First Census of Patients with Hereditary Angioedema in the Canary Islands
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Recruitment
2.2. Molecular Diagnosis
2.3. Statistical Analysis
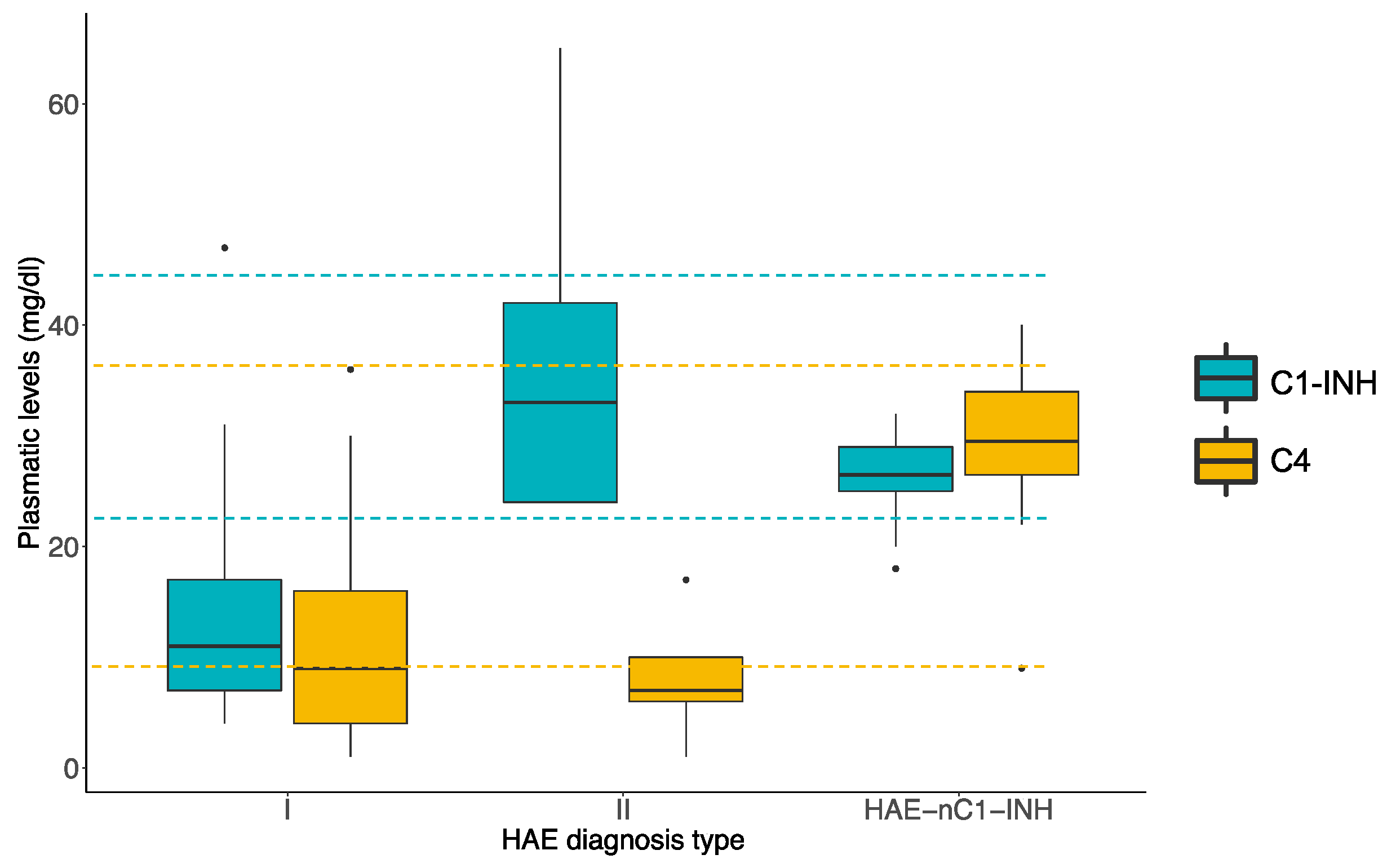
3. Results
3.1. Demographic and Clinical Patient Outcomes
3.2. Treatment Indication
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nzeako, U.C.; Frigas, E.; Tremaine, W.J. Hereditary angioedema: A broad review for clinicians. Arch. Intern. Med. 2001, 161, 2417–2429. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, A.; Aygören-Pürsün, E.; Binkley, K.E.; Blanch, A.; Bork, K.; Bouillet, L.; Bucher, C.; Castaldo, A.J.; Cicardi, M.; Davis, A.E.; et al. Hereditary and acquired angioedema: Problems and progress: Proceedings of the Third C1 Esterase Inhibitor Deficiency Workshop and Beyond. J. Allergy Clin. Immunol. 2004, 114, S51–S131. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.P.; Joseph, K.; Silverberg, M. Pathways for bradykinin formation and inflammatory disease. J. Allergy Clin. Immunol. 2002, 109, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Banday, A.Z.; Kaur, A.; Jindal, A.K.; Rawat, A.; Singh, S. An update on the genetics and pathogenesis of hereditary angioedema. Genes Dis. 2019, 7, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Caballero, T.; Baeza, M.L.; Cabañas, R.; Campos, A.; Cimbollek, S.; Gómez-Traseira, C.; González-Quevedo, T.; Guilarte, M.; Jurado-Palomo, J.; Larco, J.I.; et al. Consensus statement on the diagnosis, management, and treatment of angioedema mediated by bradykinin. Part I. treatment, follow-up and special situations. J. Investig. Allergol. Clin. Immunol. 2011, 21, 422–441. [Google Scholar]
- Zotter, Z.; Csuka, D.; Szabó, E.; Czaller, I.; Nébenführer, Z.; Temesszentandrási, G.; Fust, G.; Varga, L.; Farkas, H. The influence of trigger factors on hereditary angioedema due to c1-inhibitor deficiency. Orphanet J. Rare Dis. 2014, 9, 9–44. [Google Scholar] [CrossRef]
- Gompel, A.; Fain, O.; Boccon-Gibod, I.; Gobert, D.; Bouillet, L. Exogenous hormones and hereditary angioedema. Int. Immunopharmacol. 2019, 78, 106080. [Google Scholar] [CrossRef]
- De Campos, R.A.; Valle, S.O.R.; Toledo, E.C. Hereditary angioedema: A disease seldom diagnosed by pediatricians. J. Pediatr. 2020, 97 (Suppl. S1), S10–S16. [Google Scholar] [CrossRef]
- Uzun, T. Management of patients with hereditary angio-oedema in dental, oral and maxillofacial surgery: A Review. Br. J. Oral Maxillofac. Surg. 2019, 57, 992–997. [Google Scholar] [CrossRef]
- Zuraw, B.L. Clinical practice. hereditary angioedema. N. Engl. J. Med. 2008, 359, 1027–1036. [Google Scholar] [CrossRef]
- Marcelino-Rodriguez, I.; Callero, A.; Mendoza-Alvarez, A.; Pérez-Rodríguez, E.; Barrios-Recio, J.; García-Robaina, J.; Flores, C. Bradykinin-mediated angioedema: An update of the genetic causes and the impact of genomics. Front. Genet. 2019, 10, 900. [Google Scholar] [CrossRef] [PubMed]
- Caballero, T.; Farkas, H.; Bouillet, L.; Bowen, T.; Gompel, A.; Fagerberg, C.; Bjökander, J.; Bork, K.; Bygum, A.; Cicardi, M.; et al. International Consensus and practical guidelines on the gynecologic and obstetric management of female patients with hereditary angioedema caused by C1 inhibitor deficiency. J. Allergy Clin. Immunol. 2012, 129, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Roche, O.; Blanch, A.; Duponchel, C.; Fontán, G.; Tosi, M.; López-Trascasa, M. Hereditary Angioedema: The Mutation Spectrum of SERPING1/C1NH in a Large Spanish Cohort. Hum. Mutat. 2005, 26, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Dewald, G.; Bork, K. Missense Mutations in the Coagulation Factor XII (Hageman Factor) Gene in Hereditary Angioedema with Normal C1 Inhibitor. Biochem. Biophys. Res. Commun. 2006, 343, 1286–1289. [Google Scholar] [CrossRef]
- Zuraw, B.L. Hereditary Angioedema with Normal C1 Inhibitor: Four Types and Counting. J. Allergy Clin. Immunol. 2018, 141, 884–885. [Google Scholar] [CrossRef]
- Bafunno, V.; Firinu, D.; D’Apolito, M.; Cordisco, G.; Loffredo, S.; Leccese, A.; Bova, M.; Barca, M.P.; Santacroce, R.; Cicardi, M.; et al. Mutation of the Angiopoietin-1 gene (ANGPT1) Associates with a new type of hereditary angioedema. J. Allergy Clin. Immunol. 2018, 141, 1009–1017. [Google Scholar] [CrossRef]
- Bork, K.; Wulff, K.; Steinmüller-Magin, L.; Braenne, I.; Staubach-Renz, P.; Witzke, G.; Hardt, J. Hereditary angioedema with a mutation in the plasminogen gene. Allergy 2018, 73, 442–450. [Google Scholar] [CrossRef]
- Veronez, C.L.; Csuka, D.; Sheik, F.R.; Zuraw, B.L.; Farkas, H.; Bork, K. The expanding spectrum of mutations in hereditary angioedema. J. Allergy Clin. Immunol. Pract. 2021, 73, 442–450. [Google Scholar] [CrossRef]
- Bork, K.; Wulff, K.; Rossmann, H.; Steinmüller-Magin, L.; Braenne, I.; Witzke, G.; Hardt, J. Hereditary angioedema cosegregating with a novel kininogen1 gene mutation changing the n-terminal cleavage site of bradykinin. Allergy 2019, 74, 2479–2481. [Google Scholar] [CrossRef]
- Bork, K.; Hardt, J.; Witzke, G. Fatal laryngeal attacks and mortality in hereditary angioedema due to c1-inh deficiency. J. Allergy Clin. Immunol. 2012, 130, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.A. Severity of hereditary angioedema, prevalence and diagnostic considerations. Am. J. Manag. Care 2018, 24, S292–S298. [Google Scholar] [PubMed]
- Germenis, A.E.; Margaglione, M.; Pesquero, J.B.; Farkas, H.; Cichon, S.; Csuka, D.; Lera, A.L.; Rijavec, M.; Jolles, S.; Szilagyi, A.; et al. International consensus on the use of genetics in the management of hereditary angioedema. J. Allergy Clin. Immunol. Pr. 2020, 8, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Alvarez, A.; Muñoz-Barrera, A.; Rubio-Rodríguez, L.A.; Marcelino-Rodriguez, I.; Corrales, A.; Iñigo-Campos, A.; Callero, A.; Perez-Rodriguez, E.; Garcia-Robaina, J.C.; González-Montelongo, R.; et al. Interactive web-based resource for annotation of genetic variants causing hereditary angioedema (hada): Database development, implementation, and validation. J. Med. Internet Res. 2020, 22, e19040. [Google Scholar] [CrossRef] [PubMed]
- Patel, G.; Pongracic, J.A. Hereditary and acquired angioedema. Allergy Asthma Proc. 2019, 40, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Ponard, D.; Gaboriaud, C.; Charignon, D.; Ghannam, A.; Wagenaar-Bos, I.G.A.; Roem, D.; López-Lera, A.; López-Trascasa, M.; Tosi, M.; Drouet, C. SERPING1 mutation update: Mutation spectrum and C1 Inhibitor phenotypes. Hum. Mutat. 2019, 41, 38–57. [Google Scholar] [CrossRef]
- Henry Li, H.; Zuraw, B.; Longhurst, H.J.; Cicardi, M.; Bork, K.; Baker, J.; Lumry, W.; Bernstein, J.; Manning, M.; Levy, D.; et al. Subcutaneous C1 inhibitor for prevention of attacks of hereditary angioedema: Additional outcomes and subgroup analysis of a placebo-controlled randomized study. Allergy Asthma Clin. Immunol. 2019, 15, 1–8. [Google Scholar] [CrossRef]
- Bygum, A. Hereditary angio-oedema in Denmark: A nationwide survey. Br. J. Derm. 2009, 161, 1153–1158. [Google Scholar] [CrossRef]
- Faiyaz-Ul-Haque, M.; Al-Gazlan, S.; Abalkhail, H.A.; Al-Abdulatif, A.; Toulimat, M.; Peltekova, I.; Khaliq, A.M.R.; Al-Dayel, F.; Zaidi, S.H.E. Novel and recurrent mutations in the C1NH gene of arab patients affected with hereditary angioedema. Int. Arch. Allergy Immunol. 2010, 151, 149–154. [Google Scholar] [CrossRef]
- Kesim, B.; Uyguner, Z.O.; Gelincik, A.; Mete Gökmen, N.; Sin, A.Z.; Karakaya, G.; Erdenen, F.; Ardeniz, O.; Ozşeker, F.; Gülbahar, O.; et al. The turkish hereditary angioedema pilot study (TURHAPS): The first turkish series of hereditary angioedema. Int. Arch. Allergy Immunol. 2011, 156, 443–450. [Google Scholar] [CrossRef]
- Iwamoto, K.; Tanaka, A.; Hiragun, M.; Kawai, M.; Mihara, S.; Takenaka, M.; Shibuya, M.; Inomata, N.; Hatano, Y.; Shimizu, F.; et al. Novel and recurrent C1 inhibitor gene mutations in nine Japanese patients with hereditary angioedema. J. Derm. Sci. 2012, 68, 68–70. [Google Scholar] [CrossRef]
- Martinho, A.; Mendes, J.; Simões, O.; Nunes, R.; Gomes, J.; Dias Castro, E.; Leiria-Pinto, P.; Ferreira, M.B.; Pereira, C.; Castel-Branco, M.G.; et al. Mutations analysis of C1 inhibitor coding sequence gene among portuguese patients with hereditary angioedema. Mol. Immunol. 2013, 53, 431–434. [Google Scholar] [CrossRef]
- Psarros, F.; Koutsostathis, N.; Farmaki, E.; Speletas, M.G.; Germenis, A.E. Hereditary angioedema in Greece: The first results of the greek hereditary angioedema registry. Int. Arch. Allergy Immunol. 2014, 164, 326–332. [Google Scholar] [CrossRef]
- Johnsrud, I.; Kulseth, M.A.; Rødningen, O.K.; Landrø, L.; Helsing, P.; Waage Nielsen, E.; Heimdal, K. A nationwide study of norwegian patients with hereditary angioedema with C1 inhibitor deficiency identified six novel mutations in SERPING1. PLoS ONE 2015, 10, e0131637. [Google Scholar] [CrossRef]
- Steiner, U.C.; Keller, M.; Schmid, P.; Cichon, S.; Wuillemin, W.A. Mutational spectrum of the SERPING1 gene in swiss patients with hereditary angioedema. Clin. Exp. Immunol. 2017, 188, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Karadža-Lapić, L.; Barešić, M.; Vrsalović, R.; Ivković-Jureković, I.; Sršen, S.; Prkačin, I.; Rijavec, M.; Cikojević, D. Hereditary angioedema due to C1-Inhibitor deficiency in pediatric patients in Croatia. First national study, diagnostic and prophylactic challenges. Acta Clin. Croat. 2019, 58, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Xu, Y.; Liu, Y.; Zhi, Y. Hereditary angioedema: A Chinese perspective. Eur. J. Derm. 2019, 29, 14–20. [Google Scholar] [CrossRef]
- Gábos, G.; Moldovan, D.; Dobru, D.; Mihály, E.; Bara, N.; Nădășan, V.; Hutanu, A.; Csép, K. Mutational spectrum and genotype-phenotype relationships in a cohort of romanian hereditary angioedema patients caused by C1 inhibitor deficiency. Revista Română de Medicină de Laborator 2019, 27, 255–268. [Google Scholar] [CrossRef]
- Schöffl, C.; Wiednig, M.; Koch, L.; Blagojevic, D.; Duschet, P.; Hawranek, T.; Kinaciyan, T.; Öllinger, A.; Aberer, W. Hereditary angioedema in Austria: Prevalence and regional peculiarities. J. Dtsch. Dermatol. Ges. 2019, 17, 416–423. [Google Scholar] [CrossRef]
- Lindsay, K.; Chua, I.; Jordan, A.; Stephens, S. National audit of a hereditary and acquired angioedema cohort in New Zealand. Intern. Med. J. 2021. [Google Scholar] [CrossRef]
- Arce-Ayala, Y.M.; Diaz-Algorri, Y.; Craig, T.; Ramos-Romey, C. Clinical profile and quality of life of Puerto Ricans with hereditary angioedema. Allergy Asthma Proc. 2019, 40, 103–110. [Google Scholar] [CrossRef]
- Maurer, M.; Magerl, M.; Ansotegui, I.; Aygören-Pürsün, E.; Betschel, S.; Bork, K.; Bowen, T.; Boysen, H.B.; Farkas, H.; Grumach, A.S.; et al. The International WAO/EAACI guideline for the management of hereditary angioedema—The 2017 revision and update. World Allergy Organ. J. 2018, 11, 5. [Google Scholar] [CrossRef]
- Ferraro, M.F.; Moreno, A.S.; Castelli, E.C.; Donadi, E.A.; Palma, M.S.; Arcuri, H.A.; Lange, A.P.; Bork, K.; Sarti, W.; Arruda, L.K. A single nucleotide deletion at the C1 inhibitor gene as the cause of hereditary angioedema: Insights from a Brazilian family. Allergy 2011, 66, 1384–1390. [Google Scholar] [CrossRef]
- Betschel, S.; Badiou, J.; Binkley, K.; Borici-Mazi, R.; Hébert, J.; Kanani, A.; Keith, P.; Lacuesta, G.; Waserman, S.; Yang, B.; et al. The International/Canadian hereditary angioedema guideline. Allergy Asthma Clin. Immunol. 2019, 15, 72. [Google Scholar] [CrossRef]
- Wu, M.A.; Bova, M.; Berra, S.; Senter, R.; Parolin, D.; Caccia, S.; Cicardi, M. The central role of endothelium in hereditary angioedema due to C1 inhibitor deficiency. Int. Immunopharmacol. 2020, 82, 106304. [Google Scholar] [CrossRef]
- Bygum, A.; Aygören-Pürsün, E.; Caballero, T.; Beusterien, K.; Gholizadeh, S.; Musingarimi, P.; Wait, S.; Boysen, H. The hereditary angioedema burden of illness study in Europe (HAE-BOIS-Europe): Background and methodology. BMC Dermatol. 2012, 12, 4. [Google Scholar] [CrossRef]
- Guillen-Guio, B.; Lorenzo-Salazar, J.M.; González-Montelongo, R.; Díaz-de Usera, A.; Marcelino-Rodríguez, I.; Corrales, A.; Cabrera de León, A.; Alonso, S.; Flores, C. Genomic analyses of human European diversity at the southwestern edge: Isolation, african influence and disease associations in the Canary Islands. Mol. Biol. Evol. 2018, 35, 3010–3026. [Google Scholar] [CrossRef]
- Fragnan, N.T.M.L.; Tolentino, A.L.N.; Borba, G.B.; Oliveira, A.C.; Simões, J.A.; Palma, S.M.U.; Constantino-Silva, R.N.; Grumach, A.S. Hereditary angioedema with C1 Inhibitor (C1-INH) Deficit: The strength of recognition (51 Cases). Braz. J. Med. Biol. Res. 2018, 51, e7813. [Google Scholar] [CrossRef]
- Yip, J.; Cunliffe, W.J. Hormonally exacerbated hereditary angioedema. Australas. J. Dermatol. 1992, 33, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Bork, K.; Meng, G.; Staubach, P.; Hardt, J. Hereditary Angioedema: New findings concerning symptoms, affected organs and course. Am. J. Med. 2006, 119, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Longhurst, H.J.; Dempster, J.; Lorenzo, L.; Buckland, M.; Grigoriadou, S.; Symons, C.; Bethune, C.; Fabien, V.; Bangs, C.; Garcez, T. Real-world outcomes in hereditary angioedema: First experience from the icatibant outcome survey in the United Kingdom. Allergy Asthma Clin. Immunol. 2018, 14, 28. [Google Scholar] [CrossRef]
- Zanichelli, A.; Arcoleo, F.; Barca, M.P.; Borrelli, P.; Bova, M.; Cancian, M.; Cicardi, M.; Cillari, E.; De Carolis, C.; De Pasquale, T.; et al. A nationwide survey of hereditary angioedema due to C1 inhibitor deficiency in Italy. Orphanet J. Rare Dis. 2015, 10, 11. [Google Scholar] [CrossRef] [PubMed]
| Findings | |
|---|---|
| Recruited number | 41 |
| Age, years (mean ± SD) | 37.1 (±17.0) |
| Sex | |
| Male | 8 (20%) |
| Female | 33 (80%) |
| Clinical diagnosis | |
| HAE Type I | 22 (53.7%) |
| HAE Type II | 5 (12.2%) |
| HAE-nC1-INH | 14 (34.1%) |
| Positive family history | 13 (32.5%) |
| Age at first symptom (mean ± SD) | 23.9 (±13.9) |
| Age at diagnosis (mean ± SD) | 31.8 (±16.6) |
| Diagnostic delay (mean ± SD) | 8.1 (±12.6) |
| Pretreatment period | |
| Emergency room visits [median/IQR (25–75)] | 1.0/0.0–2.0 |
| Required hospitalization [median/IQR (25–75)] | 0.0/0.0–0.0 |
| Number of episodes by year [median/IQR (25–75)] | 5.0/1.5–5.0 |
| Airways [median/IQR (25–75)] | 0.0/0.0–1.0 |
| Abdominal [median/IQR (25–75)] | 0.0/0.0–4.7 |
| Cutaneous [median/IQR (25–75)] | 2.0/0.0–5.0 |
| Facials [median/IQR (25–75)] | 1.0/0.0–4.0 |
| Post-treatment period | |
| Emergency room visits [median/IQR (25–75)] | 0.0/0.0–0.5 |
| Required hospitalization [median/IQR (25–75)] | 0.0/0.0–0.0 |
| Number of episodes by year [median/IQR (25–75)] | 2.0/0.0–5.0 |
| Airways [median/IQR (25–75)] | 0.0/0.0–0.0 |
| Abdominal [median/IQR (25–75)] | 0.0/0.0–1.0 |
| Cutaneous [median/IQR (25–75)] | 0.0/0.0–3.7 |
| Facials [median/IQR (25–75)] | 0.0/0.0–1.0 |
| Score (n = 39) | |
| Asymptomatic (%) | 5 (12.5%) |
| Mild (%) | 20 (50.0%) |
| Moderate (%) | 10 (25.0%) |
| Severe (%) | 5 (12.5%) |
| n | |
|---|---|
| No treatment | 20 (46.5%) |
| Attenuated androgens | |
| Danazol | 5 (11.6%) |
| Estanozolol | 11 (25.6%) |
| Antifibrinolytic (tranexamic acid) | 7 (16.3%) |
| Rescue treatment (icatibant acetate) | 14 (32.6%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza-Alvarez, A.; Marcelino-Rodriguez, I.; Almeida-Quintana, L.; Martin-Fernandez, E.; Martinez-Beltran, D.; Almeida-Sanchez, Z.; Cruz-Niesvara, D.; Hernández-Santana, G.; Garcia-Robaina, J.C.; Flores, C.; et al. First Census of Patients with Hereditary Angioedema in the Canary Islands. J. Clin. Med. 2021, 10, 4711. https://doi.org/10.3390/jcm10204711
Mendoza-Alvarez A, Marcelino-Rodriguez I, Almeida-Quintana L, Martin-Fernandez E, Martinez-Beltran D, Almeida-Sanchez Z, Cruz-Niesvara D, Hernández-Santana G, Garcia-Robaina JC, Flores C, et al. First Census of Patients with Hereditary Angioedema in the Canary Islands. Journal of Clinical Medicine. 2021; 10(20):4711. https://doi.org/10.3390/jcm10204711
Chicago/Turabian StyleMendoza-Alvarez, Alejandro, Itahisa Marcelino-Rodriguez, Lourdes Almeida-Quintana, Elena Martin-Fernandez, Dara Martinez-Beltran, Zulay Almeida-Sanchez, David Cruz-Niesvara, Guacimara Hernández-Santana, Jose C. Garcia-Robaina, Carlos Flores, and et al. 2021. "First Census of Patients with Hereditary Angioedema in the Canary Islands" Journal of Clinical Medicine 10, no. 20: 4711. https://doi.org/10.3390/jcm10204711
APA StyleMendoza-Alvarez, A., Marcelino-Rodriguez, I., Almeida-Quintana, L., Martin-Fernandez, E., Martinez-Beltran, D., Almeida-Sanchez, Z., Cruz-Niesvara, D., Hernández-Santana, G., Garcia-Robaina, J. C., Flores, C., & Callero, A. (2021). First Census of Patients with Hereditary Angioedema in the Canary Islands. Journal of Clinical Medicine, 10(20), 4711. https://doi.org/10.3390/jcm10204711









