Real-World Data on Bleeding Patterns of Hemophilia A Patients Treated with Emicizumab
Abstract
:1. Introduction
2. Patients and Methods
Statistical Analysis
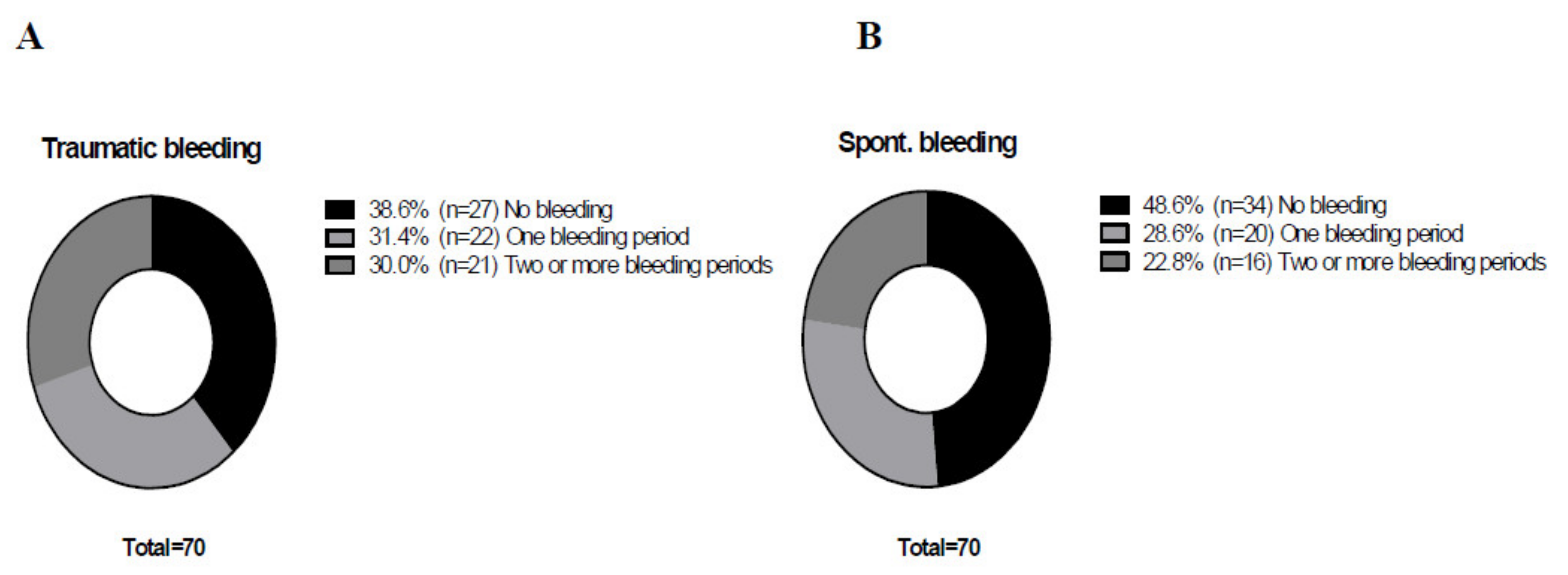
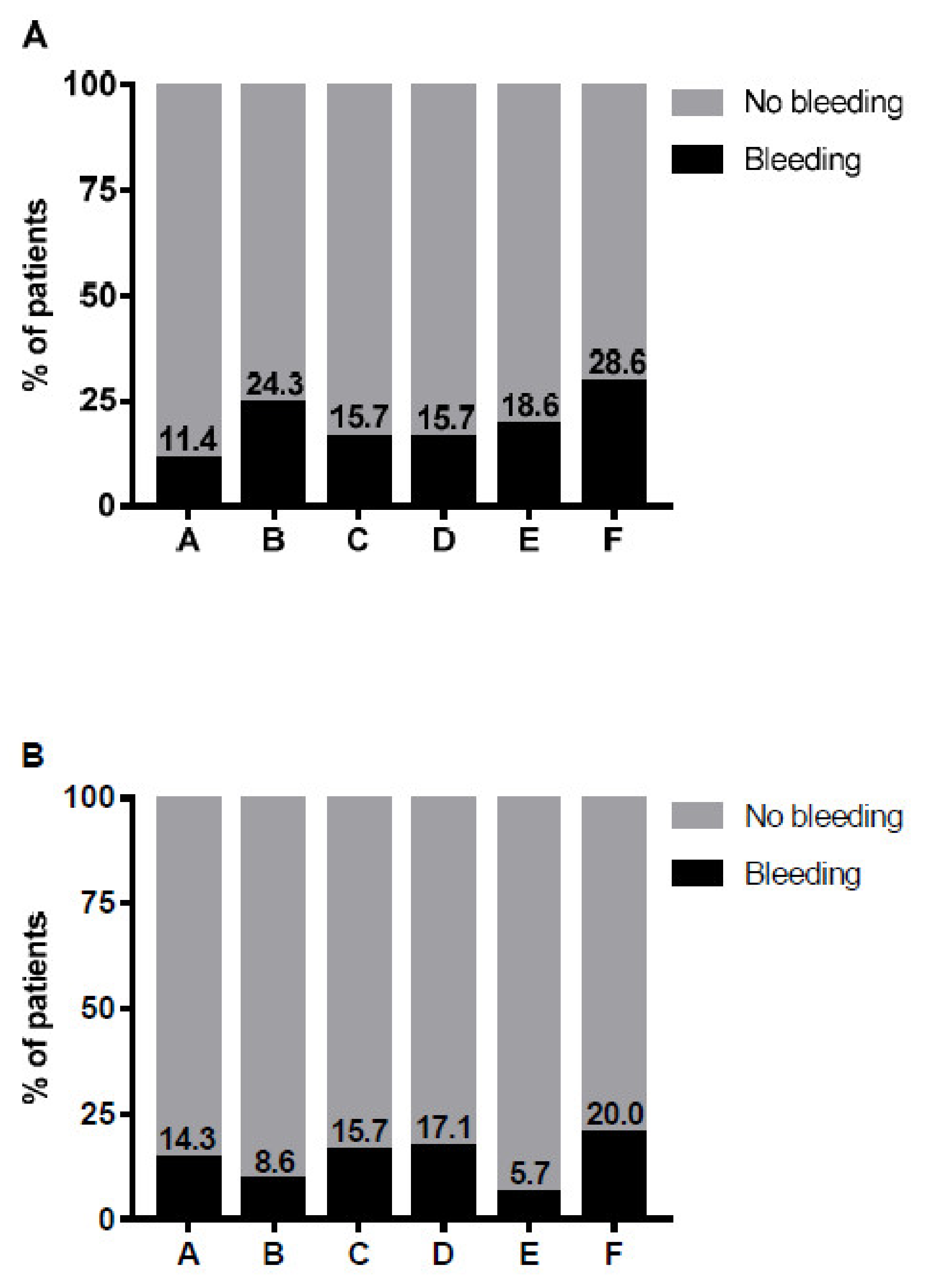
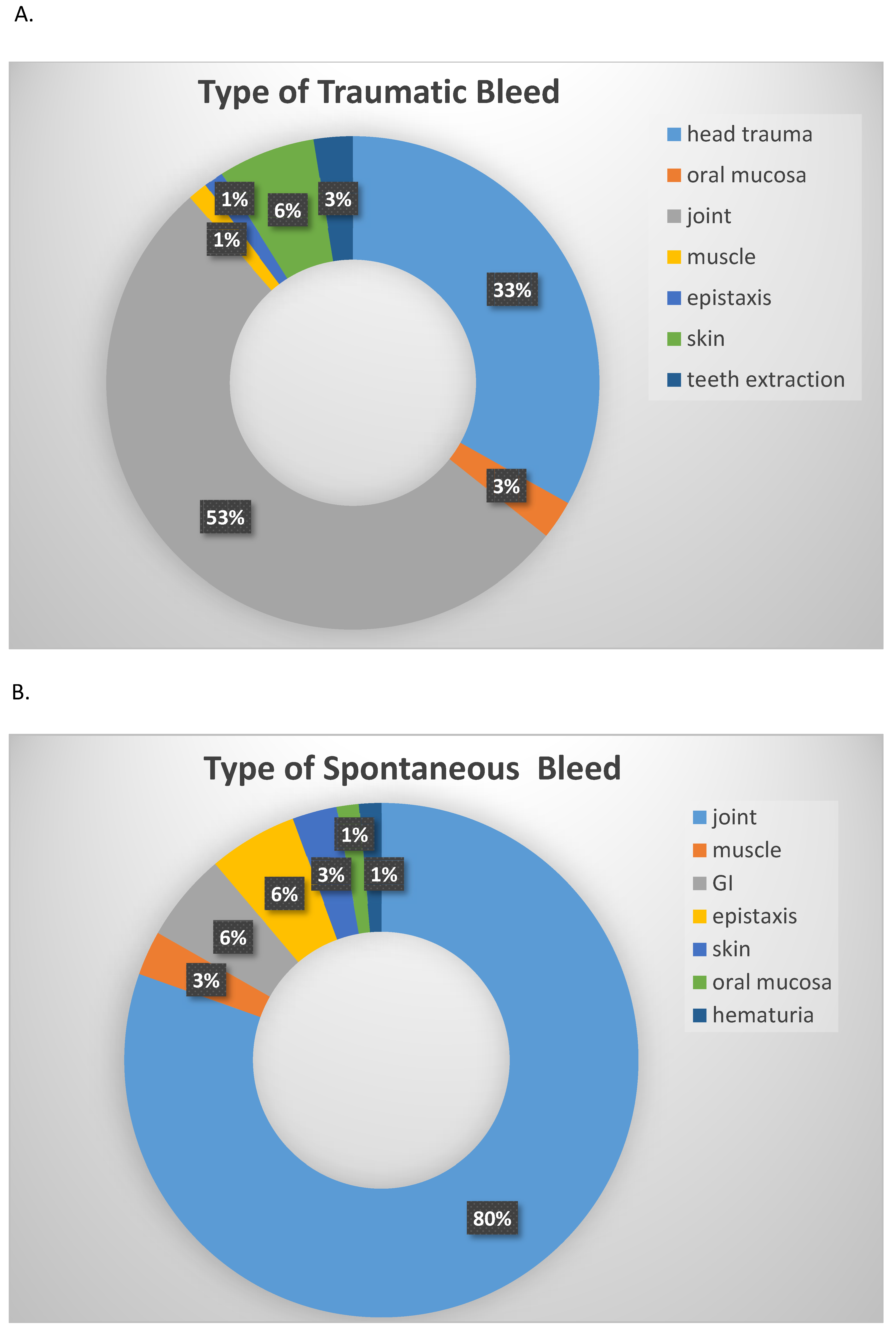
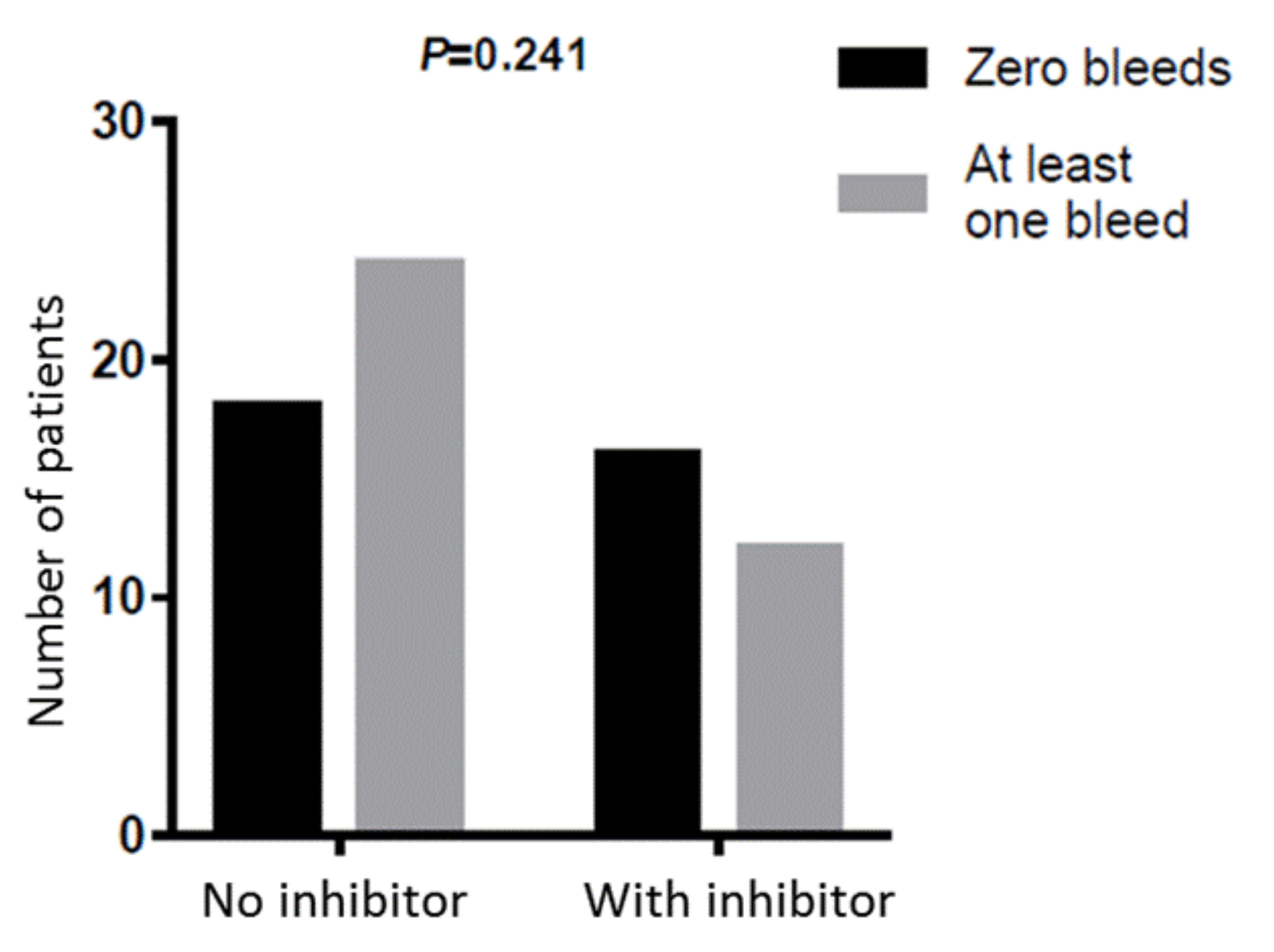
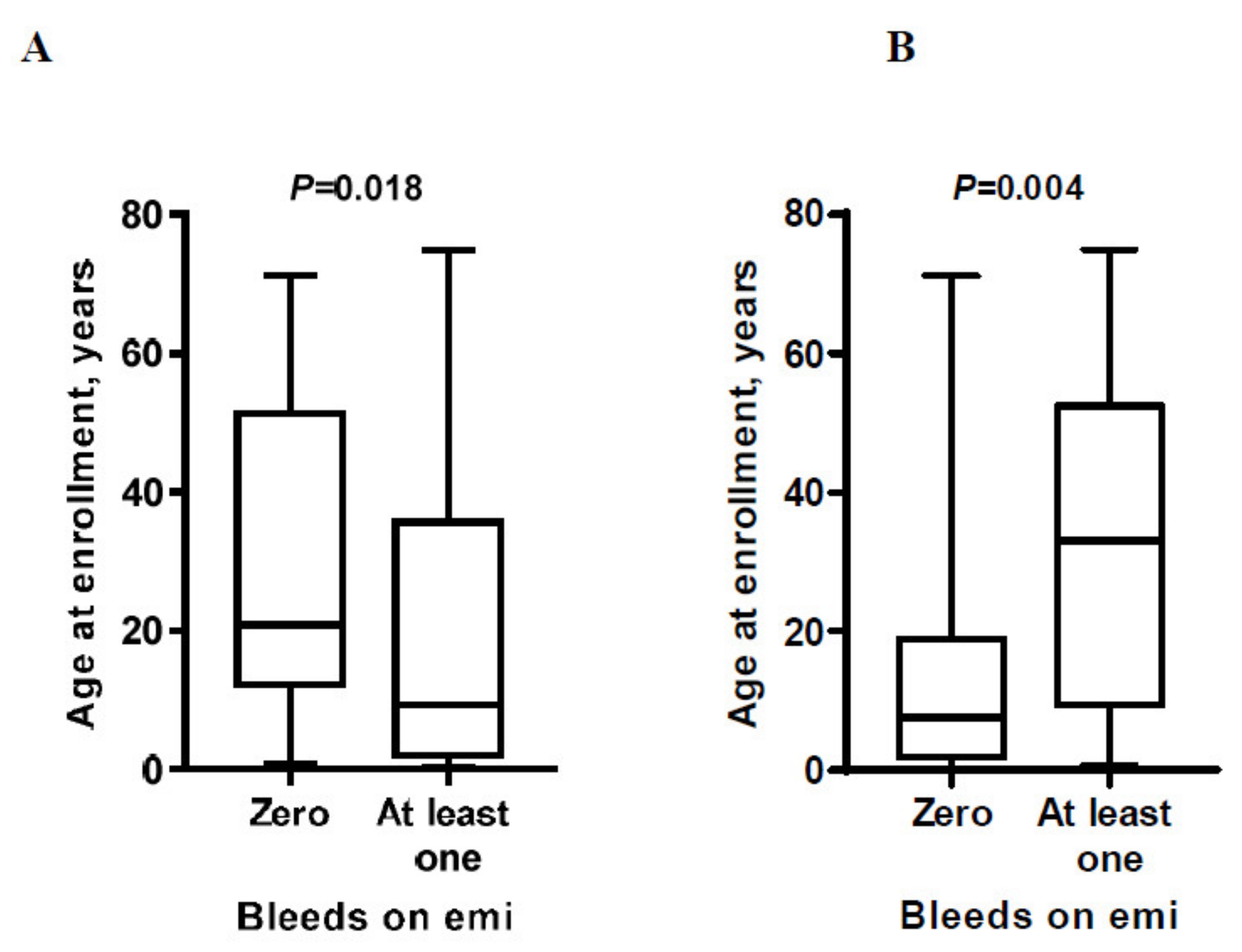
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Manco-Johnson, M.J.; Soucie, J.M.; Gill, J.C.; Joint Outcomes Committee of the Universal Data Collection; US Hemophilia Treatment Center Network. Prophylaxis usage, bleeding rates, and joint outcomes of Hemophilia, 1999 to 2010: A surveillance project. Blood 2017, 129, 2368–2374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soucie, J.M.; Miller, C.H.; Dupervil, B.; Le, B.; Buckner, T.W. Occurrence rates of haemophilia among males in the United States based on surveillance conducted in specialized haemophilia treatment centres. Haemophilia 2020, 26, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.; Adamkewicz, J.I.; Xu, J.; Petry, C.; Catalani, O.; Young, G.; Negrier, C.; Callaghan, M.U.; Levy, G.G. Pharmacokinetics and Pharmacodynamics of Emicizumab in Persons with Hemophilia A with Factor VIII Inhibitors: HAVEN 1 Study. Thromb. Haemost. 2020, 121, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Kiialainen, A.; Petry, C.; Jimenez-Yuste, V.; Santagostino, E.; Ozelo, M.; Meier, O.; Robson, S.; Castaman, G.; Klamroth, R.; Schmitt, C. Pharmacokinetics and Coagulation Biomarkers in Persons with Hemophilia A (PwHA) and FVIII Inhibitors Receiving Emicizumab in the Phase IIIb STASEY Study. Res. Pract. Thromb. Haemost. 2020, 4 (Suppl. 1), PB0942. [Google Scholar]
- Lenting, P.J.; Denis, C.V.; Christophe, O.D. Emicizumab, a bispecific antibody recognizing coagulation factors IX and X: How does it actually compare to factor VIII? Blood 2017, 130, 2463–2468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gringeri, A.; Lundin, B.; Von Mackensen, S.; Mantovani, L.; Mannucci, P.M.; ESPRIT Study Group. A randomized clinical trial of prophylaxis in children with hemophilia A (the ESPRIT Study). J. Thromb. Haemost. 2011, 9, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Shima, M.; Hanabusa, H.; Taki, M.; Matsushita, T.; Sato, T.; Fukutake, K.; Fukazawa, N.; Yoneyama, K.; Yoshida, H.; Nogami, K. Factor VIII-Mimetic Function of Humanized Bispecific Antibody in Hemophilia A. N. Engl. J. Med. 2016, 374, 2044–2053. [Google Scholar] [CrossRef]
- Oldenburg, J.; Mahlangu, J.N.; Kim, B.; Schmitt, C.; Callaghan, M.U.; Young, G.; Santagostino, E.; Kruse-Jarres, R.; Negrier, C.; Kessler, C.; et al. Emicizumab prophylaxis in hemophilia A with inhibitors. N. Engl. J. Med. 2017, 377, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Young, G.; Liesner, R.; Chang, T.; Sidonio, R.; Oldenburg, J.; Jiménez-Yuste, V.; Mahlangu, J.; Kruse-Jarres, R.; Wang, M.; Uguen, M.; et al. A multicenter, open-label phase 3 study of Emicizumab prophylaxis in children with hemophilia A with inhibitors. Blood 2019, 134, 2127–2138. [Google Scholar] [CrossRef] [PubMed]
- Pipe, S.W.; Shima, M.; Lehle, M.; Shapiro, A.; Chebon, S.; Fukutake, K.; Key, N.S.; Portron, A.; Schmitt, C.; Podolak-Dawidziak, M.; et al. Efficacy, safety, and pharmacokinetics of emicizumab prophylaxis given every 4 weeks in people with haemophilia A (HAVEN 4): A multicentre, open-label, non-randomised phase 3 study. Lancet Haematol. 2019, 6, e295–e305. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. FDA Approves Emicizumab-Kxwh for Hemophilia A with or without Factor VIII Inhibitors. Available online: www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm622564.htm (accessed on 4 October 2018).
- Callaghan, M.U.; Negrier, C.; Paz-Priel, I.; Chang, T.; Chebon, S.; Lehle, M.; Mahlangu, J.; Young, G.; Kruse-Jarres, R.; Mancuso, M.E.; et al. Long-term outcomes with emicizumab prophylaxis for hemophilia A with or without FVIII inhibitors from the HAVEN 1–4 studies. Blood 2021, 137, 2231–2242. [Google Scholar] [CrossRef] [PubMed]
- Barg, A.A.; Avishai, E.; Budnik, I.; Levy-Mendelovich, S.; Brutman-Barazani, T.; Kenet, G.; Livnat, T. Emicizumab prophylaxis among infants and toddlers with severe hemophilia A and inhibitors—A single-center cohort. Pediatric Blood Cancer 2019, 66, e27886. [Google Scholar] [CrossRef] [PubMed]
- Barg, A.A.; Livnat, T.; Budnik, I.; Avishai, E.; Brutman-Barazani, T.; Tamarin, I.; Bashari, D.; Misgav, M.; Kenet, G. Emicizumab treatment and monitoring in a paediatriccohort: Real-world data. Br. J. Haematol. 2020, 191, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Misgav, M.; Brutman-Barazani, T.; Budnik, I.; Avishai, E.; Schapiro, J.; Bashari, D.; Barg, A.A.; Lubetsky, A.; Livnat, T.; Kenet, G. Emicizumab prophylaxis in hemophilia patients older than 50 years with cardiovascular risk factors—Real world data. Haemophilia 2021, 27, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Mahlangu, J.; Oldenburg, J.; Paz-Priel, I.; Negrier, C.; Niggli, M.; Mancuso, M.E.; Schmitt, C.; Jiménez-Yuste, V.; Kempton, C.; Dhalluin, C.; et al. Emicizumab Prophylaxis in Patients Who Have Hemophilia A without Inhibitors. N. Engl. J. Med. 2018, 379, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Ebbert, P.T.; Xavier, F.; Seaman, C.D.; Ragni, M.V. Emicizumab prophylaxis in patients with haemophilia A with and without inhibitors. Haemophilia 2019, 26, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Shima, M.; Nogami, K.; Nagami, S.; Yoshida, S.; Yoneyama, K.; Ishiguro, A.; Suzuki, T.; Taki, M. A multicentre, open-label study of Emicizumab given every 2 or 4 weeks in children with severe haemophilia A without inhibitors. Haemophilia 2019, 25, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Hanley, J.; McKernan, A.; Creagh, M.D.; Classey, S.; McLaughlin, P.; Goddard, N.; Briggs, P.J.; Frostick, S.; Giangrande, P.; Wilde, J.; et al. Musculoskeletal Working Party of the UKHCDO. Guidelines for the management of acute joint bleeds and chronic synovitis in haemophilia: A United Kingdom Haemophilia Centre Doctors’ Organisation (UKHCDO) guideline. Haemophilia 2017, 23, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Wyseure, T.; Mosnier, L.O.; Von Drygalski, A. Advances and challenges in hemophilic arthropathy. Semin. Hematol. 2016, 53, 10–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Hara, J.; Walsh, S.; Camp, C.; Mazza, G.; Carroll, L.; Hoxer, C.; Wilkinson, L. The relationship between target joints and direct resource use in severe haemophilia. Health Econ. Rev. 2018, 8, 1. [Google Scholar] [CrossRef] [PubMed]
| without FVIII Inhibitors (n = 42) | with FVIII Inhibitors (n = 28) | P Value | |
|---|---|---|---|
| Age (years) | 17.2 (9.2–45.9) | 5.6 (1.0–34.5) | 0.009 a |
| Prior prophylaxis | 34 (81%) | 14 (50%) | 0.006 b |
| ITI | - | 10 (36%) | NA |
| Inhibitor (BU) | - | 12.0 (3.6–19.8) | NA |
| History of FVIII inhibitors | 7 (17%) | - | NA |
| ABR | 4 (1–12) | 6 (3–10) | 0.152 a |
| Independent Variable | B | SE of B | P Value | Odds Ratio | 95% CI for Odds Ratio |
|---|---|---|---|---|---|
| Age, years | 0.028 | 0.013 | 0.034 | 1.029 | 1.002–1.056 |
| ABR | 0.021 | 0.029 | 0.462 | 1.021 | 0.966–1.080 |
| Presence of FVIII inhibitors | −0.358 | 0.529 | 0.499 | 0.699 | 0.248–1.971 |
| Constant | −0.613 | 0.457 | 0.179 | 0.542 | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levy-Mendelovich, S.; Brutman-Barazani, T.; Budnik, I.; Avishai, E.; Barg, A.A.; Levy, T.; Misgav, M.; Livnat, T.; Kenet, G. Real-World Data on Bleeding Patterns of Hemophilia A Patients Treated with Emicizumab. J. Clin. Med. 2021, 10, 4303. https://doi.org/10.3390/jcm10194303
Levy-Mendelovich S, Brutman-Barazani T, Budnik I, Avishai E, Barg AA, Levy T, Misgav M, Livnat T, Kenet G. Real-World Data on Bleeding Patterns of Hemophilia A Patients Treated with Emicizumab. Journal of Clinical Medicine. 2021; 10(19):4303. https://doi.org/10.3390/jcm10194303
Chicago/Turabian StyleLevy-Mendelovich, Sarina, Tami Brutman-Barazani, Ivan Budnik, Einat Avishai, Assaf A. Barg, Tamara Levy, Mudi Misgav, Tami Livnat, and Gili Kenet. 2021. "Real-World Data on Bleeding Patterns of Hemophilia A Patients Treated with Emicizumab" Journal of Clinical Medicine 10, no. 19: 4303. https://doi.org/10.3390/jcm10194303
APA StyleLevy-Mendelovich, S., Brutman-Barazani, T., Budnik, I., Avishai, E., Barg, A. A., Levy, T., Misgav, M., Livnat, T., & Kenet, G. (2021). Real-World Data on Bleeding Patterns of Hemophilia A Patients Treated with Emicizumab. Journal of Clinical Medicine, 10(19), 4303. https://doi.org/10.3390/jcm10194303







