Coronary Artery Disease in Patients Older than 35 and Eligible for Cardiovascular Secondary Prevention: An Italian Retrospective Observational Analysis of Healthcare Administrative Databases
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Source
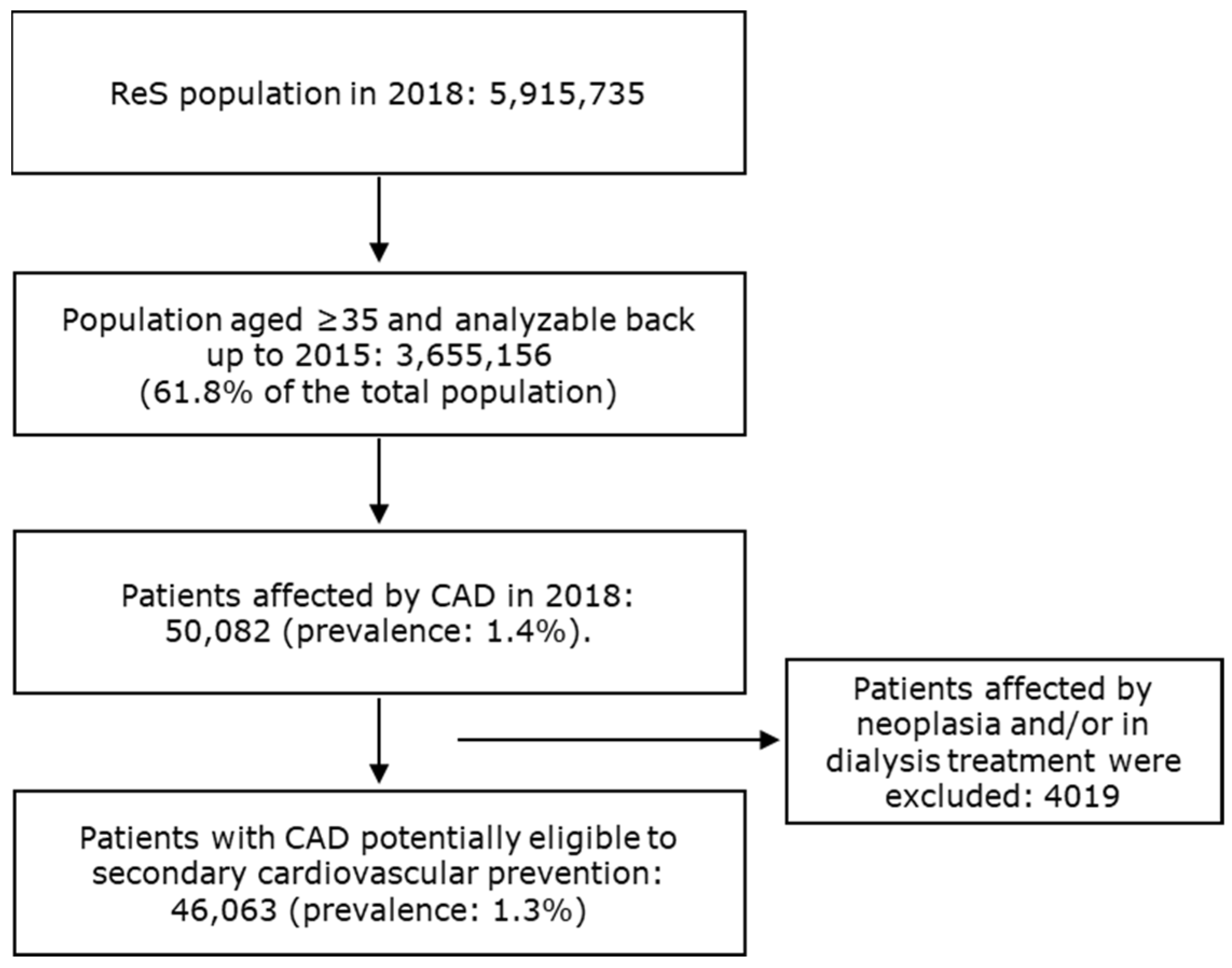
2.2. Cohort Selection
2.3. Analyses
2.3.1. Demographics and Clinical Characteristics
2.3.2. Pharmacological Treatments
- −
- Therapeutic strategies specific to secondary prevention: at least a free, filled prescription of ACE-is (plain and their combinations (ATC codes: C09A/C09B), ARBs (plain and their combinations (C09C/C09D), β-blockers (C07), lipid lowering agents (C10), and antiplatelet agents excluding heparin (B01AC).
- −
- Other CV drugs: at least a dispensation of pharmacological treatments belonging to ATC codes C and B01, different from those previously mentioned.
- −
- Non-CV drugs: at least a free, filled prescription of the drugs identified by ATC codes, different from those quoted in the above bullets.
2.3.3. Hospitalizations
- −
- Relevant CV diseases: acute coronary syndrome (ACS), angina pectoris, heart failure, haemorrhagic stroke/intracranial bleeding, ischemic stroke/transient ischemic attack (TIA).
- −
- Other CV diseases.
- −
- Non-CV diseases.
2.3.4. Healthcare Integrated Costs
- −
- Pharmaceutical costs extrapolated by the gross expenditure of local pharmacies’ sales and by the real hospital price (inclusive of value-added tax) of hospital pharmacies’ supplies.
- −
- In-hospital expenses derived from DRG (Diagnosis-Related Group) tariffs. Each hospital discharge form was linked to a DRG code which synthetizes the entire healthcare provided at admission and throughout the in-hospital stay.
- −
- Outpatient specialist care costs derived from the current National system tariffs.
2.3.5. Statistical Analyses
3. Results
3.1. Demographics and Clinical Characteristics
3.2. Pharmacological Treatments
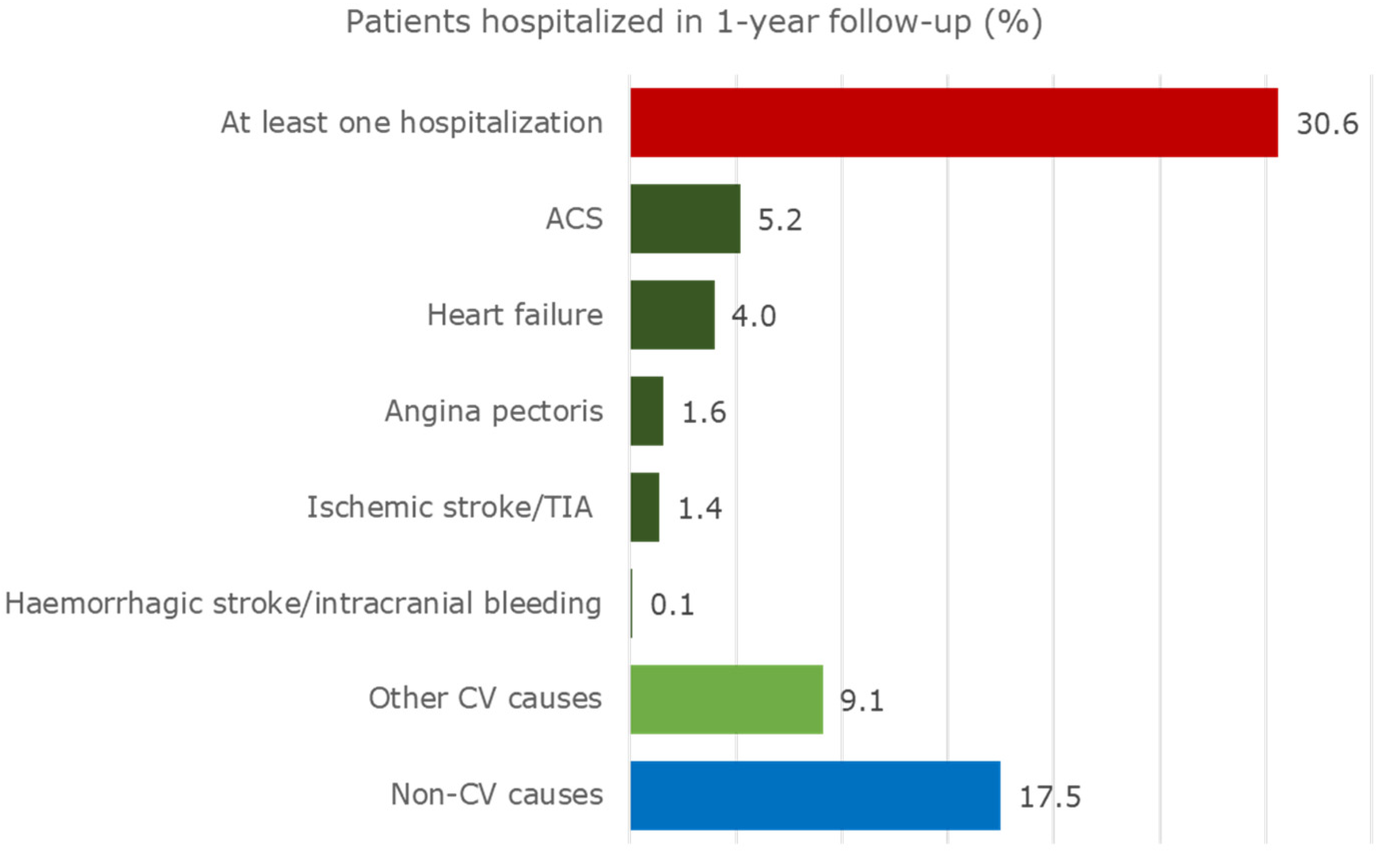
3.3. Hospitalizations
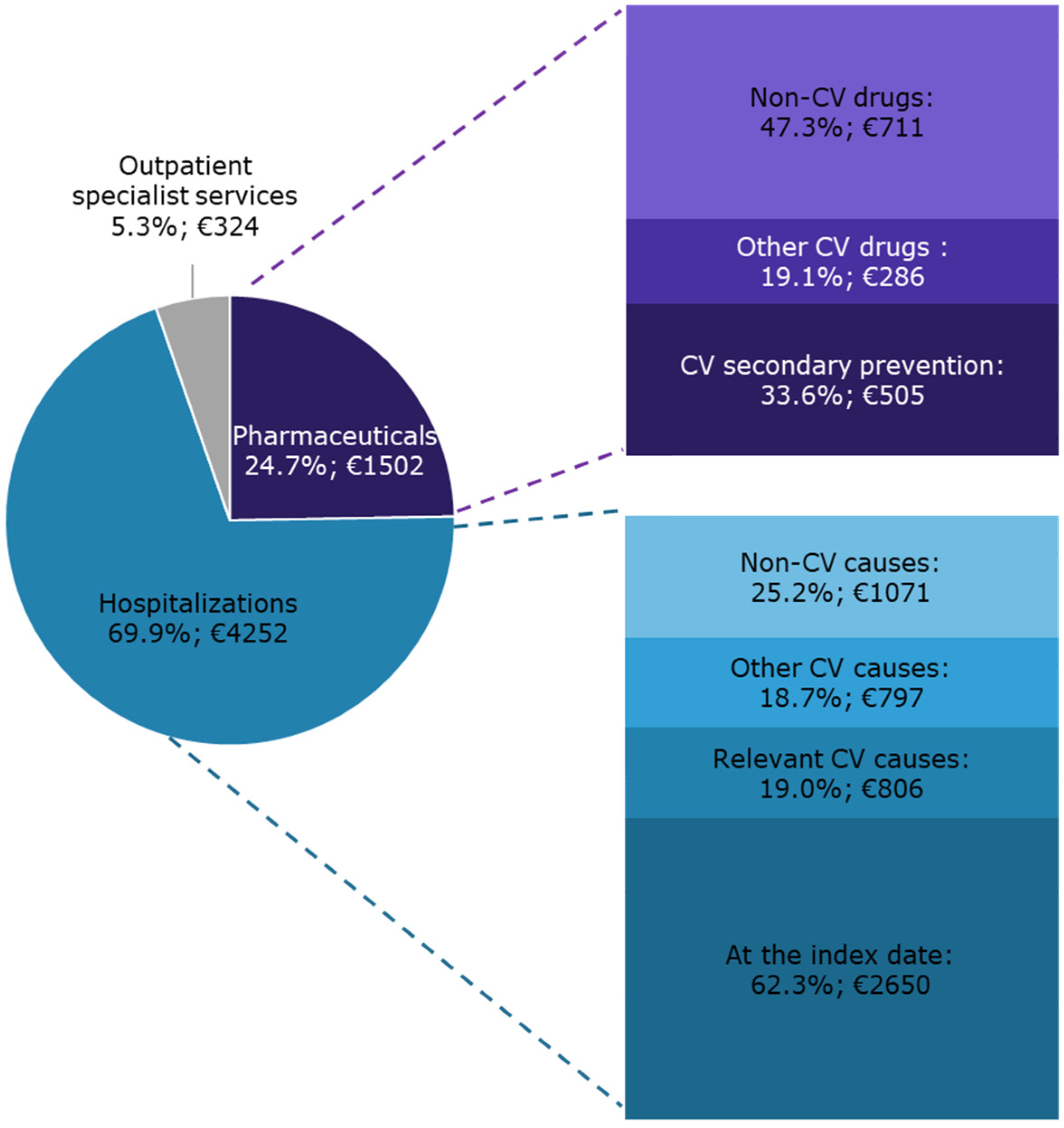
3.4. Healthcare Integrated Costs
4. Discussion
4.1. Demographics and Clinical Characteristics
4.2. Pharmacological Treatments
4.3. Hospitalizations
4.4. Healthcare Integrated Costs
4.5. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sorbets, E.; Lallmahomed, E.; Kretz, S.; Cinaud, A.; Vallée, A.; Lelong, H.; Blachier, V.; Blacher, J. Prevenzione secondaria nella malattia coronarica. EMC AKOS Trattato Med. 2021, 23, 1–11. [Google Scholar] [CrossRef]
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Bäck, M.; Benetos, A.; Biffi, A.; Boavida, J.-M.; Capodanno, D.; et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2021, 42, 3227–3337. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.A.A.; Metra, M.; Morais, J.; Atar, D. The myth of ’stable’ coronary artery disease. Nat. Rev. Cardiol. 2020, 17, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.C., Jr.; Benjamin, E.J.; Bonow, R.O.; Braun, L.T.; Creager, M.A.; Franklin, B.A.; Gibbons, R.J.; Grundy, S.M.; Hiratzka, L.F.; Jones, D.W.; et al. AHA/ACCF Secondary Prevention and Risk Reduction Therapy for Patients with Coronary and other Atherosclerotic Vascular Disease: 2011 update: A guideline from the American Heart Association and American College of Cardiology Foundation. Circulation 2011, 124, 2458–2473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuźma, Ł.; Wańha, W.; Kralisz, P.; Kazmierski, M.; Bachórzewska-Gajewska, H.; Wojakowski, W.; Dobrzycki, S. Impact of short-term air pollution exposure on acute coronary syndrome in two cohorts of industrial and non-industrial areas: A time series regression with 6,000,000 person-years of follow-up (ACS—Air Pollution Study). Environ. Res. 2021, 197, 111154. [Google Scholar] [CrossRef]
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Campana, E.; Cunha, V.; Glaveckaite, S.; Gruev, I.; Lamirault, G.; Lehmann, E.; Masi, S.; Kuate, L.M.; Mishshenko, L.; Ona, D.I.D.; et al. The use of single-pill combinations as first-line treatment for hypertension: Translating guidelines into clinical practice. J. Hypertens. 2020, 38, 2369–2377. [Google Scholar] [CrossRef] [PubMed]
- D’Anci, K.E.; Tipton, K.; Hedden-Gross, A.; Rouse, B.; Hermanson, L.; Fontanarosa, J. Effect of Intensive Blood Pressure Lowering on Cardiovascular Outcomes: A Systematic Review Prepared for the 2020 U.S. Department of Veterans Affairs/U.S. Department of Defense Guidelines. Ann. Intern. Med. 2020, 173, 895–903. [Google Scholar] [CrossRef]
- Bonaca, M.P.; Bhatt, D.L.; Cohen, M.; Steg, P.G.; Storey, R.F.; Jensen, E.C.; Magnani, G.; Bansilal, S.; Fish, M.P.; Im, K.; et al. Long-term use of ticagrelor in patients with prior myocardial infarction. N. Engl. J. Med. 2015, 372, 1791–1800. [Google Scholar] [CrossRef] [Green Version]
- Eikelboom, J.W.; Connolly, S.J.; Bosch, J.; Dagenais, G.; Dyal, L.; Lanas, F.; Metsarinne, K.; O’Donnell, M.; Dans, A.L.; Ha, J.-W.; et al. Rivaroxaban with or without Aspirin in Stable Cardiovascular Disease. N. Engl. J. Med. 2017, 377, 1319–1330. [Google Scholar] [CrossRef]
- Jortveit, J.; Halvorsen, S.; Kaldal, A.; Pripp, A.H.; Govatsmark, R.E.S.; Langørgen, J. Unsatisfactory risk factor control and high rate of new cardiovascular events in patients with myocardial infarction and prior coronary artery disease. BMC Cardiovasc. Disord. 2019, 19, 71. [Google Scholar] [CrossRef]
- Mangiapane, S.; Busse, R. Prescription prevalence and continuing medication use for secondary prevention after myocardial infarction: The reality of care revealed by claims data analysis. Dtsch. Arztebl. Int. 2011, 108, 856–862. [Google Scholar] [CrossRef]
- Maggioni, A.P.; Calabria, S.; Rossi, E.; Martini, N. Use of lipid lowering drugs in patients at very high risk of cardiovascular events: An analysis on nearly 3,000,000 Italian subjects of the ARNO Observatory. Int. J. Cardiol. 2017, 246, 62–67. [Google Scholar] [CrossRef]
- Maggioni, A.P.; Dondi, L.; Andreotti, F.; Calabria, S.; Iacoviello, M.; Gorini, M.; Gonzini, L.; Piccinni, C.; Ronconi, G.; Martini, N. Prevalence, clinical impact and costs of hyperkalaemia: Special focus on heart failure. Eur. J. Clin. Investig. 2021, 51, e13551. [Google Scholar] [CrossRef] [PubMed]
- Piccinni, C.; Cevoli, S.; Ronconi, G.; Dondi, L.; Calabria, S.; Pedrini, A.; Maggioni, A.P.; Esposito, I.; Addesi, A.; Favoni, V.; et al. Insights into real-world treatment of cluster headache through a large Italian da-tabase: Prevalence, prescription patterns, and costs. Expert Rev. Clin. Pharmacol. 2021, 14, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ronconi, G.; Dondi, L.; Calabria, S.; Piccinni, C.; Pedrini, A.; Esposito, I.; Martini, N. Real-world Prescription Pattern, Discontinuation and Costs of Ibrutinib-Naïve Patients with Chronic Lymphocytic Leukemia: An Italian Healthcare Administrative Database Analysis. Clin. Drug Investig. 2021, 41, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Ministero del Lavoro, della Salute e delle Politiche Sociali. Classificazione delle Malattie, dei Traumatismi, degli Interventi Chirurgici e delle Procedure Dagnostiche e Terapeutiche. Versione Italiana della ICD9-CM; Ministero del Lavoro, della Salute e delle Politiche Sociali: Rome, Italy, 2007.
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics-2020 Update: A Report From the American Heart Association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef] [PubMed]
- Blin, P.; Philippe, F.; Bouée, S.; Laurendeau, C.; Torreton, E.; Gourmelin, J.; Leproust, S.; Levy-Bachelot, L.; Steg, P.G. Outcomes following acute hospitalised myocardial infarction in France: An insurance claims database analysis. Int. J. Cardiol. 2016, 219, 387–393. [Google Scholar] [CrossRef]
- Shimada, K.; Hamada, S.; Sawano, M.; Yamamoto, H.; Yoshie, S.; Iijima, K.; Miyata, H. Guideline-Based Medications for Older Adults Discharged after Percutaneous Coronary Intervention in a Suburban City of Japan: A Cohort Study Using Claims Data. Tohoku J. Exp. Med. 2020, 252, 143–152. [Google Scholar] [CrossRef]
- Ulrich, R.; Pischon, T.; Robra, B.P.; Freier, C.; Heintze, C.; Herrmann, W.J. Health care utilisation and medication one year after myocardial infarction in Germany—A claims data analysis. Int. J. Cardiol. 2020, 300, 20–26. [Google Scholar] [CrossRef]
- Delate, T.; Olson, K.L.; Rasmussen, J.; Hutka, K.; Sandhoff, B.; Hornak, R.; Merenich, J.; Clinical Pharmacy Cardiac Risk Service Study Group. Reduced health care expenditures after enrollment in a collaborative cardiac care service. Pharmacotherapy 2010, 30, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- AIFA. L’uso dei Farmaci in Italia. Rapporto Nazionale Anno 2018 [The Use of Drugs in Italy. National Report Year 2018]; AIFA: Rome, Italy, 2018. [Google Scholar]
- Gini, R.; Schuemie, M.J.; Francesconi, P.; Lapi, F.; Cricelli, I.; Pasqua, A.; Gallina, P.; Donato, D.; Brugaletta, S.; Donatini, A.; et al. Can Italian healthcare administrative databases be used to compare regions with respect to compliance with standards of care for chronic diseases? PLoS ONE 2014, 9, e95419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rector, T.S.; Wickstrom, S.L.; Shah, M.; Greeenlee, N.T.; Rheault, P.; Rogowski, J.; Freedman, V.; Adams, J.; Escarce, J.J. Specificity and sensitivity of claims-based algorithms for identifying members of Medicare+Choice health plans that have chronic medical conditions. Health Serv. Res. 2004, 39, 1839–1857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saczynski, J.S.; Andrade, S.E.; Harrold, L.R.; Tjia, J.; Cutrona, S.L.; Dodd, K.S.; Goldberg, R.J.; Gurwitz, J.H. A systematic review of validated methods for identifying heart failure using administrative data. Pharmacoepidemiol. Drug Saf. 2012, 21, 129–140. [Google Scholar] [CrossRef] [PubMed]
| Demographics and Clinical Characteristics | Patients with CAD and Eligible for Secondary Prevention (n = 46,063) |
|---|---|
| Males (n; %) | 33,234; 72.1 |
| Median age (Q1; Q3) | 70 (60; 83) |
| Mean age (±SD) | 70 ± 12 |
| Distribution by age group (prevalence × 1000 subjects aged ≥ 35) | |
| 35–44 | 0.9 |
| 45–54 | 4.7 |
| 55–64 | 13.9 |
| 65–74 | 23.8 |
| 75–84 | 27.3 |
| ≥85 | 21.3 |
| Overall cohort | 12.6 |
| Comorbidities (n; %) | |
| None | 1858; 4.0 |
| 1 comorbidity | 5361; 11.6 |
| 2 comorbidities | 15,646; 34.0 |
| 3 comorbidities | 12,862; 27.9 |
| 4 or more comorbidities | 10,336; 22.4 |
| Arterial hypertension | 41,415; 89.9 |
| Dyslipidaemia | 33,332; 72.4 |
| Diabetes | 15,276; 33.2 |
| Chronic lung diseases | 9645; 20.9 |
| Heart failure | 7180; 15.6 |
| Atrial fibrillation | 5641; 12.2 |
| Depression | 4803; 10.4 |
| Cerebrovascular diseases | 2992; 6.5 |
| Chronic liver diseases | 1875; 4.1 |
| Supplied Drugs | Patients Treated (n; %) | Mean DDD Per Treated Patient |
|---|---|---|
| Drugs for CV secondary prevention (in descending order) | ||
| Antiplatelet agents (excluding heparin) | 38,201; 82.9 | 400.8 |
| Lipid lowering agents | 38,042; 82.6 | 532.1 |
| β-blockers | 33,375; 72.5 | 136.8 |
| ACE inhibitors (ACE-is) | 20,964; 45.5 | 469.4 |
| Angiotensin II receptor blockers (ARBs) | 15,564; 33.8 | 388.4 |
| At least one drug for CV secondary prevention | 44,391; 96.4 | 1261.6 |
| Concomitant CV drugs (first 10 supplied, in descending order) | ||
| Loop diuretics | 18,239; 39.6 | 300.4 |
| Antithrombotic agents (excluding antiplatelets) | 11,439; 24.8 | 173.7 |
| Selective calcium channel blockers with mainly vascular effects | 8736; 19.0 | 320.4 |
| Potassium-sparing agents | 7351; 16.0 | 126.5 |
| Other cardiac preparations | 7271; 15.8 | 202.5 |
| Nitrates | 5426; 11.8 | 338.2 |
| Anti-arrhythmics, class I and III | 4340; 9.4 | 196.5 |
| Anti-adrenergic agents, peripherally acting | 2126; 4.6 | 198.7 |
| Diuretics and potassium-sparing agents in combination | 1920; 4.2 | 95.8 |
| Selective calcium channel blockers with direct cardiac effects | 1547; 3.4 | 201.5 |
| At least one concomitant CV drug | 31,948; 69.4 | 518.3 |
| Concomitant non-CV drugs (first 10 supplied, in descending order) | ||
| Drugs for peptic ulcer and gastro-oesophageal reflux disease | 36,520; 79.3 | 230.0 |
| Anti-inflammatory and antirheumatic products, non-steroids | 17,305; 37.6 | 43.9 |
| Beta-lactam antibacterials, penicillins | 15,106; 32.8 | 14.6 |
| Quinolone antibacterials | 12,408; 26.9 | 13.1 |
| Blood glucose lowering drugs, excluding insulins | 12,293; 26.7 | 359.8 |
| Other beta-lactam antibacterials | 10,628; 23.1 | 9.3 |
| Vitamin A and D, including combinations of the two | 10,015; 21.7 | 454.0 |
| Antigout preparations | 9857; 21.4 | 155.4 |
| Corticosteroids for systemic use, plain | 9285; 20.2 | 60.1 |
| Drugs used in benign prostatic hypertrophy | 7646; 16.6 | 377.1 |
| At least one concomitant non-CV drug | 44,120; 95.8 | 861.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calabria, S.; Ronconi, G.; Dondi, L.; Piccinni, C.; Cinconze, E.; Pedrini, A.; Esposito, I.; Addesi, A.; Martini, N.; Maggioni, A.P. Coronary Artery Disease in Patients Older than 35 and Eligible for Cardiovascular Secondary Prevention: An Italian Retrospective Observational Analysis of Healthcare Administrative Databases. J. Clin. Med. 2021, 10, 4708. https://doi.org/10.3390/jcm10204708
Calabria S, Ronconi G, Dondi L, Piccinni C, Cinconze E, Pedrini A, Esposito I, Addesi A, Martini N, Maggioni AP. Coronary Artery Disease in Patients Older than 35 and Eligible for Cardiovascular Secondary Prevention: An Italian Retrospective Observational Analysis of Healthcare Administrative Databases. Journal of Clinical Medicine. 2021; 10(20):4708. https://doi.org/10.3390/jcm10204708
Chicago/Turabian StyleCalabria, Silvia, Giulia Ronconi, Letizia Dondi, Carlo Piccinni, Enrico Cinconze, Antonella Pedrini, Immacolata Esposito, Alice Addesi, Nello Martini, and Aldo Pietro Maggioni. 2021. "Coronary Artery Disease in Patients Older than 35 and Eligible for Cardiovascular Secondary Prevention: An Italian Retrospective Observational Analysis of Healthcare Administrative Databases" Journal of Clinical Medicine 10, no. 20: 4708. https://doi.org/10.3390/jcm10204708
APA StyleCalabria, S., Ronconi, G., Dondi, L., Piccinni, C., Cinconze, E., Pedrini, A., Esposito, I., Addesi, A., Martini, N., & Maggioni, A. P. (2021). Coronary Artery Disease in Patients Older than 35 and Eligible for Cardiovascular Secondary Prevention: An Italian Retrospective Observational Analysis of Healthcare Administrative Databases. Journal of Clinical Medicine, 10(20), 4708. https://doi.org/10.3390/jcm10204708







