Improved Non-Invasive Diagnosis of Bladder Cancer with an Electronic Nose: A Large Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients’ Characteristics
2.2. Sample Management
2.3. Electronic Nose
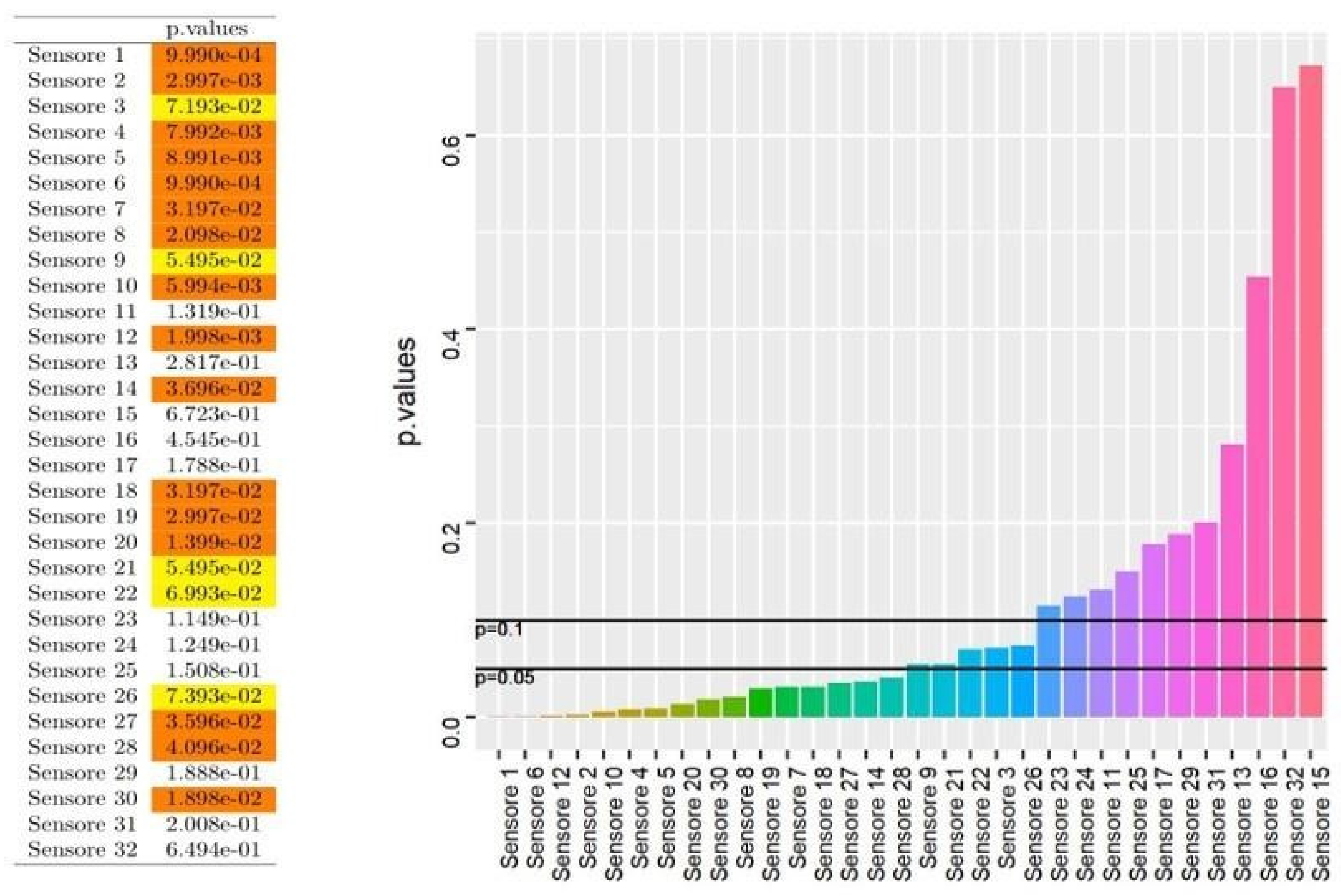
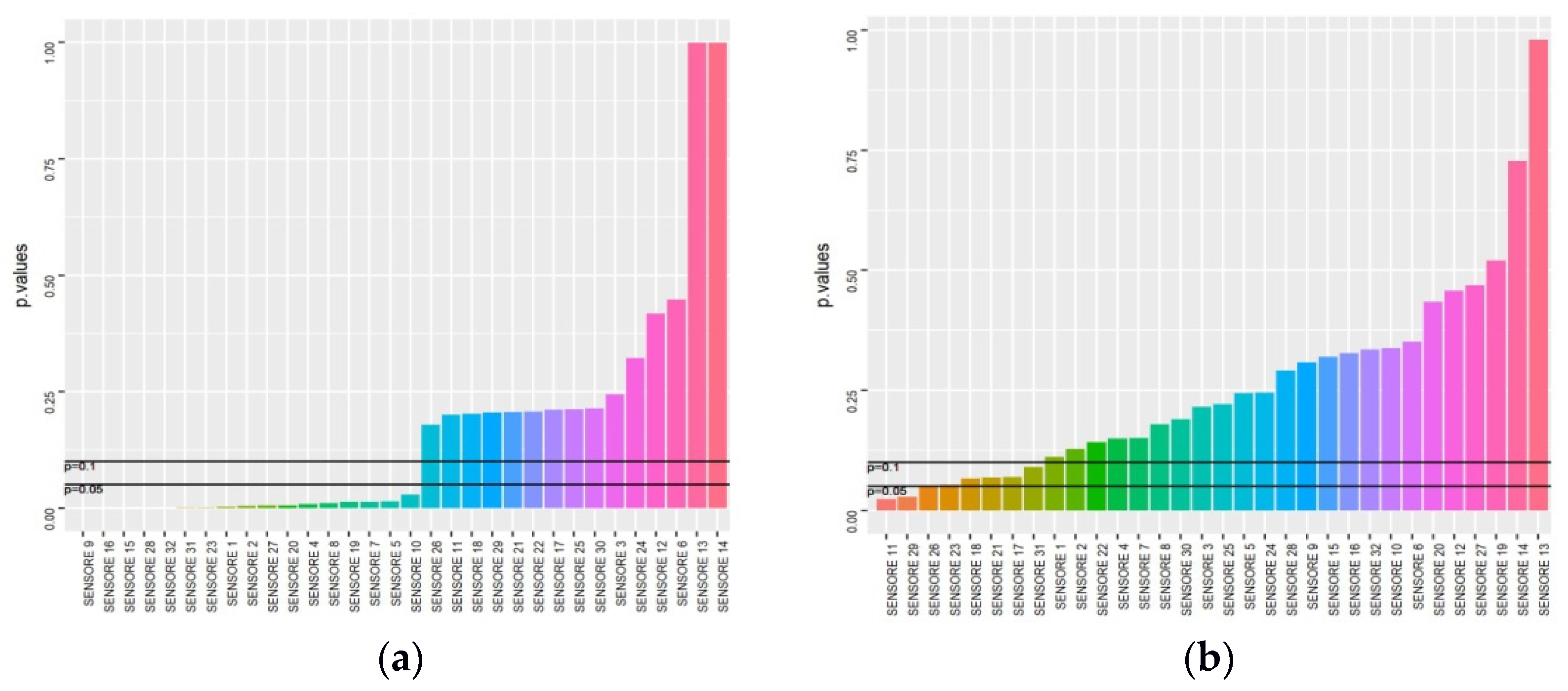
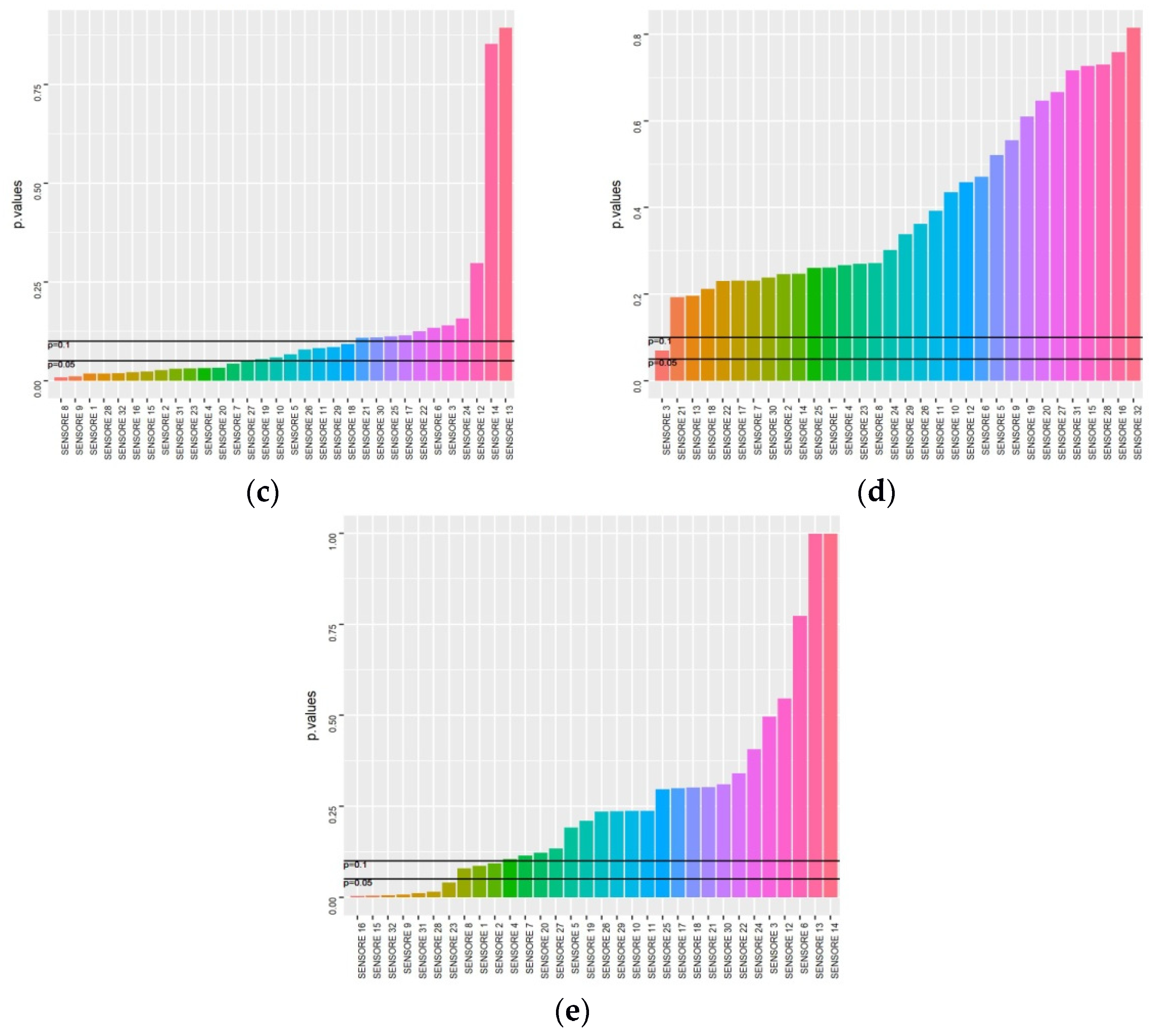
2.4. Results Management and Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Babjuk, M.; Burger, M.; Compérat, E.M.; Gontero, P.; Mostafid, A.H.; Palou, J.; van Rhijn, B.W.; Roupret, M.; Shariat, S.F.; Sylvester, R.; et al. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer (TaT1 and Carcinoma in Situ)—2019 Update. Eur. Urol. 2019, 76, 639–657. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, B.L.; Montgomery, J.S.; Zhang, Y.; Skolarus, T.A.; Weizer, A.Z.; Hollenbeck, B.K. Disparities in bladder cancer. Urol. Oncol. Semin. Orig. Investig. 2012, 30, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Gakis, G.; Kruck, S.; Stenzl, A. Can the burden of follow-up in low-grade noninvasive bladder cancer be reduced by photodynamic diagnosis, perioperative instillations, imaging, and urine markers? Curr. Opin. Urol. 2010, 20, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Lokeshwar, V.B.; Habuchi, T.; Grossman, H.B.; Murphy, W.M.; Hautmann, S.H.; Hemstreet, G.P.; Bono, A.V.; Getzenberg, R.H.; Goebell, P.; Schmitz-Dräger, B.J.; et al. Bladder tumor markers beyond cytology: International Consensus Panel on bladder tumor markers. Urology 2005, 66, 35–63. [Google Scholar] [CrossRef]
- Capuano, R.; Santonico, M.; Pennazza, G.; Ghezzi, S.; Martinelli, E.; Roscioni, C.; Lucantoni, G.; Galluccio, G.; Paolesse, R.; Di Natale, C.; et al. The lung cancer breath signature: A comparative analysis of exhaled breath and air sampled from inside the lungs. Sci. Rep. 2015, 5, 16491. [Google Scholar] [CrossRef] [Green Version]
- Phillips, M.; Herrera, J.; Krishnan, S.; Zain, M.; Greenberg, J.; Cataneo, R.N. Variation in volatile organic compounds in the breath of normal humans. J. Chromatogr. B Biomed. Sci. Appl. 1999, 729, 75–88. [Google Scholar] [CrossRef]
- Bassi, P.; De Marco, V.; De Lisa, A.; Mancini, M.; Pinto, F.; Bertoloni, R.; Longo, F. Non-Invasive Diagnostic Tests for Bladder Cancer: A Review of the Literature. Urol. Int. 2005, 75, 193–200. [Google Scholar] [CrossRef]
- Bajaj, A.; Miranda, O.R.; Kim, I.-B.; Phillips, R.L.; Jerry, D.J.; Bunz, U.H.F.; Rotello, V.M. Detection and differentiation of normal, cancerous, and metastatic cells using nanoparticle-polymer sensor arrays. Proc. Natl. Acad. Sci. USA 2009, 106, 10912–10916. [Google Scholar] [CrossRef] [Green Version]
- Willis, C.M.; Church, S.M.; Guest, C.M.; Cook, W.A.; McCarthy, N.; Bransbury, A.J.; Church, M.R.; Church, J.C. Olfactory detec-tion of human bladder cancer by dogs: Proof of principle study. BMJ 2004, 329, 712–714. [Google Scholar] [CrossRef] [Green Version]
- Willis, C.M.; Britton, L.E.; Harris, R.; Wallace, J.; Guest, C.M. Volatile organic compounds as biomarkers of bladder cancer: Sensitiv-ity and specificity using trained sniffer dogs. Cancer Biomark. 2011, 8, 145–153. [Google Scholar] [CrossRef]
- Spanel, P.; Smith, D.; Holland, T.A.; Al Singary, W.; Elder, J.B. Analysis of formaldehyde in the headspace of urine from bladder and prostate cancer patients using selected ion flow tube mass spectrometry. Rapid Commun. Mass Spectrom. 1999, 13, 1354–1359. [Google Scholar] [CrossRef]
- Weber, C.M.; Cauchi, M.; Patel, M.; Bessant, C.; Turner, C.; Britton, L.E.; Willis, C.M. Evaluation of a gas sensor array and pattern recognition for the identification of bladder cancer from urine headspace. Analyst 2011, 136, 359–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gardner, J.; Yinon, J. Electronic Noses and Sensors for the Detection of Explosives; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2004; ISBN 978-1-4020-2318-7. [Google Scholar]
- Teare, M.D.; Dimairo, M.; Shephard, N.; Hayman, A.; Whitehead, A.; Walters, S.J. Sample size requirements to estimate key design parameters from external pilot randomised controlled trials: A simulation study. Trials 2014, 15, 264. [Google Scholar] [CrossRef] [PubMed]
- Finos, L.; Salmaso, L. Weighted methods controlling the multiplicity when the number of variables is much higher than the number of observations. J. Nonparametr. Stat. 2006, 18, 245–261. [Google Scholar] [CrossRef]
- Pesarin, F.; Salmaso, L. Permutation Tests for Complex Data; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Baldini, C.; Billeci, L.; Sansone, F.; Conte, R.; Domenici, C.; Tonacci, A. Electronic Nose as a Novel Method for Diagnosing Cancer: A Systematic Review. Biosensors 2020, 10, 84. [Google Scholar] [CrossRef]
- Filipiak, W.; Sponring, A.; Filipiak, A.; Ager, C.; Schubert, J.; Miekisch, W.; Amann, A.; Troppmair, J. TD-GC-MS Analysis of Volatile Metabolites of Human Lung Cancer and Normal Cells In vitro. Cancer Epidemiol. Biomark. Prev. 2010, 19, 182–195. [Google Scholar] [CrossRef] [Green Version]
- Khalid, T.; White, P.; Costello, B.D.L.; Persad, R.; Ewen, R.; Johnson, E.; Probert, C.S.; Ratcliffe, N. A Pilot Study Combining a GC-Sensor Device with a Statistical Model for the Identification of Bladder Cancer from Urine Headspace. PLoS ONE 2013, 8, e69602. [Google Scholar] [CrossRef] [Green Version]
- Pasikanti, K.K.; Ho, P.; Chan, E. Gas chromatography/mass spectrometry in metabolic profiling of biological fluids. J. Chromatogr. B 2008, 871, 202–211. [Google Scholar] [CrossRef]
- Jobu, K.; Sun, C.; Yoshioka, S.; Yokota, J.; Onogawa, M.; Kawada, C.; Inoue, K.; Shuin, T.; Sendo, T.; Miyamura, M. Metabolomics Study on the Biochemical Profiles of Odor Elements in Urine of Human with Bladder Cancer. Biol. Pharm. Bull. 2012, 35, 639–642. [Google Scholar] [CrossRef] [Green Version]
- Gardner, J.; Bartlett, P.N. A brief history of electronic noses. Sens. Actuators B Chem. 1994, 18, 210–211. [Google Scholar] [CrossRef]
- Persaud, K.; Dodd, G. Analysis of discrimination mechanisms in the mammalian olfactory system using a model nose. Nature 1982, 299, 352–355. [Google Scholar] [CrossRef]
- Van Berkel, J.J.B.N.; Dallinga, J.W.; Möller, G.M.; Godschalk, R.W.L.; Moonen, E.J.; Wouters, E.F.M.; Van Schooten, F.J. A profile of volatile organic compounds in breath discriminates COPDpa-tients from controls. Respir. Med. 2010, 104, 557–563. [Google Scholar] [CrossRef] [Green Version]
- Bernabei, M.; Pennazza, G.; Santonico, M.; Corsi, C.; Roscioni, C.; Paolesse, R.; Di Natale, C.; D’Amico, A. A preliminary study on the possibility to diagnose urinary tract cancers by an electronic nose. Sens. Actuators B Chem. 2008, 131, 1–4. [Google Scholar] [CrossRef]
- Heers, H.; Gut, J.M.; Hegele, A.; Hofmann, R.; Boeselt, T.; Hattesohl, A.; Koczulla, A.R. Non-invasive Detection of Bladder Tumors Through Volatile Organic Compounds: A Pilot Study with an Electronic Nose. Anticancer Res. 2018, 38, 833–837. [Google Scholar] [CrossRef]
- Matsumoto, K.; Murakami, Y.; Shimizu, Y.; Hirayama, T.; Ishikawa, W.; Iwamura, M. Electronic nose to distinguish bladder cancer by urinary odour feature: A pilot study. Cancer Biomark. 2020, 28, 33–39. [Google Scholar] [CrossRef]
- Sciarra, A.; Di Lascio, G.; Del Giudice, F.; Leoncini, P.P.; Salciccia, S.; Gentilucci, A.; Porreca, A.; Chung, B.I.; Di Pierro, G.; Busetto, G.M.; et al. Comparison of the clinical usefulness of different urinary tests for the initial detection of bladder cancer: A systematic review. Curr. Urol. 2021, 15, 22–32. [Google Scholar] [CrossRef]
- Soria, F.; Droller, M.J.; Lotan, Y.; Gontero, P.; D’Andrea, D.; Gust, K.M.; Rouprêt, M.; Babjuk, M.; Palou, J.; Shariat, S. An up-to-date catalog of available urinary biomarkers for the surveillance of non-muscle invasive bladder cancer. World J. Urol. 2018, 36, 1981–1995. [Google Scholar] [CrossRef] [Green Version]
- Witjes, J.A.; Morote, J.; Cornel, E.B.; Gakis, G.; van Valenberg, F.J.P.; Lozano, F.; Sternberg, I.A.; Willemsen, E.; Hegemann, M.L.; Paitan, Y.; et al. Performance of the Bladder EpiCheck™ Methylation Test for Patients Under Surveillance for Non–muscle-invasive Bladder Cancer: Results of a Multicenter, Prospective, Blinded Clinical Trial. Eur. Urol. Oncol. 2018, 1, 307–313. [Google Scholar] [CrossRef]
- Białek, Ł.; Czerwińska, K.; Fus, Ł.; Krajewski, W.; Sadowska, A.; Radziszewski, P.; Dobruch, J.; Kryst, P.; Poletajew, S. MCM5 urine expression (ADXBLADDER) is a reliable biomarker of high-risk non-muscle-invasive bladder cancer recurrence: A prospective matched case-control study. Cancer Biomark. 2021, 30, 139–143. [Google Scholar] [CrossRef]

| Group 1 (BCa Patients) | Group 2 (Controls) | p-Value | |
|---|---|---|---|
| No. of patients | 102 | 96 | |
| Age, mean years ± SD (range) | 70.8 ± 12.4 (33–93) | 68.3 ± 10.7 (42–86) | 0.29 |
| Gender, no. of patients (%) | 0.56 | ||
| 87 (85.3%) | 79 (82.3%) | |
| 15 (14.7%) | 17 (17.7%) | |
| Smoking, no. of patients (%) | |||
| 10 (9.8%) | 16 (16.7%) | 0.15 |
| 45 (44.1%) | 31 (32.3%) | 0.16 |
| 47 (46.1%) | 49 (51%) | 0.49 |
| Comorbidities, no. of patients (%) | |||
| 56 (54.9%) | 41 (42.7%) | 0.08 |
| 22 (21.6%) | 16 (16.7%) | 0.38 |
| 21 (20.6%) | 14 (14.6%) | 0.27 |
| 9 (8.8%) | 4 (4.2%) | 0.18 |
| 16 (15.7%) | 9 (9.4%) | 0.18 |
| 4 (3.9%) | 1 (1%) | 0.20 |
| 21 (20.6%) | 12 (12.5%) | 0.13 |
| 11 (10.8%) | 6 (6.2%) | 0.25 |
| Total | NMIBC | MIBC | p-Value | |
|---|---|---|---|---|
| Histology, no. of patients (%) | 0.00 | |||
| 95 (93.1%) | 80 (98.8%) | 15 (71.4%) | |
| 7 (6.9%) | 1 (1.2%) | 6 (28.6%) | |
| Grade, no. of patients (%) | 0.00 | |||
| 31 (30.4%) | 31 (38.3%) | - | |
| 71 (69.6%) | 50 (61.7%) | 21 (100%) | |
| Stage, no. of patients (%) | ||||
| 36 (35.3%) | 36 (44.4%) | - | |
| 33 (32.3%) | 33 (40.7%) | - | |
| 9 (8.8%) | 9 (11.1%) | - | |
| 3 (2.9%) | 3 (3.7%) | - | |
| 16 (15.7%) | - | 16 (76.2%) | |
| 5 (4.9%) | - | 5 (23.8%) | |
| Focality, no. of patients (%) | 0.11 | |||
| 43 (42.2%) | 31 (38.3%) | 12 (57.1%) | |
| 59 (57.8%) | 50 (61.7%) | 9 (42.9%) | |
| Mean size, cm ± SD (range) | 2.7 ± 1.31 (1–6) | 2.4 ± 1.2 | 3.7 ± 1.3 | 0.00 |
| First diagnosis, no. of patients (%) | 43 (42.2%) | 33 (40.7%) | 10 (47.6%) | 0.12 |
| Median time from 1st diagnosis, year ± SD (range) | 1.8 ± 1.9 (0–5) | 1.9 ± 2.0 (0–5) | 1.2 ± 1.7 (0–5) | 0.43 |
| Accuracy, % | Sensitivity, % | Specificity, % | |
|---|---|---|---|
| GC-MS [8,15,16] | 70–100% | 70–100% | 42–97% |
| Sniffer dogs [5,6] | 70–90.1% | 55–86% | 56–92% |
| Electronic nose [19] | 86.7–93.3% | 93.3% | 86.7% |
| Our experience | 78.8% (71.6–87.5) | 91.1% (72.5–100) | 89.1% (81–95.8) |
| Test | Target of Measurement/Mechanism of Detection | Sensitivity, % (Range) | Specificity % (Range) |
|---|---|---|---|
| NMP22 BladderChek * [28,29] | Measurement of nuclear matrix proteins (quantitative ELISA) | (11–85.7%) | (77–100%) |
| NMP22 * [28,29] | Measurement of nuclear matrix proteins (qualitative point-of-care test) | (24–81%) | (49–100%) |
| BTA STAT * [28,29] | Measurement of human complement factor-H-related protein (point-of-care test) | (40–72%) | (29–96%) |
| BTA track * [28,29] | Measurement of human complement factor-H-related protein (quantitative ELISA) | (50–62%) | (68–87%) |
| Immunocyt * [28,29] | Fluorescent test combining 3 monoclonal antibodies (M344, LDQ10, 19A211) | (50–85%) | (62–86%) |
| UroVysion * [28,29] | Measurement of aneuploidy for chromosomes 3, 7, and 17 and loss of the 9p21 locus via fluorescence in situ hybridization (FISH) | (13–100%) | (63–100%) |
| Cxbladder monitor [28,29] | Measurement of 5 urine mRNA biomarkers and 2 clinical variables | (91–93%) | - |
| Bladder cancer (UBC) test [28,29] | Measurement of cytokeratins 8 and 18 | (12–80%) | (77.3–97%) |
| EpiCheck [28,29,30] | DNA methylation (15 biomarkers) changes | 86% (excluding Ta-LG) | 86% |
| ADXBLADDER [28,29,31] | Detection of MCM5 antibodies | 73.5% (62.7–82.6) | 33.3% (18.6–51) |
| Our experience | VOCs’ detection | 91.1% (72.5–100%) | 89.1% (81–95.8%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bassi, P.; Di Gianfrancesco, L.; Salmaso, L.; Ragonese, M.; Palermo, G.; Sacco, E.; Giancristofaro, R.A.; Ceccato, R.; Racioppi, M. Improved Non-Invasive Diagnosis of Bladder Cancer with an Electronic Nose: A Large Pilot Study. J. Clin. Med. 2021, 10, 4984. https://doi.org/10.3390/jcm10214984
Bassi P, Di Gianfrancesco L, Salmaso L, Ragonese M, Palermo G, Sacco E, Giancristofaro RA, Ceccato R, Racioppi M. Improved Non-Invasive Diagnosis of Bladder Cancer with an Electronic Nose: A Large Pilot Study. Journal of Clinical Medicine. 2021; 10(21):4984. https://doi.org/10.3390/jcm10214984
Chicago/Turabian StyleBassi, PierFrancesco, Luca Di Gianfrancesco, Luigi Salmaso, Mauro Ragonese, Giuseppe Palermo, Emilio Sacco, Rosa Arboretti Giancristofaro, Riccardo Ceccato, and Marco Racioppi. 2021. "Improved Non-Invasive Diagnosis of Bladder Cancer with an Electronic Nose: A Large Pilot Study" Journal of Clinical Medicine 10, no. 21: 4984. https://doi.org/10.3390/jcm10214984
APA StyleBassi, P., Di Gianfrancesco, L., Salmaso, L., Ragonese, M., Palermo, G., Sacco, E., Giancristofaro, R. A., Ceccato, R., & Racioppi, M. (2021). Improved Non-Invasive Diagnosis of Bladder Cancer with an Electronic Nose: A Large Pilot Study. Journal of Clinical Medicine, 10(21), 4984. https://doi.org/10.3390/jcm10214984