Continuous Remote Patient Monitoring Shows Early Cardiovascular Changes in COVID-19 Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion and Exclusion Criteria
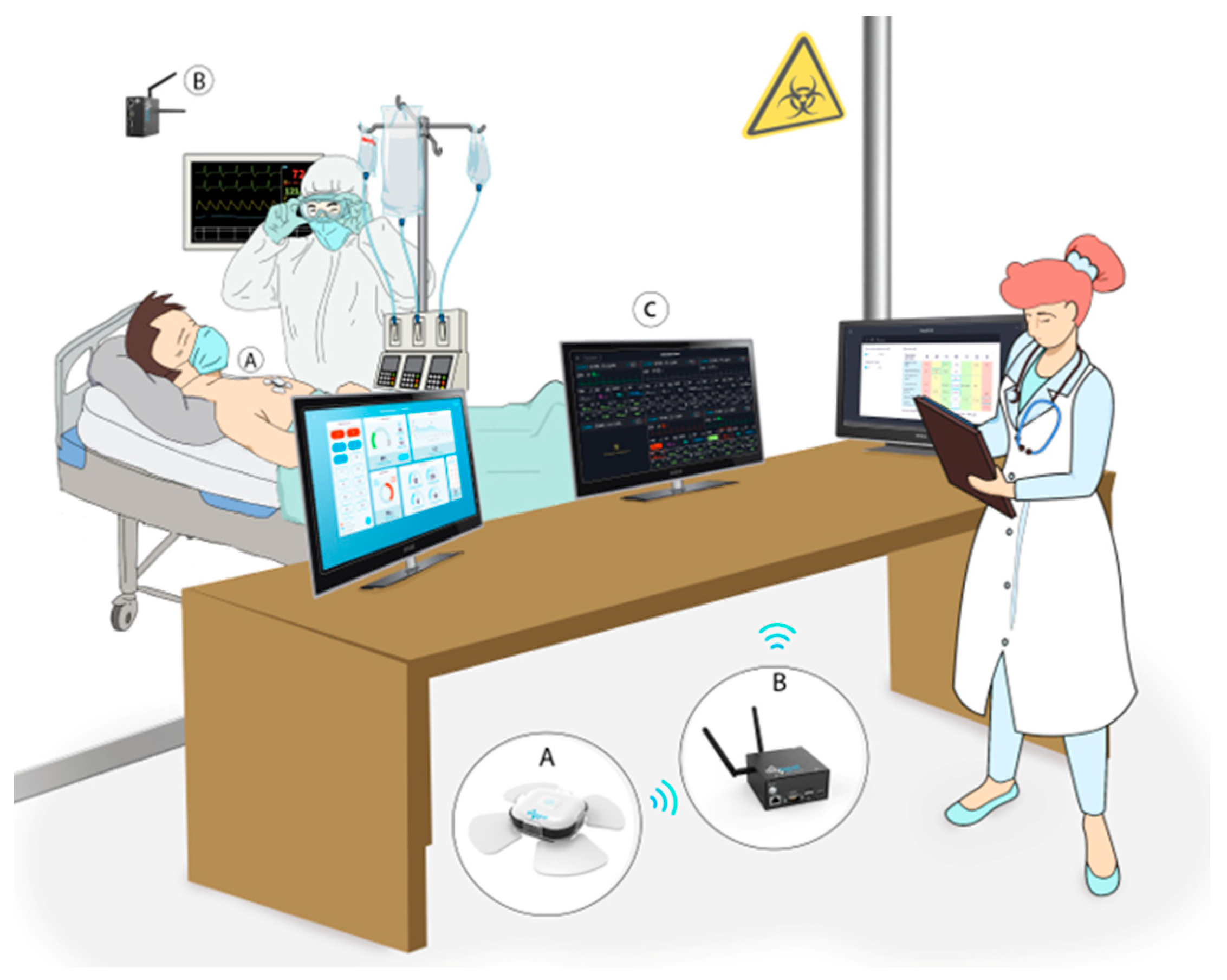
2.3. The Remote Patient Monitoring (RPM) System
2.4. Ethical Approval
2.5. Statistical Analysis
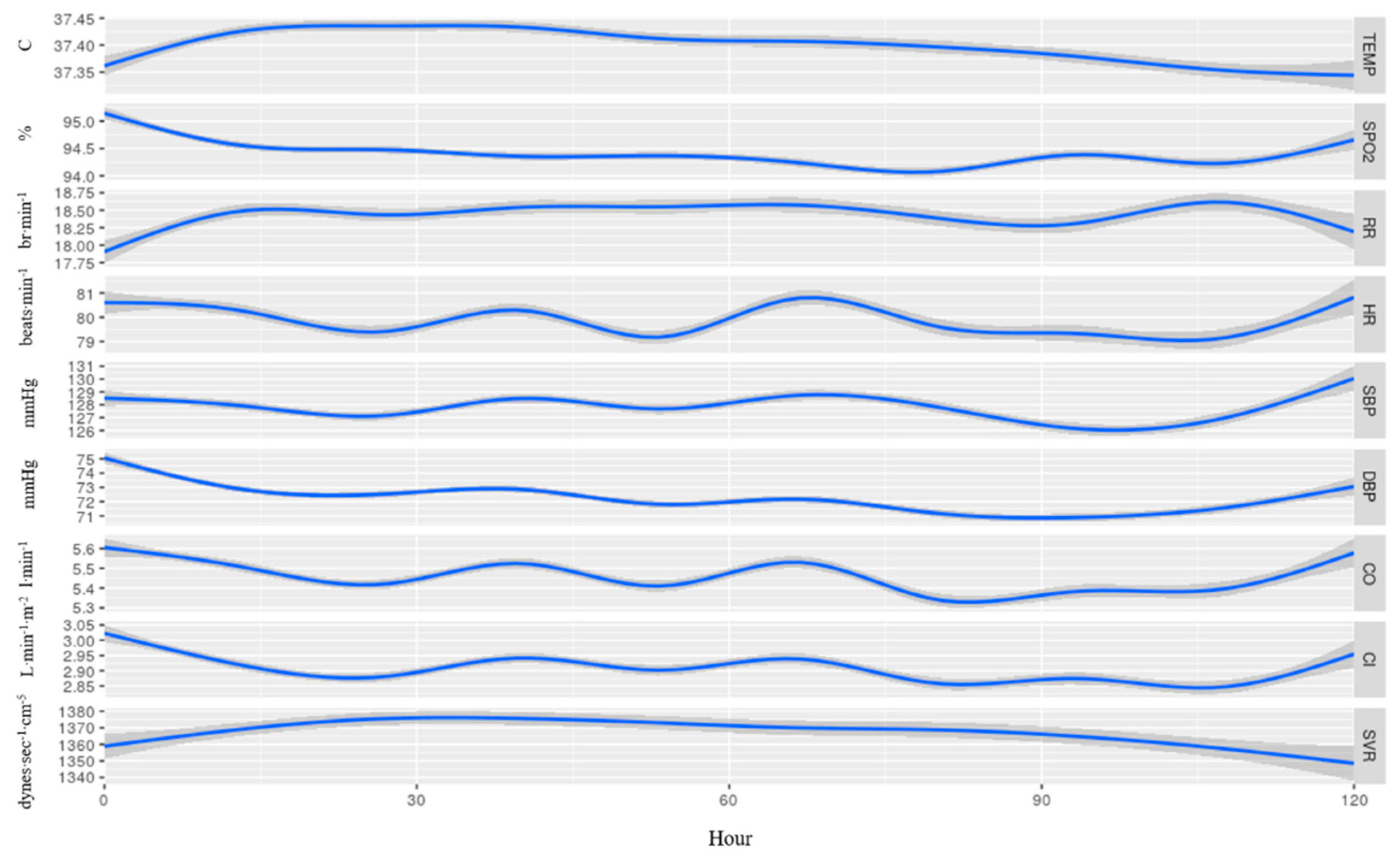
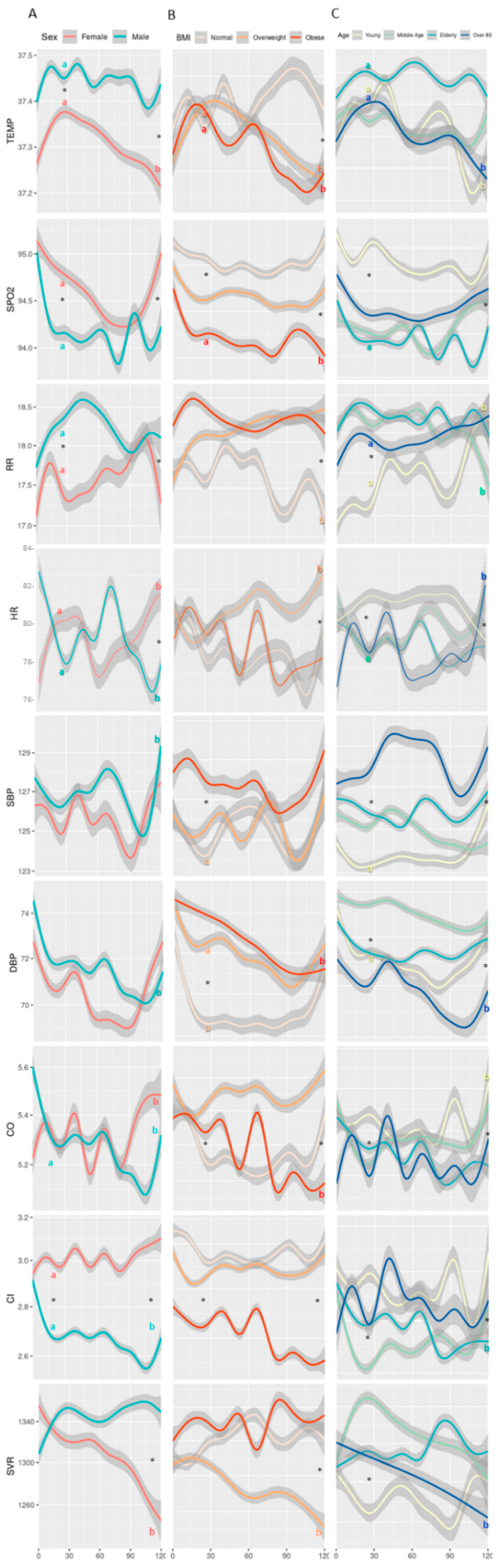
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Tikellis, C.; Thomas, M.C. Angiotensin-converting enzyme 2 (ACE2) is a key modulator of the renin angiotensin system in health and disease. Int. J. Pept. 2012, 2012, 256294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Penninger, J.M.; Li, Y.; Zhong, N.; Slutsky, A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020, 46, 586–590. [Google Scholar] [CrossRef] [Green Version]
- Bhatraju, P.K.; Ghassemieh, B.J.; Nichols, M.; Kim, R.; Jerome, K.R.; Nalla, A.K.; Greninger, A.L.; Pipavath, S.; Wurfel, M.M.; Evans, L.; et al. Covid-19 in critically ill patients in the Seattle region—Case series. N. Engl. J. Med. 2020, 382, 2012–2022. [Google Scholar] [CrossRef]
- Keesara, S.; Jonas, A.; Schulman, K. Covid-19 and health care’s digital revolution. N. Engl. J. Med. 2020, 382, e82. [Google Scholar]
- Russo, V.; Bottino, R.; Carbone, A.; Rago, A.; Papa, A.A.; Golino, P.; Nigro, G. COVID-19 and Heart: From Clinical Features to Pharmacological Implications. J. Clin. Med. 2020, 9, 1944. [Google Scholar] [CrossRef] [PubMed]
- Berlin, D.A.; Gulick, R.M.; Martinez, F.J. Severe Covid-19. N. Engl. J. Med. 2020, 383, 2451–2460. [Google Scholar] [CrossRef] [PubMed]
- Posthuma, L.M.; Downey, C.; Visscher, M.J.; Ghazali, D.A.; Joshi, M.; Ashrafian, H.; Khan, S.; Darzi, A.; Goldstone, J.; Preckel, B. Remote wireless vital signs monitoring on the ward for early detection of deteriorating patients: A case series. Int. J. Nurs. Stud. 2020, 104, 103515. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, D.R.; Davies, E.V.; Harlow, E.R.; Hsu, J.J.; Knighton, S.C.; Walker, T.A.; Voos, J.E.; Drummond, C.K. Wearable Sensors for COVID-19: A Call to Action to Harness Our Digital Infrastructure for Remote Patient Monitoring and Virtual Assessments. Front. Digit. Health 2020, 2, 8. [Google Scholar] [CrossRef]
- Russo, V.; Cassini, R.; Caso, V.; Donno, C.; Laezza, A.; Naddei, M.; Fiorelli, A.; Golino, P.; Nigro, G. Nursing Teleconsultation for the Outpatient Management of Patients with Cardiovascular Disease during COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2021, 18, 2087. [Google Scholar] [CrossRef]
- Russo, V.; Nigro, G.; D’Onofrio, A. COVID-19 and cardiac implantable electronic device remote monitoring: Crocodile tears or new opportunity? Expert Rev. Med. Devices 2020, 17, 471–472. [Google Scholar] [CrossRef]
- Magnocavallo, M.; Bernardini, A.; Mariani, M.V.; Piro, A.; Marini, M.; Nicosia, A.; Adduci, C.; Rapacciuolo, A.; Saporito, D.; Grossi, S.; et al. Home delivery of the communicator for remote monitoring of cardiac implantable devices: A multicenter experience during the covid-19 lockdown. Pacing Clin. Electrophysiol. 2021, 44, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Radin, J.M.; Quer, G.; Jalili, M.; Hamideh, D.; Steinhubl, S.R. The hopes and hazards of using personal health technologies in the diagnosis and prognosis of infections. Lancet Digit. Health 2021, 3, e455–e461. [Google Scholar] [CrossRef]
- Watkinson, P.J.; Barber, V.S.; Price, J.D.; Hann, A.; Tarassenko, L.; Young, J.D. A randomised controlled trial of the effect of continuous electronic physiological monitoring on the adverse event rate in high risk medical and surgical patients. Anaesthesia 2006, 61, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Churpek, M.M.; Adhikari, R.; Edelson, D.P. The value of vital sign trends for detecting clinical deterioration on the wards. Resuscitation 2016, 102, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taenzer, A.H.; Perreard, I.M.; MacKenzie, T.; McGrath, S.P. Characteristics of desaturation and respiratory rate in postoperative patients breathing room air versus supplemental oxygen: Are they different? Anesth. Analg. 2018, 126, 826–832. [Google Scholar] [CrossRef]
- Lonini, L.; Shawen, N.; Botonis, O.; Fanton, M.; Jayaraman, C.; Mummidisetty, C.K.; Shin, S.Y.; Rushin, C.; Jenz, S.; Xu, S.; et al. Rapid Screening of Physiological Changes Associated With COVID-19 Using Soft-Wearables and Structured Activities: A Pilot Study. IEEE J. Transl. Eng. Health Med. 2021, 9, 1–11. [Google Scholar] [CrossRef]
- Russo, V.; Rapacciuolo, A.; Pafundi, P.C.; de Divitiis, M.; Volpicelli, M.; Ruocco, A.; Rago, A.; Uran, C.; Nappi, F.; Attena, E.; et al. Cardiac implantable electronic devices replacements in patients followed by remote monitoring during COVID-19 lockdown. Eur. Heart J. Digit. Health 2021, 2, 171–174. [Google Scholar] [CrossRef]
- Chinnadurai, R.; Ogedengbe, O.; Agarwal, P.; Money-Coomes, S.; Abdurrahman, A.Z.; Mohammed, S.; Kalra, P.A.; Rothwell, N.; Pradhan, S. Older age and frailty are the chief predictors of mortality in COVID-19 patients admitted to an acute medical unit in a secondary care setting-a cohort study. BMC Geriatr. 2020, 20, 409. [Google Scholar]
- Hussain, A.; Mahawar, K.; Xia, Z.; Yang, W.; Shamsi, E.-H. Obesity and mortality of COVID-19. Meta-analysis. Obes. Res. Clin. Pract. 2020, 14, 295–300. [Google Scholar] [CrossRef]
- Popkin, B.M.; Du, S.; Green, W.D.; Beck, M.A.; Algaith, T.; Herbst, C.H.; Alsukait, R.F.; Alluhidan, M.; Alazemi, N.; Shekar, M. Individuals with obesity and COVID-19: A global perspective on the epidemiology and biological relationships. Obes. Rev. 2020, 21, e13128. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A.; Olsson, P.-E. Sex differences in severity and mortality from COVID-19: Are males more vulnerable? Biol. Sex. Differ. 2020, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Nachman, D.; Constantini, K.; Poris, G.; Wagnert-Avraham, L.; Gertz, D.S.; Littman, R.; Eisenkraft, A.; Gepner, Y. Wireless, Non-Invasive, Wearable Device for Continuous Remote Monitoring of Hemodynamic Parameters in a Swine Model of Controlled Hemorrhagic Shock. Sci. Rep. 2020, (in press). [Google Scholar] [CrossRef] [PubMed]
- Nachman, D.; Gepner, Y.; Goldstein, N.; Kabakov, E.; Ishay, A.B.; Littman, R.; Azmon, Y.; Jaffe, E.; Eisenkraft, A. Comparing blood pressure measurements between a photoplethysmography-based and a standard cuff-based manometry device. Sci. Rep. 2020, 10, 16116. [Google Scholar] [CrossRef]
- Nachman, D.; Gilan, A.; Goldstein, N.; Constantini, K.; Littman, R.; Eisenkraft, A.; Grossman, E.; Gepner, Y. Twenty-Four-Hour Ambulatory Blood Pressure Measurement Using a Novel Noninvasive, Cuffless, Wireless Device. Am. J. Hypertens. 2021, hpab095. [Google Scholar] [CrossRef]
- Kachel, E.; Constantini, K.; Nachman, D.; Carasso, S.; Littman, R.; Eisenkraft, A.; Gepner, Y. A Pilot Study of Blood Pressure Monitoring After Cardiac Surgery Using a Wearable, Non-invasive Sensor. Front. Med. 2021, 8, 693926. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014.
- Chau, N.V.V.; Lam, V.T.; Dung, N.T.; Yen, L.M.; Minh, N.N.Q.; Ngoc, N.M.; Dung, N.T.; Man, D.N.H.; Nguyet, L.A.; Ny, N.T.H. The natural history and transmission potential of asymptomatic SARS-CoV-2 infection. medRxiv 2020. [Google Scholar] [CrossRef]
- Kim, J.; Volodarskiy, A.; Sultana, R.; Pollie, M.P.; Yum, B.; Nambiar, L.; Tafreshi, R.; Mitlak, H.W.; RoyChoudhury, A.; Horn, E.M.; et al. Prognostic utility of right ventricular remodeling over conventional risk stratification in patients with COVID-19. J. Am. Coll. Cardiol. 2020, 76, 1965–1977. [Google Scholar] [CrossRef]
- Pellikka, P.A.; Naqvi, T.Z. The Right Ventricle: A Target in COVID-19 Cardiac Insult; American College of Cardiology Foundation: Washington, DC, USA, 2020. [Google Scholar]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 811–818. [Google Scholar] [CrossRef] [Green Version]
- Lindner, D.; Fitzek, A.; Bräuninger, H.; Aleshcheva, G.; Edler, C.; Meissner, K.; Scherschel, K.; Kirchhof, P.; Escher, F.; Schultheiss, H.-P. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020, 5, 1281–1285. [Google Scholar] [CrossRef]
- Zheng, Y.-Y.; Ma, Y.-T.; Zhang, J.-Y.; Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020, 17, 259–260. [Google Scholar]
- Puntmann, V.O.; Carerj, M.L.; Wieters, I.; Fahim, M.; Arendt, C.; Hoffmann, J.; Shchendrygina, A.; Escher, F.; Vasa-Nicotera, M.; Zeiher, A.M. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 1265. [Google Scholar] [CrossRef] [PubMed]
- Giustino, G.; Pinney, S.P.; Lala, A.; Reddy, V.Y.; Johnston-Cox, H.A.; Mechanick, J.I.; Halperin, J.L.; Fuster, V. Coronavirus and Cardiovascular Disease, Myocardial Injury, and Arrhythmia. J. Am. Coll. Cardiol. 2020, 76, 2011–2023. [Google Scholar] [CrossRef] [PubMed]
- Jing, Q.-L.; Liu, M.-J.; Zhang, Z.-B.; Fang, L.-Q.; Yuan, J.; Zhang, A.-R.; Dean, N.E.; Luo, L.; Ma, M.-M.; Longini, I. Household secondary attack rate of COVID-19 and associated determinants in Guangzhou, China: A retrospective cohort study. Lancet Infect. Dis. 2020, 20, 1141–1150. [Google Scholar] [CrossRef]
- Quer, G.; Radin, J.M.; Gadaleta, M.; Baca-Motes, K.; Ariniello, L.; Ramos, E.; Kheterpal, V.; Topol, E.J.; Steinhubl, S.R. Wearable sensor data and self-reported symptoms for COVID-19 detection. Nat. Med. 2020, 27, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, L.; Wymant, C.; Kendall, M.; Zhao, L.; Nurtay, A.; Abeler-Dörner, L.; Parker, M.; Bonsall, D.; Fraser, C. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science 2020, 368, eabb6936. [Google Scholar] [CrossRef] [PubMed]
| Variable | Sex | Age | BMI | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Young (18–40 Years) | Middle Age (40–60 Years) | Elderly (60–80 Years) | >80 Years | p | Normal <24.9 kg∙min−2 | Overweight 25.0–29.9 kg∙min−2 | Obese >30.0 kg∙min−2 | p | |
| n | 313 | 179 | 131 | 128 | 171 | 62 | 198 | 171 | 123 | ||
| Sex M: F | 84:47 | 89:39 | 113:58 | 27:35 | 111:87 | 122:49 | 80:43 | ||||
| Monitoring period (hours) | 95.4 ± 67.6 | 83.5 ± 62.6 | 64.9 ± 43.7 | 87.8 ± 49.8 | 101.5 ± 69.3 | 120.7 ± 97.9 | 86.6 ± 69.1 | 98.1 ± 64.1 | 88.0 ± 63.2 | ||
| Age (years) | 56 ± 17 | 60 ±22 | 31 ± 6 b,c,d | 53 ± 5 a,c,d | 69 ± 6 a,b,d | 87 ± 5 a,b,c | * | 53 ± 20 &,^ | 61 ± 18 # | 59 ± 17 # | * |
| Weight (kg) | 83.1 ± 14.5 | 71.0 ± 15.5 | 75.7 ± 17.6 b | 83.0 ± 14.6 a,c,d | 78.8 ± 16.5 b | 75.1 ± 11.1 b | * | 67.2 ± 10.3 &,^ | 80.1 ± 9.0 # | 95.3 ± 16.3 # | * |
| BMI (kg∙m−2) | 27.26 ± 4.49 | 26.83 ± 5.92 $ | 25.3 ± 5.33 b,c,d | 28.25 ± 4.49 a | 27.2 ± 5.36 a | 27.63 ± 3.81 a | * | 22.74 ± 2.18 &,^ | 27.41 ± 1.36 # | 33.67 ± 4.54 # | * |
| Body temp. (C) | 37.3 ± 0.7 | 37.2 ± 0.6 $ | 37.2 ± 0.6 d | 37.3 ± 0.7 | 37.4 ± 0.7 d | 37.2 ± 0.7 a,c | * | 37.3 ± 0.6 | 37.3 ± 0.7 | 37.3 ± 0.7 | |
| SpO2 (%) | 95.4 ±3.3 | 95.9 ±2.9 $ | 96.7 ± 2.4 b,c,d | 95.0 ± 3.3 a,c | 95.1 ± 3.2 a,b,d | 95.7 ± 3.3 a,c | * | 96.0 ± 2.9 &,^ | 95.5 ± 3.0 #,^ | 95.0 ± 3.6 #,& | * |
| RR (br∙min−1) | 17.5 ± 4.2 | 17.1 ± 3.7 | 15.7 ± 3.7 b,c,d | 18.1 ± 4.1 a,d | 18.0 ± 4.0 a | 17.1 ± 3.7 a,b | * | 17.2 ± 4.0 ^ | 17.0 ± 4.0 ^ | 18.1 ±4.2 #,& | * |
| HR (beats∙min−1) | 83 ± 14 | 81 ± 14 $ | 83 ± 15 d | 84 ± 14 d | 82 ±13 d | 79 ± 14 a,b,c | * | 83 ± 13 ^ | 83 ± 14 | 81 ± 5 # | |
| SBP (mmHg) | 129 ± 17 | 128 ± 20 | 126 ± 15 c,d | 128 ± 18 d | 130 ± 20 | 132 ± 23 a,b | * | 129 ± 18 | 128 ± 17 | 129 ± 20 | |
| DBP (mmHg) | 77 ± 13 | 75 ± 11 $ | 77 ± 11.7 c,d | 78 ± 12 d | 75 ± 12 c,d | 72 ± 13 a,b,c | * | 76 ± 12 | 76 ± 13 | 76 ± 12 | |
| CO (l∙min−1) | 5.9 ± 1.4 | 5.7± 1.4 $ | 6.0 ± 1.6 d | 5.9 ± 1.3 d | 5.8 ± 1.3 d | 5.4 ± 1.2 a,b,c | * | 5.9 ± 1.5 ^ | 5.9 ± 1.4 ^ | 5.6 ± 1.2 #,& | * |
| CI (L∙min−1∙m−2) | 3.1 ± 0.9 | 3.2 ± 0.9 | 3.3 ± 1.0 b,c,d | 3.1 ± 0.8 a | 3.2 ± 0.8 a,d | 2.9 ± 0.8 a,c | * | 3.4 ± 0.9 &,^ | 3.1 ± 0.8 #,^ | 2.8 ± 0.8 #,& | * |
| SVR (dynes∙s−1∙cm−5) | 1311 ± 277 | 1341 ± 295 | 1295 ± 291 d | 1319 ± 253 d | 1313 ± 277 d | 1401 ± 336 a,b,c | * | 1309 ± 291 | 1317 ± 284 | 1350 ± 269 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eisenkraft, A.; Maor, Y.; Constantini, K.; Goldstein, N.; Nachman, D.; Levy, R.; Halberthal, M.; Horowitz, N.A.; Golan, R.; Rosenberg, E.; et al. Continuous Remote Patient Monitoring Shows Early Cardiovascular Changes in COVID-19 Patients. J. Clin. Med. 2021, 10, 4218. https://doi.org/10.3390/jcm10184218
Eisenkraft A, Maor Y, Constantini K, Goldstein N, Nachman D, Levy R, Halberthal M, Horowitz NA, Golan R, Rosenberg E, et al. Continuous Remote Patient Monitoring Shows Early Cardiovascular Changes in COVID-19 Patients. Journal of Clinical Medicine. 2021; 10(18):4218. https://doi.org/10.3390/jcm10184218
Chicago/Turabian StyleEisenkraft, Arik, Yasmin Maor, Keren Constantini, Nir Goldstein, Dean Nachman, Ran Levy, Michael Halberthal, Netanel A. Horowitz, Ron Golan, Elli Rosenberg, and et al. 2021. "Continuous Remote Patient Monitoring Shows Early Cardiovascular Changes in COVID-19 Patients" Journal of Clinical Medicine 10, no. 18: 4218. https://doi.org/10.3390/jcm10184218
APA StyleEisenkraft, A., Maor, Y., Constantini, K., Goldstein, N., Nachman, D., Levy, R., Halberthal, M., Horowitz, N. A., Golan, R., Rosenberg, E., Lavon, E., Cohen, O., Shapira, G., Shomron, N., Ishay, A. B., Sand, E., Merin, R., Fons, M., Littman, R., & Gepner, Y. (2021). Continuous Remote Patient Monitoring Shows Early Cardiovascular Changes in COVID-19 Patients. Journal of Clinical Medicine, 10(18), 4218. https://doi.org/10.3390/jcm10184218







