Risk and Clinical Significance of Idiopathic Preterm Birth in Microvillus Inclusion Disease
Abstract
:1. Introduction
2. Methods
2.1. Collection of Case Reports
2.2. Statistics
3. Results
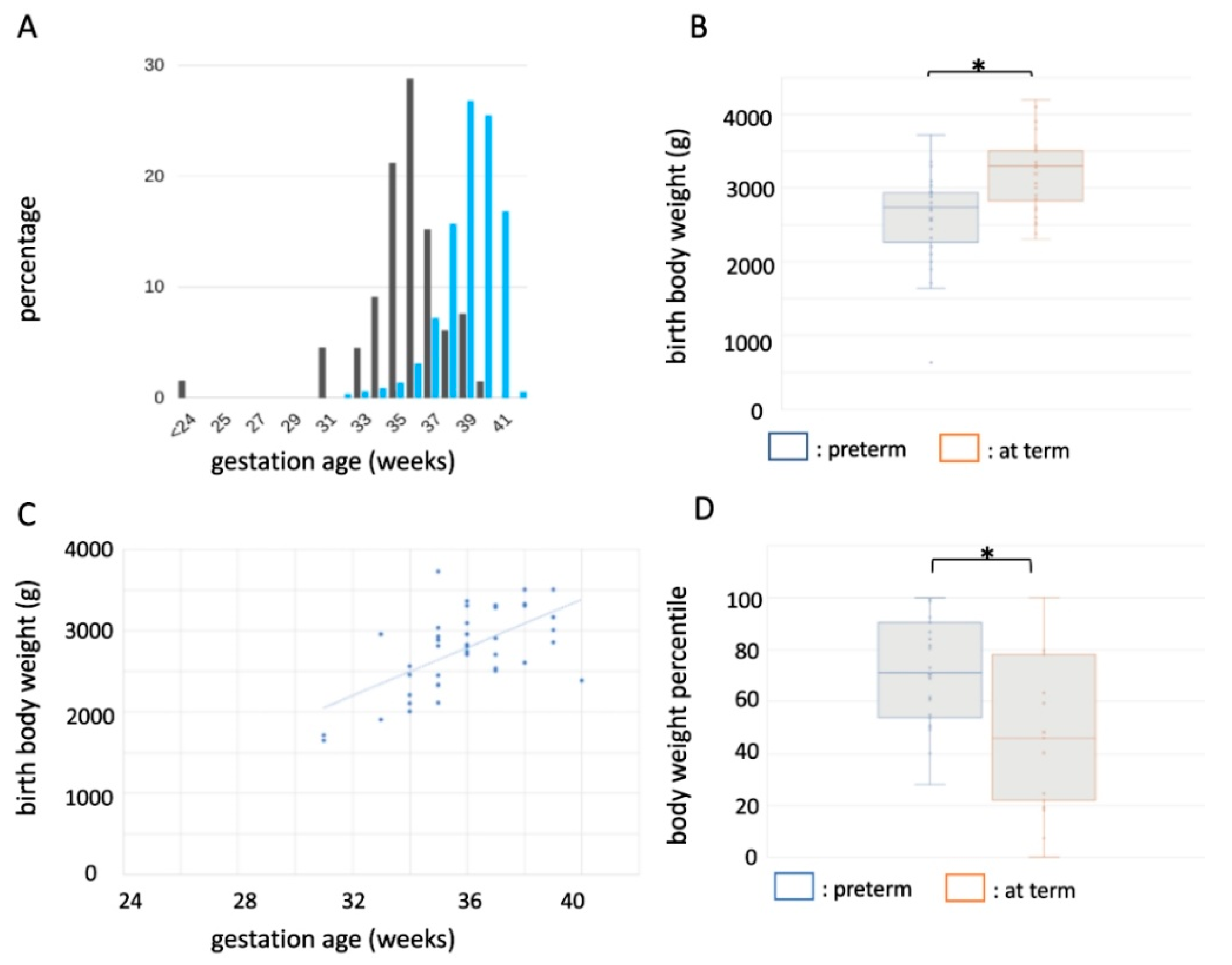
3.1. Increased Risk of Idiopathic Preterm Birth in MVID
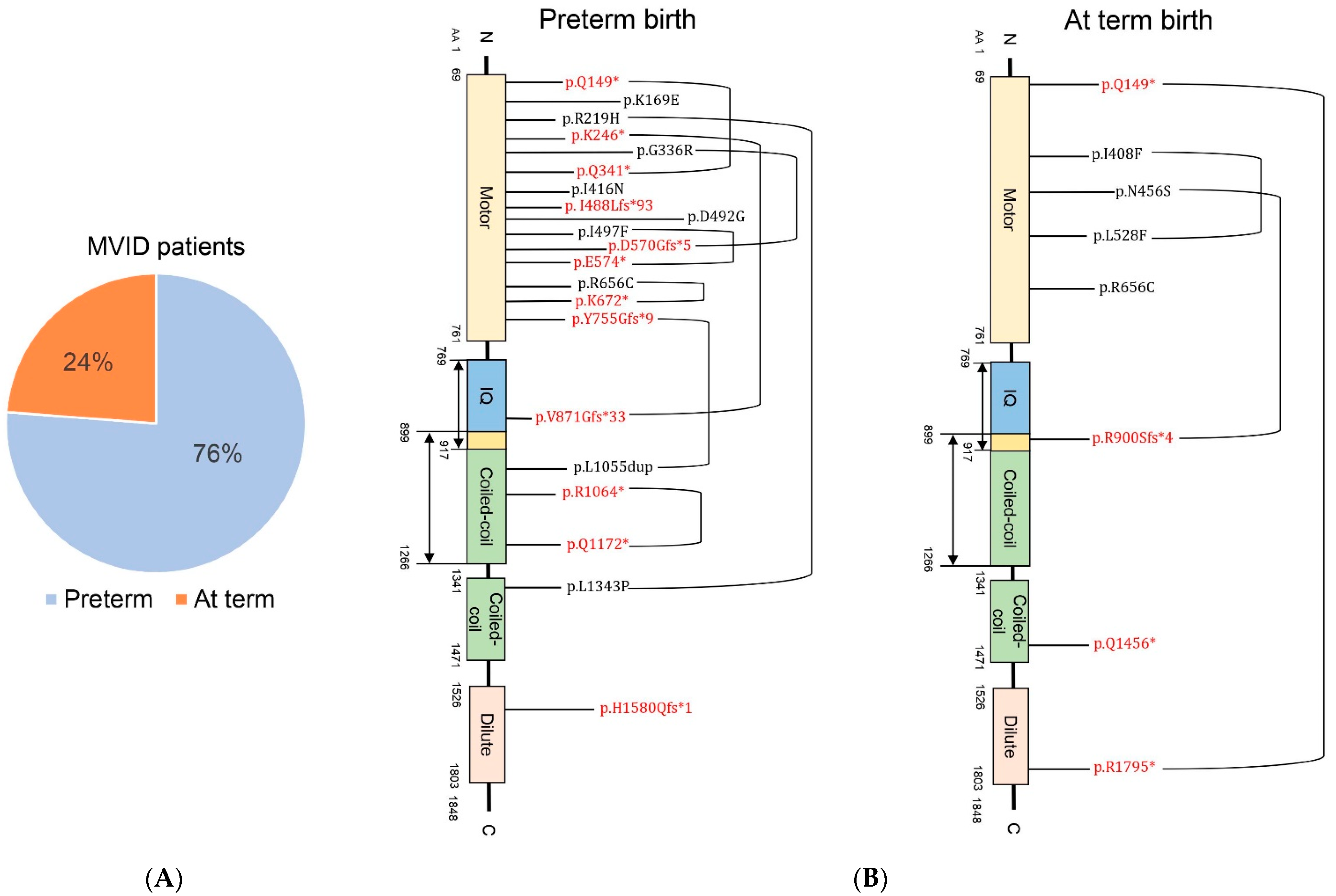
3.2. Relationship between Preterm Birth and MVID-Causing Gene Mutations
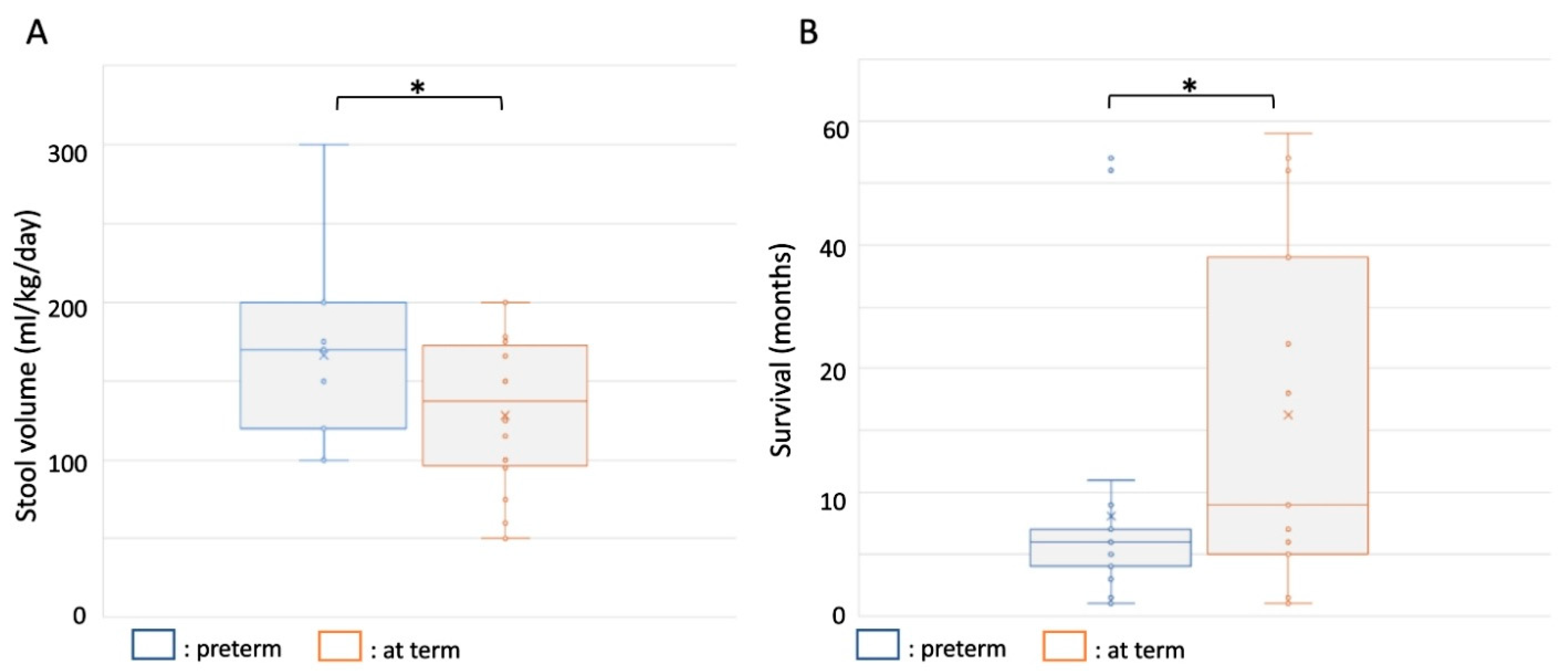
3.3. The Impact of Preterm Birth on MVID Morbidity and Mortality
3.3.1. Diarrhea and TPN Dependency
3.3.2. Respiratory Distress
3.3.3. Cholestasis
3.3.4. Mortality
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cutz, E.; Rhoads, J.M.; Drumm, B.; Sherman, P.M.; Durie, P.R.; Forstner, G.G. Microvillus Inclusion Disease: An Inherited Defect of Brush-Border Assembly and Differentiation. N. Engl. J. Med. 1989, 320, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, D.; Alrefai, W.A.; Dudeja, P.K.; Gill, R.K. Recent Advances in Understanding and Managing Malabsorption: Focus on Microvillus Inclusion Disease. F1000Research 2019, 8. [Google Scholar] [CrossRef] [Green Version]
- Müller, T.; Hess, M.W.; Schiefermeier, N.; Pfaller, K.; Ebner, H.L.; Heinz-Erian, P.; Ponstingl, H.; Partsch, J.; Röllinghoff, B.; Köhler, H.; et al. MYO5B Mutations Cause Microvillus Inclusion Disease and Disrupt Epithelial Cell Polarity. Nat. Genet. 2008, 40, 1163–1165. [Google Scholar] [CrossRef]
- Wiegerinck, C.L.; Janecke, A.R.; Schneeberger, K.; Vogel, G.F.; van Haaften-Visser, D.Y.; Escher, J.C.; Adam, R.; Thöni, C.E.; Pfaller, K.; Jordan, A.J.; et al. Loss of Syntaxin 3 Causes Variant Microvillus Inclusion Disease. Gastroenterology 2014, 147, 65–68.e10. [Google Scholar] [CrossRef] [PubMed]
- Dhekne, H.S.; Pylypenko, O.; Overeem, A.W.; Zibouche, M.; Ferreira, R.J.; van der Velde, K.J.; Rings, E.H.H.M.; Posovszky, C.; van der Sluijs, P.; Swertz, M.A.; et al. MYO5B, STX3, and STXBP2 Mutations Reveal a Common Disease Mechanism That Unifies a Subset of Congenital Diarrheal Disorders: A Mutation Update. Hum. Mutat. 2018, 39, 333–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Velde, K.J.; Dhekne, H.S.; Swertz, M.A.; Sirigu, S.; Ropars, V.; Vinke, P.C.; Rengaw, T.; van den Akker, P.C.; Rings, E.H.H.M.; Houdusse, A.; et al. An Overview and Online Registry of Microvillus Inclusion Disease Patients and Their MYO5B Mutations. Hum. Mutat. 2013, 34, 1597–1605. [Google Scholar] [CrossRef]
- Stepensky, P.; Bartram, J.; Barth, T.F.; Lehmberg, K.; Walther, P.; Amann, K.; Philips, A.D.; Beringer, O.; Zur Stadt, U.; Schulz, A.; et al. Persistent Defective Membrane Trafficking in Epithelial Cells of Patients with Familial Hemophagocytic Lymphohistiocytosis Type 5 Due to STXBP2/MUNC18-2 Mutations. Pediatr. Blood Cancer 2013, 60, 1215–1222. [Google Scholar] [CrossRef]
- Leng, C.; Rings, E.H.H.M.; de Wildt, S.N.; van IJzendoorn, S.C.D. Pharmacological and Parenteral Nutrition-Based Interventions in Microvillus Inclusion Disease. J. Clin. Med. 2020, 10, 22. [Google Scholar] [CrossRef]
- Perry, A.; Bensallah, H.; Martinez-Vinson, C.; Berrebi, D.; Arbeille, B.; Salomon, J.; Goulet, O.; Marinier, E.; Drunat, S.; Samson-Bouma, M.-E.; et al. Microvillous Atrophy: Atypical Presentations. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Croft, N.M.; Howatson, A.G.; Ling, S.C.; Nairn, L.; Evans, T.J.; Weaver, L.T. Microvillous Inclusion Disease: An Evolving Condition. J. Pediatr. Gastroenterol. Nutr. 2000, 31, 185–189. [Google Scholar] [CrossRef]
- Aldrian, D.; Vogel, G.F.; Frey, T.K.; Ayyıldız Civan, H.; Aksu, A.Ü.; Avitzur, Y.; Ramos Boluda, E.; Çakır, M.; Demir, A.M.; Deppisch, C.; et al. Congenital Diarrhea and Cholestatic Liver Disease: Phenotypic Spectrum Associated with MYO5B Mutations. J. Clin. Med. 2021, 10, 481. [Google Scholar] [CrossRef]
- Badawy, A.; Elfadul, M.; Aziabi, M.; Ageel, H.I.; Aqeel, A. Challenges of Microvillus Inclusion Disease in the NICU. Neoreviews 2020, 21, e600–e604. [Google Scholar] [CrossRef]
- Caralli, M.; Roman, C.; Coste, M.-E.; Roquelaure, B.; Buffat, C.; Bourgeois, P.; Badens, C.; Fabre, A. Genetic Enteropathies Linked to Epithelial Structural Abnormalities and Enteroendocrine Deficiency: A Systematic Review. J. Pediatr. Gastroenterol. Nutr. 2021, 72, 826–832. [Google Scholar] [CrossRef]
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, Regional, and National Causes of under-5 Mortality in 2000-15: An Updated Systematic Analysis with Implications for the Sustainable Development Goals. Lancet 2016, 388, 3027–3035. [Google Scholar] [CrossRef] [Green Version]
- Thoeni, C.E.; Vogel, G.F.; Tancevski, I.; Geley, S.; Lechner, S.; Pfaller, K.; Hess, M.W.; Müller, T.; Janecke, A.R.; Avitzur, Y.; et al. Microvillus Inclusion Disease: Loss of Myosin vb Disrupts Intracellular Traffic and Cell Polarity. Traffic 2014, 15, 22–42. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.-A.; Munoz, F.M.; Gonik, B.; Frau, L.; Cutland, C.; Mallett-Moore, T.; Kissou, A.; Wittke, F.; Das, M.; Nunes, T.; et al. Preterm Birth: Case Definition & Guidelines for Data Collection, Analysis, and Presentation of Immunisation Safety Data. Vaccine 2016, 34, 6047–6056. [Google Scholar] [CrossRef] [Green Version]
- Julia, J.; Shui, V.; Mittal, N.; Heim-Hall, J.; Blanco, C.L. Microvillus Inclusion Disease, a Diagnosis to Consider When Abnormal Stools and Neurological Impairments Run Together Due to a Rare Syntaxin 3 Gene Mutation. J. Neonatal. Perinatal. Med. 2019, 12, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Alsaleem, B.M.R.; Ahmed, A.B.M.; Fageeh, M.A. Microvillus Inclusion Disease Variant in an Infant with Intractable Diarrhea. Case Rep. Gastroenterol. 2017, 11, 647–651. [Google Scholar] [CrossRef]
- Qiu, Y.-L.; Gong, J.-Y.; Feng, J.-Y.; Wang, R.-X.; Han, J.; Liu, T.; Lu, Y.; Li, L.-T.; Zhang, M.-H.; Sheps, J.A.; et al. Defects in Myosin VB Are Associated with a Spectrum of Previously Undiagnosed Low γ-Glutamyltransferase Cholestasis. Hepatology 2017, 65, 1655–1669. [Google Scholar] [CrossRef]
- Gonzales, E.; Taylor, S.A.; Davit-Spraul, A.; Thébaut, A.; Thomassin, N.; Guettier, C.; Whitington, P.F.; Jacquemin, E. MYO5B Mutations Cause Cholestasis with Normal Serum Gamma-Glutamyl Transferase Activity in Children without Microvillous Inclusion Disease. Hepatology 2017, 65, 164–173. [Google Scholar] [CrossRef] [PubMed]
- van IJzendoorn, S.C.D.; Li, Q.; Qiu, Y.-L.; Wang, J.-S.; Overeem, A.W. Unequal Effects of Myosin 5B Mutations in Liver and Intestine Determine the Clinical Presentation of Low-Gamma-Glutamyltransferase Cholestasis. Hepatology 2020, 72, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Girard, M.; Lacaille, F.; Verkarre, V.; Mategot, R.; Feldmann, G.; Grodet, A.; Sauvat, F.; Irtan, S.; Davit-Spraul, A.; Jacquemin, E.; et al. MYO5B and Bile Salt Export Pump Contribute to Cholestatic Liver Disorder in Microvillous Inclusion Disease. Hepatology 2014, 60, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Halac, U.; Lacaille, F.; Joly, F.; Hugot, J.-P.; Talbotec, C.; Colomb, V.; Ruemmele, F.M.; Goulet, O. Microvillous Inclusion Disease: How to Improve the Prognosis of a Severe Congenital Enterocyte Disorder. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sun, Y.; van IJzendoorn, S.C.D. A Link between Intrahepatic Cholestasis and Genetic Variations in Intracellular Trafficking Regulators. Biology 2021, 10, 119. [Google Scholar] [CrossRef]
- Blencowe, H.; Cousens, S.; Oestergaard, M.Z.; Chou, D.; Moller, A.-B.; Narwal, R.; Adler, A.; Vera Garcia, C.; Rohde, S.; Say, L.; et al. National, Regional, and Worldwide Estimates of Preterm Birth Rates in the Year 2010 with Time Trends since 1990 for Selected Countries: A Systematic Analysis and Implications. Lancet 2012, 379, 2162–2172. [Google Scholar] [CrossRef] [Green Version]
- Raju, T.N.K. The “Late Preterm” Birth-Ten Years Later. Pediatrics 2017, 139, e20163331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yadav, S.; Lee, B.; Kamity, R. Neonatal Respiratory Distress Syndrome. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Many, A.; Hill, L.M.; Lazebnik, N.; Martin, J.G. The Association between Polyhydramnios and Preterm Delivery. Obstet. Gynecol. 1995, 86, 389–391. [Google Scholar] [CrossRef]
- Lampl, M.; Kusanovic, J.P.; Erez, O.; Espinoza, J.; Gotsch, F.; Goncalves, L.; Hassan, S.; Gomez, R.; Nien, J.K.; Frongillo, E.A.; et al. Early Rapid Growth, Early Birth: Accelerated Fetal Growth and Spontaneous Late Preterm Birth. Am. J. Hum. Biol. 2009, 21, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Elenis, E.; Wikström, A.-K.; Simic, M. Accelerated Fetal Growth in Early Pregnancy and Risk of Preterm Birth: A Prospective Cohort Study. BMC Pregnancy Childbirth 2020, 20, 764. [Google Scholar] [CrossRef]
- Overeem, A.W.; Posovszky, C.; Rings, E.H.M.M.; Giepmans, B.N.G.; van IJzendoorn, S.C.D. The Role of Enterocyte Defects in the Pathogenesis of Congenital Diarrheal Disorders. Dis. Model. Mech. 2016, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Wedenoja, S.; Höglund, P.; Holmberg, C. Review Article: The Clinical Management of Congenital Chloride Diarrhoea. Aliment. Pharmacol. Ther. 2010, 31, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Kravtsov, D.V.; Ahsan, M.K.; Kumari, V.; van Ijzendoorn, S.C.D.; Reyes-Mugica, M.; Kumar, A.; Gujral, T.; Dudeja, P.K.; Ameen, N.A. Identification of Intestinal Ion Transport Defects in Microvillus Inclusion Disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G142–G155. [Google Scholar] [CrossRef] [PubMed]
- Strauss, J.F.; Romero, R.; Gomez-Lopez, N.; Haymond-Thornburg, H.; Modi, B.P.; Teves, M.E.; Pearson, L.N.; York, T.P.; Schenkein, H.A. Spontaneous Preterm Birth: Advances toward the Discovery of Genetic Predisposition. Am. J. Obstet. Gynecol. 2018, 218, 294–314.e2. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Feenstra, B.; Bacelis, J.; Liu, X.; Muglia, L.M.; Juodakis, J.; Miller, D.E.; Litterman, N.; Jiang, P.-P.; Russell, L.; et al. Genetic Associations with Gestational Duration and Spontaneous Preterm Birth. N. Engl. J. Med. 2017, 377, 1156–1167. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leng, C.; Sun, Y.; van IJzendoorn, S.C.D. Risk and Clinical Significance of Idiopathic Preterm Birth in Microvillus Inclusion Disease. J. Clin. Med. 2021, 10, 3935. https://doi.org/10.3390/jcm10173935
Leng C, Sun Y, van IJzendoorn SCD. Risk and Clinical Significance of Idiopathic Preterm Birth in Microvillus Inclusion Disease. Journal of Clinical Medicine. 2021; 10(17):3935. https://doi.org/10.3390/jcm10173935
Chicago/Turabian StyleLeng, Changsen, Yue Sun, and Sven C. D. van IJzendoorn. 2021. "Risk and Clinical Significance of Idiopathic Preterm Birth in Microvillus Inclusion Disease" Journal of Clinical Medicine 10, no. 17: 3935. https://doi.org/10.3390/jcm10173935
APA StyleLeng, C., Sun, Y., & van IJzendoorn, S. C. D. (2021). Risk and Clinical Significance of Idiopathic Preterm Birth in Microvillus Inclusion Disease. Journal of Clinical Medicine, 10(17), 3935. https://doi.org/10.3390/jcm10173935





