Peripheral Retinal Degenerations and Idiopathic Epiretinal Membrane: Analysis with Ultra-Wide-Field Scanning Laser Ophthalmoscopy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Examination Procedure
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
- In most people, ERM coexists with peripheral retinal changes.
- Some of these changes promote retinal detachment. Thus, surgeons should consider the risk of retinal tear during vitrectomy, which increases the scope of surgery and may adversely affect prognosis. According to the results obtained in our study, we strongly suggest examining patients before the vitrectomy (ILM peeling) procedure with a Goldmann triple mirror.
- Ultra-wide field ophthalmoscopy is a valuable diagnostic method. It enabled the detection of nearly 87% of all peripheral retinal lesions in patients with ERM, but it cannot replace fundus examination with a Goldmann triple mirror or ophthalmoscopy with scleral indentation.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ERM | Epiretinal Membrane |
| OCT | Optical Coherence Tomography |
| OCT-A | Optical Coherence Tomography Angiography |
| UWFP | Ultra-Wide Field Laser Opthalmoscopy |
| BCVA | Best Corrected Visual Acuity |
| ILM | Internal Limiting Membrane |
| AMD | Age-Related Macular Degeneration |
| DR | Diabetic Retinopathy |
| RD | Retinal Detachment |
References
- Yoshida, M.; Kunikata, H.; Kunimatsu-Sanuki, S.; Nakazawa, T. Efficacy of 27-Gauge Vitrectomy with Internal Limiting Membrane Peeling for Epiretinal Membrane in Glaucoma Patients. J. Ophthalmol. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Smiddy, W.E.; Maguire, A.M.; Green, W.R.; Michels, R.G.; De La Cruz, Z.; Enger, C.; Jaeger, M.; Rice, T.A. Idiopathic epiretinal membranes. Ultrastructural characteristics and clinicopathologic correlation. Ophthalmology 1989, 96, 811–820. [Google Scholar] [CrossRef]
- Chang, W.-C.; Lin, C.; Lee, C.-H.; Sung, T.-L.; Tung, T.-H.; Liu, J.-H. Vitrectomy with or without internal limiting membrane peeling for idiopathic epiretinal membrane: A meta-analysis. PLoS ONE 2017, 12, e0179105. [Google Scholar] [CrossRef] [PubMed]
- Hardin, J.S.; Gauldin, D.W.; Soliman, M.K.; Chu, C.J.; Yang, Y.C.; Sallam, A.B. Cataract Surgery Outcomes in Eyes with Primary Epiretinal Membrain. JAMA Ophthalmol. 2018, 136, 148–154. [Google Scholar] [CrossRef]
- Kumar, D.A.; Maitra, P.; Agarwal, A. Epiretinal membrane profile on spectral domain optical coherence tomography in patients with uveitis. Indian J. Ophthalmol. 2019, 67, 376. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, C. Evidence-based analysis of prophylactic treatment of asymptomatic retinal breaks and lattice degeneration. Ophthalmology 2000, 107, 12–15. [Google Scholar] [CrossRef]
- Folk, J.; Arrindell, E.L.; Klugman, M.R. The Fellow Eye of Patients with Phakic Lattice Retinal Detachment. Ophthalmology 1989, 96, 72–79. [Google Scholar] [CrossRef]
- Smolin, G. Statistical analysis of retinal holes and tears. Am. J. Ophthalmol. 1965, 60, 1055–1059. [Google Scholar] [CrossRef]
- Everett, W.G. Bilateral retinal detachment and degenerations. Trans. Am. Ophthalmol. Soc. 1966, 64, 543–585. [Google Scholar]
- Hirano, T.; Imai, A.; Kasamatsu, H.; Kakihara, S.; Toriyama, Y.; Murata, T. Assessment of diabetic retinopathy using two ultra-wide-field fundus imaging systems, the Clarus® and Optos™ systems. BMC Ophthalmol. 2018, 18, 332. [Google Scholar] [CrossRef] [Green Version]
- Domalpally, A.; Clemons, T.E.; Danis, R.P.; Sadda, S.R.; Cukras, C.A.; Toth, C.A.; Friberg, T.R.; Chew, E.Y. Peripheral Retinal Changes Associated with Age-Related Macular Degeneration in the Age-Related Eye Disease Study 2. Ophthalmology 2017, 124, 479–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kritzenberger, M.; Junglas, B.; Framme, C.; Helbig, H.; Gabel, V.P.; Fuchshofer, R.; Tamm, E.R.; Hillenkamp, J. Different collagen types define two types of idiopathic epiretinal membranes. Histopathology 2011, 58, 953–965. [Google Scholar] [CrossRef] [Green Version]
- Snead, D.R.J.; Cullen, N.; James, S.; Poulson, A.V.; Morris, A.H.C.; Lukaris, A.; Scott, J.D.; Richards, A.J.; Snead, M.P. Hyperconvolution of the inner limiting membrane in vitreomaculopathies. Graefe’s Arch. Clin. Exp. Ophthalmol. 2004, 242, 853–862. [Google Scholar] [CrossRef]
- Vatavuk, Z.; Derk, B.A.; Knežević, T.; Belak, M.; Milošević, M.; Friberg, T.R. Morphological and Angiographic Peripheral Retinal Changes in Patients with Age-Related Macular Degeneration. Ophthalmol. Retin. 2018, 2, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.; Tan, S.-P.; Lee, S.Y.; Cheung, G.C.M.; Tan, G.; Kumar, N.; Cheng, C.-Y.; Wong, T.Y. Prevalence and risk factors for epiretinal membrane: The Singapore Epidemiology of Eye Disease study. Br. J. Ophthalmol. 2016, 101, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Kadonosono, K. Macular Diseases: Epiretinal Membrane. Microincision Vitr. Surg. 2014, 54, 159–163. [Google Scholar] [CrossRef]
- Forshaw, T.R.J.; Minör, Å.S.; Subhi, Y.; Sørensen, T.L. Peripheral Retinal Lesions in Eyes with Age-Related Macular Degeneration Using Ultra-Widefield Imaging: A Systematic Review with Meta-analyses. Ophthalmol Retin. 2019, 3, 734–743. [Google Scholar] [CrossRef]
- Wilkinson, C.P. Interventions for asymptomatic retinal breaks and lattice degeneration for preventing retinal detachment. Cochrane Database Syst. Rev. 2014, CD003170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kottow, M. Peripheral retinal degenerations and breaks. Graefe’s Arch. Clin. Exp. Ophthalmol. 1980, 214, 53–60. [Google Scholar] [CrossRef]
- Byer, N.E. Lattice degeneration of the retina. Surv. Ophthalmol. 1979, 23, 213–248. [Google Scholar] [CrossRef]
- Byer, N.E. Clinical study of lattice degeneration of the retina. Trans. Am. Acad. Ophthalmol. Otolaryngol. 1965, 69, 1065–1081. [Google Scholar] [PubMed]
- Liu, L.; Wang, F.; Xu, D.; Xie, C.; Zou, J. The application of wide-field laser ophthalmoscopy in fundus examination before myopic refractive surgery. BMC Ophthalmol. 2017, 17, 250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Malley, P.; Allen, R.A.; Traatsma, B.R.S.; O’Malley, C.C. Paving-Stone Degeneration of the Retina. Arch. Ophthalmol. 1965, 73, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Zudaire, E.; Gambardella, L.; Kurcz, C.; Vermeren, S. A Computational Tool for Quantitative Analysis of Vascular Networks. PLoS ONE 2011, 6, e27385. [Google Scholar] [CrossRef] [Green Version]
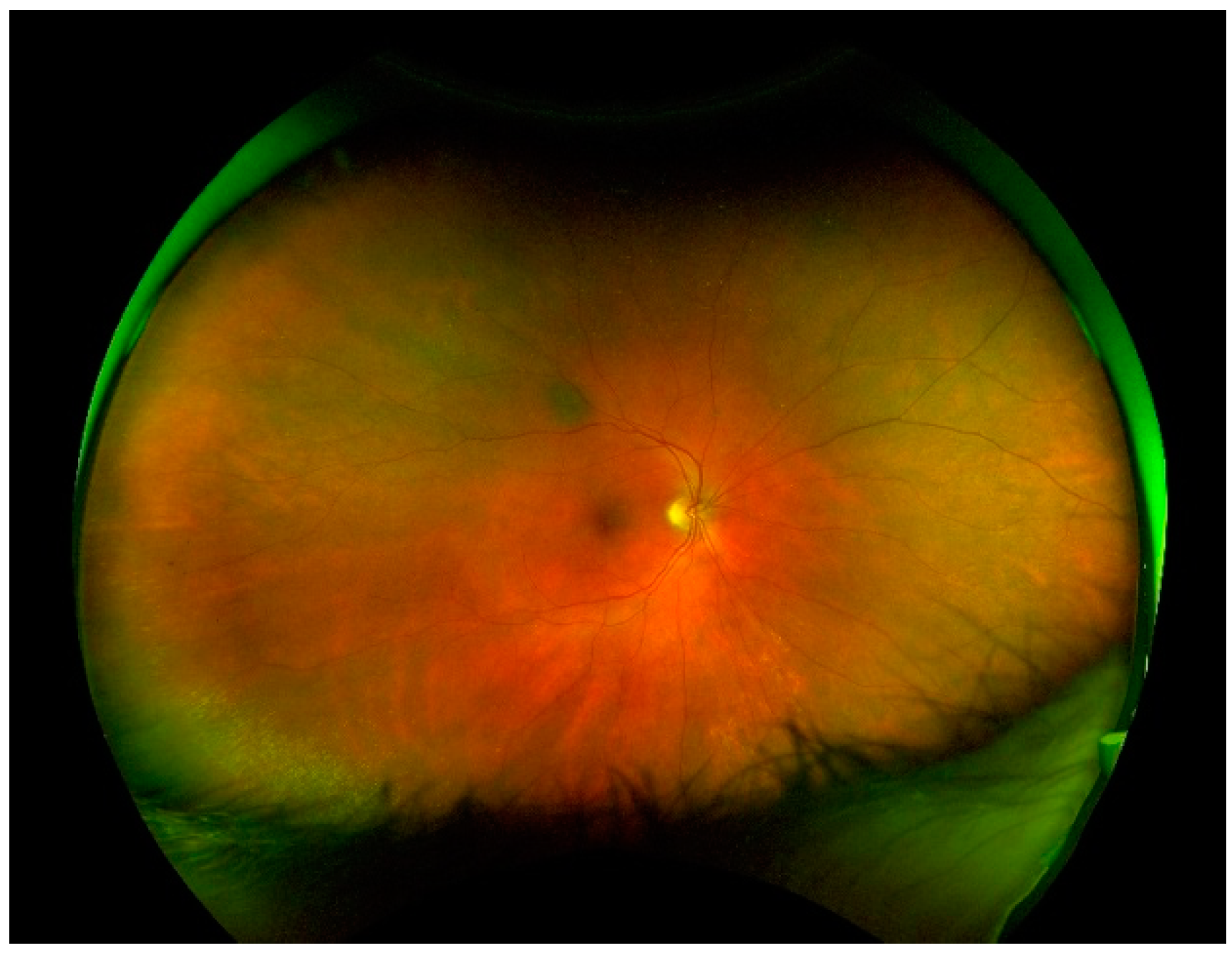
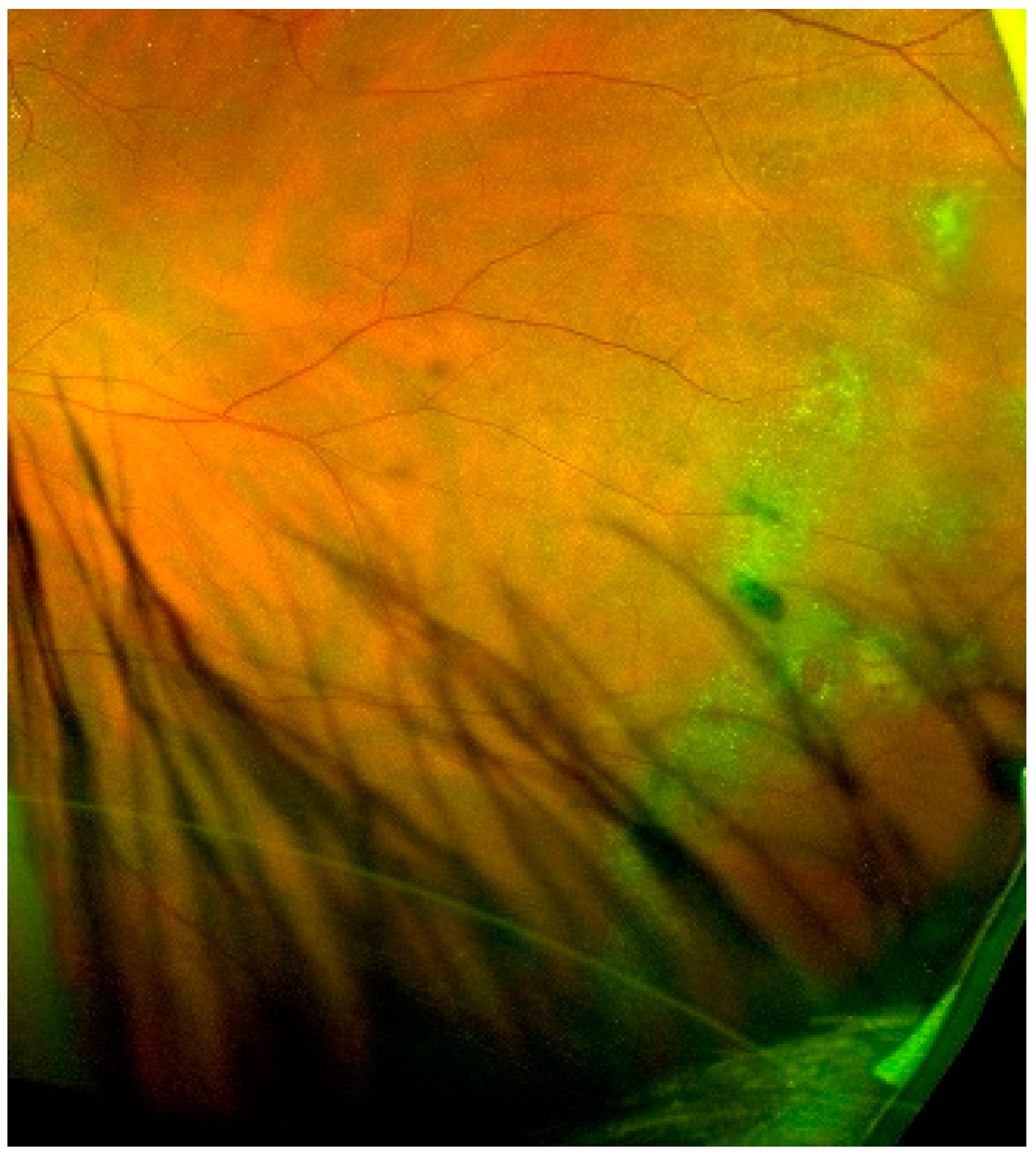
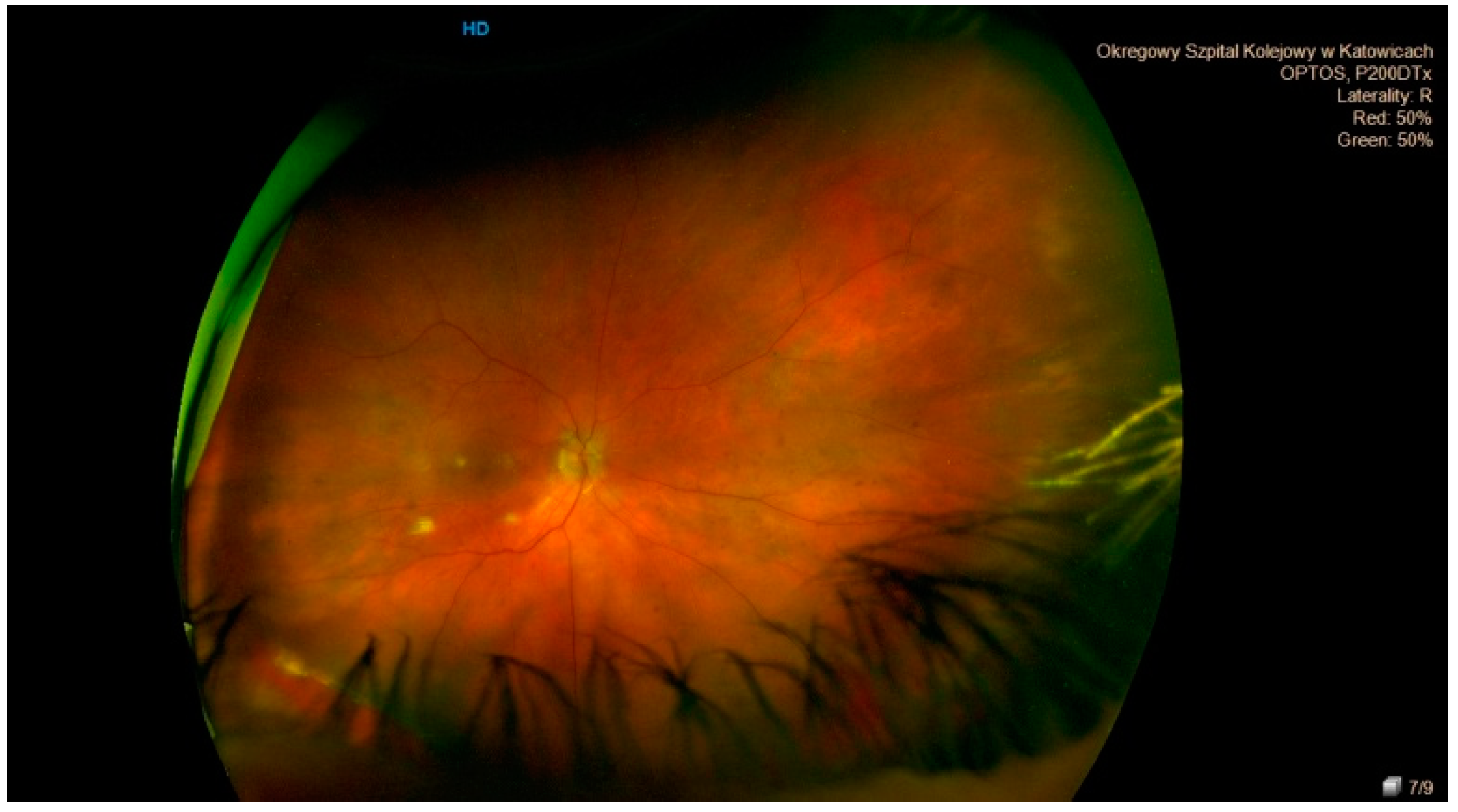
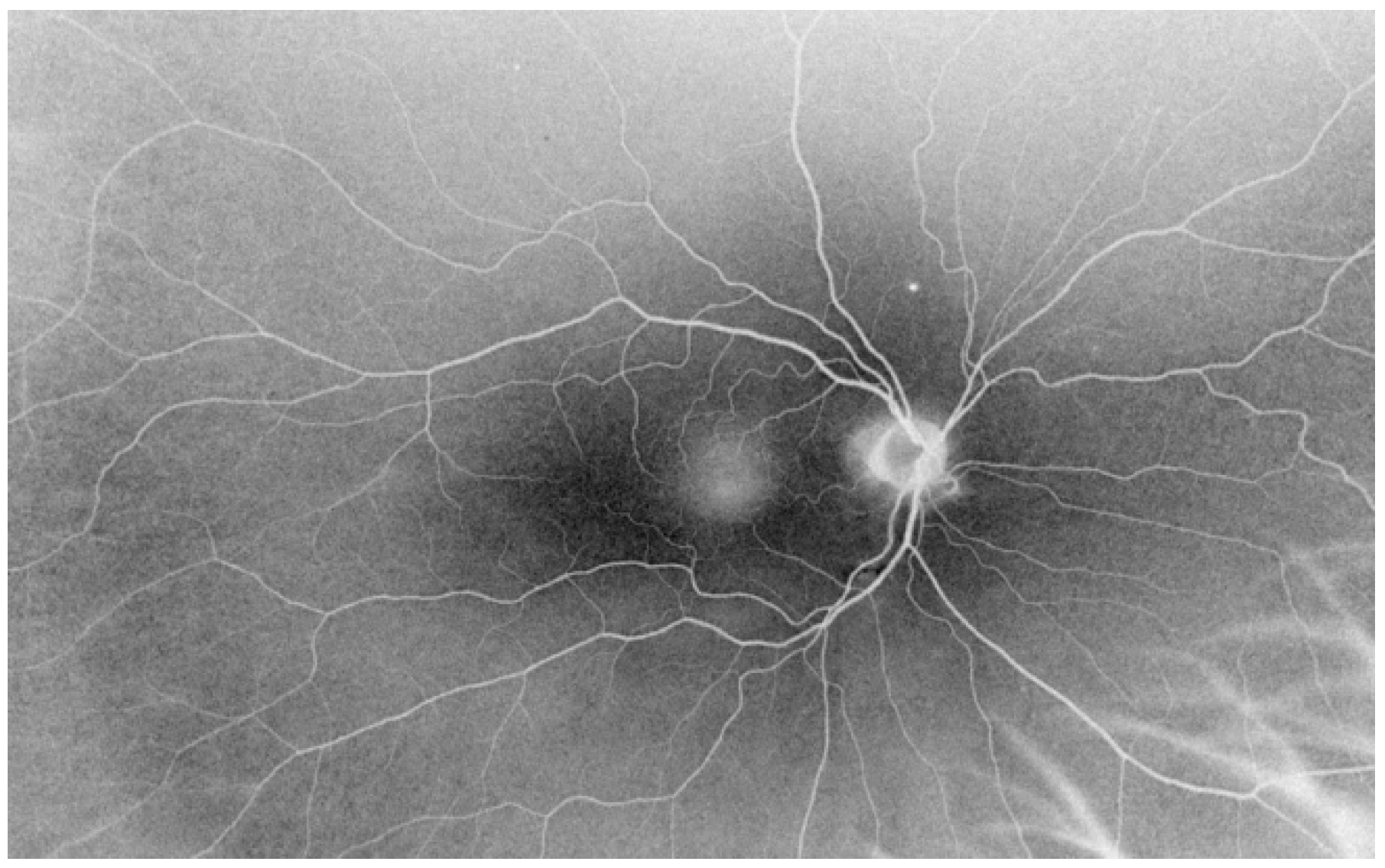
| Lattice Degeneration | Snail Track Degeneration | Paving Stone Degeneration | Retinoschisis | Microcystoid Degeneration | Retinal Break | |
|---|---|---|---|---|---|---|
| ERM group | 14 (14.58%) | 12 (12.5%) | 33 (34.37%) | 7 (7.29%) | 11 (11.45%) | 19 (19.79%) |
| Control group | 7 (16.66%) | 6 (14.28%) | 15 (35.71%) | 5 (11.90%) | 5 (11.90%) | 4 (9.52%) |
| Statistical comparison | p = 0.29 | p = 0.33 | p = 0.047 | p = 0.85 | p = 0.28 | p = 0.006 |
| Lattice Degeneration | Snail Track Degeneration | Paving Stone Degeneration | Retinoschisis | Microcystoid Degeneration | Retinal Break | |
|---|---|---|---|---|---|---|
| ERM group | 11 (13.0%) | 9 (10.7%) | 31 (36.90%) | 5 (5.95%) | 10 (11.90%) | 18 (21.42%) |
| Control group | 4 (14.28%) | 2 (7.14%) | 11 (39.28%) | 3 (10.71%) | 5 (17.85%) | 4 (14.28%) |
| Statistical comparison | p = 0.16 | p = 0.077 | p = 0.01 | p = 0.40 | p = 0.38 | p = 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulfik-Dembska, K.; Teper, S.; Dembski, M.; Nowińska, A.; Wylęgała, E. Peripheral Retinal Degenerations and Idiopathic Epiretinal Membrane: Analysis with Ultra-Wide-Field Scanning Laser Ophthalmoscopy. J. Clin. Med. 2021, 10, 3876. https://doi.org/10.3390/jcm10173876
Ulfik-Dembska K, Teper S, Dembski M, Nowińska A, Wylęgała E. Peripheral Retinal Degenerations and Idiopathic Epiretinal Membrane: Analysis with Ultra-Wide-Field Scanning Laser Ophthalmoscopy. Journal of Clinical Medicine. 2021; 10(17):3876. https://doi.org/10.3390/jcm10173876
Chicago/Turabian StyleUlfik-Dembska, Klaudia, Sławomir Teper, Michał Dembski, Anna Nowińska, and Edward Wylęgała. 2021. "Peripheral Retinal Degenerations and Idiopathic Epiretinal Membrane: Analysis with Ultra-Wide-Field Scanning Laser Ophthalmoscopy" Journal of Clinical Medicine 10, no. 17: 3876. https://doi.org/10.3390/jcm10173876
APA StyleUlfik-Dembska, K., Teper, S., Dembski, M., Nowińska, A., & Wylęgała, E. (2021). Peripheral Retinal Degenerations and Idiopathic Epiretinal Membrane: Analysis with Ultra-Wide-Field Scanning Laser Ophthalmoscopy. Journal of Clinical Medicine, 10(17), 3876. https://doi.org/10.3390/jcm10173876








