Shadow Burden of Undiagnosed Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) on Society: Retrospective and Prospective—In Light of COVID-19
Abstract
:1. Introduction
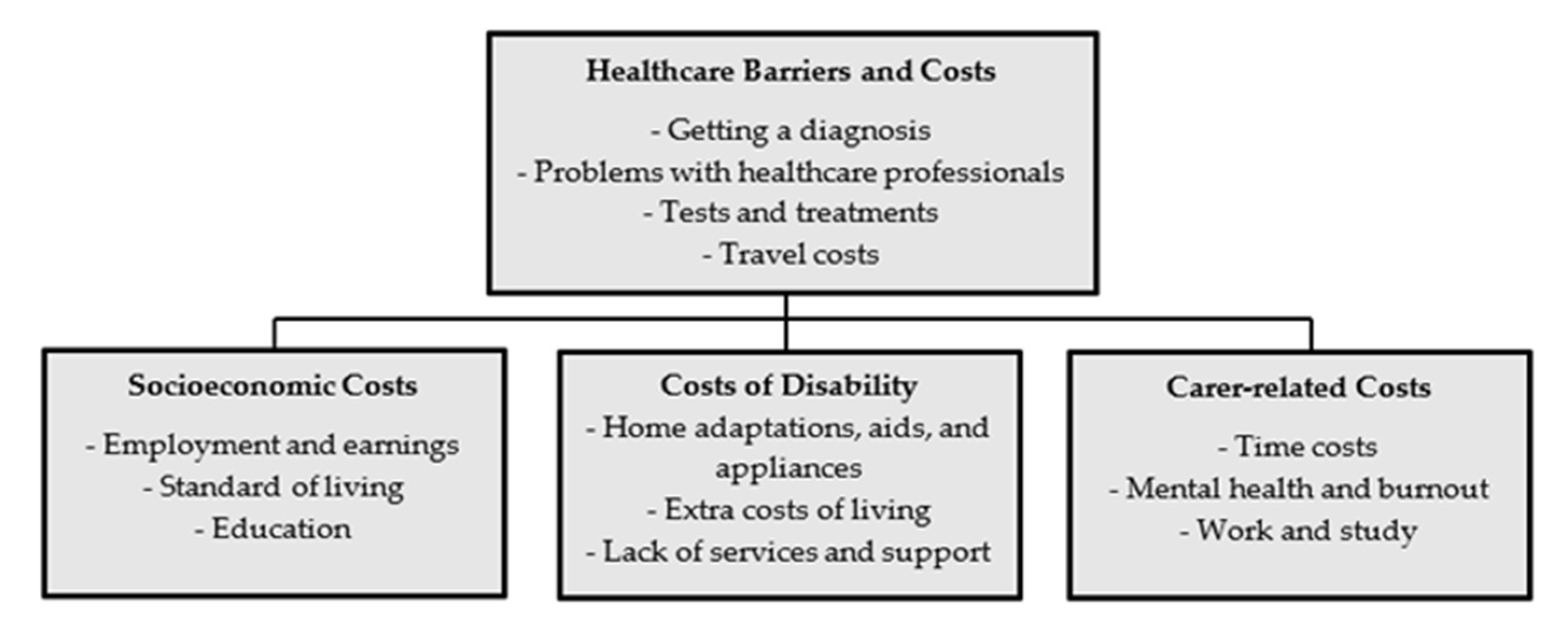
- Estimate the literature on classic factors presumably affecting the diagnostic scope of ME/CFS, and their ascription to Latvian circumstances, as well as conducting a literature review to assess the potential relationship between ME/CFS and COVID-19 as a new contributing agent, and its reflection in scientific literature;
- Analyse data from the survey performed both for ME/CFS patients and for persons experiencing symptoms similar to those of ME/CFS, but without a diagnosis (prior to the COVID-19 pandemic), in order to compare the certain socioeconomic and disease management aspects for patients and potential undiagnosed patients. Data from the ME/CFS patients’ survey were previously analysed in a comparative study with Italy and the UK. Conversely, the data from undiagnosed patients were not analysed previously; nevertheless, these data create significant potential for assessing the shadow impact of ME/CFS;
- Test the possible interaction between COVID-19 and ME/CFS in Latvian circumstances, by conducting a survey of former COVID-19 patients on the presence of ME/CFS symptoms;
- Make preliminary predictions on the potential shadow impact of ME/CFS on society, limiting this study to direct costs for patients.
2. Theoretical Background and Literature Review
- ME/CFS (Canadian Consensus Criteria (CCC, 2003) [14]), and;
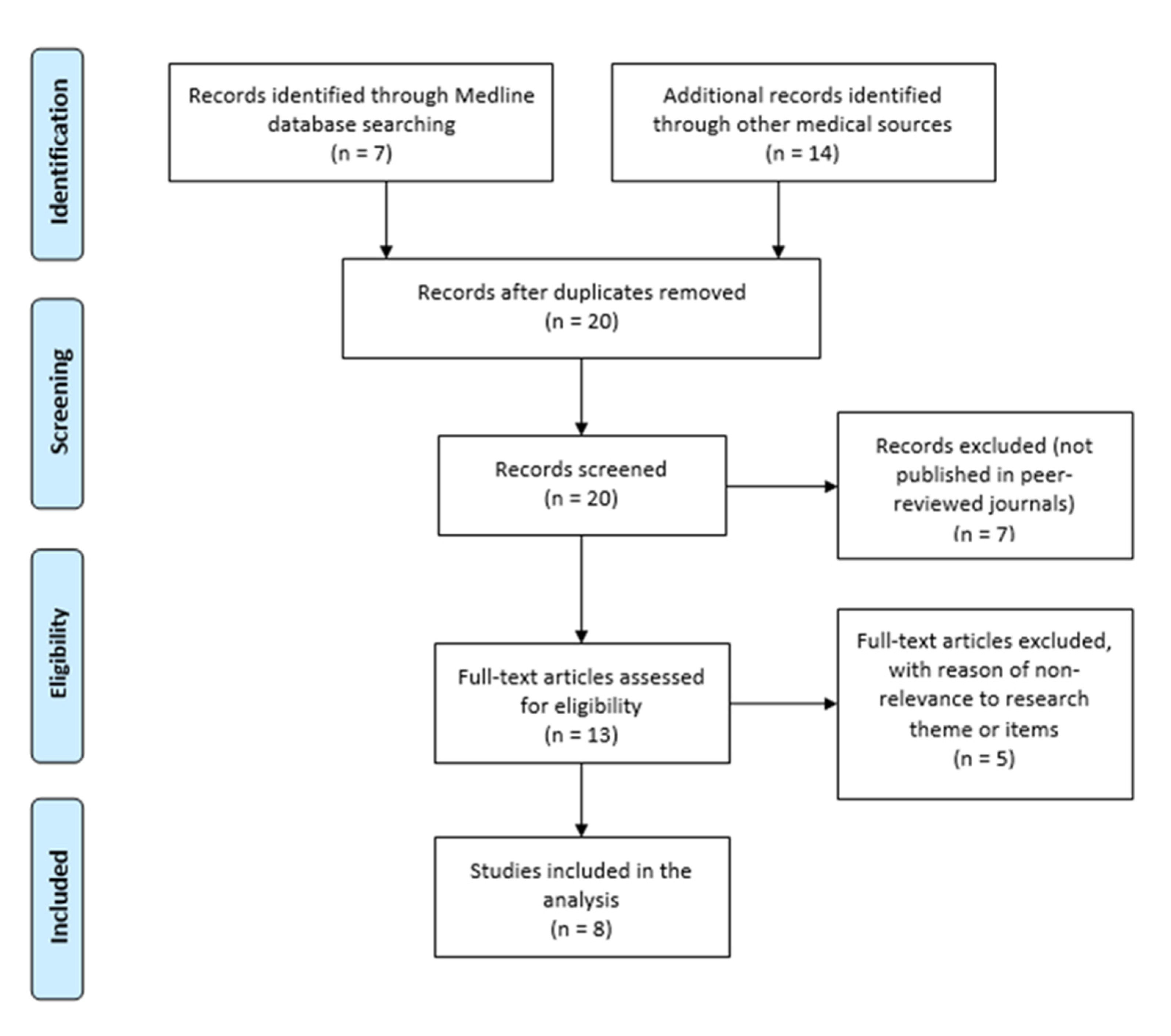
3. Materials and Methods of Empirical Research
- (1)
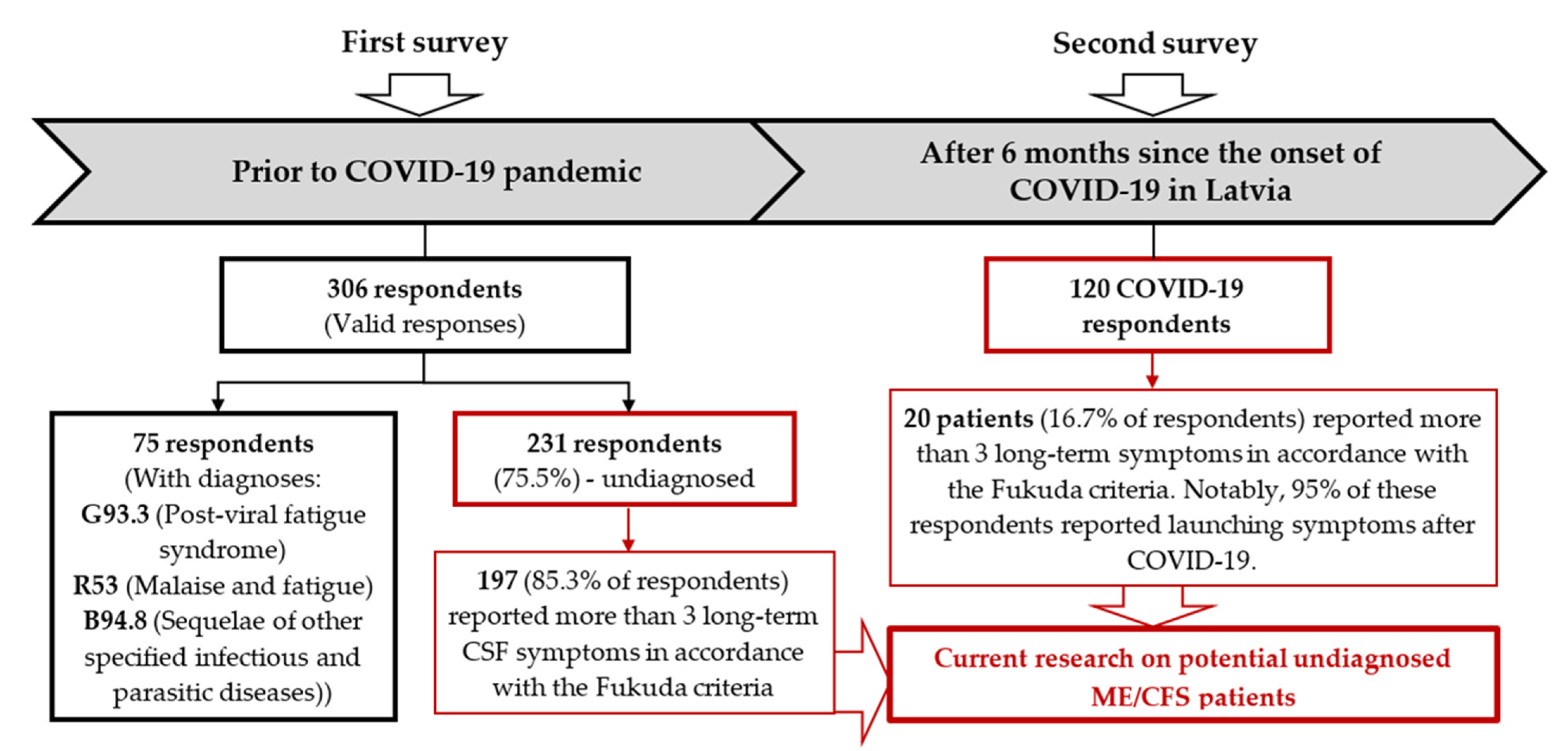
- The first part was dedicated to comparison of self-reported data from ME/CFS patients with those from persons experiencing symptoms similar to those of ME/CFS, but without a diagnosis, obtained by the survey performed prior to the COVID-19 pandemic. This part envisaged the creation of an assumption on the ME/CFS shadow burden “status quo”—not addressing the impact of COVID-19—in Latvia.
- (2)
- The second part aimed to investigate the data from COVID-19 patients’ surveyed on the presence of ME/CFS symptoms, 6 months after being affected by COVID-19, in Latvia.
4. Results
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pheby, D.F.H.; Araja, D.; Berkis, U.; Brenna, E.; Cullinan, J.; de Korwin, J.-D.; Gitto, L.; Hughes, D.A.; Hunter, R.M.; Trepel, D.; et al. The development of a consistent Europe-wide approach to investigating the economic impact of myalgic encephalomyelitis (ME/CFS): A report from the European Network on ME/CFS (EUROMENE). Healthcare 2020, 8, 88. [Google Scholar] [CrossRef] [Green Version]
- Brenna, E.; Gitto, L. The economic burden of Chronic Fatigue Syndrome/Myalgic Encephalomyelitis (CFS/ME): An initial summary of the existing evidence and recommendations for further research. Eur. J. Pers. Cent. Healthc. 2017, 5, 413–420. [Google Scholar] [CrossRef]
- Lim, E.-J.; Ahn, Y.-C.; Jang, E.-S.; Lee, S.-W.; Lee, S.-H.; Son, C.-G. Systematic review and meta-analysis of the prevalence of chronic fatigue syndrome/ myalgic encephalomyelitis (CFS/ME). J. Transl. Med. 2020, 18, 100. [Google Scholar] [CrossRef] [PubMed]
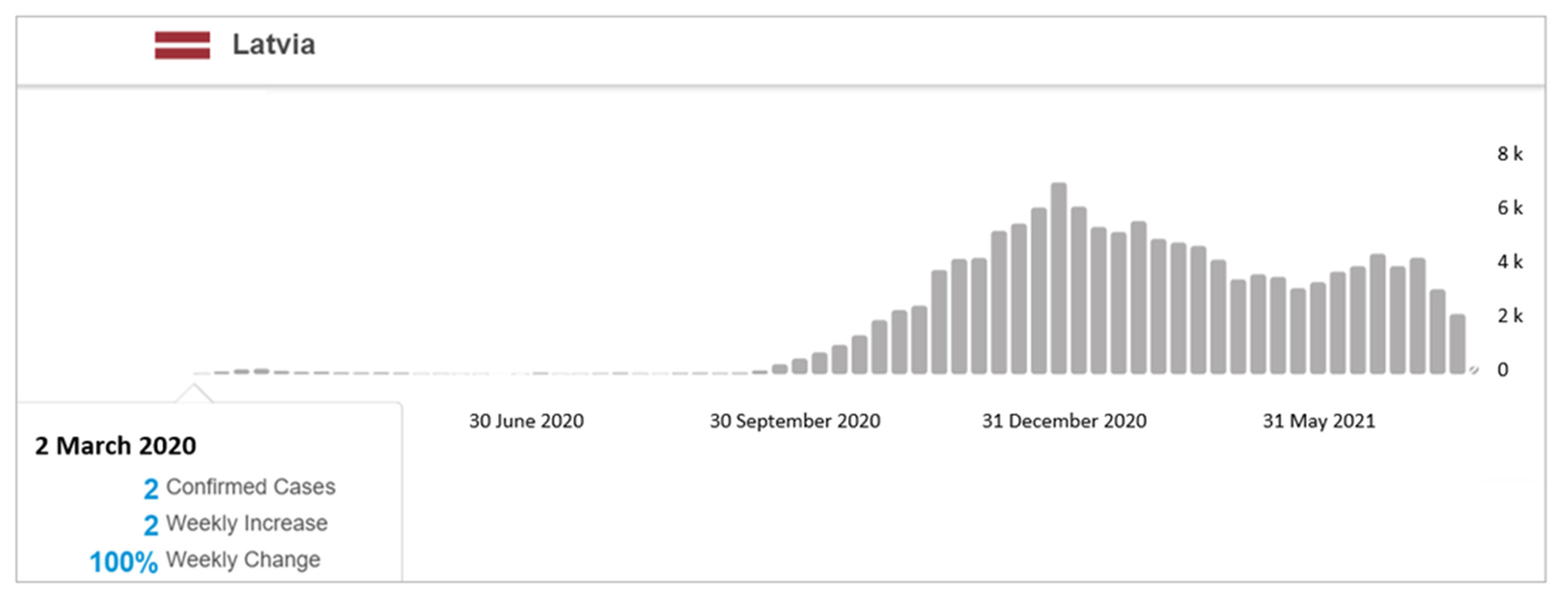
- World Health Organisation Coronavirus (COVID-19) Dashboard with Vaccination Data, Confirmed Cases in Latvia, March 2020–May 2021. Available online: https://covid19.who.int/region/euro/country/lv (accessed on 2 June 2021).
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems 10th Revision. Available online: https://icd.who.int/browse10/2019/en (accessed on 16 March 2021).
- Twisk, F. Myalgic Encephalomyelitis (ME) or What? An Operational Definition. Diagnostics 2018, 8, 64. [Google Scholar] [CrossRef] [Green Version]
- Lim, E.-J.; Son, C.-G. Review of case definitions for myalgic encephalomyelitis/ chronic fatigue syndrome (ME/CFS). J. Transl. Med. 2020, 18, 289. [Google Scholar] [CrossRef]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.; Dobbins, J.G.; Komaroff, A.L. The chronic fatigue syndrome: A comprehensive approach to its definition and study. Ann. Intern. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Holmes, G.P.; Kaplan, J.E.; Gantz, N.M.; Komaroff, A.L.; Schonberger, L.B.; Straus, S.E.; Jones, J.F.; Dubois, R.E.; Cunningham-Rundles, C.; Pahwa, S.; et al. Chronic fatigue syndrome: A working case definition. Ann. Intern Med. 1988, 108, 387–389. [Google Scholar] [CrossRef]
- Lloyd, A.R.; Hickie, I.; Boughton, C.R.; Spencer, O.; Wakefield, D. Prevalence of chronic fatigue syndrome in an Australian population. Med. J. Aust. 1990, 153, 522–528. [Google Scholar] [CrossRef]
- Sharpe, M.C.; Archard, L.C.; Banatvala, J.E.; Borysiewicz, L.K.; Clare, A.W.; David, A.; Edwards, R.H.; Hawlon, K.E.; Lambert, H.P.; Lane, R.J. A report–chronic fatigue syndrome: Guidelines for research. J. R. Soc. Med. 1991, 84, 118–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramsay, A.M.; Dowsett, E.G. Myalgic Encephalomyelitis: Then and Now. In Clinical and Scientific Basis of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome, 1st ed.; Hyde, B.M., Goldstein, J., Levine, P., Eds.; The Nightingale Research Foundation: Ottawa, ON, Canada, 1992; pp. 569–595. ISBN 978-0969566205. [Google Scholar]
- Carruthers, B.M.; Van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International Consensus Criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef] [Green Version]
- Carruthers, B.M.; Van de Sande, M.I. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Clinical Case Definition and Guidelines for Medical Practitioners. An Overview of the Canadian Consensus Document. The National Library of Canada, 2005. Available online: https://www.investinme.org/Documents/PDFdocuments/Canadian_ME_Overview_A4.pdf (accessed on 4 March 2021).
- Institute of Medicine. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome; Redefining an Illness. The National Academies Press, 2015. Available online: www.nap.edu (accessed on 12 February 2021).
- Twisk, F.N.M. Myalgic Encephalomyelitis, Chronic Fatigue Syndrome, and Systemic Exertion Intolerance Disease: Three Distinct Clinical Entities. Challenges 2018, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Germain, A.; Ruppert, D.; Levine, S.M.; Hanson, M.R. Prospective Biomarkers from Plasma Metabolomics of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Implicate Redox Imbalance in Disease Symptomatology. Metabolites 2018, 8, 90. [Google Scholar] [CrossRef] [Green Version]
- Almenar-Pérez, E.; Sánchez-Fito, T.; Ovejero, T.; Nathanson, L.; Oltra, E. Impact of Polypharmacy on Candidate Biomarker miRNomes for the Diagnosis of Fibromyalgia and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Striking Back on Treatments. Pharmaceutics 2019, 11, 126. [Google Scholar] [CrossRef] [Green Version]
- Sweetman, E.; Noble, A.; Edgar, C.; Mackay, A.; Helliwell, A.; Vallings, R.; Ryan, M.; Tale, W. Current Research Provides Insight into the Biological Basis and Diagnostic Potential for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Diagnostics 2019, 9, 73. [Google Scholar] [CrossRef] [Green Version]
- Scheibenbogen, C.; Freitag, H.; Blanco, J.; Capelli, E.; Lacerda, E.; Authier, J.; Meeus, M.; Castro Marrero, J.; Nora-Krukle, Z.; Oltra, E.; et al. The European ME/CFS Biomarker Landscape project: An initiative of the European network EUROMENE. J. Transl. Med. 2017, 15, 162. [Google Scholar] [CrossRef] [Green Version]
- Pheby, D.F.H.; Araja, D.; Berkis, U.; Brenna, E.; Cullinan, J.; de Korwin, J.-D.; Gitto, L.; Hughes, D.A.; Hunter, R.M.; Trepel, D.; et al. A Literature Review of GP Knowledge and Understanding of ME/CFS: From the Socioeconomic Working Group of the European Network on ME/CFS (EUROMENE). Medicina 2021, 57, 7. [Google Scholar] [CrossRef]
- Cullinan, J.; Pheby, D.; Araja, D.; Berkis, U.; Brenna, E.; de Korwin, J.-D.; Gitto, L.; Hughes, D.; Hunter, R.; Trepel, D.; et al. Perceptions of European ME/CFS Experts Concerning Knowledge and Understanding of ME/CFS among Primary Care Physicians in Europe: A Report from the European ME/CFS Research Network (EUROMENE). Medicina 2021, 57, 208. [Google Scholar] [CrossRef] [PubMed]
- Brenna, E.; Araja, D.; Pheby, D.F.H. Comparative Survey of People with ME/CFS in Italy, Latvia, and the UK: A Report on behalf of the Socioeconomics Working Group of the European ME/CFS Research Network (EUROMENE). Medicina 2021, 57, 300. [Google Scholar] [CrossRef] [PubMed]
- Haywood, K.L.; Staniszewska, S.; Sarah Chapman, S. Quality and acceptability of patient-reported outcome measures used in chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME): A systematic review. Qual. Life. Res. 2012, 21, 35–52. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-Y.; Lee, J.-S.; Son, C.-G. Systematic Review of Primary Outcome Measurements for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis (CFS/ME) in Randomized Controlled Trials. J. Clin. Med. 2020, 9, 3463. [Google Scholar] [CrossRef] [PubMed]
- Araja, D.; Berkis, U.; Castro Marrero, J.; Ivanovs, A.; Krumina, A.; Lunga, A.; Murovska, M.; Svirskis, S.; Zalewski, P. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) patients’ engagement in outcome measuring and disease management through Information Technology. ICHOM Congr. (Present.) 2020. [Google Scholar] [CrossRef]
- Islam, M.F.; Cotler, J.; Jason, L.A. Post-viral fatigue and COVID-19: Lessons from past epidemics. Fatigue Biomed. Health Behav. 2020, 8, 61–69. [Google Scholar] [CrossRef]
- Muller, A.E.; Tveito, K.; Bakken, I.J.; Flottorp, S.A.; Mjaaland, S.; Larun, L. Potential causal factors of CFS/ME: A concise and systematic scoping review of factors researched. J. Transl. Med. 2020, 18, 484. [Google Scholar] [CrossRef] [PubMed]
- Cullinan, J.; Ní Chomhraí, O.; Kindlon, T.; Black, L.; Casey, B. Understanding the economic impact of myalgic encephalomyelitis/chronic fatigue syndrome in Ireland: A qualitative study [version 1; peer review: 2 approved]. HRB Open Res. 2020, 3, 88. [Google Scholar] [CrossRef]
- Strayer, D.R.; Young, D.; Mitchell, W.M. Effect of disease duration in a randomized Phase III trial of rintatolimod, an immune modulator for Myalgic Encephalomyelitis/ Chronic Fatigue Syndrome. PLoS ONE 2020, 15, e0240403. [Google Scholar] [CrossRef]
- Gaber, T. Assessment and management of post-COVID fatigue. Prog. Neurol. Psychiatry 2021, 25, 36–39. [Google Scholar] [CrossRef]
- Friedman, K.J.; Murovska, M.; Pheby, D.F.H.; Zalewski, P. Our Evolving Understanding of ME/CFS. Medicina 2021, 57, 200. [Google Scholar] [CrossRef]
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med. Virol. 2021, 93, 1013–1022. [Google Scholar] [CrossRef]
- Simani, L.; Ramezani, M.; Darazam, I.A.; Sagharichi, M.; Aalipour, M.A.; Ghorbani, F.; Pakdaman, H. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. J. Neuro Virol. 2021. [Google Scholar] [CrossRef]
- Townsend, L.; Moloney, D.; Finucane, C.; McCarthy, K.; Bergin, C.; Bannan, C.; Kenny, R.-A. Fatigue following COVID-19 infection is not associated with autonomic dysfunction. PLoS ONE 2021, 16, e0247280. [Google Scholar] [CrossRef]
- Graham, E.L.; Clark, J.R.; Orban, Z.S.; Lim, P.H.; Szymanski, A.L.; Taylor, C.; DiBiase, R.M.; Jia, D.T.; Balabanov, R.; Ho, S.U.; et al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized Covid-19 ‘long haulers’. Ann. Clin. Transl. Neurol. 2021, 8, 1073–1085. [Google Scholar] [CrossRef] [PubMed]
- Toogood, P.L.; Clauw, D.J.; Phadke, S.; Hoffman, D. Myalgic encephalomyelitis/ chronic fatigue syndrome (ME/CFS): Where will the drugs come from? Pharmacol. Res. 2021, 165, 105465. [Google Scholar] [CrossRef] [PubMed]
- Rovite, V.; Wolff-Sagi, Y.; Zaharenko, L.; Nikitina-Zake, L.; Grens, E.; Klovins, J. Genome Database of the Latvian Population (LGDB): Design, Goals, and Primary Results. J. Epidemiol. 2018, 28, 353–360.b. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Close, S.; Marshall-Gradisnik, S.; Byrnes, J.; Smith, P.; Nghiem, S.; Staines, D. The Economic Impacts of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in an Australian Cohort. Front. Public Health 2020, 8, 420. [Google Scholar] [CrossRef] [PubMed]
- Households’ Disposable Income per Household Member, in 2019. Central Statistical Bureau (Republic of Latvia), 2020. Available online: https://www.csb.gov.lv/en/statistics/statistics-by-theme/social-conditions/household-budget/tables/iig040/households-disposable-income-euro-month (accessed on 12 February 2021).
- Central Statistical Bureau of Latvia. Population in Latvia, at the Beginning of 2020. Available online: https://www.csb.gov.lv/en/statistics/statistics-by-theme/population/number-and-change/search-in-theme/2694 (accessed on 12 April 2021).
- Pheby, D.F.H.; Araja, D.; Berkis, U.; Brenna, E.; Cullinan, J.; de Korwin, J.; Gitto, L.; Hughes, D.A.; Hunter, R.M.; Trepel, D.; et al. The Role of Prevention in Reducing the Economic Impact of ME/CFS in Europe: A Report from the Socioeconomics Working Group of the European Network on ME/CFS (EUROMENE). Medicina 2021, 57, 388. [Google Scholar] [CrossRef] [PubMed]
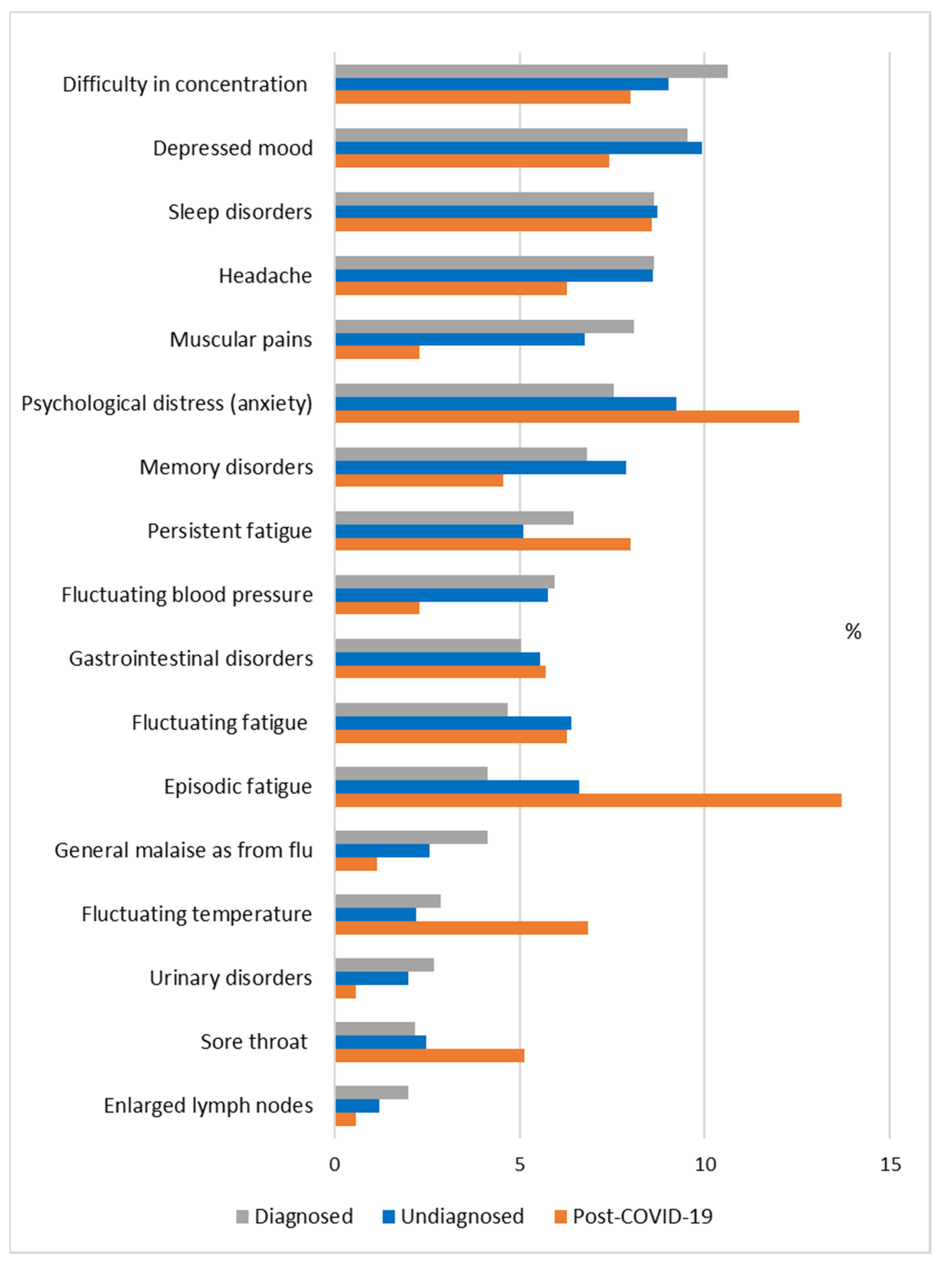
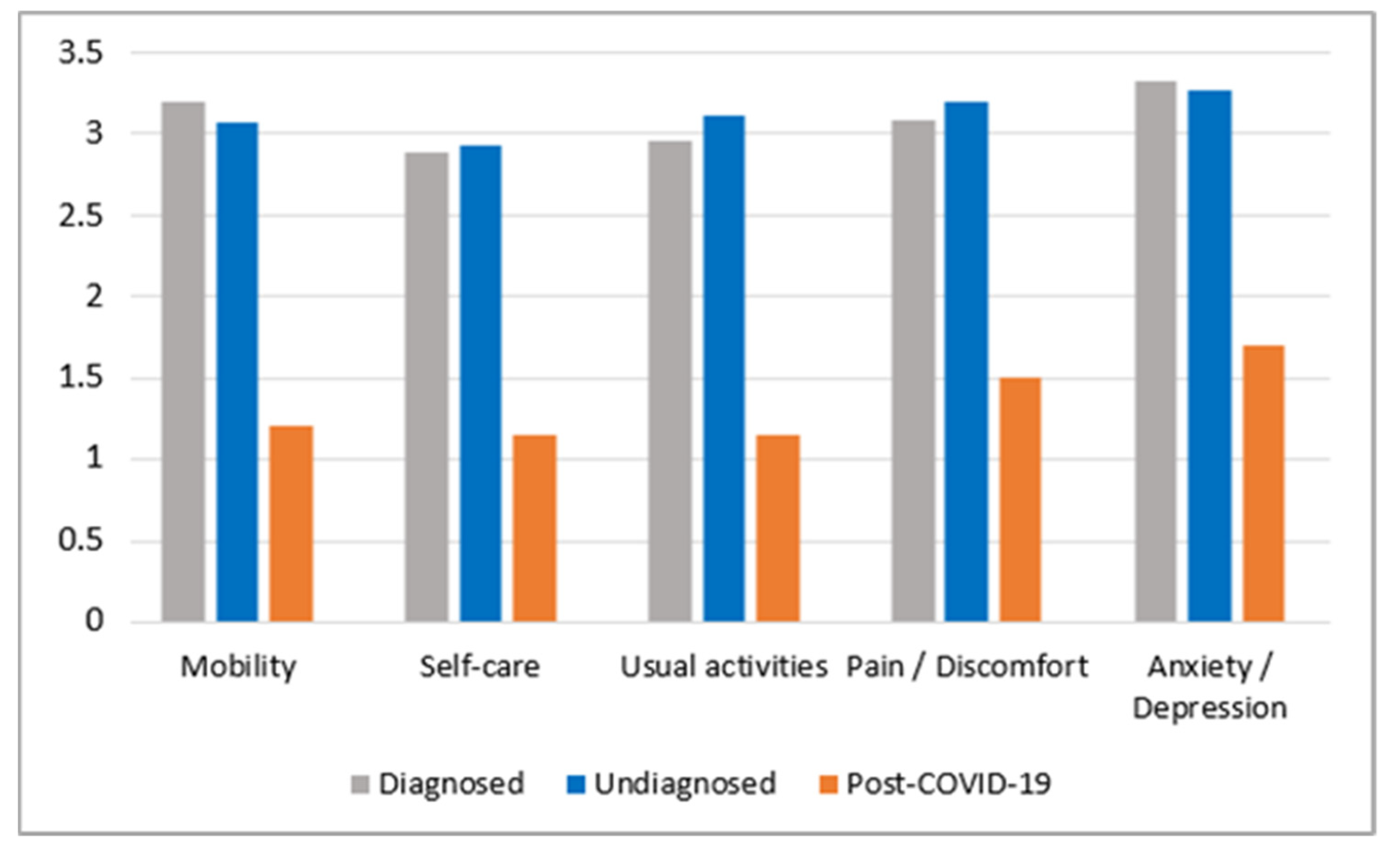
| Authors | Type of Research | Main Results and Conclusions |
|---|---|---|
| Strayer et al. (Oct 2020) [30] | Research Article | The results may have direct relevance to the cognitive impairment and fatigue being experienced by patients clinically recovered from COVID-19 and free of detectable SARS-CoV-2. |
| Gaber (Jan 2021) [31] | Review | Post-viral fatigue is the most common long-term health issue facing survivors of COVID-19, according to initial reports. The author discusses the risk, diagnosis, and principles of management of post-viral fatigue and its chronic variant—ME/CFS—within the context of the pandemic, and highlights that further research is urgently needed to guide clinical practice. Several symptoms are classically associated with post-viral fatigue and ME/CFS, including physical pain, recurrent headaches, malaise, cognitive impairment, unrefreshing sleep, recurrent sore throats, and lymphadenopathy. These symptoms are strongly associated with the post-exertional phase of the boom-and-bust cycle. Identification of the post-COVID patients needing support and treatment should be a part of the overall COVID-19 response globally. |
| Friedman et al. (Feb 2021) [32] | Opinion | The similarity and overlap of ME/CFS and long-haul COVID-19 symptoms suggest similar pathological processes. A unifying hypothesis explains the precipitating events, such as viral triggers and other documented exposures; for their overlap in symptoms, ME/CFS and long-haul COVID-19 should be described as post-active-phase-of-infection syndromes (PAPISs). The authors further propose that the underlying biochemical pathways and pathophysiological processes of similar symptoms are similar regardless of the initiating trigger. The authors caution that failure to meet the now combined challenges of ME/CFS and long-haul COVID-19 will impose serious socioeconomic as well as clinical consequences for patients, the families of patients, and society as a whole. |
| Halpin et al. (Feb 2021) [33] | Research Article | There is currently very limited information on the nature and prevalence of post-COVID-19 symptoms after hospital discharge. In this research, a purposive sample of 100 survivors discharged from a large university hospital was assessed 4–8 weeks after discharge by a multidisciplinary team of rehabilitation professionals. Participants were between 29 and 71 days (mean 48 days) post-discharge from hospital; 32 participants required treatment in an intensive care unit (ICU group), and 68 were managed in hospital wards without needing ICU care (ward group). New illness-related fatigue was the most commonly reported symptom—by 72% of participants in the ICU group and 60.3% in the ward group. There was a clinically significant drop in EQ5D, of 68.8% in the ICU group and 45.6% in the ward group. The authors recommend planning rehabilitation services to manage post-discharge symptoms appropriately and maximize the functional return of COVID-19 survivors. |
| Simani et al. (Feb 2021) [34] | Research Article | The obtained data revealed the prevalence of CFS among patients with COVID-19, which is almost similar to CFS prevalence in the general population. Moreover, post-traumatic stress disorder (PTSD) in patients with COVID-19 is not associated with an increased risk of CFS. This study suggests that medical institutions should pay attention to the psychological consequences of the COVID-19 outbreak. |
| Townsend et al. (Feb 2021) [35] | Research Article | The results demonstrate the significant burden of fatigue, symptoms of autonomic dysfunction, and anxiety in the aftermath of COVID-19 infection but, reassuringly, do not demonstrate pathological findings on autonomic testing. |
| Graham et al. (Mar 2021) [36] | Research Article | A prospective study of the first 100 consecutive patients (50 SARS-CoV-2 laboratory-positive (SARS-CoV-2+) and 50 laboratory-negative (SARS-CoV-2−) individuals) presenting to the Neuro-Covid-19 clinic between May and November 2020 concluded that non-hospitalized COVID-19 “long-haulers” experience prominent and persistent “brain fog” and fatigue that affect their cognition and quality of life. |
| Toogood et al. (Mar 2021) [37] | Review | Viral infection is an established trigger for the onset of ME/CFS symptoms, raising the possibility of an increase in ME/CFS prevalence resulting from the ongoing COVID-19 pandemic. |
| Item | Persons’ Group | No. of Respondents | Mean | Standard Deviation (SD) | No. Responding “Yes” | % | 95% Confidence Interval (%) |
|---|---|---|---|---|---|---|---|
| Age (Years) | Diagnosed | 75 | 50.0 | 14.7 | 46.6–53.3 | ||
| Undiagnosed | 222 | 45.1 | 12.9 | 43.4–46.8 | |||
| Gender (No. Females) | Diagnosed | 75 | 62 | 82.7 | 74.1–91.2 | ||
| Undiagnosed | 226 | 187 | 82.7 | 77.8–87.7 | |||
| Education (No. with Higher Education) | Diagnosed | 74 | 32 | 43.2 | 32.0–54.5 | ||
| Undiagnosed | 225 | 115 | 51.1 | 44.6–57.6 | |||
| No. Living Alone | Diagnosed | 74 | 25 | 33.8 | 23.0–44.6 | ||
| Undiagnosed | 224 | 69 | 30.8 | 24.8–36.9 | |||
| Household Income, per Member (EUR, p.a.) | Diagnosed | 65 | 5364.4 | 2991.1 | 4637.3–6091.55 | ||
| Undiagnosed | 213 | 6365.5 | 3819.7 | 5852.5–6878.5 | |||
| No. Symptoms | Diagnosed | 75 | 7.5 | 2.5 | 6.9–8.1 | ||
| Undiagnosed | 231 | 6.3 | 2.8 | 5.9–6.7 | |||
| Variability of Symptoms | Diagnosed | 75 | 53 | 70.7 | 60.4–81.0 | ||
| Undiagnosed | 231 | 178 | 77.1 | 71.6–82.5 | |||
| No. Investigations | Diagnosed | 75 | 5.7 | 3.2 | 5.0–6.4 | ||
| Undiagnosed | 124 | 4.7 | 2.5 | 4.3–5.1 | |||
| Out-of-Pocket Spending, to Mitigate Symptoms (EUR, p.a.) | Diagnosed | 75 | 1143.0 | 125.1 | 1114.7–1171.3 | ||
| Undiagnosed | 209 | 979.2 | 156.1 | 958.0–1000.4 | |||
| Difficulty Explaining Illness to | |||||||
| Physicians | Diagnosed | 75 | 20 | 26.7 | 16.7–36.7 | ||
| Undiagnosed | 231 | 76 | 32.9 | 26.8–39.0 | |||
| Family | Diagnosed | 75 | 35 | 46.7 | 35.4–58.0 | ||
| Undiagnosed | 231 | 100 | 43.3 | 36.9–49.7 | |||
| Friends | Diagnosed | 75 | 20 | 26.7 | 16.7–36.7 | ||
| Undiagnosed | 231 | 70 | 30.3 | 24.4–36.2 | |||
| Employers | Diagnosed | 75 | 30 | 40.0 | 28.9–51.1 | ||
| Undiagnosed | 231 | 81 | 35.1 | 28.9–41.2 | |||
| Quality of Life: | |||||||
| Prior to Illness | Diagnosed | 74 | 74.6 | 24.0 | 69.0–80.2 | ||
| Undiagnosed | 212 | 74.1 | 22.0 | 71.1–77.1 | |||
| In Past Year | Diagnosed | 74 | 57.3 | 16.3 | 53.5–61.1 | ||
| Undiagnosed | 219 | 58.1 | 16.8 | 55.9–60.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araja, D.; Berkis, U.; Lunga, A.; Murovska, M. Shadow Burden of Undiagnosed Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) on Society: Retrospective and Prospective—In Light of COVID-19. J. Clin. Med. 2021, 10, 3017. https://doi.org/10.3390/jcm10143017
Araja D, Berkis U, Lunga A, Murovska M. Shadow Burden of Undiagnosed Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) on Society: Retrospective and Prospective—In Light of COVID-19. Journal of Clinical Medicine. 2021; 10(14):3017. https://doi.org/10.3390/jcm10143017
Chicago/Turabian StyleAraja, Diana, Uldis Berkis, Asja Lunga, and Modra Murovska. 2021. "Shadow Burden of Undiagnosed Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) on Society: Retrospective and Prospective—In Light of COVID-19" Journal of Clinical Medicine 10, no. 14: 3017. https://doi.org/10.3390/jcm10143017
APA StyleAraja, D., Berkis, U., Lunga, A., & Murovska, M. (2021). Shadow Burden of Undiagnosed Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) on Society: Retrospective and Prospective—In Light of COVID-19. Journal of Clinical Medicine, 10(14), 3017. https://doi.org/10.3390/jcm10143017







