The Role of Antioxidants on Wound Healing: A Review of the Current Evidence
Abstract
:1. Introduction
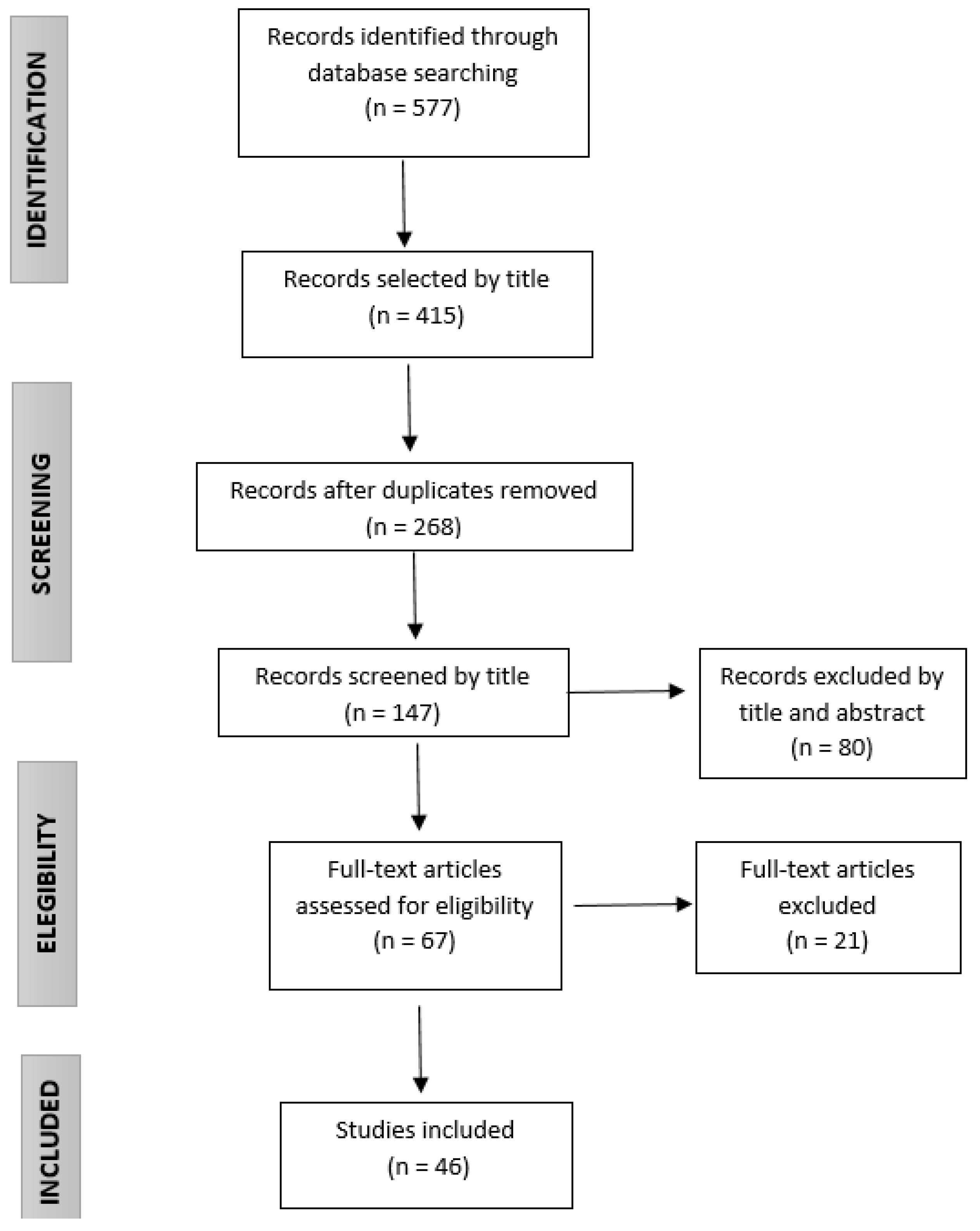
2. Methods
3. Results
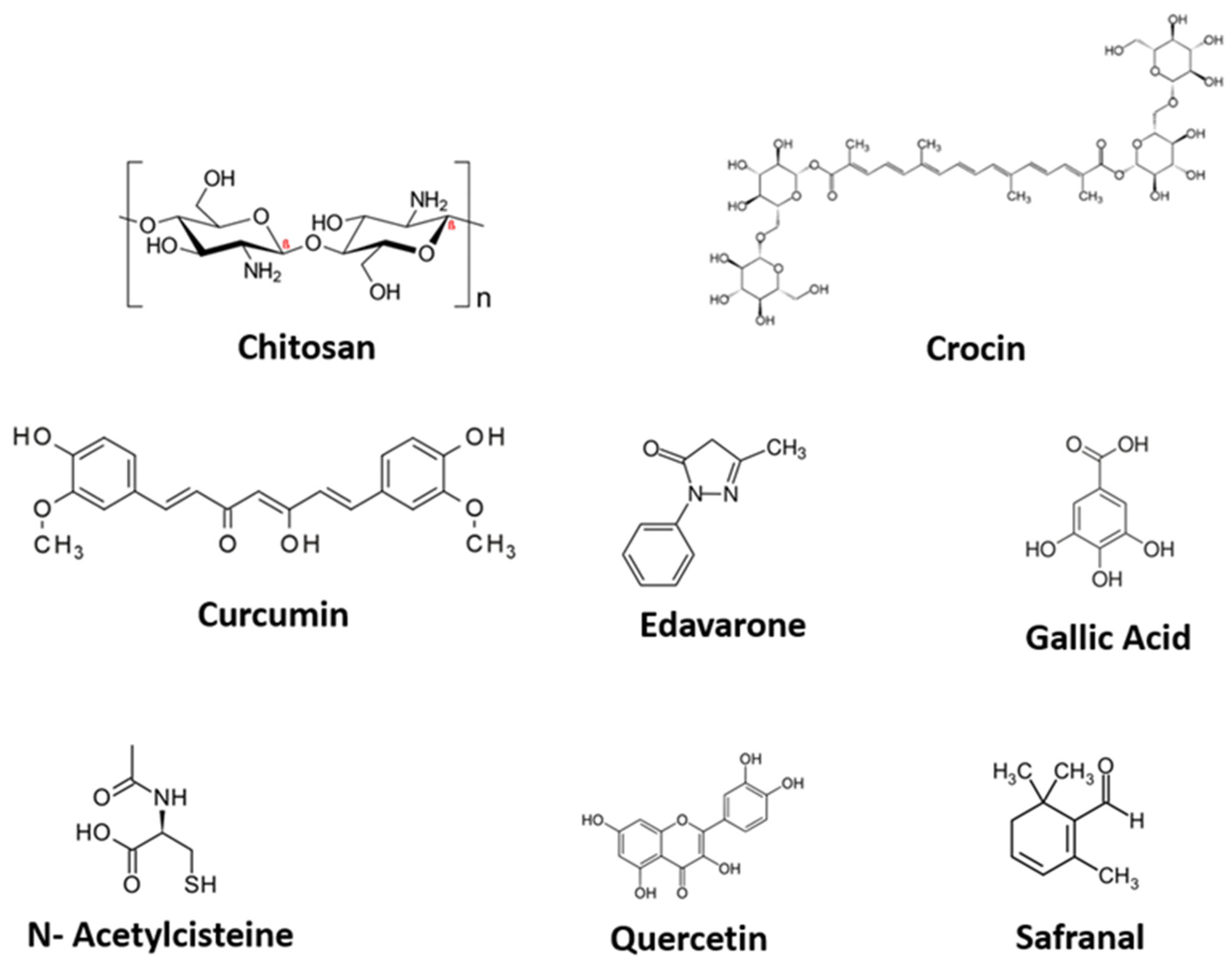
3.1. Compounds with Antioxidant Activity
3.1.1. Curcumin
3.1.2. N-Acetyl Cysteine
3.1.3. Chitosan
3.1.4. Gallic Acid
3.1.5. Edaravone
3.1.6. Crocin and Safranal
3.1.7. Quercetin
3.2. Effects on the Healing Process of Antioxidant Compounds
3.2.1. Curcumin
3.2.2. Curcumin and Chitosan
3.2.3. Curcumin and N-Acetyl Cysteine
3.2.4. N-Acetyl Cysteine and α-Tocopherol
3.2.5. N-Acetyl Cysteine
3.2.6. Chitosan
3.2.7. Chitosan and Plant Extracts
3.2.8. Chitosan and Gallic Acid
3.2.9. Edaravone
3.2.10. Crocin and Safranal
3.2.11. Quercetin
3.2.12. Quercetin and Oleic Acid
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Proksch, E.; Brandner, J.M.; Jensen, J.M. The skin: An indispensable barrier. Exp. Dermatol. 2008, 17, 1063–1072. [Google Scholar] [CrossRef]
- Enoch, S.; Leaper, D.J. Basic science of wound healing. Surgery 2008, 26, 31–37. [Google Scholar] [CrossRef]
- Rosenbaum, A.J.; Banerjee, S.; Rezak, K.M.; Uhl, R.L. Advances in Wound Management. J. Am. Acad. Orthop. Surg. 2018, 26, 833–843. [Google Scholar] [CrossRef]
- Velnar, T.; Bailey, T.; Smrkolj, V. The wound healing process: An overview of the cellular and molecular mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef] [PubMed]
- Atkin, L.; Bućko, Z.; Conde Montero, E.; Cutting, K.; Moffatt, C.; Probst, A.; Romanelli, M.; Schultz, G.S.; Tettelbach, W. Implementing TIMERS: The race against hard-to-heal wounds. J. Wound Care. 2019, 28, 1–49. [Google Scholar] [CrossRef] [Green Version]
- Scialò, F.; Fernández-Ayala, D.J.; Sanz, A. Role of Mitochondrial Reverse Electron Transport in ROS Signaling: Potential Roles in Health and Disease. Front. Physiol. 2017, 8, 428. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.Z.; Jiang, S.; Zhang, L.; Yu, Z.B. Mitochondrial electron transport chain, ROS generation and uncoupling (Review). Int. J. Mol. Med. 2019, 44, 3–15. [Google Scholar] [CrossRef] [Green Version]
- Beckman, K.B.; Ames, B.N. The free radical theory of aging matures. Physiol. Rev. 1998, 78, 547–581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez, P.G.; Felix, F.N.; Woodley, D.T.; Shim, E.K. The role of oxygen in wound healing: A review of the literature. Dermatol. Surg. 2008, 34, 1159–1169. [Google Scholar] [CrossRef]
- Cano, S.M.; Lancel, S.; Boulanger, E.; Neviere, R. Targeting Oxidative Stress and Mitochondrial Dysfunction in the Treatment of Impaired Wound Healing: A Systematic Review. Antioxidants 2018, 7, 98. [Google Scholar] [CrossRef] [Green Version]
- Ponugoti, B.; Xu, F.; Zhang, C.; Tian, C.; Pacios, S.; Graves, D.T. FOXO1 promotes wound healing through the up-regulation of TGF-beta1 and prevention of oxidative stress. J. Cell Biol. 2013, 203, 327–343. [Google Scholar] [CrossRef] [Green Version]
- Johnson, J.B.; Broszczak, D.A.; Mani, J.S.; Anesi, J.; Naiker, M. A cut above the rest: Oxidative stress in chronic wounds and the potential role of polyphenols as therapeutics. J. Pharm. Pharmacol. 2021, 3, rgab038. [Google Scholar] [CrossRef] [PubMed]
- Andre-Levigne, D.; Modarressi, A.; Pepper, M.S.; Pittet-Cuenod, B. Reactive oxygen species and nox enzymes are emerging as key players in cutaneous wound repais. Int. J. Med. Sci. 2017, 18, 2149. [Google Scholar] [CrossRef] [Green Version]
- Klyubin, I.V.; Kirpichnikova, K.M.; Gamaley, I.A. Hydrogen peroxide-induced chemotaxis of mouse peritoneal neutrophils. Eur. J. Cell Biol. 1996, 70, 347–351. [Google Scholar]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D. Georgopoulos NT.Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef] [Green Version]
- Fitzmaurice, S.D.; Sivamani, R.K.; Isseroff, R.R. Antioxidant therapies for wound healing: A clinical guide to currently commercially available products. Skin Pharmacol. Physiol. 2011, 24, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Nqoro, X.; Aderibigbe, B.A. Polymer-Based Materials Loaded with Curcumin for Wound Healing Applications. Polymers 2020, 12, 2286. [Google Scholar] [CrossRef]
- Zhao, Y.; Dai, C.; Wang, Z.; Chen, W.; Liu, J.; Zhuo, R.; Yu, A.; Huang, S. A novel curcumin-loaded composite dressing facilitates wound healing due to its natural antioxidant effect. Drug Des. Devel Ther. 2019, 13, 3269–3280. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, N.; Wong, S.K.; Mohamed, I.N.; Mohamed, N.; Chin, K.Y.; Ima-Nirwana, S.; Shuid, A.N. Wound Healing Properties of Selected Natural Products. Int. J. Environ Res. Public Health 2018, 15, 2360. [Google Scholar] [CrossRef] [Green Version]
- Merrell, J.G.; McLaughlin, S.W.; Tie, L.; Laurencin, C.T.; Chen, A.F.; Nair, L.S. Curcumin-loaded poly(epsilon-caprolactone) nanofibres: Diabetic wound dressing with anti-oxidant and anti-inflammatory properties. Clin. Exp. Pharmacol. Physiol. 2009, 36, 1149–1156. [Google Scholar] [CrossRef] [Green Version]
- Kregiel, D.; Rygala, A.; Kolesinska, B.; Nowacka, M.; Herc, A.S.; Kowalewska, A. Antimicrobial and Antibiofilm N-acetyl-L-cysteine Grafted Siloxane Polymers with Potential for Use in Water Systems. Int. J. Mol. Sci. 2019, 20, 2011. [Google Scholar] [CrossRef] [Green Version]
- Ozkaya, H.; Omma, T.; Bag, Y.M.; Uzunoglu, K.; Isildak, M.; Duymus, M.E.; Kismet, K.; Senes, M.; Fidanci, V.; Celepli, P.; et al. Topical and Systemic Effects of N-acetyl Cysteine on Wound Healing in a Diabetic Rat Model. Wounds 2019, 31, 91–96. [Google Scholar]
- Tsai, M.L.; Huang, H.P.; Hsu, J.D.; Lai, Y.R.; Hsiao, Y.P.; Lu, F.J.; Chang, H.R. Topical N-acetylcysteine accelerates wound healing in vitro and in vivo via the PKC/Stat3 pathway. Int. J. Mol. Sci. 2014, 15, 7563–7578. [Google Scholar] [CrossRef] [Green Version]
- Schoukens, G. Bioactive Dressings to Promote Wound Healing. In Woodhead Publishing Series in Textiles, Advanced Textiles for Wound Care, 2nd ed.; Rajendran, S., Ed.; Woodhead: London, UK, 2009; pp. 114–152. [Google Scholar] [CrossRef]
- Patrulea, V.; Ostafe, V.; Borchard, G.; Jordan, O. Chitosan as a starting material for wound healing applications. Eur. J. Pharm. Biopharm. 2015, 97, 417–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colobatiu, L.; Gavan, A.; Mocan, A.; Bogdan, C.; Mirel, S.; Tomuta, I. Development of bioactive compounds-loaded chitosan films by using a QbD approach—A novel and potential wound dressing material. React. Funct. Polym. 2019, 138, 46–54. [Google Scholar] [CrossRef]
- Colobatiu, L.; Gavan, A.; Potarniche, A.V.; Rus, V.; Diaconesa, Z.; Mocan, A.; Tomuta, I.; Mirel, S.; Mihaiu, M. Evaluation of bioactive compounds-loaded chitosan films as a novel and potential diabetic wound dressing material. React. Funct. Polym. 2019, 145, 104369. [Google Scholar] [CrossRef]
- Liu, H.; Qu, X.; Kim, E.; Lei, M.; Dai, K.; Tan, X.; Xu, M.; Li, J.; Liu, Y.; Shi, X.; et al. Bio-inspired redox-cycling antimicrobial film for sustained generation of reactive oxygen species. Biomaterials 2018, 162, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Stefanov, I.; Pérez-Rafael, S.; Hoyo, J.; Cailloux, J.; Santana Pérez, O.O.; Hinojosa-Caballero, D.; Tzanov, T. Multifunctional Enzymatically Generated Hydrogels for Chronic Wound Application. Biomacromolecules 2017, 18, 1544–1555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thi, P.L.; Lee, Y.; Tran, D.L.; Thi, T.T.H.; Kang, J.I.; Park, K.M.; Park, K.D. In situ forming and reactive oxygen species-scavenging gelatin hydrogels for enhancing wound healing efficacy. Acta Biomater. 2020, 103, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Wu, W.; Lei, Y.; Gaucher, C.; Pei, S.; Zhang, J.; Xia, X. Edaravone-Loaded Alginate-Based Nanocomposite Hydrogel Accelerated Chronic Wound Healing in Diabetic Mice. Mar. Drugs 2019, 17, 285. [Google Scholar] [CrossRef] [Green Version]
- Tamer, T.M.; Valachová, K.; Hassan, M.A.; Omer, A.M.; El-Shafeey, M.; Mohy Eldin, M.S.; Šoltés, L. Chitosan/hyaluronan/edaravone membranes for anti-inflammatory wound dressing: In vitro and in vivo evaluation studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 90, 227–235. [Google Scholar] [CrossRef]
- Khorasani, G.; Hosseinimehr, S.J.; Zamani, P.; Ghasemi, M.; Ahmadi, A. The effect of saffron (Crocus sativus) extract for healing of second-degree burn wounds in rats. Keio J. Med. 2008, 57, 190–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alemzadeh, E.; Oryan, A. Effectiveness of a Crocus sativus Extract on Burn Wounds in Rats. Planta Med. 2018, 84, 1191–1200. [Google Scholar] [CrossRef]
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, inflammation and immunity. Nutrients 2016, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Aceituno-Medina, M.; Mendoza, S.; Rodrıguez, B.A.; Lagaron, J.M.; López-Rubio, A. Improved antioxidant capacity of quer-cetin and ferulic acid during in-vitro digestion through encapsulation within food-grade electrospun fibers. J. Funct. Foods 2015, 12, 332–341. [Google Scholar] [CrossRef] [Green Version]
- Tran, H.A.; Ly, K.L.; Fox, K.E.; Tran, P.A.; Nguyen, T.H. Immobilization of Antimicrobial Silver and Antioxidant Flavonoid as a Coating for Wound Dressing Materials. Int. J. Nanomed. 2019, 17, 9929–9939. [Google Scholar] [CrossRef] [Green Version]
- Gomathi, K.; Gopinath, D.; Rafiuddin Ahmed, M.; Jayakumar, R. Quercetin incorporated collagen matrices for dermal wound healing processes in rat. Biomaterials 2003, 24, 2767–2772. [Google Scholar] [CrossRef]
- Yin, G.; Wang, Z.; Wang, Z.; Wang, X. Topical application of quercetin improves wound healing in pressure ulcer lesions. Exp. Dermatol. 2018, 27, 779–786. [Google Scholar] [CrossRef]
- Liu, J.; Chen, Z.; Wang, J.; Li, R.; Li, T.; Chang, M.; Yan, F.; Wang, Y. Encapsulation of Curcumin Nanoparticles with MMP9-Responsive and Thermos-Sensitive Hydrogel Improves Diabetic Wound Healing. ACS Appl. Mater. Interfaces 2018, 10, 16315–16326. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.; Wu, Q.; Wang, Y.; Zhang, D.; Luo, F.; Zhao, X.; Wei, Y.; Qian, Z. A biodegradable hydrogel system containing curcumin encapsulated in micelles for cutaneous wound healing. Biom6aterials 2013, 34, 6377–6387. [Google Scholar] [CrossRef]
- Kant, V.; Gopal, A.; Pathak, N.N.; Kumar, P.; Tandan, S.K.; Kumar, D. Antioxidant and anti-inflammatory potential of curcumin accelerated the cutaneous wound healing in streptozotocin-induced diabetic rats. Int. Immunopharmacol. 2014, 20, 322–330. [Google Scholar] [CrossRef]
- Pandey, V.K.; Ajmal, G.; Upadhyay, S.N.; Mishra, P.K. Nano-fibrous scaffold with curcumin for anti-scar wound healing. Int. J. Pharm. 2020, 589, 119858. [Google Scholar] [CrossRef]
- Abbas, M.; Hussain, T.; Arshad, M.; Ansari, A.R.; Irshad, A.; Nisar, J.; Hussain, F.; Masood, N.; Nazir, A.; Iqbal, M. Wound healing potential of curcumin cross-linked chitosan/polyvinyl alcohol. Int. J. Biol. Macromol. 2019, 140, 871–876. [Google Scholar] [CrossRef]
- Mei, L.; Fan, R.; Li, X.; Wang, Y.; Han, B.; Gu, Y.; Zhou, L.; Zheng, Y.; Tong, A.; Guo, G. Nanofibers for improving the wound repair process: The combination of a grafted chitosan and an antioxidant agent. Polym. Chem. 2017, 8, 1664–1671. [Google Scholar] [CrossRef]
- Castro, B.; Palomares, T.; Azcoitia, I.; Bastida, F.; del Olmo, M.; Soldevilla, J.J.; Alonso-Varona, A. Development and preclinical evaluation of a new galactomannan-based dressing with antioxidant properties for wound healing. Histol. Histopathol. 2015, 30, 1499–1512. [Google Scholar] [CrossRef] [PubMed]
- Castro, B.; Bastida, F.D.; Segovia, T.; López Casanova, P.; Soldevilla, J.J.; Verdú-Soriano, J. The use of an antioxidant dressing on hard-to-heal wounds: A multicentre, prospective case series. J. Wound Care 2017, 26, 742–750. [Google Scholar] [CrossRef]
- Jiménez-García, J.F.; Abad García, M.M.; Ortiz Villegas, L.; Zarco Marín, J.M.; Expósito Torres, R.; García Fernández, F.P. Cicatrización de úlceras venosas complejas mediante el control del estrés oxidativo. Rev. ROL Enferm. 2018, 41, 739–744. [Google Scholar]
- Jiménez-García, J.F.; Aguilera-Manrique, G.; Parra-Anguita, L.; Torra-Bou, J.E.; Villegas-Tripiana, I.; García-Fernández, F.P. An antioxidant approach directed to non-healing wounds activation-A case report study. Clin. Case Rep. Rev. 2021, 7, 1–6. [Google Scholar] [CrossRef]
- Dhall, S.; Do, D.C.; Garcia, M.; Kim, J.; Mirebrahim, S.H.; Lyubovitsky, J.; Lonardi, S.; Nothnagel, E.A.; Schiller, N.; Martins-Green, M. Generating and reversing chronic wounds in diabetic mice by manipulating wound redox parameters. J. Diabetes Res. 2014, 2014, 562625. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Kim, J.; Wu, J.; Ahamed, A.; Wang, Y.; Martins-Green, M. N-Acetyl-cysteine and Mechanisms Involved in Resolution of Chronic Wound Biofil. J. Diabetes Res. 2020, 2020, 9589507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oguz, A.; Uslukaya, O.; Alabalık, U.; Turkoglu, A.; Kapan, M.; Bozdag, Z. Topical N-acetylcysteine improves wound healing comparable to dexpanthenol: An experimental study. Int. Surg. 2015, 100, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Aktunc, E.; Ozacmak, V.H.; Ozacmak, H.S.; Barut, F.; Buyukates, M.; Kandemir, O.; Demircan, N. N-acetyl cysteine promotes angiogenesis and clearance of free oxygen radicals, thus improving wound healing in an alloxan-induced diabetic mouse model of incisional wound. Clin. Exp. Dermatol. 2010, 35, 902–909. [Google Scholar] [CrossRef]
- Hou, J.; Chen, L.; Liu, Z.; Li, J.; Yang, J.; Zhong, A.; Zhou, M.; Sun, Y.; Guo, L.; Yang, Y.; et al. Sustained release of N-acetylcysteine by sandwich structured polycaprolactone/collagen scaffolds for wound healing. J. Biomed. Mater. Res. A 2019, 107, 1414–1424. [Google Scholar] [CrossRef]
- Hou, J.; Chen, L.; Zhou, M.; Li, J.; Liu, J.; Fang, H.; Zeng, Y.; Sun, J.; Wang, Z. Multi-Layered Polyamide/Collagen Scaffolds with Topical Sustained Release of N-Acetylcysteine for Promoting Wound Healing. Int. J. Nanomed. 2020, 15, 1349–1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiahui, H.; Yongping, L.; Mengting, S.; Baolin, G. Anti-oxidant electroactive and antibacterial nanofibrous wound dressings based on poly(ε-caprolactone)/quaternized chitosan-graft-polyaniline for full-thickness skin wound healing. Chem. Eng. J. 2020, 385, 123464. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, Y.; Pan, X.; Chen, S.; Zhuang, H.; Wang, S. A composite hydrogel of chitosan/heparin/poly (γ-glutamic acid) loaded with superoxide dismutase for wound healing. Carbohydr. Polym. 2018, 180, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, H.; Guo, B.; Dong, R.; Qiu, Y.; Ma, P.X. Antibacterial anti-oxidant electroactive injectable hydrogel as self-healing wound dressing with hemostasis and adhesiveness for cutaneous wound healing. Biomaterials 2017, 122, 34–47. [Google Scholar] [CrossRef]
- Mo, X.; Cen, J.; Gibson, E.; Wang, R.; Percival, S.L. An open multicenter comparative randomized clinical study on chitosan. Wound Repair Regen. 2015, 23, 518–524. [Google Scholar] [CrossRef]
- Halim, A.S.; Nor, F.M.; Mat Saad, A.Z.; Mohd Nasir, N.A.; Norsa’adah, B.; Ujang, Z. Efficacy of chitosan derivative films versus hydrocolloid dressing on superficial wounds. J. Taibah Univ. Med. Sci. 2018, 13, 512–520. [Google Scholar] [CrossRef]
- Rocasalbas, G.; Francesko, A.; Touriño, S.; Fernández-Francos, X.; Guebitz, G.M.; Tzanov, T. Laccase-assisted formation of bioactive chitosan/gelatin hydrogel stabilized with plant polyphenols. Carbohydr. Polym. 2013, 92, 989–996. [Google Scholar] [CrossRef]
- Díaz-González, M.; Rocasalbas, G.; Francesko, A.; Touriño Eirín, S.; Torres, J.L.; Tzanov, T. Inhibition of deleterious chronic wound enzymes with plant polyphenols. Biocatal. Biotransform. 2012, 1, 102–110. [Google Scholar] [CrossRef]
- Bektas, N.; Şenel, B.; Yenilmez, E.; Özatik, O.; Arslana, R. Evaluation of wound healing effect of chitosan-based gel formulation containing vitexin. Saudi Pharm. J. 2020, 28, 87–94. [Google Scholar] [CrossRef]
- Singh, M.P.; Gupta, A.; Sisodia, S.S. Wound healing activity of Terminalia bellerica Roxb. and gallic acid in experimentally induced diabetic animals. J. Complement. Integr. Med. 2019, 17. [Google Scholar] [CrossRef]
- Yang, D.J.; Moh, S.H.; Son, D.H.; You, S.; Kinyua, A.W.; Ko, C.M.; Song, M.; Yeo, J.; Choi, Y.H.; Kim, K.W. Gallic Acid Promotes Wound Healing in Normal and Hyperglucidic Conditions. Molecules 2016, 21, 899. [Google Scholar] [CrossRef] [Green Version]
- Naito, R.; Nishinakamura, H.; Watanabe, T.; Nakayama, J.; Kodama, S. Edaravone, a free radical scavenger, accelerates wound healing in diabetic mice. Wounds 2014, 26, 163–171. [Google Scholar] [PubMed]
- Zeka, K.; Ruparelia, K.C.; Sansone, C.; Macchiarelli, G.; Continenza, M.A.; Arroo, R.R.J. New Hydrogels Enriched with Antioxidants from Saffron Crocus Can Find Applications in Wound Treatment and/or Beautification. Skin Pharmacol. Physiol. 2018, 31, 95–98. [Google Scholar] [CrossRef]
- Ajmal, G.; Bonde, G.V.; Thokala, S.; Mittal, P.; Khan, G.; Singh, J.; Pandey, V.K.; Mishra, B. Ciprofloxacin HCl and quercetin functionalized electrospun nanofiber membrane: Fabrication and its evaluation in full thickness wound healing. Artif. Cells Nanomed. Biotechnol. 2019, 47, 228–240. [Google Scholar] [CrossRef]
- Ajmal, G.; Bonde, G.V.; Mittal, P.; Khan, G.; Pandey, V.K.; Bakade, B.V.; Mishra, B. Biomimetic PCL-gelatin based nanofibers loaded with ciprofloxacin hydrochloride and quercetin: A potential antibacterial and anti-oxidant dressing material for accelerated healing of a full thickness wound. Int. J. Pharm. 2019, 567, 118480. [Google Scholar] [CrossRef] [PubMed]
- Kant, V.; Jangir, B.L.; Kumar, V.; Nigam, A.; Sharma, V. Quercetin accelerated cutaneous wound healing in rats by modulation of different cytokines and growth factors. Growth Factors 2020, 38, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Gallelli, G.; Cione, E.; Serra, R.; Leo, A.; Citraro, R.; Matricardi, P.; Di, M.C.; Bisceglia, F.; Caroleo, M.; Basile, S.; et al. Nano-hydrogel embedded with quercetin and oleic acid as a new formulation in the treatment of diabetic foot ulcer: A pilot study. Int. Wound J. 2020, 17, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Comino-Sanz, I.M.; López-Franco, M.D.; Castro, B.; Pancorbo-Hidalgo, P.L. Antioxidant dressing therapy versus standard wound care in chronic wounds (the REOX study): Study protocol for a randomized controlled trial. Trials 2020, 21, 505. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, J.; Li, P.; Ning, N. The Effect of Chitosan in Wound Healing: A Systematic Review. Adv. Skin Wound Care 2021, 34, 262–266. [Google Scholar] [CrossRef]
- Dryden, M. Reactive oxygen species: A novel antimicrobial. Int. J. Antimicrob. Agents 2018, 51, 299–303. [Google Scholar] [CrossRef] [PubMed]
| Compound | Type | Chemical Properties | Biological Properties | Origin |
|---|---|---|---|---|
| Curcumin | Polyphenolic | Non-water-soluble | Anti-inflammatory, antibacterial and antioxidant | Vegetal Curcuma longa rhizome |
| Chitosan | Polysaccharide (derived from chitin) | Water-soluble | Highly biocompatible Hemostatic, antibacterial and antioxidant | Animal Exoskeleton from crustaceans |
| N-acetyl Cysteine (NAC) | Sulfhydryl | Water-soluble | Precursor in the formation of glutathione (GSH), antioxidant | Modified form of the amino acid l-cysteine |
| Gallic Acid | Polyphenol | Soluble in alcohol, ether, acetone | Antioxidant, anti-inflammatory, analgesic | Vegetables, fruits, leaves and wildflowers. |
| Edaravone | 3-methyl-1-phenyl-2-pyrazolin-5-one | Soluble in hot water and hot alcohol | Free radical scavenger, antioxidant | Chemical synthesis |
| Crocin and Safranal | Carotenoid | Lipophilic (poor water solubility) | Antioxidant, anti-inflammatory, antitumoral | Vegetal of saffron crocus (Crocus Sativus) |
| Quercetin | Flavonoid (polyphenol) | Non-water-soluble | Antioxidant, anti-inflammatory | Vegetables and fruits |
| Compound | Author | Year | Carrier | Type of Study | ||
|---|---|---|---|---|---|---|
| In Vitro | Animal Model | Human | ||||
| Curcumin | Merrell [21] | 2009 | Polycaprolactone nanofibers | X | X | |
| Liu [41] | 2018 | Gelatin microspheres | X | X | ||
| Gong [42] | 2013 | Hydrogel | X | X | ||
| Kant [43] | 2014 | X | ||||
| Pandey [44] | 2020 | Polyvinyl pyrrolidone nanofibers | X | X | ||
| Curcumin + Chitosan | Zhao [19] | 2019 | β-cyclodextrin | X | X | |
| Abbas [45] | 2019 | X | ||||
| Mei [46] | 2017 | Nanofibers polypropylene | X | X | ||
| Curcumin + N-acetyl-cysteine | Castro [47] | 2015 | Galactomannan | X | X | |
| Castro [48] | 2017 | Galactomannan | X (Obs) | |||
| Jimenez-Garcia [49] | 2018 | Galactomannan | X (Obs) | |||
| Jimenez-Garcia [50] | 2021 | Galactomannan | X (Obs) | |||
| N-acetyl cysteine + α-tocopherol | Dhall [51] | 2014 | X | X | ||
| Li [52] | 2020 | Microbiota | X | |||
| N-acetyl cysteine | Ozkaya [23] | 2019 | X | |||
| Oguz [53] | 2015 | X | ||||
| Tsai [24] | 2014 | X | X | |||
| Aktunc [54] | 2010 | X | X | |||
| Hou [55] | 2019 | polycaprolactone | X | X | ||
| Hou [56] | 2020 | polyamide | X | X | ||
| Chitosan | Jiahui [57] | 2020 | polycaprolactone | X | X | |
| Zhang [58] | 2018 | hydrogel poly (γ-glutamic acid) and heparin | X | X | ||
| Liu [29] | 2018 | catechol-modified chitosan film | X | X | ||
| Zhao [59] | 2017 | Hydrogel (polyaniline) | X | X | ||
| Mo [60] | 2015 | X (Exp) | ||||
| Halim [61] | 2018 | X (Exp) | ||||
| Chitosan + plan extracts | Colobatiu [27,28] | 2019 | Polymer and polyvinyl alcohol | X | X | |
| Rocasalbas [62] | 2013 | Gelatin hydrogel | X | |||
| Díaz-González [63] | 2012 | X | ||||
| Bektas N [64] | 2020 | Hydrogel | X | X | ||
| Chitosan + gallic acid | Thi [31] | 2020 | Gelatin hydrogel | X | X | |
| Singh [65] | 2019 | X | ||||
| Yang [66] | 2016 | X | ||||
| Stefanov [30] | 2016 | X | X (ExV) | |||
| Edaravone | Fan [32] | 2019 | Alginate hydrogel | X | X | |
| Naito [67] | 2014 | X | ||||
| Crocin and Safranal | Zeka [68] | 2017 | hydrogel | X | ||
| Khorasani [34] | 2008 | X | ||||
| Alemzadeh [35] | 2018 | X | ||||
| Quercetin | Ajmal [69] | 2019 | poly (e-caprolactone) | X | ||
| Ajmal [70] | 2019 | poly (e-caprolactone) | X | |||
| Tran [38] | 2019 | X | ||||
| Gomathi [39] | 2002 | X | ||||
| Yin [40] | 2018 | X | ||||
| Kant [71] | 2020 | X | ||||
| Quercetin + Oleic acid | Gallelli [72] | 2020 | X (Exp) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Comino-Sanz, I.M.; López-Franco, M.D.; Castro, B.; Pancorbo-Hidalgo, P.L. The Role of Antioxidants on Wound Healing: A Review of the Current Evidence. J. Clin. Med. 2021, 10, 3558. https://doi.org/10.3390/jcm10163558
Comino-Sanz IM, López-Franco MD, Castro B, Pancorbo-Hidalgo PL. The Role of Antioxidants on Wound Healing: A Review of the Current Evidence. Journal of Clinical Medicine. 2021; 10(16):3558. https://doi.org/10.3390/jcm10163558
Chicago/Turabian StyleComino-Sanz, Inés María, María Dolores López-Franco, Begoña Castro, and Pedro Luis Pancorbo-Hidalgo. 2021. "The Role of Antioxidants on Wound Healing: A Review of the Current Evidence" Journal of Clinical Medicine 10, no. 16: 3558. https://doi.org/10.3390/jcm10163558
APA StyleComino-Sanz, I. M., López-Franco, M. D., Castro, B., & Pancorbo-Hidalgo, P. L. (2021). The Role of Antioxidants on Wound Healing: A Review of the Current Evidence. Journal of Clinical Medicine, 10(16), 3558. https://doi.org/10.3390/jcm10163558







