.; Van Mieghem, C.; Erglis, A.; Lin, F.Y.; Dunning, A.M.; Apruzzese, P.; et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA 2012, 3082, 1237–1245. [Google Scholar] [CrossRef]
- Lin, J.; Jiang, A.; Ling, M.; Mo, Y.; Li, M.; Zhao, J. Prediction of neurologic deterioration based on support vector machine algorithms and serum osmolarity equations. Brain Behav. 2018, 8, e01023. [Google Scholar] [CrossRef] [PubMed]
- Harrer, S.; Shah, P.; Antony, B.; Hu, J. Artificial Intelligence for Clinical Trial Design. Trends Pharmacol. Sci. 2019, 40, 577–591. [Google Scholar] [CrossRef]
- Pappalardo, F.; Russo, G.; Tshinanu, F.M.; Viceconti, M. In silico clinical trials: Concepts and early adoptions. Brief. Bioinform. 2018, 20, 1699–1708. [Google Scholar] [CrossRef]
- Abedi, V.; Lu, P.; Hontecillas, R.; Verma, M.; Vess, G.; Philipson, C.; Carbo, A.; Leber, A.; Juni, N.; Hoops, S.; et al. Phase III Placebo-Controlled, Randomized Clinical Trial with Synthetic Crohn’s Disease Patients to Evaluate Treatment Response. Emerg. Trends Appl. Infrastruct. Comput. Biol. Bioinform. Syst. Biol. 2015, 2, 169. [Google Scholar]
- Li, N.Y.K.; Verdolini, K.; Clermont, G.; Mi, Q.; Rubinstein, E.N.; Hebda, P.A.; Vodovotz, Y. A patient-specific in silico model of inflammation and healing tested in acute vocal fold injury. PLoS ONE 2008, 3, e2789. [Google Scholar] [CrossRef]
- Valerio, L.G. Application of advanced in silico methods for predictive modeling and information integration. Expert Opin. Drug Metab. Toxicol. 2012, 8, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Filipiak-Strzecka, D.; Kasprzak, J.D.; Szymczyk, E.; Wejner-Mik, P.; Lipiec, P. Bedside screening with the use of pocket-size imaging device can be useful for ruling out carotid artery stenosis in patients scheduled for cardiac surgery. Echocardiography 2017, 34, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Osheroff, J.A.; Teich, J.M.; Middleton, B.; Steen, E.B.; Wright, A.; Detmer, D.E. A Roadmap for National Action on Clinical Decision Support. J. Am. Med. Inform. Assoc. 2007, 14, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Varonen, H.; Kortteisto, T.; Kaila, M. What may help or hinder the implementation of computerized decision support systems (CDSSs): A focus group study with physicians. Fam. Pract. 2008, 25, 162–167. [Google Scholar] [CrossRef]
- Bouaud, J.; Spano, J.-P.; Lefranc, J.-P.; Cojean-Zelek, I.; Blaszka-Jaulerry, B.; Zelek, L.; Durieux, A.; Tournigand, C.; Rousseau, A.; Vandenbussche, P.-Y.; et al. Physicians’ Attitudes Towards the Advice of a Guideline-Based Decision Support System: A Case Study with OncoDoc2 in the Management of Breast Cancer Patients. Stud. Health Technol. Inform. 2015, 216, 264–269. [Google Scholar] [PubMed]
- Van Der Sijs, H.; Mulder, A.; van Gelder, T.; Aarts, J.; Berg, M.; Vulto, A. Drug safety alert generation and overriding in a large Dutch university medical centre. Pharmacoepidemiol. Drug Saf. 2009, 180, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Van Der Sijs, H.; Aarts, J.; Vulto, A.; Berg, M. Overriding of drug safety alerts in computerized physician order entry. J. Am. Med. Inform. Assoc. 2006, 13, 138–147. [Google Scholar] [CrossRef]
- Bergman, L.G.; Fors, U.G.H. Computer-aided DSM-IV-diagnostics—Acceptance, use and perceived usefulness in relation to users’ learning styles. BMC Med. Inform. Decis. Mak. 2005, 5, 1. [Google Scholar] [CrossRef]
- Curry, L.; Reed, M.H. Electronic decision support for diagnostic imaging in a primary care setting. J. Am. Med. Inform. Assoc. 2011, 18, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Padman, R.; Johnson, M.; Diamond, H.S. Understanding technology adoption in clinical care: Clinician adoption behavior of a point-of-care reminder system. Int. J. Med. Inform. 2005, 74, 535–543. [Google Scholar] [CrossRef]
- Rousseau, N.; McColl, E.; Newton, J.; Grimshaw, J.; Eccles, M. Practice based, longitudinal, qualitative interview study of computerised evidence based guidelines in primary care. Br. Med. J. 2003, 326, 314–318. [Google Scholar] [CrossRef][Green Version]
- Johnson, M.; Zheng, K.; Padman, R. Modeling the longitudinality of user acceptance of technology with an evidence-adaptive clinical decision support system. Decis. Support Syst. 2014, 57, 444–453. [Google Scholar] [CrossRef]
- Sousa, V.E.C.; Lopez, K.D.; Febretti, A.; Stifter, J.; Yao, Y.; Johnson, A.; Wilkie, D.J.; Keenan, G.M. Use of simulation to study nurses’ acceptance and nonacceptance of clinical decision support suggestions. CIN Comput. Inform. Nurs. 2015, 330, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Terraz, O.; Wietlisbach, V.; Jeannot, J.-G.; Burnand, B.; Froehlich, F.; Gonvers, J.-J.; Harris, J.K.; Vader, J.-P. The EPAGE internet guideline as a decision support tool for determining the appropriateness of colonoscopy. Digestion 2005, 71, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Gadd, C.S.; Baskaran, P.; Lobach, D.F. Identification of design features to enhance utilization and acceptance of systems for Internet-based decision support at the point of care. Proc. AMIA Symp. 1998, 91–95. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2232383/ (accessed on 15 November 2021).
- Khalifa, M. Clinical Decision Support: Strategies for Success. Procedia Comput. Sci. 2014, 37, 422–427. [Google Scholar] [CrossRef]
- McCoy, A.B.; Thomas, E.J.; Krousel-Wood, M.; Sittig, D.F. Clinical decision support alert appropriateness: A review and proposal for improvement. Ochsner J. 2014, 14, 195–202. [Google Scholar]
- Aakre, C.A.; Dziadzko, M.A.; Herasevich, V. Towards automated calculation of evidence-based clinical scores. World J. Methodol. 2017, 7, 16. [Google Scholar] [CrossRef]
- Khairat, S.; Marc, D.; Crosby, W.; al Sanousi, A. Reasons for Physicians Not Adopting Clinical Decision Support Systems: Critical Analysis. JMIR Med. Inform. 2018, 6, e24. [Google Scholar] [CrossRef] [PubMed]
- Kanstrup, A.M.; Christiansen, M.B.; Nøhr, C. Four principles for user interface design of computerised clinical decision support systems. Stud. Health Technol. Inform. 2011, 166, 65–73. [Google Scholar]
- Venkatesh, V.; Morris, M.G.; Davis, G.B.; Davis, F.D. User acceptance of information technology: Toward a unified view. MIS Q. Manag. Inf. Syst. 2003, 27, 425–478. [Google Scholar] [CrossRef]
- Peleg, M.; Shachak, A.; Wang, D.; Karnieli, E. Using multi-perspective methodologies to study users’ interactions with the prototype front end of a guideline-based decision support system for diabetic foot care. Int. J. Med. Inform. 2009, 78, 482–493. [Google Scholar] [CrossRef]
- Rudin, R.S.; Fischer, S.H.; Shi, Y.; Shekelle, P.; Amill-Rosario, A.; Damberg, C.L. Trends in the Use of Clinical Decision Support by Health System–Affiliated Ambulatory Clinics in the United States, 2014–2016. Am. J. Acc. Care 2019, 7, 4–10. [Google Scholar]
- Parikh, R.B.; Teeple, S.; Navathe, A.S. Addressing Bias in Artificial Intelligence in Health Care. JAMA 2019, 322, 2377–2378. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.; Probst, J.; Brock-Martin, A.; Bennett, K.; Glover, S.; Hardin, J. Association between clinical decision support system use and rural quality disparities in the treatment of pneumonia. J. Rural Health 2014, 30, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Samal, L.; Lipsitz, S.R.; Hicks, L.R.S. Impact of electronic health records on racial and ethnic disparities in blood pressure control at US primary care visits. Arch. Intern. Med. 2012, 172, 75–76. [Google Scholar] [CrossRef][Green Version]
- Sintchenko, V.; Coiera, E.; Iredell, J.R.; Gilbert, G.L. Comparative Impact of Guidelines, Clinical Data, and Decision Support on Prescribing Decisions: An Interactive Web Experiment with Simulated Cases. J. Am. Med. Inform. Assoc. 2004, 11, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Cui, Y. Deep transfer learning for reducing health care disparities arising from biomedical data inequality. Nat. Commun. 2020, 11, 5131. [Google Scholar] [CrossRef]
- Cirillo, D.; Catuara-Solarz, S.; Morey, C.; Guney, E.; Subirats, L.; Mellino, S.; Gigante, A.; Valencia, A.; Rementeria, M.J.; Chadha, A.S.; et al. Sex and gender differences and biases in artificial intelligence for biomedicine and healthcare. NPJ Digit. Med. 2020, 3, 81. [Google Scholar] [CrossRef] [PubMed]
- Abedi, V.; Li, J.; Shivakumar, M.K.; Avula, V.; Chaudhary, D.P.; Shellenberger, M.J.; Khara, H.S.; Zhang, Y.; Lee, M.T.M.; Wolk, D.M.; et al. Increasing the Density of Laboratory Measures for Machine Learning Applications. J. Clin. Med. 2020, 10, 103. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yan, X.S.; Chaudhary, D.; Avula, V.; Mudiganti, S.; Husby, H.; Shahjouei, S.; Afshar, A.; Stewart, W.F.; Yeasin, M.; et al. Imputation of missing values for electronic health record laboratory data. NPJ Digit. Med. 2021, 4, 147. [Google Scholar] [CrossRef] [PubMed]

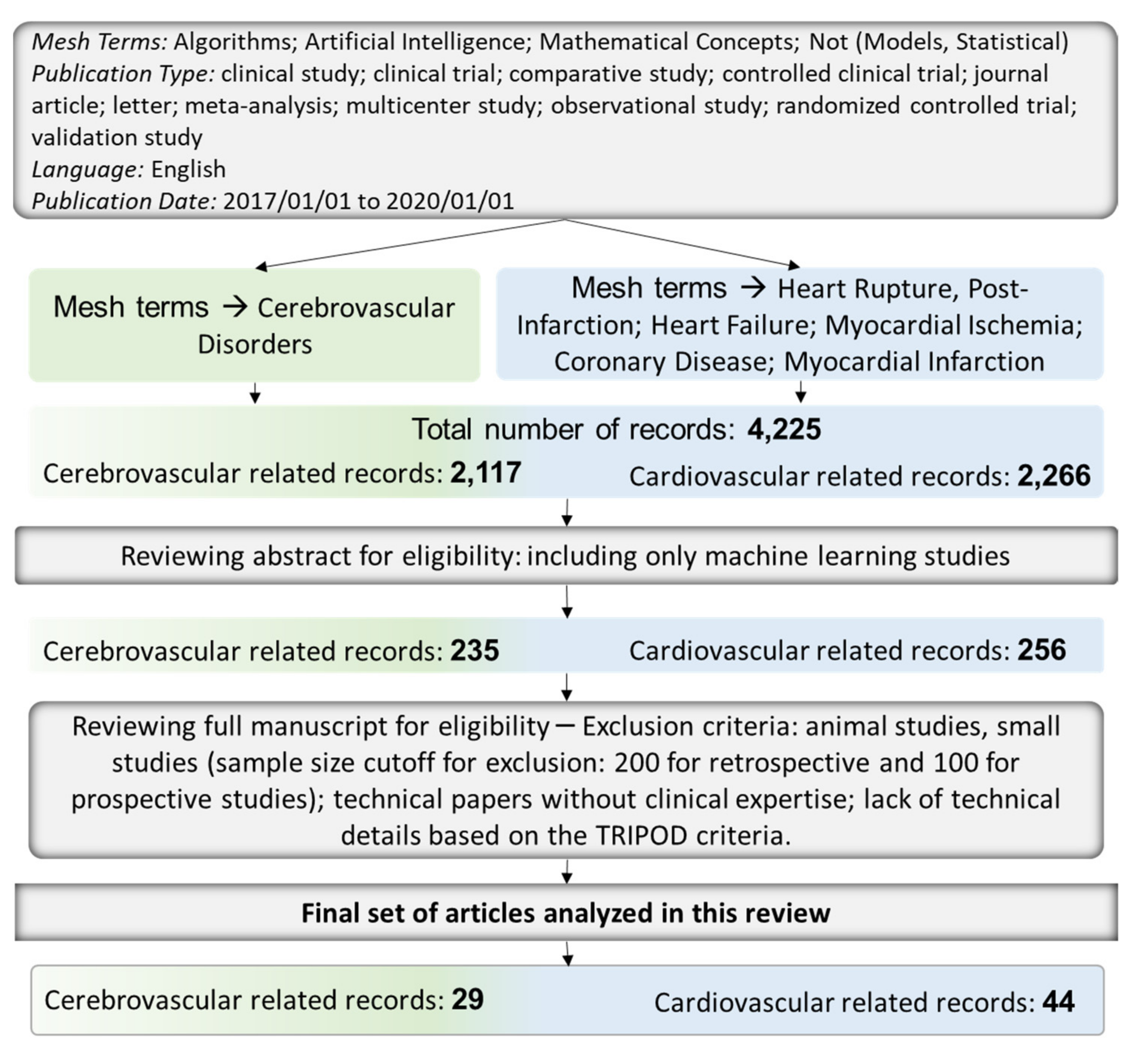
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abedi, V.; Razavi, S.-M.; Khan, A.; Avula, V.; Tompe, A.; Poursoroush, A.; Vafaei Sadr, A.; Li, J.; Zand, R. Artificial Intelligence: A Shifting Paradigm in Cardio-Cerebrovascular Medicine. J. Clin. Med. 2021, 10, 5710. https://doi.org/10.3390/jcm10235710
Abedi V, Razavi S-M, Khan A, Avula V, Tompe A, Poursoroush A, Vafaei Sadr A, Li J, Zand R. Artificial Intelligence: A Shifting Paradigm in Cardio-Cerebrovascular Medicine. Journal of Clinical Medicine. 2021; 10(23):5710. https://doi.org/10.3390/jcm10235710
Chicago/Turabian StyleAbedi, Vida, Seyed-Mostafa Razavi, Ayesha Khan, Venkatesh Avula, Aparna Tompe, Asma Poursoroush, Alireza Vafaei Sadr, Jiang Li, and Ramin Zand. 2021. "Artificial Intelligence: A Shifting Paradigm in Cardio-Cerebrovascular Medicine" Journal of Clinical Medicine 10, no. 23: 5710. https://doi.org/10.3390/jcm10235710
APA StyleAbedi, V., Razavi, S.-M., Khan, A., Avula, V., Tompe, A., Poursoroush, A., Vafaei Sadr, A., Li, J., & Zand, R. (2021). Artificial Intelligence: A Shifting Paradigm in Cardio-Cerebrovascular Medicine. Journal of Clinical Medicine, 10(23), 5710. https://doi.org/10.3390/jcm10235710









