Health, Wellbeing, and Prognosis of Australian Adolescents with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Case-Controlled Follow-Up Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Measures
2.4. Statistical Analysis
3. Results
3.1. Participant Characteristics
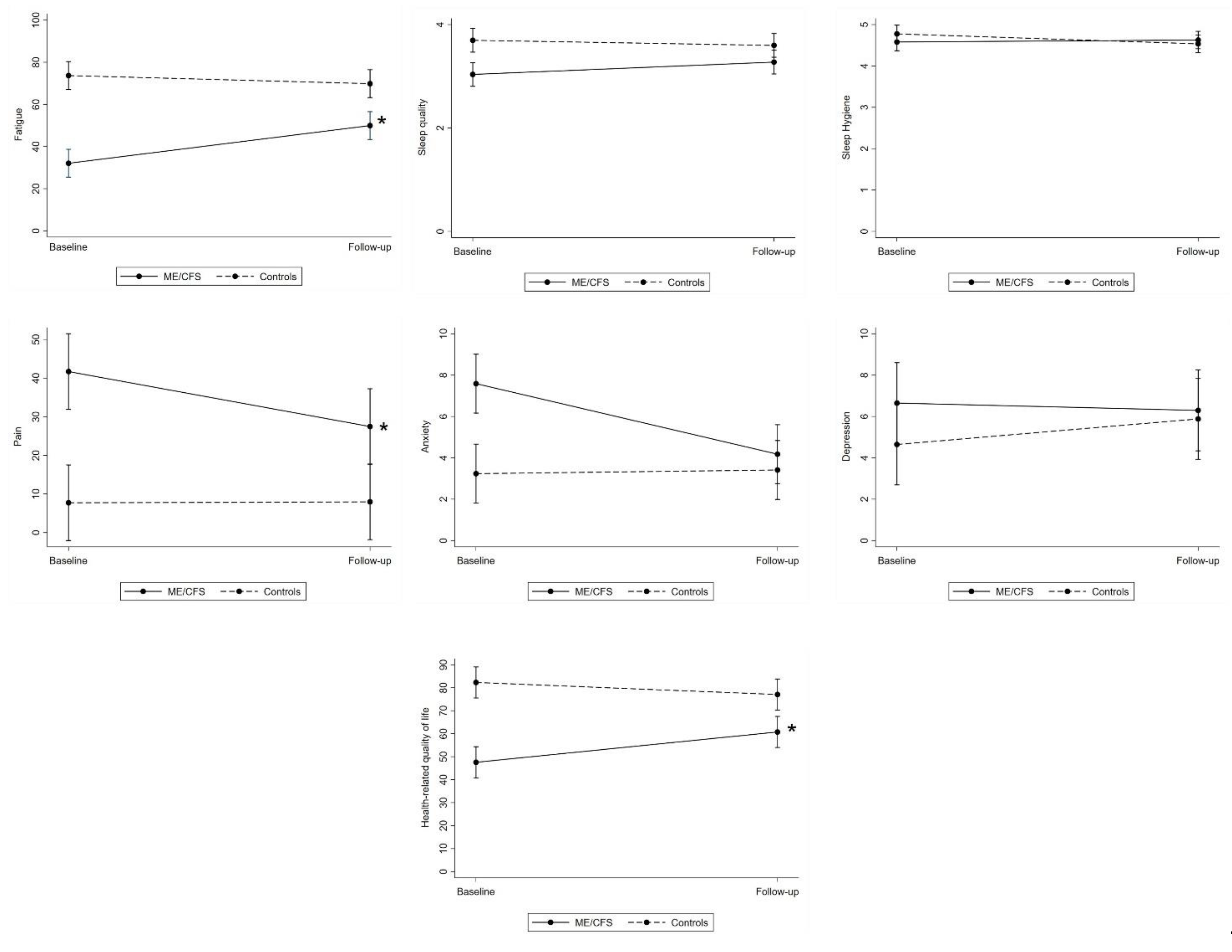
3.2. Group Differences in Trajectories of Health and Psychological Wellbeing from Baseline to Follow-Up
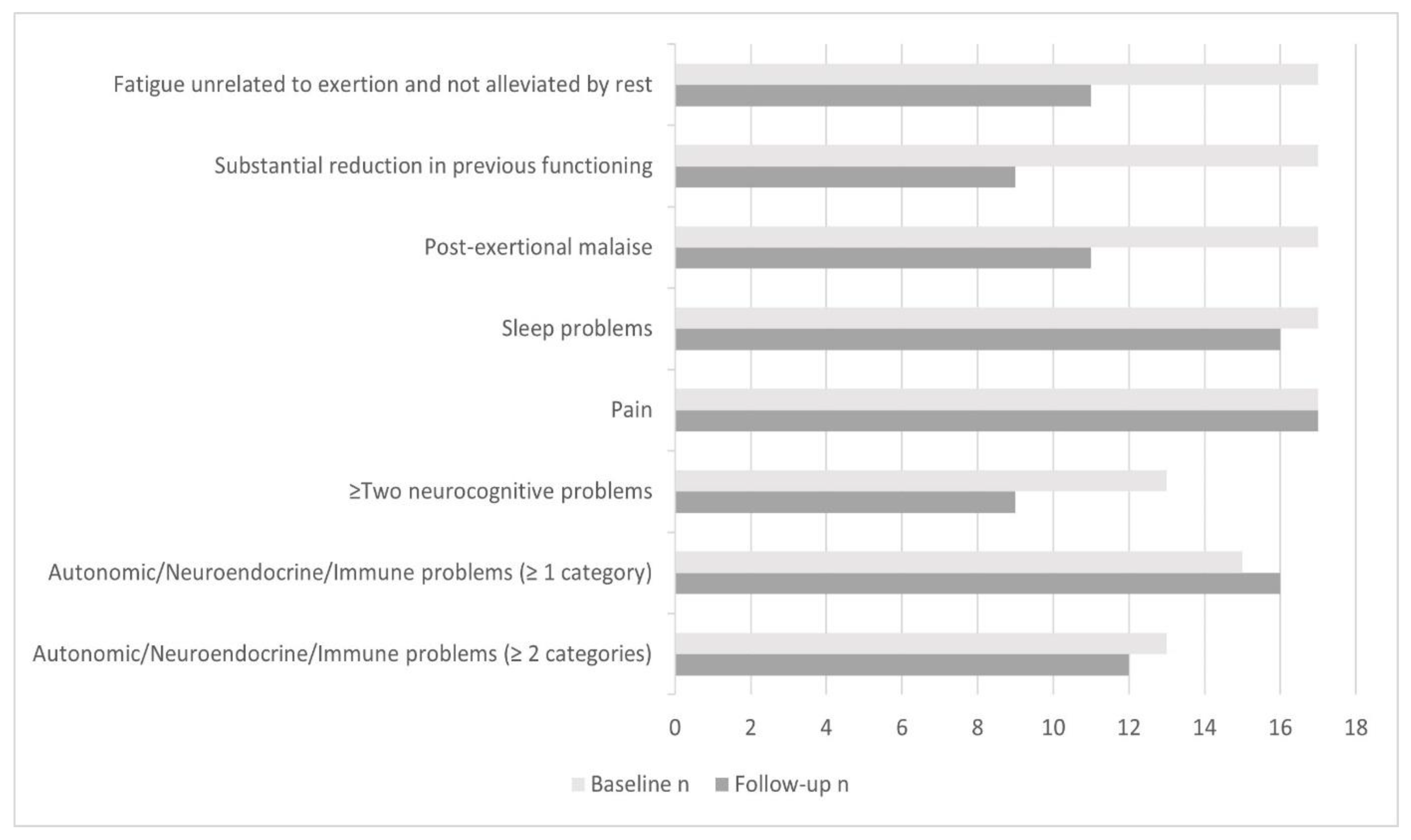
3.3. ME/CFS Symptomatology and Fulfilment of ME/CFS Diagnostic Criteria
3.4. Predictors of ME/CFS Criteria Fulfilment at Follow-Up
4. Discussion
4.1. Trajectories of Health and Psychological Wellbeing from Baseline to Follow-Up
4.2. Fulfilment of Paediatric ME/CFS Diagnostic Criteria at Follow-Up
4.3. Predictors of ME/CFS Criteria Fulfilment
4.4. Study Limitations and Strengths
4.5. Clinical Implications and Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crawley, E. Pediatric chronic fatigue syndrome: Current perspectives. Pediatric Health Med. Ther. 2017, 9, 27. [Google Scholar] [CrossRef]
- Kennedy, G.; Underwood, C.; Belch, J.J. Physical and functional impact of chronic fatigue syndrome/myalgic encephalomyelitis in childhood. Pediatrics 2010, 125, e1324–e1330. [Google Scholar] [CrossRef]
- Knight, S.; Harvey, A.; Lubitz, L.; Rowe, K.; Reveley, C.; Veit, F.; Hennel, S.; Scheinberg, A. Paediatric chronic fatigue syndrome: Complex presentations and protracted time to diagnosis. J. Paediatr. Child Health 2013, 49, 919–924. [Google Scholar] [CrossRef]
- Knight, S.; Politis, J.; Garnham, C.; Scheinberg, A.; Tollit, M. School functioning in adolescents with Chronic Fatigue Syndrome. Front. Pediatrics 2018, 6, 302. [Google Scholar] [CrossRef]
- Ausubel, D.P. Theory and Problems of Adolescent Development; iUniverse: Bloomington, Indiana, 2002. [Google Scholar]
- Bould, H.; Collin, S.; Lewis, G.; Rimes, K.; Crawley, E. Depression in paediatric chronic fatigue syndrome. Arch. Dis. Child. 2013, 98, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Crawley, E.; Hunt, L.; Stallard, P. Anxiety in children with CFS/ME. Eur. Child Adolesc. Psychiatry 2009, 18, 683. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.; Waters, E.; Mackinnon, A.; Reddihough, D.; Graham, H.K.; Mehmet-Radji, O.; Boyd, R. Paediatric quality of life instruments: A review of the impact of the conceptual framework on outcomes. Dev. Med. Child Neurol. 2006, 48, 311–318. [Google Scholar] [CrossRef]
- Josev, E.; Jackson, M.; Bei, B.; Trinder, J.; Harvey, A.; Clarke, C.; Snodgrass, K.; Scheinberg, A.; Knight, S. Sleep quality in adolescents with chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). J. Clin. Sleep Med. 2017, 13, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Parslow, R.M.; Anderson, N.; Byrne, D.; Shaw, A.; Haywood, K.L.; Crawley, E. Adolescent’s descriptions of fatigue, fluctuation and payback in chronic fatigue syndrome/myalgic encephalopathy (CFS/ME): Interviews with adolescents and parents. BMJ Paediatr. Open 2018, 2, e000281. [Google Scholar] [CrossRef]
- Rowe, K. Long Term Follow up of Young People with Chronic Fatigue Syndrome Attending a Pediatric Outpatient Service. Front. Pediatrics 2019, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Sankey, A.; Hill, C.M.; Brown, J.; Quinn, L.; Fletcher, A. A follow-up study of chronic fatigue syndrome in children and adolescents: Symptom persistence and school absenteeism. Clin. Child Psychol. Psychiatry 2006, 11, 126–138. [Google Scholar] [CrossRef]
- Winger, A.; Kvarstein, G.; Wyller, V.B.; Ekstedt, M.; Sulheim, D.; Fagermoen, E.; Småstuen, M.C.; Helseth, S. Health related quality of life in adolescents with chronic fatigue syndrome: A cross-sectional study. Health Qual. Life Outcomes 2015, 13, 96. [Google Scholar] [CrossRef] [PubMed]
- Short, M.A.; Gradisar, M.; Lack, L.C.; Wright, H.R.; Dohnt, H. The sleep patterns and well-being of Australian adolescents. J. Adolesc. 2013, 36, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Rimes, K.A.; Goodman, R.; Hotopf, M.; Wessely, S.; Meltzer, H.; Chalder, T. Incidence, prognosis, and risk factors for fatigue and chronic fatigue syndrome in adolescents: A prospective community study. Pediatrics 2007, 119, e603–e609. [Google Scholar] [CrossRef] [PubMed]
- Van Oort, F.; Greaves-Lord, K.; Verhulst, F.; Ormel, J.; Huizink, A. The developmental course of anxiety symptoms during adolescence: The TRAILS study. J. Child Psychol. Psychiatry 2009, 50, 1209–1217. [Google Scholar] [CrossRef]
- Merikangas, K.R.; He, J.-p.; Burstein, M.; Swanson, S.A.; Avenevoli, S.; Cui, L.; Benjet, C.; Georgiades, K.; Swendsen, J. Lifetime prevalence of mental disorders in US adolescents: Results from the National Comorbidity Survey Replication–Adolescent Supplement (NCS-A). J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 980–989. [Google Scholar] [CrossRef]
- Lawrence, D.; Johnson, S.; Hafekost, J.; de Haan, K.B.; Sawyer, M.; Ainley, J.; Zubrick, S.R. The Mental Health of Children and Adolescents: Report on the Second Australian Child and Adolescent Survey of Mental Health and Wellbeing; Department of Health: Canberra, Australia, 2015. [Google Scholar]
- Bisegger, C.; Cloetta, B.; Von Bisegger, U.; Abel, T.; Ravens-Sieberer, U. Health-related quality of life: Gender differences in childhood and adolescence. Soz.-Und Präventivmedizin 2005, 50, 281–291. [Google Scholar] [CrossRef]
- Goldbeck, L.; Schmitz, T.G.; Besier, T.; Herschbach, P.; Henrich, G. Life satisfaction decreases during adolescence. Qual. Life Res. 2007, 16, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Meuleners, L.B.; Lee, A.H. Adolescent quality of life: A school-based cohort study in Western Australia. Pediatrics Int. 2003, 45, 706–711. [Google Scholar] [CrossRef]
- Smith, M.S.; Glover, D.; Mitchell, J.; McCauley, E.A.; Corey, L.; Gold, D.; Tenover, F.C. Chronic fatigue in adolescents. Pediatrics 1991, 88, 195–202. [Google Scholar]
- Krilov, L.R.; Fisher, M.; Friedman, S.B.; Reitman, D.; Mandel, F.S. Course and outcome of chronic fatigue in children and adolescents. Pediatrics 1998, 102, 360–366. [Google Scholar] [CrossRef]
- Bell, D.; Jordan, K.; Robinson, M. Thirteen-year follow-up of children and adolescents with chronic fatigue syndrome. Pediatrics 2001, 107, 994–998. [Google Scholar] [PubMed]
- Andersen, M.; Permin, H.; Albrecht, F. Illness and disability in Danish chronic fatigue syndrome patients at diagnosis and 5-year follow-up. J. Psychosom. Res. 2004, 56, 217–229. [Google Scholar] [CrossRef]
- van Geelen, S.M.; Bakker, R.J.; Kuis, W.; van de Putte, E.M. Adolescent chronic fatigue syndrome: A follow-up study. Arch. Pediatrics Adolesc. Med. 2010, 164, 810–814. [Google Scholar] [CrossRef]
- Rangel, L.; Garralda, M.; Levin, M.; Roberts, H. The course of severe chronic fatigue syndrome in childhood. J. R. Soc. Med. 2000, 93, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Norris, T.; Collin, S.M.; Tilling, K.; Nuevo, R.; Stansfeld, S.A.; Sterne, J.A.; Heron, J.; Crawley, E. Natural course of chronic fatigue syndrome/myalgic encephalomyelitis in adolescents. Arch. Dis. Child. 2017, 102, 522–528. [Google Scholar] [CrossRef]
- Lim, A.; Lubitz, L. Chronic fatigue syndrome: Successful outcome of an intensive inpatient programme. J. Paediatr. Child Health 2002, 38, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Moore, Y.; Serafimova, T.; Anderson, N.; King, H.; Richards, A.; Brigden, A.; Sinai, P.; Higgins, J.; Ascough, C.; Clery, P.; et al. Recovery from chronic fatigue syndrome: A systematic review—Heterogeneity of definition limits study comparison. Arch. Dis. Child. 2021. Epub ahead of print: 12 April 2021. [Google Scholar] [CrossRef]
- Crawley, E. The epidemiology of chronic fatigue syndrome/myalgic encephalitis in children. Arch. Dis. Child. 2014, 99, 171–174. [Google Scholar] [CrossRef]
- ter Wolbeek, M.; Van Doornen, L.J.; Kavelaars, A.; Heijnen, C.J. Predictors of persistent and new-onset fatigue in adolescent girls. Pediatrics 2008, 121, e449–e457. [Google Scholar] [CrossRef]
- Viner, R.M.; Clark, C.; Taylor, S.J.; Bhui, K.; Klineberg, E.; Head, J.; Booy, R.; Stansfeld, S.A. Longitudinal risk factors for persistent fatigue in adolescents. Arch. Pediatrics Adolesc. Med. 2008, 162, 469–475. [Google Scholar] [CrossRef]
- Carter, B.D.; Marshall, G.S. New developments: Diagnosis and management of chronic fatigue in children and adolescents. Curr. Probl. Pediatrics 1995, 25, 281–293. [Google Scholar] [CrossRef]
- Josev, E.K.; Malpas, C.B.; Seal, M.L.; Scheinberg, A.; Lubitz, L.; Rowe, K.; Knight, S.J. Resting-state functional connectivity, cognition, and fatigue in response to cognitive exertion: A novel study in adolescents with chronic fatigue syndrome. Brain Imaging Behav. 2020, 14, 1815–1830. [Google Scholar] [CrossRef]
- Jason, L.; Jordan, K.; Miike, T.; Bell, D.; Lapp, C.; Torres-Harding, S.; Rowe, K.; Gurwitt, A.; De Meirleir, K.; Van Hoof, E. A pediatric case definition for myalgic encephalomyelitis and chronic fatigue syndrome. J. Chronic Fatigue Syndr. 2006, 13, 1–44. [Google Scholar] [CrossRef]
- Jason, L.; Porter, N.; Shelleby, E.; Till, L.; Bell, D.; Lapp, C.; Rowe, K.; Meirleir, K.D. Examining criteria to diagnose ME/CFS in pediatric samples. J. Behav. Health Med. 2010, 1, 186. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- Cohen, L.L.; Lemanek, K.; Blount, R.L.; Dahlquist, L.M.; Lim, C.S.; Palermo, T.M.; McKenna, K.D.; Weiss, K.E. Evidence-based assessment of pediatric pain. J. Pediatric Psychol. 2008, 33, 939–957. [Google Scholar] [CrossRef]
- Crichton, A.; Knight, S.; Oakley, E.; Babl, F.; Anderson, V. Fatigue in child chronic health conditions: A systematic review of assessment instruments. Pediatrics 2015, 135, e1015–e1031. [Google Scholar] [CrossRef]
- Deale, A.; Wessely, S. Patients’ perceptions of medical care in chronic fatigue syndrome. Soc. Sci. Med. 2001, 52, 1859–1864. [Google Scholar] [CrossRef]
- Huber, N.L.; Nicoletta, A.; Ellis, J.M.; Everhart, D.E. Validating the Adolescent Sleep Wake Scale for use with young adults. Sleep Med. 2020, 69, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.; Harvey, A.; Hennel, S.; Lubitz, L.; Rowe, K.; Reveley, C.; Dean, N.; Clarke, C.; Scheinberg, A. Measuring quality of life and fatigue in adolescents with chronic fatigue syndrome: Estimates of feasibility, internal consistency and parent–adolescent agreement of the PedsQL™. Fatigue Biomed. Health Behav. 2015, 3, 220–234. [Google Scholar] [CrossRef]
- Storfer-Isser, A.; Lebourgeois, M.K.; Harsh, J.; Tompsett, C.J.; Redline, S. Psychometric properties of the A dolescent S leep H ygiene S cale. J. Sleep Res. 2013, 22, 707–716. [Google Scholar] [CrossRef]
- White, D.; Leach, C.; Sims, R.; Atkinson, M.; Cottrell, D. Validation of the Hospital Anxiety and Depression Scale for use with adolescents. Br. J. Psychiatry 1999, 175, 452–454. [Google Scholar] [CrossRef]
- Varni, J.W.; Seid, M.; Kurtin, P.S. PedsQL™ 4.0: Reliability and validity of the Pediatric Quality of Life Inventory™ Version 4.0 Generic Core Scales in healthy and patient populations. Med. Care 2001, 39, 800–812. [Google Scholar] [CrossRef]
- Varni, J.W.; Limbers, C.A. The PedsQL Multidimensional Fatigue Scale in young adults: Feasibility, reliability and validity in a University student population. Qual. Life Res. 2008, 17, 105–114. [Google Scholar] [CrossRef]
- Varni, J.W.; Burwinkle, T.M.; Katz, E.R.; Meeske, K.; Dickinson, P. The PedsQL™ in pediatric cancer: Reliability and validity of the pediatric quality of life inventory™ generic core scales, multidimensional fatigue scale, and cancer module. Cancer 2002, 94, 2090–2106. [Google Scholar] [CrossRef] [PubMed]
- LeBourgeois, M.K.; Giannotti, F.; Cortesi, F.; Wolfson, A.R.; Harsh, J. The relationship between reported sleep quality and sleep hygiene in Italian and American adolescents. Pediatrics 2005, 115, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Thompson, K.L.; Hanson, V. The Varni/Thompson Pediatric Pain Questionnaire. I. Chronic musculoskeletal pain in juvenile rheumatoid arthritis. Pain 1987, 28, 27–38. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- Jason, L.; Evans, M.; Porter, N.; Brown, M.; Brown, A.; Hunnell, J.; Anderson, V.; Lerch, A.; Meirleir, K.D.; Friedberg, F. The Development of a Revised Canadian Myalgic Encephalomyelitis Chronic Fatigue Syndrome Case Definition. Am. J. Biochem. Biotechnol. 2010, 6, 120–135. [Google Scholar] [CrossRef]
- Jason, L.; Porter, N.; Shelleby, E.; Till, L.; Bell, D.S.; Lapp, C.W.; Rowe, K.; De Meirleir, K. Severe Versus Moderate Criteria for the New Pediatric Case Definition for ME/CFS. Child Psychiatry Hum. Dev. 2009, 40, 609–620. [Google Scholar] [CrossRef]
- Wechsler, D. Wechsler Abbreviated Scale of Intelligence (WASI-II), 2nd ed.; The Psychological Corporation: San Antonio, TX, USA, 2011. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Department of Psychology. New York University: New York, NY, USA, 1988; pp. 1–567. [Google Scholar]
- Sawilowsky, S.S. New effect size rules of thumb. J. Mod. Appl. Stat. Methods 2009, 8, 26. [Google Scholar] [CrossRef]
- Rosenthal, J.A. Qualitative Descriptors of Strength of Association and Effect Size. J. Soc. Serv. Res. 1996, 21, 37–59. [Google Scholar] [CrossRef]
- Snodgrass, K.; Harvey, A.; Scheinberg, A.; Knight, S. Sleep disturbances in pediatric chronic fatigue syndrome: A review of current research. J. Clin. Sleep Med. 2015, 11, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Roma, M.; Marden, C.; Flaherty, M.; Jasion, S.; Cranston, E.; Rowe, P. Impaired health-related quality of life in adolescent myalgic encephalomyelitis/chronic fatigue syndrome: The impact of core symptoms. Front. Pediatrics 2019, 7, 26. [Google Scholar] [CrossRef]
- Bell, D.; Bell, K.; Cheney, P. Primary juvenile fibromyalgia syndrome and chronic fatigue syndrome in adolescents. Clin. Infect. Dis. 1994, 18, S21–S23. [Google Scholar] [CrossRef]
- Jackson, M.L.; Bruck, D. Sleep abnormalities in chronic fatigue syndrome/myalgic encephalomyelitis: A review. J. Clin. Sleep Med. 2012, 8, 719–728. [Google Scholar] [CrossRef]
- Garralda, E.; Rangel, L.; Levin, M.; Roberts, H.; Ukoumunne, O. Psychiatric adjustment in adolescents with a history of chronic fatigue syndrome. J. Am. Acad. Child Adolesc. Psychiatry 1999, 38, 1515–1521. [Google Scholar] [CrossRef]
- Smith, M.S.; Martin-Herz, S.P.; Womack, W.M.; Marsigan, J.L. Comparative study of anxiety, depression, somatization, functional disability, and illness attribution in adolescents with chronic fatigue or migraine. Pediatrics 2003, 111, e376–e381. [Google Scholar] [CrossRef]
- van Middendorp, H.; Geenen, R.; Kuis, W.; Heijnen, C.J.; Sinnema, G. Psychological adjustment of adolescent girls with chronic fatigue syndrome. Pediatrics 2001, 107, E35. [Google Scholar] [CrossRef] [PubMed]
- Sulheim, D.; Hurum, H.; Helland, I.B.; Thaulow, E.; Wyller, V.B. Adolescent chronic fatigue syndrome; a follow-up study displays concurrent improvement of circulatory abnormalities and clinical symptoms. Biopsychosoc. Med. 2012, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Walford, G.; Nelson, W.; McCluskey, D. Fatigue, depression, and social adjustment in chronic fatigue syndrome. Arch. Dis. Child. 1993, 68, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Adamczyk, L.; Burgess, M.; Chalder, T. Psychological and demographic factors associated with fatigue and social adjustment in young people with severe chronic fatigue syndrome/myalgic encephalomyelitis: A preliminary mixed-methods study. J. Behav. Med. 2019, 42, 898–910. [Google Scholar] [CrossRef]
- Loades, M.E.; Rimes, K.A.; Ali, S.; Chalder, T. Depressive symptoms in adolescents with chronic fatigue syndrome (CFS): Are rates higher than in controls and do depressive symptoms affect outcome? Clin. Child Psychol. Psychiatry 2019, 24, 580–592. [Google Scholar] [CrossRef]
- Williams, T.E.; Chalder, T.; Sharpe, M.; White, P.D. Heterogeneity in chronic fatigue syndrome–empirically defined subgroups from the PACE trial. Psychol. Med. 2017, 47, 1454–1465. [Google Scholar] [CrossRef]
- Morris, G.; Maes, M. Case definitions and diagnostic criteria for Myalgic Encephalomyelitis and Chronic fatigue Syndrome: From clinical-consensus to evidence-based case definitions. Neuroendocrinol. Lett. 2013, 34, 185–199. [Google Scholar]
- Collin, S.M.; Nuevo, R.; Van De Putte, E.M.; Nijhof, S.L.; Crawley, E. Chronic fatigue syndrome (CFS) or myalgic encephalomyelitis (ME) is different in children compared to in adults: A study of UK and Dutch clinical cohorts. BMJ Open 2015, 5, e008830. [Google Scholar] [CrossRef]
- May, M.; Emond, A.; Crawley, E. Phenotypes of chronic fatigue syndrome in children and young people. Arch. Dis. Child. 2010, 95, 245–249. [Google Scholar] [CrossRef]
- van de Putte, E.M.; Engelbert, R.; Kuis, W.; Kimpen, J.; Uiterwaal, C.S. How fatigue is related to other somatic symptoms. Arch. Dis. Child. 2006, 91, 824–827. [Google Scholar] [CrossRef][Green Version]
- Winger, A.; Kvarstein, G.; Wyller, V.B.; Sulheim, D.; Fagermoen, E.; Småstuen, M.C.; Helseth, S. Pain and pressure pain thresholds in adolescents with chronic fatigue syndrome and healthy controls: A cross-sectional study. BMJ Open 2014, 4, e005920. [Google Scholar] [CrossRef][Green Version]
- Nijhof, S.L.; Priesterbach, L.P.; Bleijenberg, G.; Engelbert, R.H.; van de Putte, E.M. Functional improvement is accompanied by reduced pain in adolescent chronic fatigue syndrome. Pain Med. 2013, 14, 1435–1438. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lievesley, K.; Rimes, K.A.; Chalder, T. A review of the predisposing, precipitating and perpetuating factors in Chronic Fatigue Syndrome in children and adolescents. Clin. Psychol. Rev. 2014, 34, 233–248. [Google Scholar] [CrossRef]
- Collin, S.M.; Norris, T.; Gringras, P.; Blair, P.S.; Tilling, K.; Crawley, E. Childhood sleep and adolescent chronic fatigue syndrome (CFS/ME): Evidence of associations in a UK birth cohort. Sleep Med. 2018, 46, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Collin, S.M.; Norris, T.; Joinson, C.; Loades, M.E.; Lewis, G.; Stansfeld, S.A.; Crawley, E. Depressive symptoms at age 9–13 and chronic disabling fatigue at age 16: A longitudinal study. J. Adolesc. 2019, 75, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Barnett, A.G.; Van Der Pols, J.C.; Dobson, A.J. Regression to the mean: What it is and how to deal with it. Int. J. Epidemiol. 2005, 34, 215–220. [Google Scholar] [CrossRef]
- Ter Wolbeek, M.; Van Doornen, L.J.; Kavelaars, A.; Tersteeg-Kamperman, M.D.; Heijnen, C.J. Fatigue, depressive symptoms, and anxiety from adolescence up to young adulthood: A longitudinal study. Brain Behav. Immun. 2011, 25, 1249–1255. [Google Scholar] [CrossRef]
| Measure Domain | Name of Measure | Description of Measure |
|---|---|---|
| Fatigue | PedsQL™ Multidimensional Fatigue Scale [47,48] | 18-item Likert-rated scale (from ‘Never’ or 0 to ‘Almost always’ or 4) that assesses level of subjective fatigue over the past month. Items reversed scored, linearly transformed to a 0–100 scale, and summed over the number of items answered to form a Total Fatigue score. Higher total fatigue scores reflected fewer problems related to fatigue. |
| Sleep quality and sleep hygiene | Adolescent Sleep Wake Scale (ASWS) and Adolescent Sleep Hygiene Scale (ASHS) [49] | Two 28-item instruments that assess aspects of sleep over past month: ASWS assesses subjective sleep quality including evaluation of sleep initiation and maintenance; ASHS assesses sleep hygiene and sleep practices. Items measured on a 6-point Likert scale (1 = always; 6 = never). Higher total scores indicate better sleep quality and hygiene. |
| Pain | PedsQL™ Pediatric Pain Questionnaire Visual Analogue Scale [50] | Self-rated 100 mm scale to measure intensity of present pain, from ‘not hurting’ or ‘no pain’ (0) to ‘hurting a whole lot’ or ‘severe pain’ (100). |
| Depression and Anxiety | Hospital Anxiety and Depression Scale [51] | Consists of 14 items (7 in each subscale) and each item is scored from 0 to 3. Higher total scores indicate greater levels of depression and anxiety. |
| Health-related quality of life (HRQOL) | PedsQL™ Core Generic Module [46,47] | Widely-used measure of health-related quality of life (HRQOL) assessing subjective impact of health status on wellbeing and life satisfaction. Respondents rate 23 items on 5-point Likert scale (0 = never a problem; 4 = almost always a problem) according to how much of a problem each item has been over the previous month. Items reversed scored and linearly transformed to create a total score ranging between 0 and 100. Higher total scores indicate better perceived HRQOL. |
| Participant Characteristics | ME/CFS (n = 17) | Controls (n = 17) | Independent t-test | p-Value |
|---|---|---|---|---|
| Age in years ((M (SD; range)) | ||||
| Baseline | 15.99 (1.59; 13.42–18.92) | 15.90 (1.60; 13.33–18.08) | 0.17 | 0.86 |
| Follow-up | 18.78 (1.63; 15.5–21.58) | 18.20 (1.56; 15.58–20.58) | 1.07 | 0.29 |
| Female sex (%, n) | 82%, 14 | 65%, 11 | Χ2 = 1.36 | 0.24 |
| Socio-economic Indexes for Areas (SEIFA) (M (range)) * | 7.12 (1–10) | 7.81 (1–10) | −0.73 | 0.47 |
| Follow-up time interval in years (M (SD; range)) | 2.75 (0.81; 1.83–4.58) | 2.27 (0.43; 1.67–3) | 2.14 | 0.04 |
| Estimated FSIQ (M (SD; range)) ** | ||||
| Baseline | 103.75 (13.67; 86–145) | 107.71 (12.50; 89–130) | −0.87 | 0.39 |
| Follow-up | 105.56 (11.41; 90–136) | 109.94 (12.98; 81–129) | −1.03 | 0.31 |
| Time from symptom onset to diagnosis (study enrolment) (%, n) | - | - | - | |
| 3–6 months | 24%, 4 | - | - | - |
| 7–12 months | 29%, 5 | - | - | - |
| 13–24 months | 24%, 4 | - | - | - |
| >24 months | 24%, 4 | - | - | - |
| Perceived illness trigger at study enrolment (%, n) ** × | ||||
| Infectious Illness | 41%, 7 | - | - | - |
| Accident | 12%, 2 | - | - | - |
| Severe stress | 12%, 2 | - | - | - |
| Immunisation | 6%, 1 | - | - | - |
| Trip or vacation | 0%, 0 | - | - | - |
| No identifiable trigger | 24%, 4 | - | - | - |
| Visited health professional or specialist between baseline and follow-up × | ||||
| No | 24%, 4 | |||
| Yes | 76%, 13 | |||
| General Practitioner | 47%, 8 | |||
| Paediatrician | 41%, 7 | |||
| Physiotherapist | 29%, 5 | |||
| Psychologist | 29%, 5 | |||
| Cardiologist | 12%, 2 | |||
| Gynaecologist | 12%, 2 | |||
| Psychiatrist | 6%, 1 | |||
| Neurologist | 6%, 1 | |||
| Gastroenterologist | 6%, 1 | |||
| Naturopath | 6%, 1 | |||
| Number of visits to that health professional or specialist between baseline and follow-up | ||||
| 0 visits | 24%, 4 | |||
| 1 visit | 0%, 0 | |||
| 2 visits | 12%, 2 | |||
| 3 visits | 0%, 0 | |||
| >3 visits | 65%, 11 | |||
| Measures of Health and Psychological Wellbeing | Estimated Mean Difference (b) with 95% CIs | SE | p-Value | Effect Size (Cohen’s d) |
|---|---|---|---|---|
| Fatigue | ||||
| Time | 17.89 (8.83, 26.96) | 4.62 | <0.001 | 0.66 |
| Group | 41.62 (32.10, 51.14) | 4.86 | <0.001 | 1.47 |
| Time × Group | −21.73 (−34.55, −8.91) | 6.54 | 0.001 | 0.57 |
| Sleep quality | ||||
| Time | 0.24 (0.02, 0.45) | 0.11 | 0.03 | 0.37 |
| Group | 0.66 (0.33, 0.99) | 0.17 | <0.001 | 0.67 |
| Time × Group | −0.34 (−0.64, −0.03) | 0.16 | 0.03 | 0.37 |
| Sleep hygiene | ||||
| Time | 0.05 (−0.13, 0.23) | 0.09 | 0.60 | 0.09 |
| Group | 0.20 (−0.11, 0.50) | 0.16 | 0.20 | 0.22 |
| Time × Group | −0.29 (−0.55, −0.03) | 0.13 | 0.03 | 0.38 |
| Pain | ||||
| Time | −14.24 (−24.23, −4.24) | 5.10 | <0.01 | 0.48 |
| Group | −33.97 (−48.14, −19.80) | 7.23 | <0.001 | 0.81 |
| Time × Group | 14.57 (0.33, 28.61) | 7.21 | 0.045 | 0.34 |
| Anxiety | ||||
| Time | −3.41 (−4.81, −2.02) | 0.71 | <0.001 | 0.82 |
| Group | −4.35 (−6.42, −2.29) | 1.05 | <0.001 | 0.71 |
| Time × Group | 3.59 (1.62, 5.56) | 1.01 | <0.001 | 0.61 |
| Depression | ||||
| Time | −0.35 (−2.18, 1.48) | 0.93 | 0.71 | 0.06 |
| Group | −2.00 (−4.84, 0.84) | 1.45 | 0.17 | 0.24 |
| Time × Group | 1.59 (−1.00, 4.18) | 1.32 | 0.23 | 0.21 |
| HRQOL | ||||
| Time | 13.19 (6.48, 19.91) | 3.43 | <0.001 | 0.66 |
| Group | 34.75 (24.95, 44.55) | 5.00 | <0.001 | 1.19 |
| Time × Group | −18.43 (−27.93, −8.94) | 4.84 | <0.001 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Josev, E.K.; Cole, R.C.; Scheinberg, A.; Rowe, K.; Lubitz, L.; Knight, S.J. Health, Wellbeing, and Prognosis of Australian Adolescents with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Case-Controlled Follow-Up Study. J. Clin. Med. 2021, 10, 3603. https://doi.org/10.3390/jcm10163603
Josev EK, Cole RC, Scheinberg A, Rowe K, Lubitz L, Knight SJ. Health, Wellbeing, and Prognosis of Australian Adolescents with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Case-Controlled Follow-Up Study. Journal of Clinical Medicine. 2021; 10(16):3603. https://doi.org/10.3390/jcm10163603
Chicago/Turabian StyleJosev, Elisha K., Rebecca C. Cole, Adam Scheinberg, Katherine Rowe, Lionel Lubitz, and Sarah J. Knight. 2021. "Health, Wellbeing, and Prognosis of Australian Adolescents with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Case-Controlled Follow-Up Study" Journal of Clinical Medicine 10, no. 16: 3603. https://doi.org/10.3390/jcm10163603
APA StyleJosev, E. K., Cole, R. C., Scheinberg, A., Rowe, K., Lubitz, L., & Knight, S. J. (2021). Health, Wellbeing, and Prognosis of Australian Adolescents with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Case-Controlled Follow-Up Study. Journal of Clinical Medicine, 10(16), 3603. https://doi.org/10.3390/jcm10163603






