Age-Specific Normative Values of Lumbar Spine Trabecular Bone Score (TBS) in Taiwanese Men and Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Study Population
2.2. Demographic Data, Areal Bone Mineral Density Assessment and Trabecular Bone Score
2.3. Statistical Analysis
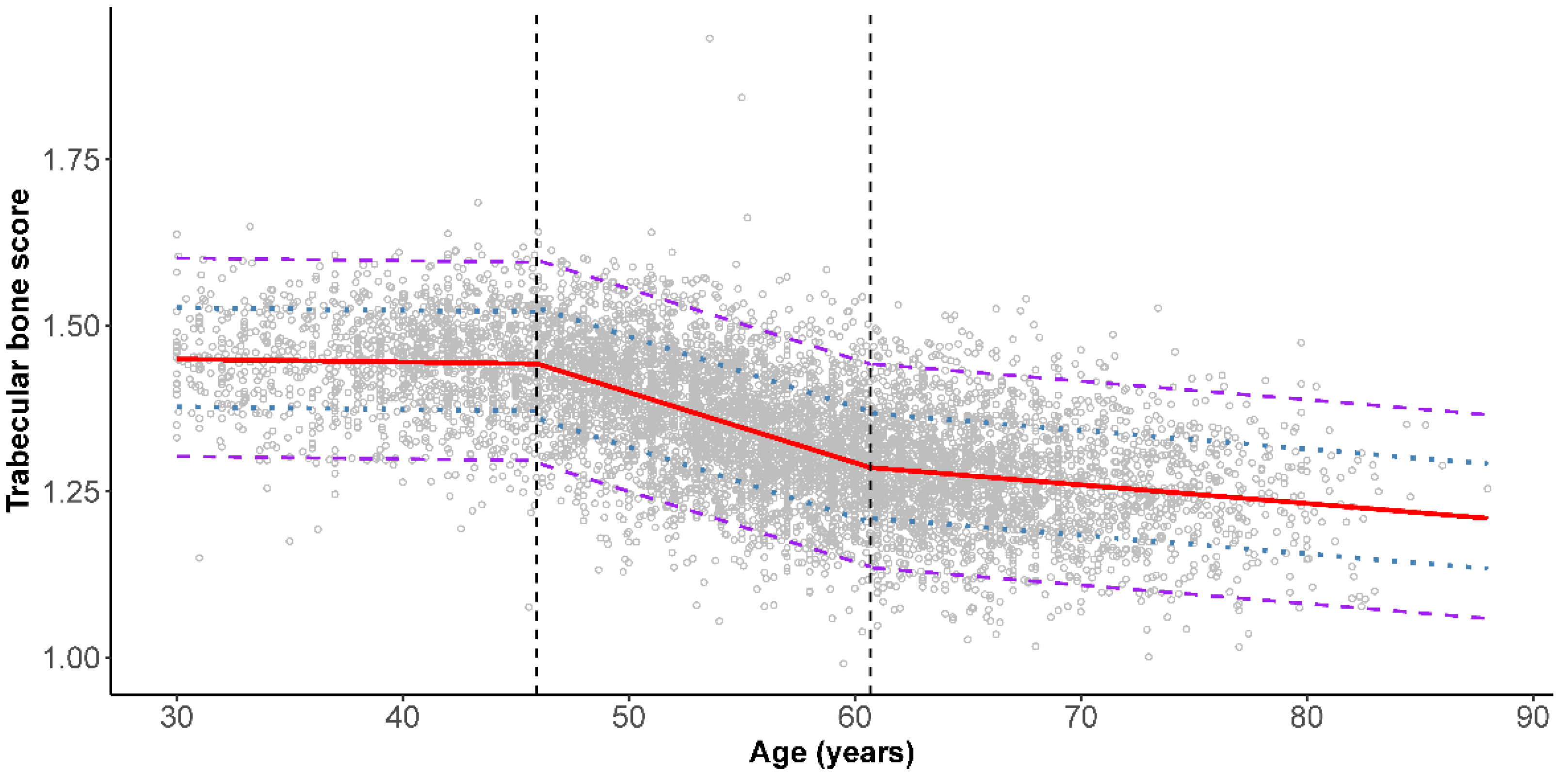
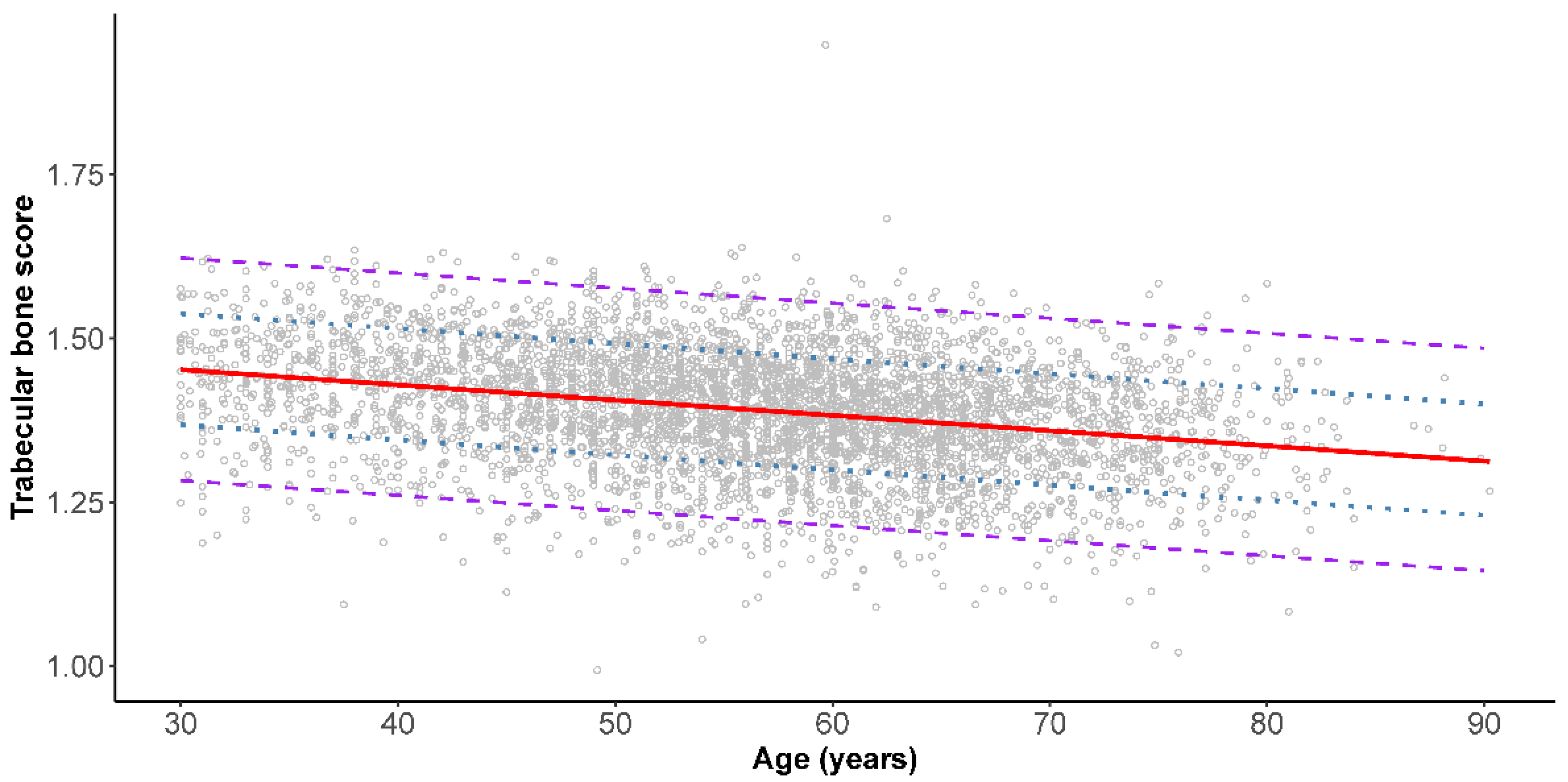
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clynes, M.A.; Harvey, N.C.; Curtis, E.M.; Fuggle, N.R.; Dennison, E.M.; Cooper, C. The epidemiology of osteoporosis. Br. Med. Bull. 2020, 133, 105–117. [Google Scholar] [CrossRef]
- Wade, S.W.; Strader, C.; Fitzpatrick, L.A.; Anthony, M.S.; O’Malley, C.D. Estimating prevalence of osteoporosis: Examples from industrialized countries. Arch. Osteoporos. 2014, 9, 182. [Google Scholar] [CrossRef]
- Delmas, P.D.; Seeman, E. Changes in bone mineral density explain little of the reduction in vertebral or nonvertebral fracture risk with anti-resorptive therapy. Bone 2004, 34, 599–604. [Google Scholar] [CrossRef]
- Hernandez, C.J.; van der Meulen, M.C. Understanding bone strength is not enough. J. Bone Miner. Res. 2017, 32, 1157–1162. [Google Scholar] [CrossRef] [Green Version]
- Pothuaud, L.; Carceller, P.; Hans, D. Correlations between grey-level variations in 2D projection images (TBS) and 3D microarchitecture: Applications in the study of human trabecular bone microarchitecture. Bone 2008, 42, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Hans, D.; Barthe, N.; Boutroy, S.; Pothuaud, L.; Winzenrieth, R.; Krieg, M.A. Correlations between trabecular bone score, measured using anteroposterior dual-energy X-ray absorptiometry acquisition, and 3-dimensional parameters of bone microarchitecture: An experimental study on human cadaver vertebrae. J. Clin. Densitom. 2011, 14, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Leung, J.; Hans, D.; Lamy, O.; Kwok, T. The added value of trabecular bone score to FRAX(R) to predict major osteoporotic fractures for clinical use in Chinese older people: The Mr. OS and Ms. OS cohort study in Hong Kong. Osteoporos. Int. 2017, 28, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Hans, D.; Goertzen, A.L.; Krieg, M.A.; Leslie, W.D. Bone microarchitecture assessed by TBS predicts osteoporotic fractures independent of bone density: The Manitoba study. J. Bone Miner. Res. 2011, 26, 2762–2769. [Google Scholar] [CrossRef]
- Boutroy, S.; Hans, D.; Sornay-Rendu, E.; Vilayphiou, N.; Winzenrieth, R.; Chapurlat, R. Trabecular bone score improves fracture risk prediction in non-osteoporotic women: The OFELY study. Osteoporos. Int. 2013, 24, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Iki, M.; Fujita, Y.; Tamaki, J.; Kouda, K.; Yura, A.; Sato, Y.; Moon, J.S.; Winzenrieth, R.; Okamoto, N.; Kurumatani, N. Trabecular bone score may improve FRAX(R) prediction accuracy for major osteoporotic fractures in elderly Japanese men: The Fujiwara-kyo Osteoporosis Risk in Men (FORMEN) Cohort Study. Osteoporos. Int. 2015, 26, 1841–1848. [Google Scholar] [CrossRef]
- Hans, D.; Stenova, E.; Lamy, O. The Trabecular Bone Score (TBS) Complements DXA and the FRAX as a fracture risk assessment tool in routine clinical practice. Curr. Osteoporos. Rep. 2017, 15, 521–531. [Google Scholar] [CrossRef]
- McCloskey, E.V.; Oden, A.; Harvey, N.C.; Leslie, W.D.; Hans, D.; Johansson, H.; Barkmann, R.; Boutroy, S.; Brown, J.; Chapurlat, R.; et al. A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J. Bone Miner. Res. 2016, 31, 940–948. [Google Scholar] [CrossRef]
- Lee, K.A.; Kim, J.; Kim, H.J.; Kim, H.S. Discriminative ability of trabecular bone score over bone mineral density for vertebral and fragility fracture in patients treated with long-term and low-dose glucocorticoid. Int. J. Rheum. Dis. 2021, 24, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.B.; Holloway-Kew, K.L.; Hans, D.; Kotowicz, M.A.; Hyde, N.K.; Pasco, J.A. Reference ranges for trabecular bone score in Australian men and women: A cross-sectional study. JBMR Plus 2019, 3, e10133. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Qi, H.M.; Di, W.J.; Liu, J.; Yu, J.; Lv, S.; Shen, Y.L.; Zha, J.M.; Cai, J.M.; Lai, B.; et al. Establishment of TBS reference plots and correlation between TBS and BMD in healthy mainland Chinese women. Arch. Osteoporos. 2016, 11, 5. [Google Scholar] [CrossRef]
- Dufour, R.; Winzenrieth, R.; Heraud, A.; Hans, D.; Mehsen, N. Generation and validation of a normative, age-specific reference curve for lumbar spine trabecular bone score (TBS) in French women. Osteoporos. Int. 2013, 24, 2837–2846. [Google Scholar] [CrossRef] [PubMed]
- Shafiee, G.; Sharifi, F.; Heshmat, R.; Ostovar, A.; Ebrahimpur, M.; Sheidaei, A.; Nabipour, I.; Larijani, B. The reference value of trabecular bone score (TBS) in the Iranian population. J. Diabetes Metab. Disord. 2020, 19, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Bazzocchi, A.; Ponti, F.; Diano, D.; Amadori, M.; Albisinni, U.; Battista, G.; Guglielmi, G. Trabecular bone score in healthy ageing. Br. J. Radiol. 2015, 88, 20140865. [Google Scholar] [CrossRef] [Green Version]
- Iki, M.; Tamaki, J.; Kadowaki, E.; Sato, Y.; Dongmei, N.; Winzenrieth, R.; Kagamimori, S.; Kagawa, Y.; Yoneshima, H. Trabecular bone score (TBS) predicts vertebral fractures in Japanese women over 10 years independently of bone density and prevalent vertebral deformity: The Japanese Population-Based Osteoporosis (JPOS) cohort study. J. Bone Miner. Res. 2014, 29, 399–407. [Google Scholar] [CrossRef]
- Guagnelli, M.A.; Winzenrieth, R.; Deleze, M.; Cons-Molina, F.; Clark, P. Description of normative spine TBS data for men and women in Mexican population. J. Clin. Densitom. 2021, 24, 129–134. [Google Scholar] [CrossRef]
- Simonelli, C.; Leib, E.; Mossman, N.; Winzenrieth, R.; Hans, D.; McClung, M. Creation of an age-adjusted, dual-energy x-ray absorptiometry-derived trabecular bone score curve for the lumbar spine in non-Hispanic US White women. J. Clin. Densitom. 2014, 17, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Rathnayake, H.; Lekamwasam, S.; Wickramatilake, C.; Lenora, J. Trabecular bone score and bone mineral density reference data for women aged 20-70 years and the effect of local reference data on the prevalence of postmenopausal osteoporosis: A cross-sectional study from Sri Lanka. Arch. Osteoporos. 2019, 14, 91. [Google Scholar] [CrossRef] [PubMed]
- Sritara, C.; Thakkinstian, A.; Ongphiphadhanakul, B.; Amnuaywattakorn, S.; Utamakul, C.; Akrawichien, T.; Vathesatogkit, P.; Sritara, P. Age-adjusted dual x-ray absorptiometry-derived trabecular bone score curve for the lumbar spine in Thai females and males. J. Clin. Densitom. 2016, 19, 494–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 14 October 2021).
- RStudio Team. Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2021; Available online: http://www.rstudio.com/ (accessed on 14 October 2021).
- Muggeo, V.M. Segmented: An R package to fit regression models with broken-line relationships. R News 2008, 8, 20–25. [Google Scholar]
- Iki, M.; Tamaki, J.; Sato, Y.; Winzenrieth, R.; Kagamimori, S.; Kagawa, Y.; Yoneshima, H. Age-related normative values of trabecular bone score (TBS) for Japanese women: The Japanese Population-based Osteoporosis (JPOS) study. Osteoporos. Int. 2019, 26, 245–252, Correction in 2019, 30, 253–255. [Google Scholar] [CrossRef]
- Jain, R.K.; Narang, D.K.; Hans, D.; Vokes, T.J. Ethnic differences in trabecular bone score. J. Clin. Densitom. 2017, 20, 172–179. [Google Scholar] [CrossRef]
- Castro, C.H.M.; Szejnfeld, V.L. Fracture risk differentially modulated by trabecular bone score according to ethnicity. J. Clin. Densitom. 2017, 20, 132–133. [Google Scholar] [CrossRef]
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Shoumura, S.; Emura, S.; Bunai, Y. Regional variations of vertebral trabecular bone microstructure with age and gender. Osteoporos. Int. 2008, 19, 1473–1483. [Google Scholar] [CrossRef]
- Shen, T.Y.; Strong, C.; Yu, T. Age at menopause and mortality in Taiwan: A cohort analysis. Maturitas 2020, 136, 42–48. [Google Scholar] [CrossRef]
- Mazzetti, G.; Berger, C.; Leslie, W.D.; Hans, D.; Langsetmo, L.; Hanley, D.A.; Kovacs, C.S.; Prior, J.C.; Kaiser, S.M.; Davison, K.S.; et al. Densitometer-specific differences in the correlation between body mass index and lumbar spine trabecular bone score. J. Clin. Densitom. 2017, 20, 233–238. [Google Scholar] [CrossRef] [PubMed]
| Age Group (Years) | Body Mass Index | |||||||
|---|---|---|---|---|---|---|---|---|
| Total | Male | Female | ||||||
| n | (%) | n | (%) | Mean (SD) | n | (%) | Mean (SD) | |
| 30–34 | 395 | (3.3) | 185 | (4.1) | 24.7 (4.4) | 210 | (2.8) | 22.2 (3.9) |
| 35–39 | 575 | (4.8) | 259 | (5.7) | 25.9 (3.9) | 316 | (4.2) | 22.6 (3.5) |
| 40–44 | 955 | (7.9) | 335 | (7.4) | 25.5 (3.6) | 620 | (8.3) | 23.3 (3.8) |
| 45–49 | 1455 | (12.1) | 502 | (11.1) | 25.3 (3.5) | 953 | (12.7) | 23.2 (3.2) |
| 50–54 | 2016 | (16.8) | 705 | (15.6) | 25.1 (3.0) | 1311 | (17.5) | 23.3 (3.2) |
| 55–59 | 2654 | (22.1) | 959 | (21.2) | 24.8 (2.9) | 1695 | (22.6) | 23.6 (3.2) |
| 60–64 | 1535 | (12.8) | 572 | (12.6) | 24.7 (2.9) | 963 | (12.8) | 23.8 (3.3) |
| 65–69 | 1336 | (11.1) | 521 | (11.5) | 24.4 (3.3) | 815 | (10.9) | 24.0 (3.5) |
| 70–74 | 705 | (5.9) | 303 | (6.7) | 24.2 (2.9) | 402 | (5.4) | 23.8 (3.0) |
| 75–79 | 316 | (2.6) | 146 | (3.2) | 23.9 (2.8) | 170 | (2.3) | 24.2 (3.2) |
| 80–90 | 86 | (0.7) | 46 | (1.0) | 23.8 (3.3) | 40 | (0.5) | 23.9 (3.7) |
| Total | 12,028 | (100) | 4533 | (37.7) | 24.9 (3.3) | 7495 | (62.3) | 23.5 (3.3) |
| Age Group (Years) | Lumbar Spine Areal BMD | Lumbar Spine T-Score | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Q1 | Median | Q3 | Mean (SD) | Q1 | Median | Q3 | ||
| Male | |||||||||
| 30–34 | 1.000 (0.129) | 0.923 | 1.000 | 1.080 | −0.232 (1.093) | −0.90 | −0.20 | 0.40 | |
| 35–39 | 1.022 (0.133) | 0.927 | 1.009 | 1.111 | −0.051 (1.132) | −0.90 | −0.10 | 0.70 | |
| 40–44 | 1.001 (0.131) | 0.917 | 0.991 | 1.086 | −0.229 (1.074) | −1.00 | −0.30 | 0.50 | |
| 45–49 | 0.986 (0.135) | 0.892 | 0.975 | 1.069 | −0.376 (1.154) | −1.20 | −0.40 | 0.40 | |
| 50–54 | 0.985 (0.136) | 0.883 | 0.976 | 1.075 | −0.403 (1.170) | −1.20 | −0.50 | 0.40 | |
| 55–59 | 0.976 (0.150) | 0.875 | 0.965 | 1.064 | −0.458 (1.254) | −1.30 | −0.60 | 0.30 | |
| 60–64 | 0.989 (0.162) | 0.883 | 0.975 | 1.088 | −0.357 (1.383) | −1.30 | −0.40 | 0.50 | |
| 65–69 | 0.988 (0.163) | 0.868 | 0.977 | 1.085 | −0.353 (1.374) | −1.30 | −0.40 | 0.50 | |
| 70–74 | 0.983 (0.175) | 0.854 | 0.977 | 1.088 | −0.413 (1.497) | −1.50 | −0.40 | 0.50 | |
| 75–79 | 0.993 (0.172) | 0.885 | 0.990 | 1.087 | −0.323 (1.462) | −1.20 | −0.30 | 0.50 | |
| 80–90 | 1.059 (0.224) | 0.926 | 1.011 | 1.230 | 0.220 (1.891) | −0.85 | −0.15 | 1.48 | |
| Female | |||||||||
| 30–34 | 0.995 (0.111) | 0.920 | 0.988 | 1.065 | −0.132 (0.978) | −0.80 | −0.20 | 0.50 | |
| 35–39 | 1.003 (0.117) | 0.924 | 1.003 | 1.086 | −0.056 (1.034) | −0.70 | −0.10 | 0.68 | |
| 40–44 | 1.008 (0.116) | 0.933 | 1.003 | 1.083 | −0.005 (1.018) | −0.70 | 0.00 | 0.60 | |
| 45–49 | 0.992 (0.133) | 0.904 | 0.988 | 1.080 | −0.158 (1.170) | −0.90 | −0.20 | 0.60 | |
| 50–54 | 0.920 (0.139) | 0.819 | 0.916 | 1.013 | −0.789 (1.219) | −1.70 | −0.80 | 0.00 | |
| 55–59 | 0.847 (0.128) | 0.754 | 0.837 | 0.925 | −1.420 (1.129) | −2.20 | −1.50 | −0.70 | |
| 60–64 | 0.816 (0.135) | 0.718 | 0.804 | 0.902 | −1.689 (1.187) | −2.60 | −1.80 | −1.00 | |
| 65–69 | 0.809 (0.138) | 0.717 | 0.799 | 0.892 | −1.737 (1.200) | −2.50 | −1.80 | −1.00 | |
| 70–74 | 0.790 (0.132) | 0.694 | 0.776 | 0.882 | −1.924 (1.164) | −2.80 | −2.00 | −1.20 | |
| 75–79 | 0.792 (0.131) | 0.697 | 0.785 | 0.868 | −1.899 (1.145) | −2.70 | −2.00 | −1.20 | |
| 80–90 | 0.758 (0.165) | 0.656 | 0.713 | 0.825 | −2.220 (1.449) | −3.08 | −2.60 | −1.63 | |
| Age Group (Years) | Trabecular Bone Score | |||||||
|---|---|---|---|---|---|---|---|---|
| Male | Female | |||||||
| Mean (SD) | Q1 | Median | Q3 | Mean (SD) | Q1 | Median | Q3 | |
| 30–34 | 1.438 (0.086) | 1.392 | 1.439 | 1.498 | 1.448 (0.072) | 1.401 | 1.451 | 1.495 |
| 35–39 | 1.440 (0.087) | 1.386 | 1.447 | 1.500 | 1.444 (0.075) | 1.398 | 1.449 | 1.497 |
| 40–44 | 1.427 (0.081) | 1.372 | 1.435 | 1.482 | 1.446 (0.073) | 1.399 | 1.449 | 1.496 |
| 45–49 | 1.412 (0.084) | 1.362 | 1.418 | 1.469 | 1.423 (0.082) | 1.374 | 1.429 | 1.478 |
| 50–54 | 1.400 (0.083) | 1.348 | 1.408 | 1.456 | 1.375 (0.087) | 1.317 | 1.375 | 1.436 |
| 55–59 | 1.388 (0.086) | 1.333 | 1.390 | 1.446 | 1.314 (0.084) | 1.260 | 1.313 | 1.368 |
| 60–64 | 1.375 (0.086) | 1.324 | 1.380 | 1.427 | 1.282 (0.081) | 1.232 | 1.282 | 1.331 |
| 65–69 | 1.365 (0.082) | 1.315 | 1.369 | 1.421 | 1.265 (0.082) | 1.211 | 1.264 | 1.314 |
| 70–74 | 1.347 (0.086) | 1.298 | 1.347 | 1.411 | 1.252 (0.083) | 1.197 | 1.248 | 1.307 |
| 75–79 | 1.349 (0.087) | 1.297 | 1.348 | 1.403 | 1.246 (0.082) | 1.189 | 1.248 | 1.305 |
| 80–90 | 1.344 (0.095) | 1.282 | 1.356 | 1.413 | 1.225 (0.084) | 1.154 | 1.237 | 1.287 |
| Total | 1.392 (0.089) | 1.335 | 1.396 | 1.453 | 1.344 (0.107) | 1.267 | 1.342 | 1.424 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuang, T.-L.; Chuang, M.-H.; Wang, Y.-F.; Koo, M. Age-Specific Normative Values of Lumbar Spine Trabecular Bone Score (TBS) in Taiwanese Men and Women. J. Clin. Med. 2021, 10, 4740. https://doi.org/10.3390/jcm10204740
Chuang T-L, Chuang M-H, Wang Y-F, Koo M. Age-Specific Normative Values of Lumbar Spine Trabecular Bone Score (TBS) in Taiwanese Men and Women. Journal of Clinical Medicine. 2021; 10(20):4740. https://doi.org/10.3390/jcm10204740
Chicago/Turabian StyleChuang, Tzyy-Ling, Mei-Hua Chuang, Yuh-Feng Wang, and Malcolm Koo. 2021. "Age-Specific Normative Values of Lumbar Spine Trabecular Bone Score (TBS) in Taiwanese Men and Women" Journal of Clinical Medicine 10, no. 20: 4740. https://doi.org/10.3390/jcm10204740
APA StyleChuang, T.-L., Chuang, M.-H., Wang, Y.-F., & Koo, M. (2021). Age-Specific Normative Values of Lumbar Spine Trabecular Bone Score (TBS) in Taiwanese Men and Women. Journal of Clinical Medicine, 10(20), 4740. https://doi.org/10.3390/jcm10204740







