A Prospective, Single Arm, Multi-Center Study Evaluating the Clinical Outcomes of Ventral Hernias Treated with OviTex® 1S Permanent Reinforced Tissue Matrix: The BRAVO Study 12-Month Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion/Exclusion Criteria
2.3. Surgery
2.4. Follow-Up
2.5. Primary Endpoint
2.6. Secondary Endpoints
2.7. Statistical Analysis
3. Results
3.1. Ninety-Day Outcomes
3.2. Twelve-Month Outcomes
3.3. Quality of Life Survey
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Faylona, J.M. Evolution of ventral hernia repair. Asian J. Endosc. Surg. 2017, 10, 252–258. [Google Scholar] [CrossRef]
- Holihan, J.L.; Hannon, C.; Goodenough, C.; Flores-Gonzalez, J.R.; Itani, K.M.; Olavarria, O.; Mo, J.; Ko, T.C.; Kao, L.S.; Liang, M.K. Ventral Hernia Repair: A Meta-Analysis of Randomized Controlled Trials. Surg. Infect. 2017, 18, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.W.; Luijendijk, R.W.; Hop, W.C.; Halm, J.A.; Verdaasdonk, E.G.; Jeekel, J. Long-term Follow-up of a Randomized Controlled Trial of Suture Versus Mesh Repair of Incisional Hernia. Ann. Surg. 2004, 240, 578–585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luijendijk, R.W.; Hop, W.C.; Tol, M.P.V.D.; De Lange, D.C.; Braaksma, M.M.; Ijzermans, J.N.; Boelhouwer, R.U.; De Vries, B.C.; Salu, M.K.; Wereldsma, J.C.; et al. A Comparison of Suture Repair with Mesh Repair for Incisional Hernia. N. Engl. J. Med. 2000, 343, 392–398. [Google Scholar] [CrossRef]
- Kokotovic, D.; Bisgaard, T.; Helgstrand, F. Long-term Recurrence and Complications Associated With Elective Incisional Hernia Repair. JAMA 2016, 316, 1575–1582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- 2019 AHS Annual Meeting. Hernia 2019, 23, 1–109. [CrossRef] [PubMed]
- Hawn, M.T.; Gray, S.; Snyder, C.W.; Graham, L.; Finan, K.R.; Vick, C.C. Predictors of mesh explantation after incisional hernia repair. Am. J. Surg. 2011, 202, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Klinge, B.K.U. Foreign Body Reaction to Meshes Used for the Repair of Abdominal Wall Hernias. Eur. J. Surg. 1999, 165, 665–673. [Google Scholar] [CrossRef]
- Novus Scientific. 2020. Available online: https://novusscientific.com/row/products/tigr-matrix/ (accessed on 9 June 2020).
- SutureOnline.com. Resorption Times Sutures. 2020. Available online: https://www.sutureonline.com/resorption-times-sutures (accessed on 9 June 2020).
- Overbeck, N.; Nagvajara, G.M.; Ferzoco, S.; May, B.C.H.; Beierschmitt, A.; Qi, S. In-vivo evaluation of a reinforced ovine biologic: A comparative study to available hernia mesh repair materials. Hernia 2020, 24, 1293–1306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosen, M.J.; Bauer, J.J.; Harmaty, M.; Carbonell, A.M.; Cobb, W.S.; Matthews, B.; Goldblatt, M.I.; Selzer, D.J.; Poulose, B.K.; Hansson, B.M.E.; et al. Multicenter, Prospective, Longitudinal Study of the Recurrence, Surgical Site Infection, and Quality of Life After Contaminated Ventral Hernia Repair Using Biosynthetic Absorbable Mesh. Ann. Surg. 2017, 265, 205–211. [Google Scholar] [CrossRef] [Green Version]
- Butler, C.E.; Prieto, V.G. Reduction of Adhesions with Composite AlloDerm/Polypropylene Mesh Implants for Abdominal Wall Reconstruction. Plast. Reconstr. Surg. 2004, 114, 464–473. [Google Scholar] [CrossRef]
- Kumar, A.S.; Fitzgerald, J.F. Biologic versus Synthetic Mesh Reinforcement: What are the Pros and Cons? Clin. Colon Rectal Surg. 2014, 27, 140–148. [Google Scholar] [CrossRef] [Green Version]
- Roth, J.S.; Anthone, G.J.; Selzer, D.J.; Poulose, B.K.; Bittner, J.G.; Hope, W.W.; Dunn, R.M.; Martindale, R.G.; Goldblatt, M.I.; Earle, D.B.; et al. Prospective evaluation of poly-4-hydroxybutyrate mesh in CDC class I/high-risk ventral and incisional hernia repair: 18-month follow-up. Surg. Endosc. 2018, 32, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Rastegarpour, A.; Cheung, M.; Vardhan, M.; Ibrahim, M.M.; Butler, C.E.; Levinson, H. Surgical mesh for ventral incisional hernia repairs: Understanding mesh design. Plast. Surg. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Chamieh, J.; Tan, W.H.; Ramirez, R.; Nohra, E.; Apakama, C.; Symons, W. Synthetic versus Biologic Mesh in Single-Stage Repair of Complex Abdominal Wall Defects in a Contaminated Field. Surg. Infect. 2017, 18, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Bellows, C.F.; Shadduck, P.; Helton, W.S.; Martindale, R.; Stouch, B.C.; Fitzgibbons, R. Early report of a randomized comparative clinical trial of Strattice™ reconstructive tissue matrix to lightweight synthetic mesh in the repair of inguinal hernias. Hernia 2014, 18, 221–230. [Google Scholar] [CrossRef]
- Garvey, P.B.; Giordano, S.; Baumann, D.P.; Liu, J.; Butler, C.E. Long-Term Outcomes after Abdominal Wall Reconstruction with Acellular Dermal Matrix. J. Am. Coll. Surg. 2017, 224, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jasbon, F.; Norrby, J.; Ivarsson, M.-L.; Björck, S. Inguinal hernia repair using a synthetic long-term resorbable mesh: Results from a 3-year prospective safety and performance study. Hernia 2014, 18, 723–730. [Google Scholar] [CrossRef] [Green Version]
- Itani, K.M.; Rosen, M.; Vargo, D.; Awad, S.S.; DeNoto, G.; Butler, C.E. Prospective study of single-stage repair of contaminated hernias using a biologic porcine tissue matrix: The RICH Study. Surgery 2012, 152, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Zerbib, P.; Caiazzo, R.; Piessen, G.; Rogosnitzky, M.; Séquier, C.; Koriche, D.; Truant, S.; Boleslawski, E.; Chambon, J.P.; Pruvot, F.R. Outcome in porcine acellular dermal matrix reinforcement of infected abdominal wall defects: A prospective study. Hernia 2015, 19, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Fafaj, A.; Petro, C.C.; Tastaldi, L.; Alkhatib, H.; AlMarzooqi, R.; Olson, M.A.; Stewart, T.G.; Prabhu, A.; Krpata, D.; Rosen, M.J. Intraperitonealversusretromuscular mesh placement for open incisional hernias: An analysis of the Americas Hernia Society Quality Collaborative. BJS 2020, 107, 1123–1129. [Google Scholar] [CrossRef]
- Roth, J.S.; Anthone, G.J.; Selzer, D.J.; Poulose, B.K.; Pierce, R.A.; Bittner, J.G.; Hope, W.W.; Dunn, R.M.; Martindale, R.G.; Goldblatt, M.I.; et al. Prospective, multicenter study of P4HB (Phasix™) mesh for hernia repair in cohort at risk for complications: 3-Year follow-up. Ann. Med. Surg. 2021, 61, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.J.; Barrio, M.; House, M.G.; Socas, J.; Reed, R.L.; Nakeeb, A.; Ceppa, E.P. Reinforced BioScaffold Mesh Lowers Recurrent Hernia Rate in High-Risk Ventral Hernia Repair With Surgical Site Occurrences. In Proceedings of the Americas Hernia Society (AHS) Annual Meeting, Las Vegas, NV, USA, 11–14 March 2019. [Google Scholar]
- Holihan, J.L.; Nguyen, D.H.; Nguyen, M.T.; Mo, J.; Kao, L.S.; Liang, M.K. Mesh Location in Open Ventral Hernia Repair: A Systematic Review and Network Meta-analysis. World J. Surg. 2016, 40, 89–99. [Google Scholar] [CrossRef]
- Dempsey, S.; Miller, C.H.; Hill, R.C.; Hansen, K.C.; May, B.C.H. Functional Insights from the Proteomic Inventory of Ovine Forestomach Matrix. J. Proteome Res. 2019, 18, 1657–1668. [Google Scholar] [CrossRef] [PubMed]
- Orenstein, S.B.; Saberski, E.R.; Kreutzer, D.L.; Novitsky, Y.W. Comparative Analysis of Histopathologic Effects of Synthetic Meshes Based on Material, Weight, and Pore Size in Mice. J. Surg. Res. 2012, 176, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Deeken, C.R.; Lake, S.P. Mechanical properties of the abdominal wall and biomaterials utilized for hernia repair. J. Mech. Behav. Biomed. Mater. 2017, 74, 411–427. [Google Scholar] [CrossRef] [PubMed]
- Vauclair, E.; Bert, M.; Facy, O.; Cheynel, N.; Rat, P.; Ortega-Deballon, P. What results can be expected one year after complex incisional hernia repair with biosynthetic mesh? J. Visc. Surg. 2021, 158, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Krpata, D.M.; Petro, C.C.; Prabhu, A.S.; Tastaldi, L.; Zolin, S.; Fafaj, A.; Rosenblatt, S.; Poulose, B.K.; Pierce, R.A.; Warren, J.A.; et al. Effect of Hernia Mesh Weights on Postoperative Patient-Related and Clinical Outcomes After Open Ventral Hernia Repair. JAMA Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Rognoni, C.; Cuccurullo, D.; Borsoi, L.; Bonavina, L.; Asti, E.; Crovella, F.; Bassi, U.A.; Carbone, G.; Guerini, F.; De Paolis, P.; et al. Clinical outcomes and quality of life associated with the use of a biosynthetic mesh for complex ventral hernia repair: Analysis of the “Italian Hernia Club” registry. Sci. Rep. 2020, 10, 10706. [Google Scholar] [CrossRef] [PubMed]
- Gillion, J.F.; Lepere, M.; Barrat, C.; Cas, O.; Dabrowski, A.; Jurczak, F.; Khalil, H.; Zaranis, C.; The French Club Hernie Study Group. Two-year patient-related outcome measures (PROM) of primary ventral and incisional hernia repair using a novel three-dimensional composite polyester monofilament mesh: The SymCHro registry study. Hernia 2019, 23, 767–781. [Google Scholar] [CrossRef] [Green Version]

| Preoperative Variables | 90 Days | 12 Months |
|---|---|---|
| Subjects Enrolled—92 | ||
| Subjects Included, n (%) | 84 (91%) | 76 (83%) |
| Sex, n (%) | ||
| Male | 34 (40%) | 31 (41%) |
| Female | 50 (60%) | 45 (59%) |
| Age (years), range | 62 (33.8–84.8) | 62 (33.8–84.8) |
| Body Mass Index (kg/m2), mean (range) | 31 (19.5–39.7) | 31 (21.6–39.5) |
| Comorbid Conditions | ||
| Obesity, n (%) | 46 (55%) | 44 (58%) |
| Patients with Prior VH Repairs, n (%) | 31 (37%) | 26 (34%) |
| Number of repairs, mean | 2 (0%) | 2 (0%) |
| Prior SSI, n (%) | 4 (5%) | 3 (4%) |
| History of Surgical Infection, n (%) | 14 (17%) | 13 (17%) |
| Perioperative Variables | ||
| CDC Wound Class, n (%) | ||
| CDC Class I Clean | 68 (81%) | 61 (80%) |
| CDC Class II Clean–Contaminated | 12 (14%) | 11 (14%) |
| CDC Class III Contaminated | 4 (5%) | 4 (5%) |
| VHWG, n (%) | ||
| Grade I | 21 (25%) | 16 (21%) |
| Grade II | 44 (52%) | 43 (57%) |
| Grade III | 19 (23%) | 17 (22%) |
| Operative Characteristics | 90 Days | 12 Months |
|---|---|---|
| Hernia Defect Size (cm2), mean (range) | 101 (1–384) | 101 (1–384) |
| Mesh Size at Implantation (cm2), mean (range) | 279 (30–400) | 277 (30–400) |
| Approach, n (%) | ||
| Open | 55 (65%) | 47 (62%) |
| Laparoscopic | 11 (13%) | 11 (14%) |
| Robotic | 18 (21%) | 18 (24%) |
| Plane of Placement, n (%) | ||
| Retrorectus | 35 (42%) | 29 (38%) |
| Intraperitoneal | 38 (45%) | 36 (47%) |
| TAR | 9 (11%) | 9 (12%) |
| Retrofascial/Pre-Peritoneal | 1 (1%) | 1 (1%) |
| Onlay | 1 (1%) | 1 (1%) |
| Primary Closure, n (%) | 78 (93%) | 70 (92%) |
| Component Separation, n (%) | 43 (51%) | 41 (54%) |
| Time in Surgery (Hours), mean (range) | 2.62 (0.70–9.68) | 2.62 (0.70–9.68) |
| Hospital Stay (Days), mean (range) | 4.46 (0–18) | 4.45 (0–18) |
| Adverse Events | 90 Days | 12 Months |
|---|---|---|
| Total Patients | 84 | 76 |
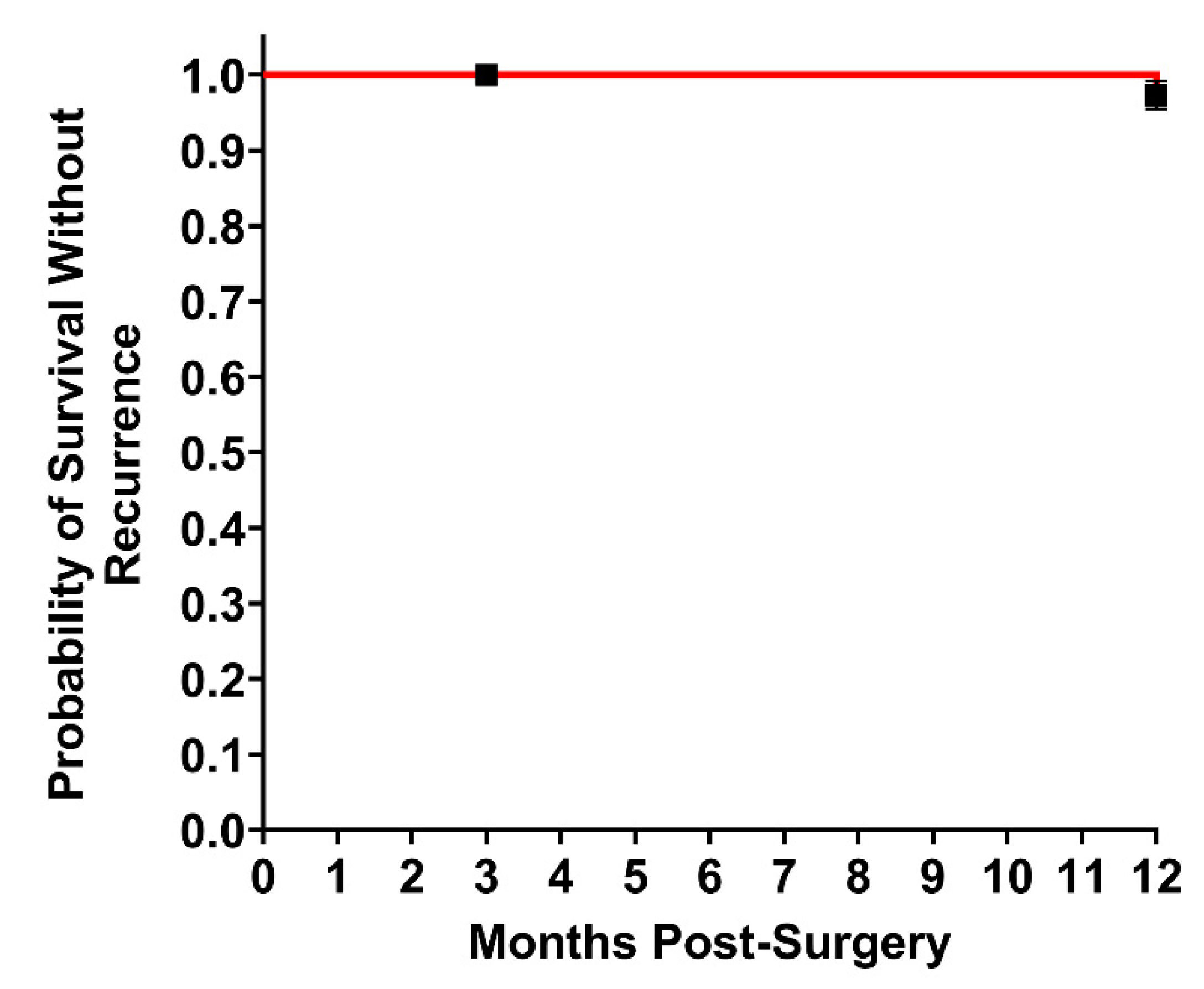
| Hernia Recurrence, n (%) | - | 2 (3%) |
| SSO (patients), n (%) | 21 (25%) | 20 (26%) |
| Seroma (requiring intervention) | 3 (4%) | 1 (1%) |
| Hematoma | 4 (5%) | 4 (5%) |
| Wound Dehiscence | 1 (1%) | 1 (1%) |
| Skin Necrosis | 1 (1%) | 1 (1%) |
| Fistulae | 2 (2%) | 2 (3%) |
| Superficial Infection | 8 (10%) | 7 (9%) |
| Deep/Abscess Infection | 4 (5%) | 6 (8%) |
| Organ Space Infection | - | - |
| SSO/SSI Requiring Procedural Intervention | 11 (13%) | 10 (13%) |
| Complications | ||
| Bowel Obstruction | 1 (1%) | 1 (1%) |
| DVT/PE | 3 (4%) | 3 (4%) |
| Ileus | 7 (8%) | 6 (8%) |
| Malignancy | 1 (1%) | 2 (3%) |
| Any other non-surgery or hernia-related complications | 15 (18%) | 15 (20%) |
| Full Mesh Removal | * | 1 (1%) |
| Risk Factor | Number of Patients |
|---|---|
| BMI between 30 and 40 | 44 (58%) |
| Active smokers | 5 (7%) |
| COPD | 3 (4%) |
| Diabetes mellitus | 18 (24%) |
| Coronary artery disease | 5 (7%) |
| Advanced age (>/=75 yrs) | 9 (12%) |
| Total High Risk (at least one) | 60 (79%) |
| Quality of Life Surveys | 90 Days | 12 Months |
|---|---|---|
| EQ-5D | ||
| Change (Baseline to Time point), mean | −0.26 | −0.91 |
| Interpretation | 2.3% improvement | 11% improvement |
| Patients, n | 76 | 69 |
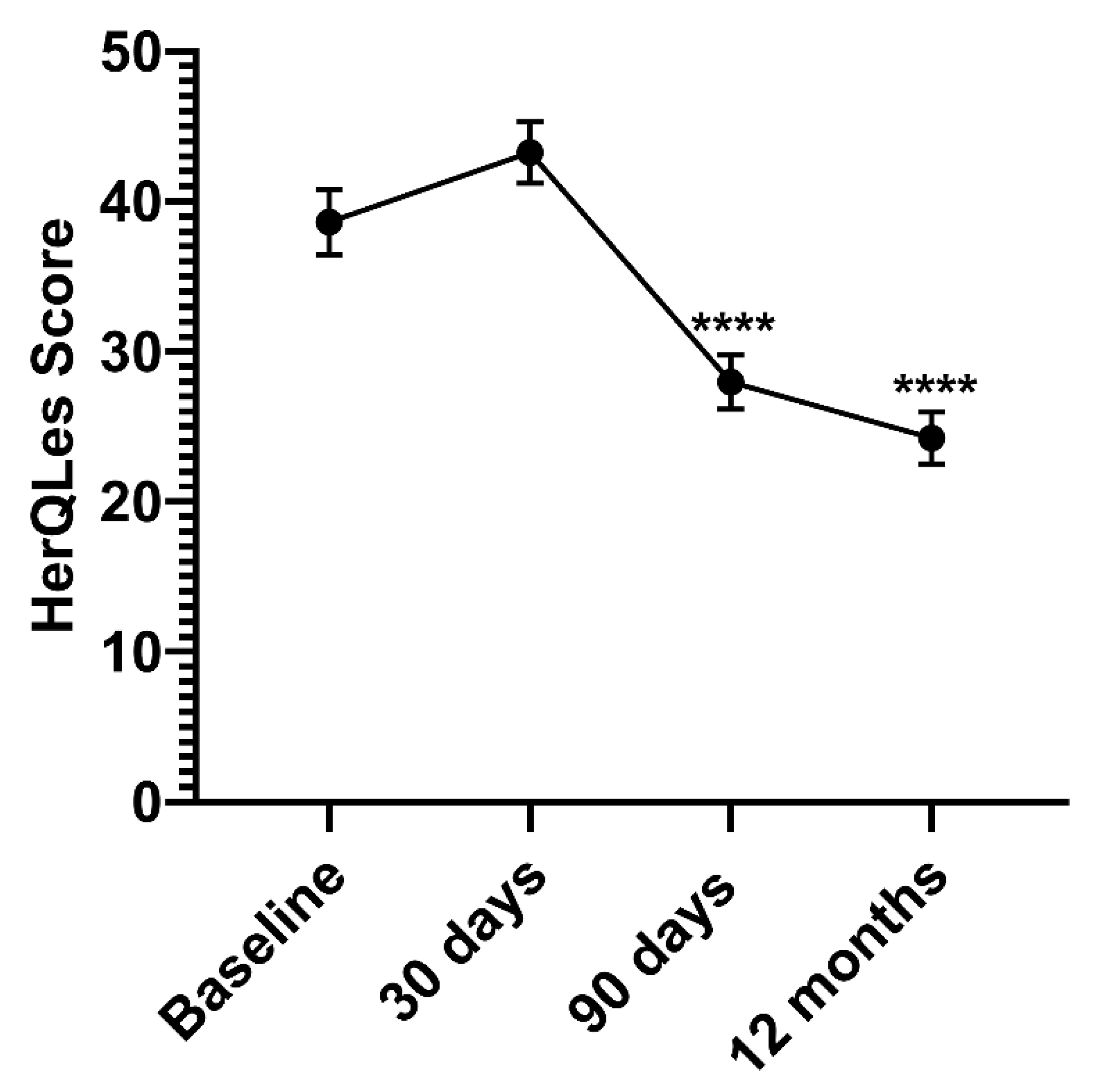
| HerQLes | ||
| Change (Baseline to Time point), mean | −10.63 | −15.46 |
| Interpretation | 25.9% improvement | 38.8% improvement |
| Patients, n | 75 | 69 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
DeNoto, G., III; Ceppa, E.P.; Pacella, S.J.; Sawyer, M.; Slayden, G.; Takata, M.; Tuma, G.; Yunis, J. A Prospective, Single Arm, Multi-Center Study Evaluating the Clinical Outcomes of Ventral Hernias Treated with OviTex® 1S Permanent Reinforced Tissue Matrix: The BRAVO Study 12-Month Analysis. J. Clin. Med. 2021, 10, 4998. https://doi.org/10.3390/jcm10214998
DeNoto G III, Ceppa EP, Pacella SJ, Sawyer M, Slayden G, Takata M, Tuma G, Yunis J. A Prospective, Single Arm, Multi-Center Study Evaluating the Clinical Outcomes of Ventral Hernias Treated with OviTex® 1S Permanent Reinforced Tissue Matrix: The BRAVO Study 12-Month Analysis. Journal of Clinical Medicine. 2021; 10(21):4998. https://doi.org/10.3390/jcm10214998
Chicago/Turabian StyleDeNoto, George, III, Eugene P. Ceppa, Salvatore J. Pacella, Michael Sawyer, Geoffrey Slayden, Mark Takata, Gary Tuma, and Jonathan Yunis. 2021. "A Prospective, Single Arm, Multi-Center Study Evaluating the Clinical Outcomes of Ventral Hernias Treated with OviTex® 1S Permanent Reinforced Tissue Matrix: The BRAVO Study 12-Month Analysis" Journal of Clinical Medicine 10, no. 21: 4998. https://doi.org/10.3390/jcm10214998
APA StyleDeNoto, G., III, Ceppa, E. P., Pacella, S. J., Sawyer, M., Slayden, G., Takata, M., Tuma, G., & Yunis, J. (2021). A Prospective, Single Arm, Multi-Center Study Evaluating the Clinical Outcomes of Ventral Hernias Treated with OviTex® 1S Permanent Reinforced Tissue Matrix: The BRAVO Study 12-Month Analysis. Journal of Clinical Medicine, 10(21), 4998. https://doi.org/10.3390/jcm10214998






