Application of Tissue Aspirate Parathyroid Hormone Assay for Imaging Suspicious Neck Lesions in Patients with Complicated Recurrent or Persistent Renal Hyperparathyroidism
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
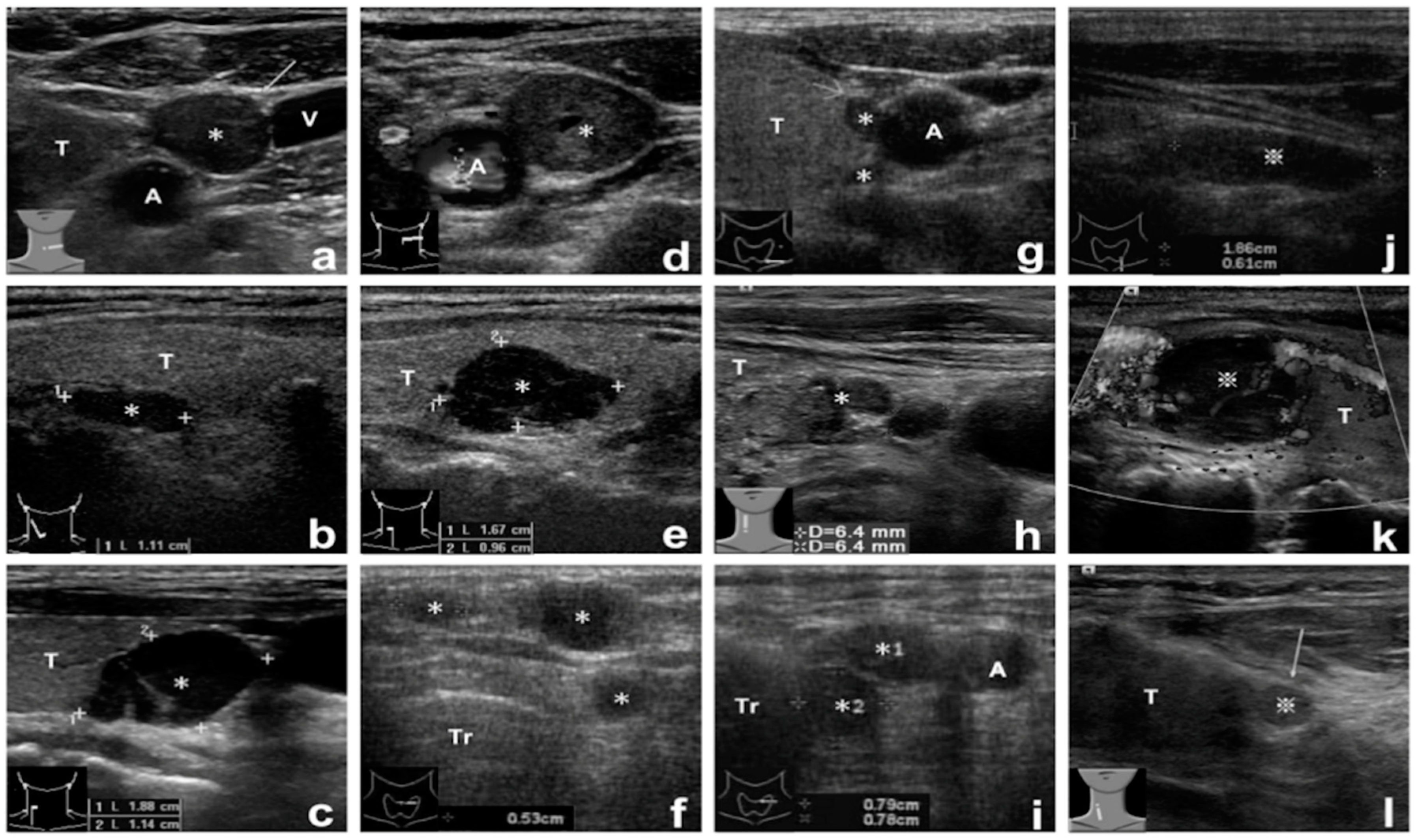
2.2. Tissue Aspirate PTH Assay
2.3. Data Analysis
2.4. Statistical Analysis
3. Results
3.1. Patient Demographics
3.2. Comparison between Different Assay Results
3.3. Comparisons with Localization Studies
3.4. Surgical Intervention
3.5. Reoperative Findings
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PTH | parathyroid hormone |
References
- Yuen, N.K.; Ananthakrishnan, S.; Campbell, M.J. Hyperparathyroidism of renal disease. Perm. J. 2016, 20, 15–127. [Google Scholar] [CrossRef] [PubMed]
- Chou, F.F.; Lee, C.H.; Chen, H.Y.; Chen, J.B.; Hsu, K.T.; Sheen-Chen, S.M. Persistent and recurrent hyperparathyroidism after total parathyroidectomy with autotransplantation. Ann. Surg. 2002, 235, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Hibi, Y.; Tominaga, Y.; Sato, T. Reoperation for renal hyperparathyroidism. World J. Surg. 2002, 26, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Jofre, R.; Lopez Gomez, J.M.; Menarguez, J. Parathyroidectomy: Whom and when? Kidney Int. Suppl. 2003, S97–S100. [Google Scholar] [CrossRef][Green Version]
- Patow, C.A.; Norton, J.A.; Brennan, M.F. Vocal cord paralysis and reoperative parathyroidectomy. A prospective study. Ann. Surg. 1986, 203, 282–285. [Google Scholar] [CrossRef]
- Alkhalili, E.; Tasci, Y.; Aksoy, E. The utility of neck ultrasound and sestamibi scans in patients with secondary and tertiary hyperparathyroidism. World J. Surg. 2015, 39, 701–705. [Google Scholar] [CrossRef]
- Lai, E.C.; Ching, A.S.; Leong, H.T. Secondary and tertiary hyperparathyroidism: Role of preoperative localization. ANZ J. Surg. 2007, 77, 880–882. [Google Scholar] [CrossRef]
- Neumann, D.R.; Esselstyn, C.B., Jr.; Madera, A.M. Sestamibi/iodine subtraction single photon emission computed tomography in reoperative secondary hyperparathyroidism. Surgery 2000, 128, 22–28. [Google Scholar] [CrossRef]
- Irvin, G.L., III; Molinari, A.S.; Figueroa, C.; Carneiro, D.M. Improved success rate in reoperative parathyroidectomy with intraoperative PTH assay. Ann. Surg. 1999, 229, 874–878, discussion 878–879. [Google Scholar] [CrossRef]
- Miller, D.L. Pre-operative localization and interventional treatment of parathyroid tumors: When and how? World J. Surg. 1991, 15, 706–715. [Google Scholar]
- Neumann, D.R.; Esselstyn, C.B., Jr.; Madera, A.; Wong, C.O.; Lieber, M. Parathyroid detection in secondary hyperparathyroidism with 123I/99mTc-sestamibi subtraction single photon emission computed tomography. J. Clin. Endocrinol. Metab. 1998, 83, 3867–3871. [Google Scholar] [CrossRef] [PubMed]
- Dotzenrath, C.; Cupisti, K.; Goretzki, E. Operative treatment of renal autonomous hyperparathyroidism: Cause of persistent or recurrent disease in 304 patients. Langenbeck’s Arch. Surg. 2003, 387, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Rodriquez, J.M.; Tezelman, S.; Siperstein, A.E. Localization procedures in patients with persistent or recurrent hyperparathyroidism. Arch. Surg. 1994, 129, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Kunstman, J.W.; Kirsch, J.D.; Mahajan, A.; Udelsman, R. Clinical review: Parathyroid localization and implications for clinical management. J. Clin. Endocrinol. Metab. 2013, 98, 902–912. [Google Scholar] [CrossRef]
- Seehofer, D.; Steinmuller, T.; Rayes, N. Parathyroid hormone venous sampling before reoperative surgery in renal hyperparathyroidism: Comparison with noninvasive localization procedures and review of the literature. Arch. Surg. 2004, 139, 1331–1338. [Google Scholar] [CrossRef]
- Doppman, J.L.; Krudy, A.G.; Marx, S.J.; Saxe, A.; Schneider, P.; Norton, J.A.; Spiegel, A.M.; Downs, R.W.; Schaaf, M.; Brennan, M.E.; et al. Aspiration of enlarged parathyroid glands for parathyroid hormone assay. Radiology 1983, 148, 31–35. [Google Scholar] [CrossRef]
- Abdelghani, R.; Noureldine, S.; Abbas, A.; Moroz, K.; Kandil, E. The diagnostic value of parathyroid hormone washout after fine-needle aspiration of suspicious cervical lesions in patients with hyperparathyroidism. Laryngoscope 2013, 123, 1310–1313. [Google Scholar] [CrossRef]
- MacFarlane, M.P.; Fraker, D.L.; Shawker, T.H. Use of preoperative fine-needle aspiration in patients undergoing reoperation for primary hyperparathyroidism. Surgery 1994, 116, 959–965. [Google Scholar]
- Stephen, A.E.; Milas, M.; Garner, C.N.; Wagner, K.E.; Siperstein, A.E. Use of surgeon-performed office ultrasound and parathyroid fine needle aspiration for complex parathyroid localization. Surgery 2005, 138, 1143–1151. [Google Scholar] [CrossRef]
- Kim, J.; Horowitz, G.; Hong, M.; Orsini, M.; Asa, S.L.; Higgins, K. The dangers of parathyroid biopsy. J. Otolaryngol. Head Neck Surg. = Le J. D’oto-Rhino-Laryngol. Et De Chir. Cervico-Faciale 2017, 46, 4. [Google Scholar] [CrossRef]
- Frasoldati, A.; Pesenti, M.; Toschi, E.; Azzarito, C.; Zini, M.; Valcavi, R. Detection and diagnosis of parathyroid incidentalomas during thyroid sonography. J. Clin. Ultrasound. 1999, 27, 492–498. [Google Scholar] [CrossRef]
- Sacks, B.; Pallotta, J.; Cole, A.; Hurwitz, J. Diagnosis of parathyroid adenomas: Efficacy of measuring parathormone levels in needle aspirates of cervical masses. AJR Am. J. Roentgenol. 1994, 163, 1223–1226. [Google Scholar] [CrossRef] [PubMed]
- Maser, C.; Donovan, P.; Santos, F. Sonographically guided fine needle aspiration with rapid parathyroid hormone assay. Ann. Surg. Oncol. 2006, 13, 1690. [Google Scholar] [CrossRef] [PubMed]
| Tissue Aspirate PTH Assay | Negative | Positive | p Value |
|---|---|---|---|
| Number (n = 50) | 11 | 39 | |
| Tissue aspirate PTH level (pg/mL), median (range) | 3.9 (1.0–28.6) | 2500 (38.9–44470.0) | <0.001 |
| Serum systemic PTH level (pg/mL), median (range) | 428.2 (214.8–1817.8) | 869.7 (227.8 ≥ 2500.0) | 0.137 |
| Serum calcium level (mg/dL), median (range) | 10.0 (9.4–12.5) | 10.5 (8.8–12.5) | 0.218 |
| Parathyroid scintigraphy, n (%) | 0.008 | ||
| Negative | 9 (81.8%) | 13 (33.3%) | |
| Suspicious | 0 (0.0%) | 18 (46.2%) | |
| Positive | 2 (18.2%) | 8 (20.5%) | |
| Parathyroid sonography, n (%) | 0.114 | ||
| Suspicious | 11 (100%) | 28 (71.8%) | |
| Positive | 0 (0%) | 11 (28.2%) | |
| Neck lesion explored, n (%) | 3 (27.3%) † | 35 (89.76%) ‡ | <0.001 |
| Pathology proved parathyroid lesions, n | 1 | 35 |
| Locolization Studies | Parathyroid Sonography | |||||
|---|---|---|---|---|---|---|
| Suspicious (n = 39) | Positive (n = 11) | |||||
| Parathyroid Scintigraphy | Tissue Aspirate PTH Assay | Negative (n = 11) | Positive (n = 28) | Negative (n = 0) | Positive (n = 11) | |
| Negative (n = 11) Positive (n = 39) | ||||||
| Negative (n = 22) | Negative (n = 9) | 9 | 0 | |||
| Positive (n = 13) | 9 | 4 | ||||
| Suspicious (n = 18) | Negative (n = 0) | 0 | 0 | |||
| Positive (n = 18) | 14 | 4 | ||||
| Positive (n = 10) | Negative (n = 2) | 2 * | 0 | |||
| Positive (n = 8) | 5 | 3 | ||||
| Location | Lesions (n = 36) | Patients (n = 26) * |
|---|---|---|
| Ectopic | ||
| Undescended | 3 (8.3%) | 3 |
| Intrathymic | 1 (2.8%) | 1 |
| Carotid sheath | 2 (5.6%) | 2 |
| Parathyromatosis | 9 (25.0%) | 4 |
| Intrathyroid | 8 (22.2%) | 7 |
| Neck (usual parathyroid area) | ||
| LS | 4 (11.1%) | 4 |
| LI | 1 (2.8%) | 1 |
| RS | 4 (11.1%) | 4 |
| RI | 4 (11.1%) | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, C.-L.; Hsu, Y.-C.; Huang, S.-M.; Hung, C.-J. Application of Tissue Aspirate Parathyroid Hormone Assay for Imaging Suspicious Neck Lesions in Patients with Complicated Recurrent or Persistent Renal Hyperparathyroidism. J. Clin. Med. 2021, 10, 329. https://doi.org/10.3390/jcm10020329
Hung C-L, Hsu Y-C, Huang S-M, Hung C-J. Application of Tissue Aspirate Parathyroid Hormone Assay for Imaging Suspicious Neck Lesions in Patients with Complicated Recurrent or Persistent Renal Hyperparathyroidism. Journal of Clinical Medicine. 2021; 10(2):329. https://doi.org/10.3390/jcm10020329
Chicago/Turabian StyleHung, Chien-Ling, Yu-Chen Hsu, Shih-Ming Huang, and Chung-Jye Hung. 2021. "Application of Tissue Aspirate Parathyroid Hormone Assay for Imaging Suspicious Neck Lesions in Patients with Complicated Recurrent or Persistent Renal Hyperparathyroidism" Journal of Clinical Medicine 10, no. 2: 329. https://doi.org/10.3390/jcm10020329
APA StyleHung, C.-L., Hsu, Y.-C., Huang, S.-M., & Hung, C.-J. (2021). Application of Tissue Aspirate Parathyroid Hormone Assay for Imaging Suspicious Neck Lesions in Patients with Complicated Recurrent or Persistent Renal Hyperparathyroidism. Journal of Clinical Medicine, 10(2), 329. https://doi.org/10.3390/jcm10020329






