Prognostic Implications of Chronic Heart Failure and Utility of NT-proBNP Levels in Heart Failure Patients with SARS-CoV-2 Infection
Abstract
1. Introduction
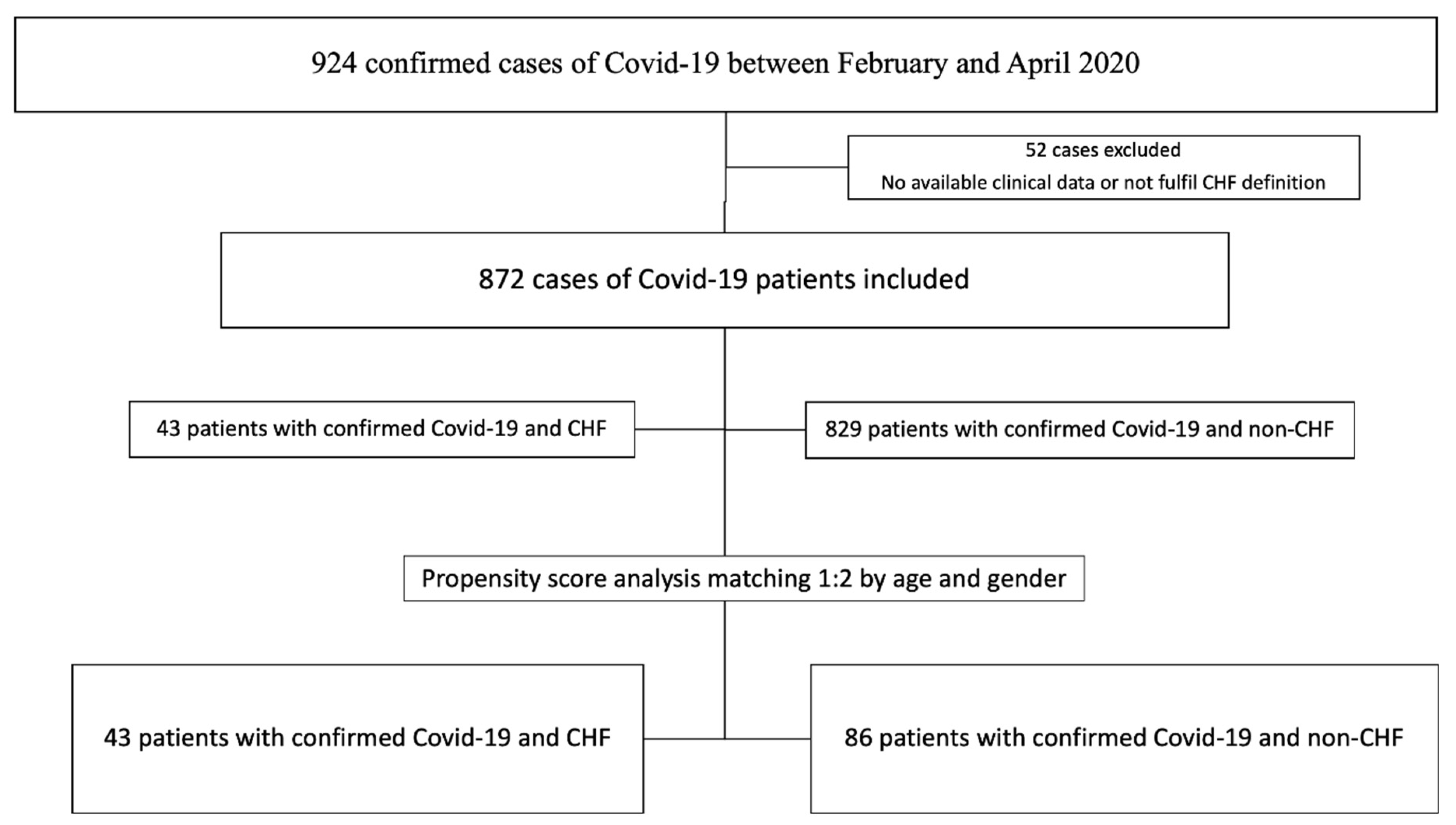
2. Materials and Methods
2.1. Study Design and Data Collection
2.2. Statistical Analysis
2.3. Ethics Considerations
3. Results
3.1. Characteristics of Chronic Heart Failure (CHF) versus Non-CHF
3.2. Outcomes in CHF vs. Non-CHF
3.3. CHF Cohort
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Bi, Z.; Zhao, Y. Prevalence and Impact of Cardiovascular Metabolic Diseases on COVID-19 in China. Clin. Res. Cardiol. 2020, 109, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72,314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.P.; Blet, A.; Smyth, D.; Li, H. The Science Underlying COVID-19: Implications for the Cardiovascular System. Circulation 2020, 142, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Hendren, N.S.; Drazner, M.H.; Bozkurt, B.; Cooper, L.T. Description and Proposed Management of the Acute COVID-19 Cardiovascular Syndrome. Circulation 2020, 141, 1903–1914. [Google Scholar] [CrossRef] [PubMed]
- Inciardi, R.M.; Lupi, L.; Zaccone, G.; Italia, L.; Raffo, M.; Tomasoni, D.; Cani, D.S.; Cerini, M.; Farina, D.; Gavazzi, E.; et al. Cardiac Involvement in a Patient with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Alon, D.; Stein, G.Y.; Korenfeld, R.; Fuchs, S. Predictors and Outcomes of Infection-Related Hospital Admissions of Heart Failure Patients. PLoS ONE 2013, 8, e72476. [Google Scholar] [CrossRef]
- Chioncel, O.; Mebazaa, A.; Harjola, V.-P.; Coats, A.J.; Piepoli, M.F.; Crespo-Leiro, M.G.; Laroche, C.; Seferovic, P.M.; Anker, S.D.; Ferrari, R.; et al. Clinical Phenotypes and Outcome of Patients Hospitalized for Acute Heart Failure: The ESC Heart Failure Long-Term Registry. Eur. J. Heart Fail. 2017, 19, 1242–1254. [Google Scholar] [CrossRef]
- Platz, E.; Jhund, P.S.; Claggett, B.L.; Pfeffer, M.A.; Swedberg, K.; Granger, C.B.; Yusuf, S.; Solomon, S.D.; McMurray, J.J. Prevalence and Prognostic Importance of Precipitating Factors Leading to Heart Failure Hospitalization: Recurrent Hospitalizations and Mortality. Eur. J. Heart Fail. 2018, 20, 295–303. [Google Scholar] [CrossRef]
- Tralhão, A.; Póvoa, P. Cardiovascular Events after Community-Acquired Pneumonia: A Global Perspective with Systematic Review and Meta-Analysis of Observational Studies. J. Clin. Med. 2020, 9, 414. [Google Scholar] [CrossRef]
- Rizzo, P.; Vieceli Dalla Sega, F.; Fortini, F.; Marracino, L.; Rapezzi, C.; Ferrari, R. COVID-19 in the Heart and the Lungs: Could We “Notch” the Inflammatory Storm? Basic Res. Cardiol. 2020, 115. [Google Scholar] [CrossRef]
- Alvarez-Garcia, J.; Lee, S.; Gupta, A.; Cagliostro, M.; Joshi, A.A.; Rivas-Lasarte, M.; Contreras, J.; Mitter, S.S.; LaRocca, G.; Tlachi, P.; et al. Prognostic Impact of Prior Heart Failure in Patients Hospitalized with COVID-19. J. Am. Coll. Cardiol. 2020, 76, 2334–2348. [Google Scholar] [CrossRef] [PubMed]
- Rey, J.R.; Caro-Codón, J.; Rosillo, S.O.; Iniesta, Á.M.; Castrejón-Castrejón, S.; Marco-Clement, I.; Martín-Polo, L.; Merino-Argos, C.; Rodríguez-Sotelo, L.; García-Veas, J.M.; et al. Heart Failure in COVID-19 Patients: Prevalence, Incidence and Prognostic Implications. Eur. J. Heart Fail. 2020. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Meng, K.; He, M.; Zhu, R.; Guan, H.; Ke, Z.; Leng, L.; Wang, X.; Liu, B.; Hu, C.; et al. Clinical Characteristics and Prognosis of 244 Cardiovascular Patients Suffering from Coronavirus Disease in Wuhan, China. J. Am. Heart Assoc. 2020, e016796. [Google Scholar] [CrossRef] [PubMed]
- Inciardi, R.M.; Adamo, M.; Lupi, L.; Cani, D.S.; Di Pasquale, M.; Tomasoni, D.; Italia, L.; Zaccone, G.; Tedino, C.; Fabbricatore, D.; et al. Characteristics and Outcomes of Patients Hospitalized for COVID-19 and Cardiac Disease in Northern Italy. Eur. Heart J. 2020, 41, 1821–1829. [Google Scholar] [CrossRef] [PubMed]
- Dong, N.; Cai, J.; Zhou, Y.; Liu, J.; Li, F. End-Stage Heart Failure With COVID-19: Strong Evidence of Myocardial Injury by 2019-NCoV. JACC Heart Fail. 2020, 8, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Farré, N.; Vela, E.; Clèries, M.; Bustins, M.; Cainzos-Achirica, M.; Enjuanes, C.; Moliner, P.; Ruiz, S.; Verdú-Rotellar, J.M.; Comín-Colet, J. Real World Heart Failure Epidemiology and Outcome: A Population-Based Analysis of 88,195 Patients. PLoS ONE 2017, 12, e0172745. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.-P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC)Developed with the Special Contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [CrossRef]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020. [Google Scholar] [CrossRef]
- Groenewegen, A.; Rutten, F.H.; Mosterd, A.; Hoes, A.W. Epidemiology of Heart Failure. Eur. J. Heart Fail. 2020, 22, 1342–1356. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical Course and Risk Factors for Mortality of Adult Inpatients with COVID-19 in Wuhan, China: A Retrospective Cohort Study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y. Prevalence of Comorbidities and Its Effects in Patients Infected with SARS-CoV-2: A Systematic Review and Meta-Analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Baroutjian, A.; Sanchez, C.; Boneva, D.; McKenney, M.; Elkbuli, A. SARS-CoV-2 Pharmacologic Therapies and Their Safety/Effectiveness According to Level of Evidence. Am. J. Emerg. Med. 2020, 38, 2405–2415. [Google Scholar] [CrossRef] [PubMed]
- Cantini, F.; Goletti, D.; Petrone, L.; Najafi Fard, S.; Niccoli, L.; Foti, R. Immune Therapy, or Antiviral Therapy, or Both for COVID-19: A Systematic Review. Drugs 2020, 80, 1929–1946. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; DeSantis, C.; Virgo, K.; Stein, K.; Mariotto, A.; Smith, T.; Cooper, D.; Gansler, T.; Lerro, C.; Fedewa, S.; et al. Cancer Treatment and Survivorship Statistics, 2012. CA Cancer J. Clin. 2012, 62, 220–241. [Google Scholar] [CrossRef]
- Abubakar, A.R.; Sani, I.H.; Godman, B.; Kumar, S.; Islam, S.; Jahan, I.; Haque, M. Systematic Review on the Therapeutic Options for COVID-19: Clinical Evidence of Drug Efficacy and Implications. Infect. Drug Resist. 2020, 13, 4673–4695. [Google Scholar] [CrossRef]
- Poulakou, G.; Dimakakos, E.; Kollias, A.; Kyriakoulis, K.G.; Rapti, V.; Trontzas, I.; Thanos, C.; Abdelrasoul, M.; Vantana, T.; Leontis, K.; et al. Beneficial Effects of Intermediate Dosage of Anticoagulation Treatment on the Prognosis of Hospitalized COVID-19 Patients: The ETHRA Study. In Vivo 2021, 35, 653–661. [Google Scholar] [CrossRef]
- Patell, R.; Chiasakul, T.; Bauer, E.; Zwicker, J.I. Pharmacologic Thromboprophylaxis and Thrombosis in Hospitalized Patients with COVID-19: A Pooled Analysis. Thromb. Haemost. 2020. [Google Scholar] [CrossRef]
- Kytömaa, S.; Hegde, S.; Claggett, B.; Udell, J.A.; Rosamond, W.; Temte, J.; Nichol, K.; Wright, J.D.; Solomon, S.D.; Vardeny, O. Association of Influenza-like Illness Activity With Hospitalizations for Heart Failure: The Atherosclerosis Risk in Communities Study. JAMA Cardiol. 2019, 4, 363–369. [Google Scholar] [CrossRef]
- Long, B.; Brady, W.J.; Koyfman, A.; Gottlieb, M. Cardiovascular Complications in COVID-19. Am. J. Emerg. Med. 2020, 38, 1504–1507. [Google Scholar] [CrossRef]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical Characteristics of 113 Deceased Patients with Coronavirus Disease 2019: Retrospective Study. BMJ 2020, 368, m1091. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Fernández, A.; Izquierdo, A.; Subirana, I.; Farré, N.; Vila, J.; Durán, X.; García-Guimaraes, M.; Valdivielso, S.; Cabero, P.; Soler, C.; et al. Markers of Myocardial Injury in the Prediction of Short-Term COVID-19 Prognosis. Revista Española de Cardiología 2020. [Google Scholar] [CrossRef]
- Zhang, Y.; Coats, A.J.S.; Zheng, Z.; Adamo, M.; Ambrosio, G.; Anker, S.D.; Butler, J.; Xu, D.; Mao, J.; Khan, M.S.; et al. Management of Heart Failure Patients with COVID-19: A Joint Position Paper of the Chinese Heart Failure Association & National Heart Failure Committee and the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2020, 22, 941–956. [Google Scholar] [CrossRef] [PubMed]
- Sisti, N.; Valente, S.; Mandoli, G.E.; Santoro, C.; Sciaccaluga, C.; Franchi, F.; Cameli, P.; Mondillo, S.; Cameli, M. COVID-19 in Patients with Heart Failure: The New and the Old Epidemic. Postgrad. Med. J. 2020. [Google Scholar] [CrossRef]
- Mehra, M.R.; Desai, S.S.; Kuy, S.; Henry, T.D.; Patel, A.N. Cardiovascular Disease, Drug Therapy, and Mortality in Covid-19. N. Engl. J. Med. 2020, 382, e102. [Google Scholar] [CrossRef]
- Bader, F.; Manla, Y.; Atallah, B.; Starling, R.C. Heart Failure and COVID-19. Heart Fail. Rev. 2020. [Google Scholar] [CrossRef]
- Nagarajan, V.; Hernandez, A.V.; Tang, W.H.W. Prognostic Value of Cardiac Troponin in Chronic Stable Heart Failure: A Systematic Review. Heart 2012, 98, 1778–1786. [Google Scholar] [CrossRef]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef]
- Sandoval, Y.; Januzzi, J.L.; Jaffe, A.S. Cardiac Troponin for the Diagnosis and Risk-Stratification of Myocardial Injury in COVID-19: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2020. [Google Scholar] [CrossRef]
- Akpınar, E.E.; Hoşgün, D.; Akpınar, S.; Ateş, C.; Baha, A.; Gülensoy, E.S.; Ogan, N.; Akpınar, E.E.; Hoşgün, D.; Akpınar, S.; et al. Do N-Terminal pro-Brain Natriuretic Peptide Levels Determine the Prognosis of Community Acquired Pneumonia? J. Bras. Pneumol. 2019, 45. [Google Scholar] [CrossRef]
- Gao, L.; Jiang, D.; Wen, X.-S.; Cheng, X.-C.; Sun, M.; He, B.; You, L.-N.; Lei, P.; Tan, X.-W.; Qin, S.; et al. Prognostic Value of NT-ProBNP in Patients with Severe COVID-19. Respir. Res. 2020, 21, 83. [Google Scholar] [CrossRef] [PubMed]

| Total (n = 129) | CHF (n = 43) | Non-CHF (n = 86) | p Value | |
|---|---|---|---|---|
| Women | 66 (51.2) | 22 (51.2) | 44 (51.2) | 1 |
| Age, years | 80.3 (±12) | 80.3 (±12.1) | 80.4 (±12.1) | 0.984 |
| Influenza vaccination | 27 (34.6) | 10 (43.5) | 17 (30.9) | 0.287 |
| Smoking | 31 (24) | 9 (20.9) | 22 (25.6) | 0.832 |
| Diabetes | 49 (38) | 21 (48.8) | 28 (32.6) | 0.073 |
| Hypertension | 101 (78.3) | 38 (88.4) | 63 (73.3) | 0.050 |
| Dyslipemia | 67 (51.9) | 27 (62.8) | 40 (46.5) | 0.081 |
| Obesity | 29 (26.4) | 14 (32.6) | 15 (17.4) | 0.018 |
| Ischemic cardiac disease | 22 (17.1) | 10 (23.3) | 12 (14) | 0.185 |
| AF or flutter | 31 (24) | 22 (51.2) | 9 (10.5) | 0.001 |
| Valvular heart disease | 19 (14.7) | 14 (32.6) | 5 (5.8) | 0.001 |
| ACEI or ARB II | 58 (45) | 23 (53.5) | 35 (40.7) | 0.169 |
| COPD | 14 (10.9) | 8 (18.6) | 6 (7.0) | 0.069 |
| Previous Cancer | 33 (25.6) | 10 (23.3) | 23 (26.7) | 0.669 |
| CKD | 34 (26.4) | 18 (41.9) | 16 (18.6) | 0.005 |
| Peripheral vascular disease | 17 (13.2) | 7 (16.3) | 10 (11.6) | 0.462 |
| Stroke | 21 (16.3) | 9 (20.9) | 12 (14.0) | 0.312 |
| Asthma | 14 (10.9) | 9 (20.9) | 5 (5.8) | 0.015 |
| LVEF, % | 57 (±11.9) | 54 (±12.6) | 63 (±6.9) | 0.002 |
| Cardiac Biomarkers * | ||||
| Hs-TnT > 14 ng/L | 80 (77.6) | 27 (84.4) | 53 (74.6) | 0.273 |
| Hs-TnT, ng/L | 25.4 (14.5–47.4) | 41.6 (21.4–69) | 22.8 (14–34.2) | 0.003 |
| NT-proBNP, pg/mL | 841 (228–3785) | 3423 (616–10,400) | 558 (213–1692) | 0.002 |
| Outcomes | ||||
| Intensive care unit admission | 7 (5.4) | 1 (2.3) | 6 (7.0) | 0.423 |
| Advanced ventilatory support | 16 (12.4) | 4 (9.3) | 12 (14.0) | 0.450 |
| Hospital length of stay, days | 12 (3–24) | 17 (8–31) | 10 (1–20) | 0.023 |
| Clinical worsening during admission | 39 (30.2) | 14 (32.6) | 25 (29) | 0.749 |
| Overall death | 47 (36.4) | 22 (51.2) | 25 (29.1) | 0.014 |
| Overall CV death | 4 (3.1) | 4 (9.3) | 0 (0) | 0.019 |
| Acute HF during admission | 12 (9.3) | 9 (21) | 3 (3.5) | 0.004 |
| Total (n = 129) | CHF (n = 43) | Non-HF (n = 86) | p Value | |
|---|---|---|---|---|
| Clinical findings | ||||
| Fever | 62 (48) | 23 (53.5) | 39 (45.3) | 0.214 |
| Dyspnea with or without cough | 64 (49.6) | 27 (62.8) | 37 (43) | 0.038 |
| SBP, mmHg | 133 (±21.4) | 133 (±22.7) | 133 (±20.9) | 0.995 |
| DBP, mmHg | 74 (±15.2) | 73 (±15.7) | 74 (±15.1) | 0.786 |
| Heart rate, bpm | 88 (±16.7) | 89 (±20) | 89.5 (±19.9) | 0.564 |
| Respiratory rate, rpm | 27 (±6.8) | 27 (±7.5) | 27 (±6.5) | 0.902 |
| Oxygen saturation, % | 94 (±5.7) | 94 (±6.8) | 94 (±5) | 0.331 |
| Initial FiO2, % | 31 (±23.7) | 32 (±25) | 30 (±23.2) | 0.671 |
| PaO2/FiO2 | 271 (±127.6) | 312 (±135.4) | 256 (±121.9) | 0.080 |
| PaO2/FiO2 < 300 | 50 (38.8) | 13 (30.2) | 37 (43) | 0.387 |
| Laboratory findings * | ||||
| Hemoglobin, g/dL | 12.5 (±2.0) | 11.7 (±2.3) | 12.8 (±1.8) | 0.008 |
| WBCC, per µL | 8.3 (±5.0) | 9.3 (±6.4) | 7.8 (±4.1) | 0.177 |
| Lymphocytes, per µL | 1.3 (±1.6) | 1.5 (±2.4) | 1.2 (±1) | 0.299 |
| Platelet count | 215 (±99) | 233 (±131) | 206 (±80) | 0.243 |
| Creatinine, mg/dL | 1.3 (±0.9) | 1.6 (±1.2) | 1.2 (±0.6) | 0.024 |
| eGFR, mL/min/1.73m2 | 62 (±28) | 53 (±25.8) | 67 (±28.4) | 0.008 |
| AST, U/L | 41 (±58) | 39 (±53) | 42 (±59) | 0.870 |
| ALT, U/L | 35 (±61) | 39 (±53) | 41 (±65) | 0.611 |
| Bilirubin, mg/dL | 1.1 (±6.3) | 2.7 (±11.7) | 0.4 (±0.17) | 0.293 |
| CPK, U/L | 87 (51–194) | 59 (41.5–167.5) | 98 (58.8–209) | 0.144 |
| Serum lactate, mmol/L | 2.2 (1.1–1.8) | 1.4 (1.1–2.1) | 1.5 (1.1–1.7) | 0.840 |
| Baseline CRP, mg/dL | 9.6 (5.2–17) | 8.8 (3.9–12.7) | 10.6 (5.3–19.5) | 0.122 |
| Procalcitonin, ng/mL | 0.18 (0.1–0.4) | 0.2 (0.1–0.5) | 0.17 (0.1–0.4) | 0.675 |
| LDH, U/L | 345 (±154) | 339 (±130) | 348 (±163) | 0.811 |
| D-dimer, ng/mL | 876 (402–1440) | 880 (560–1995) | 835 (575–1535) | 0.936 |
| Abnormal chest radiography | 109 (84.5) | 35 (81.4) | 74 (86) | 1 |
| In-hospital treatment | ||||
| Lopinavir/Ritonavir | 13 (10.1) | 3 (7) | 10 (11.6) | 0.542 |
| Darunavir/Ritonavir | 2 (1.6) | 0 (0) | 13 (15.1) | 0.552 |
| Corticosteroid | 47 (36.4) | 13 (30.2) | 34 (39.5) | 0.294 |
| Tocilizumab | 13 (10.1) | 3 (7) | 10 (11.6) | 0.542 |
| Hydroxychloroquine ± Azithromycin | 114 (88.4) | 35 (81.4) | 79 (91.9) | 0.080 |
| Enoxaparin (prophylaxis or treatment doses) | 74 (57.4) | 19 (44.2) | 55 (64) | 0.136 |
| Vitamin D | 25 (19.4) | 6 (14) | 19 (22.1) | 0.270 |
| Ceftriaxone | 79 (61.2) | 21 (48.8) | 58 (67.4) | 0.041 |
| Respiratory support | ||||
| Oxygen support | 95 (73.6) | 35 (81.4) | 60 (69.8) | 0.436 |
| High Flow Nasal Cannula | 1 (0.8) | 0 (0) | 1 (1.2) | |
| Non-invasive ventilation | 8 (6.2) | 3 (7) | 5 (5.8) | |
| Intubation and invasive ventilation | 7 (5.4) | 1 (2.3) | 6 (7) | |
| Alive (n = 82) | Dead (n = 47) | p Value | |
|---|---|---|---|
| Previous CHF | 21 (25.6) | 22 (46.8) | 0.014 |
| Women | 37 (45.1) | 29 (61.7) | 0.070 |
| Age, years | 77.4 (±13.7) | 85.5 (±5.5) | 0.001 |
| Smoking | 20 (24.4) | 11 (23.4) | 0.508 |
| Diabetes | 21 (25.6) | 28 (59.6) | 0.001 |
| Hypertension | 60 (73.2) | 41 (87.2) | 0.062 |
| Dyslipemia | 40 (48.8) | 27 (57.4) | 0.343 |
| Obesity | 16 (19.5) | 13 (27.7) | 0.175 |
| Ischemic cardiac disease | 9 (11.0) | 13 (27.7) | 0.015 |
| AF or flutter | 15 (18.3) | 16 (34) | 0.044 |
| Valvular heart disease | 10 (12.2) | 9 (19.1) | 0.283 |
| ACEI or ARA II | 37 (45.1) | 21 (44.7) | 0.961 |
| COPD | 8 (9.8) | 6 (12.8) | 0.597 |
| Previous Cancer | 21 (25.6) | 12 (25.5) | 0.992 |
| Stroke | 14 (17.1) | 7 (14.9) | 0.747 |
| Asthma | 11 (13.4) | 3 (6.4) | 0.217 |
| CKD | 13 (15.9) | 21 (44.7) | 0.001 |
| Peripheral vascular disease | 7 (8.5) | 10 (21.3) | 0.040 |
| Influenza vaccination | 16 (19.5) | 11 (23.4) | 0.313 |
| Hs-TnT *, ng/L | 20.5 (14–30.5) | 44 (26–78) | 0.001 |
| NT-proBNP *, pg/mL | 482 (180–893) | 3786 (1391–10,400) | 0.001 |
| LVEF, % | 59.5 (±8.8) | 53.7 (±14.7) | 0.44 |
| Univariate HR (95%CI) | p Value | Adjusted * HR (95%CI) | p Value | |
|---|---|---|---|---|
| Previous CHF | 1.76 (1.13–2.73) | 0.014 | 2.3 (1.26–4.2) | 0.007 |
| Age (per every year) | 1.1 (1.03–1.12) | 0.002 | 1.08 (1.03–1.14) | 0.001 |
| Diabetes | 4.41 (1.52–3.82) | 0.001 | 3.04 (1.65–5.6) | 0.001 |
| Peripheral vascular disease | 1.78 (1.11–2.87) | 0.040 | 2.49 (1.17–5.3) | 0.018 |
| Ischemic cardiac disease | 1.86 (1.19–2.9) | 0.015 | - | |
| AF or flutter | 1.63 (1.04–2.55) | 0.044 | - | |
| CKD | 2.26 (1.48–3.44) | 0.001 | - |
| LVEF | |
| Preserved | 30 (69.8) |
| Mid-range | 2 (4.7) |
| Reduced | 9 (20.9) |
| NYHA | |
| I | 12 (27.9) |
| II | 21 (48.8) |
| III | 8 (18.6) |
| HF etiology | |
| Ischemic | 5 (11.6) |
| Non-ischemic | 18 (41.9) |
| Hypertensive | 17 (39.5) |
| Background medical therapy | |
| RAAS inhibition | 23 (53.5) |
| Beta-blockers | 14 (32.6) |
| ARNI | 4 (9.3) |
| MRA | 4 (9.3) |
| Loop diuretics | 17 (39.5) |
| Thiazides | 3 (7) |
| Anticoagulant | 14 (32.6) |
| Antiplatelet | 9 (20.9) |
| Statins | 15 (34.9) |
| Alive (n = 21) | Dead (n = 22) | p Value | Univariate HR (95%CI) | |
|---|---|---|---|---|
| Age (years) | 76 (±15.1) | 84.4 (±6.3) | 0.035 | 1.1 (1–1.1) |
| LVEF ≤ 40% | 1 (4.8) | 7 (31.8) | 0.047 | 1.97 (1.2–3.2) |
| AF or flutter | 7 (33.3) | 15 (68.2) | 0.022 | 2.1 (1.1–4) |
| CKD | 5 (23.8) | 15 (59.1) | 0.019 | 2 (1.1–3.6) |
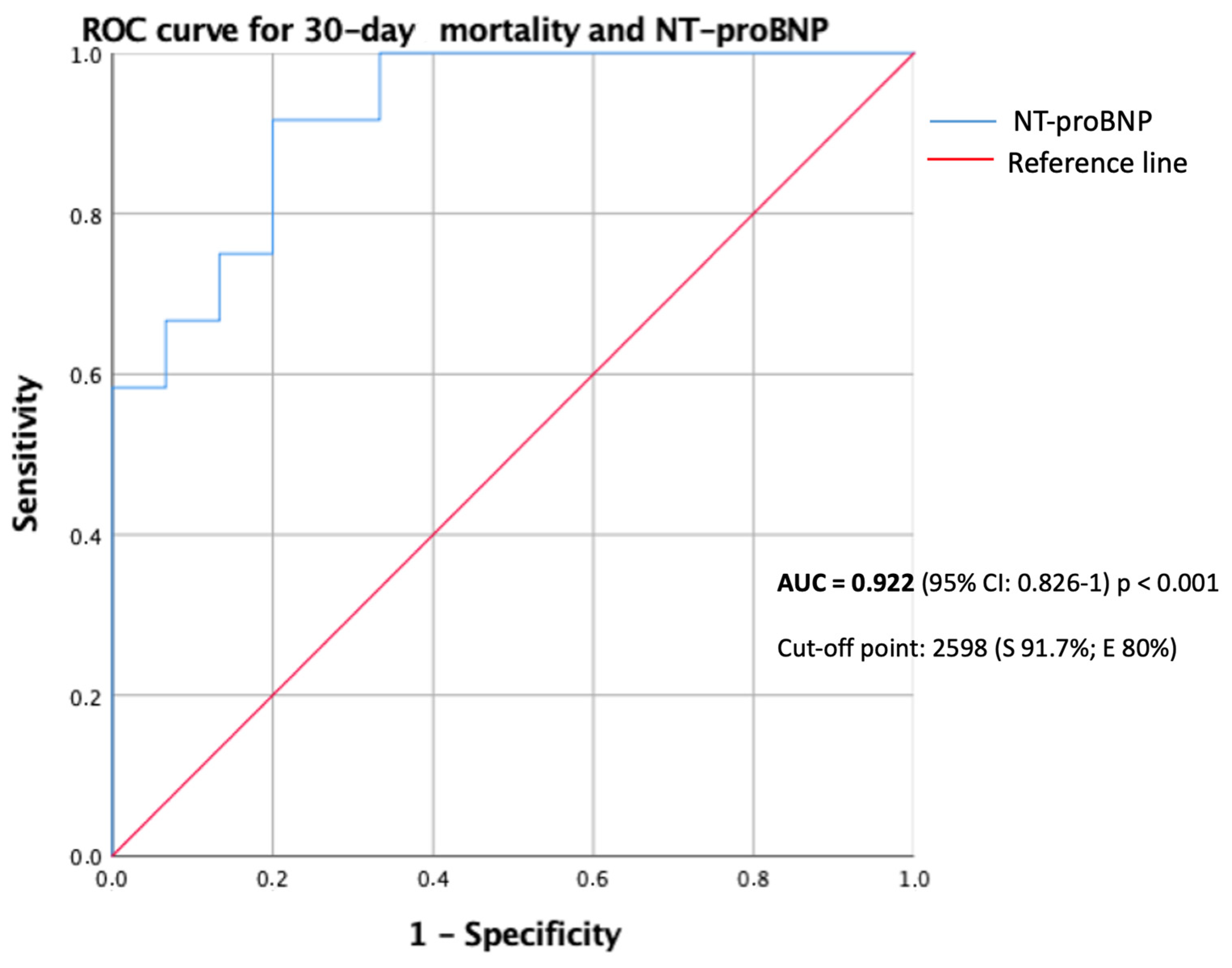
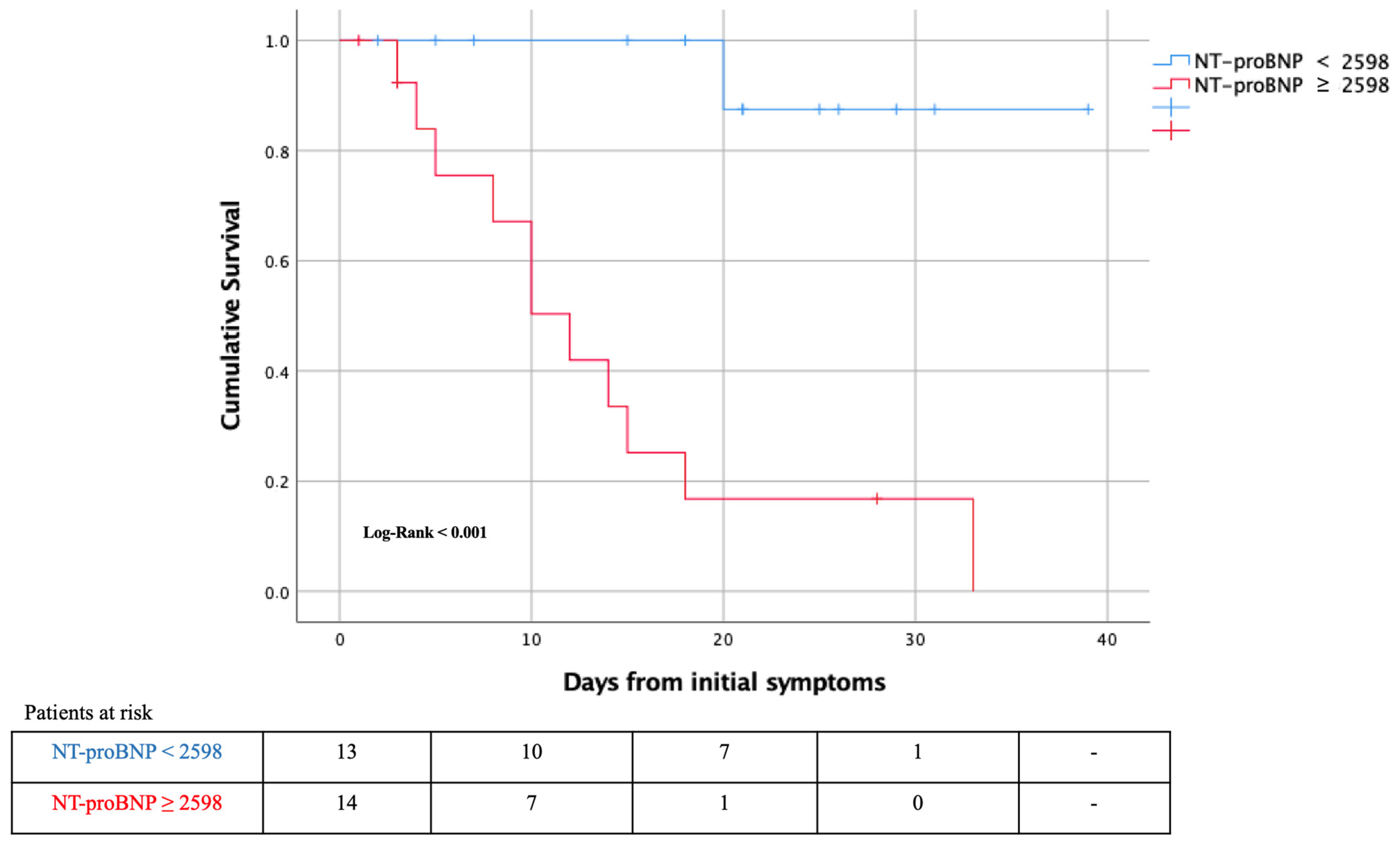
| NT-proBNP *, pg/mL | 747 (182–1773) | 10,966 (4539–16,094) | 0.003 | 1.065 † (1.02–1.11) |
| NT-proBNP * ≥ 2598 pg/mL | 3 (20) | 11 (91.7) | 0.001 | 10.2 (1.5–68.5) |
| Hs-TnT * < 14 ng/L | 5 (27.8) | 0 (0) | 0.052 | 0.48 (0.33–0.71) |
| NYHA ≥ III (advanced HF) | 4 (19) | 4 (18.1) | 0.522 | - |
| Women | 10 (47.6) | 12 (54.5) | 0.65 | - |
| Smoking | 3 (14.3) | 6 (27.2) | 0.231 | - |
| DM | 8 (38.1) | 13 (59.1) | 0.169 | - |
| Hypertension | 19 (90.5) | 19 (86.4) | 1 | - |
| Dyslipemia | 11 (52.4) | 16 (72.7) | 0.168 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belarte-Tornero, L.C.; Valdivielso-Moré, S.; Vicente Elcano, M.; Solé-González, E.; Ruíz-Bustillo, S.; Calvo-Fernández, A.; Subinara, I.; Cabero, P.; Soler, C.; Cubero-Gallego, H.; et al. Prognostic Implications of Chronic Heart Failure and Utility of NT-proBNP Levels in Heart Failure Patients with SARS-CoV-2 Infection. J. Clin. Med. 2021, 10, 323. https://doi.org/10.3390/jcm10020323
Belarte-Tornero LC, Valdivielso-Moré S, Vicente Elcano M, Solé-González E, Ruíz-Bustillo S, Calvo-Fernández A, Subinara I, Cabero P, Soler C, Cubero-Gallego H, et al. Prognostic Implications of Chronic Heart Failure and Utility of NT-proBNP Levels in Heart Failure Patients with SARS-CoV-2 Infection. Journal of Clinical Medicine. 2021; 10(2):323. https://doi.org/10.3390/jcm10020323
Chicago/Turabian StyleBelarte-Tornero, Laia C., Sandra Valdivielso-Moré, Miren Vicente Elcano, Eduard Solé-González, Sonia Ruíz-Bustillo, Alicia Calvo-Fernández, Isaac Subinara, Paula Cabero, Cristina Soler, Héctor Cubero-Gallego, and et al. 2021. "Prognostic Implications of Chronic Heart Failure and Utility of NT-proBNP Levels in Heart Failure Patients with SARS-CoV-2 Infection" Journal of Clinical Medicine 10, no. 2: 323. https://doi.org/10.3390/jcm10020323
APA StyleBelarte-Tornero, L. C., Valdivielso-Moré, S., Vicente Elcano, M., Solé-González, E., Ruíz-Bustillo, S., Calvo-Fernández, A., Subinara, I., Cabero, P., Soler, C., Cubero-Gallego, H., Vaquerizo, B., & Farré, N. (2021). Prognostic Implications of Chronic Heart Failure and Utility of NT-proBNP Levels in Heart Failure Patients with SARS-CoV-2 Infection. Journal of Clinical Medicine, 10(2), 323. https://doi.org/10.3390/jcm10020323





